Transgenesis and web resources in quail
Figures
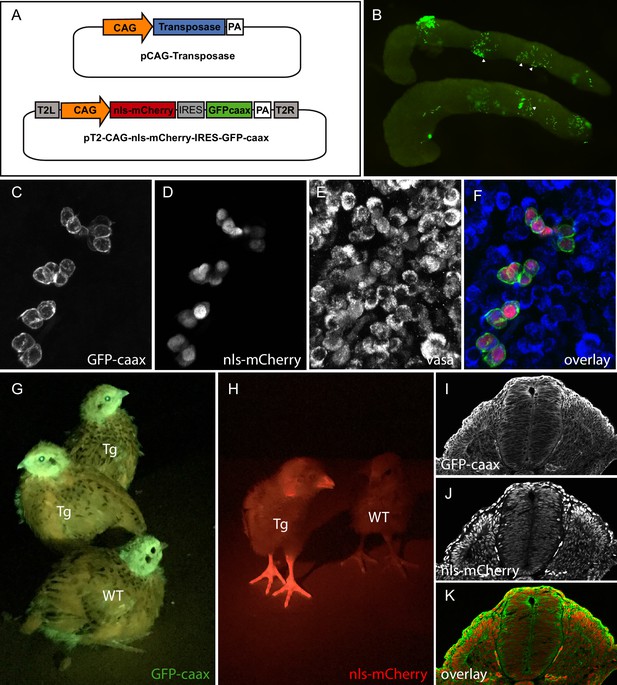
PGC transfection in vivo and generation of a quail line ubiquitously expressing membranal GFP and nuclear mCherry.
(A) Vectors used in the injection mix. (B) Gonads from E7 embryo dissected 5 days after in vivo PGC transfection, showing GFP-positive transfected PGCs (arrowheads). (C–F) Confocal views of transfected (GFP- and mCherry-positive) PGCs among non-transfected PGCs, in the gonads of E7 injected embryo. PGCs are recognized by their expression of the Vasa marker (E,F). (G–H) Transgenic (Tg) and wild-type (WT) chicks showing ubiquitous expression of membranal GFP and nuclear mCherry when observed with UV goggles. (I–J) cross-section of an E3 transgenic quail embryo, showing strong and ubiquitous expression of the transgenes in all cells of the embryo.
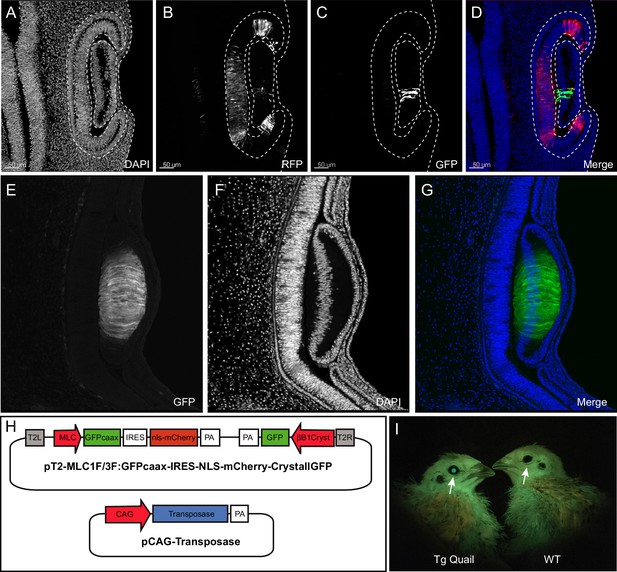
Design and use of the CrystallGFP mini gene.
(A–D) Cross-section of the head of an E4 embryo, electroporated one day earlier in the optic cup with a CrystallGFP minigene. (A) DAPI, (B) electroporation marker CAG-RFP plasmid, (C) GFP, (D) overlay. (E–G) Cross-section of the head of a 3-day-old embryo of the Tg(MLC:GFP-IRES-NLS-mCherry,CRYBB1:GFP) transgenic line showing the specific expression of GFP throughout the lens. (H) Electroporation constructs used to express the CrystallGFP minigene in a muscle-specific transgenic line (see Figure 4). (I) Transgenic and WT adults of the muscle-specific transgenic line showing GFP expression in lens.
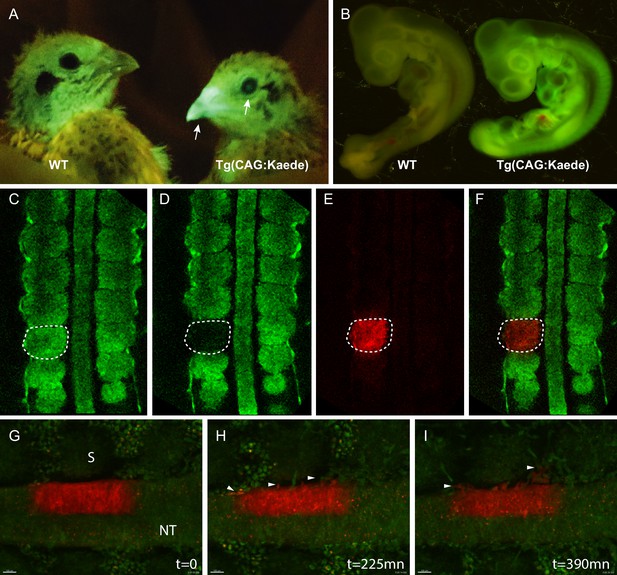
Generation of the photoconvertible Kaede transgenic quail line TgT2(CAG:Kaede).
(A) Two-week-old WT and transgenic quails showing the ubiquitous expression of the green fluorescent Kaede in the beak and eye (arrows). (B) WT and transgenic 3-day-old embryos showing strong ubiquitous expression of the protein. (C–F) A newly formed somite before (C) and after (D–F) photoconversion. (G–I) Snapshots from a time-lapse video (see Video 3) showing the morphogenic movements of photoconverted neural tube cells. Arrowheads in H and I show neural crest cells initiating their lateral migration. NT: Neural Tube, S: Somite.
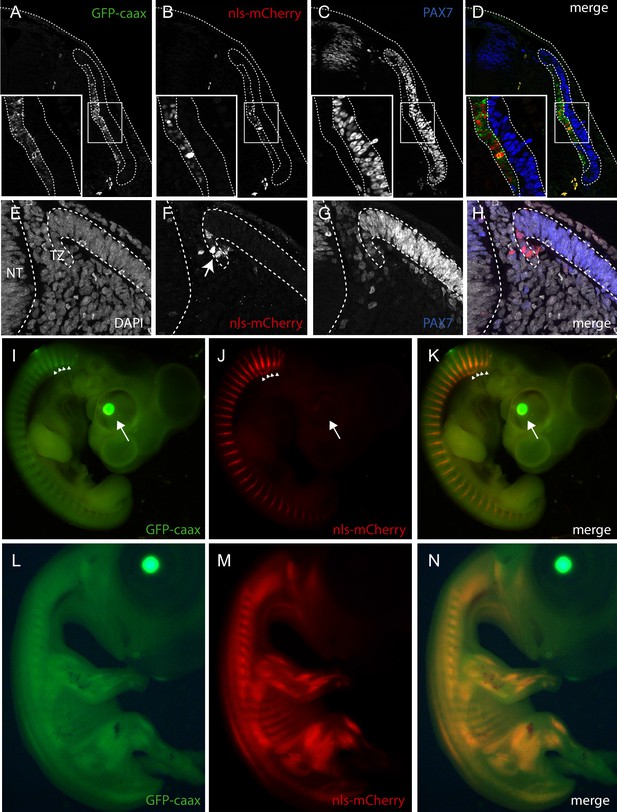
Description of muscle specific transgenic quail TgT2(Mmu.MLC1F/3F:GFP-CAAX-IRES-NLS-mCherry,Gga.CRYBB1:GFP).
(A–D) Cross-section of E3 transgenic embryo stained for the indicated markers, showing the expression of the transgene throughout the primary myotome. (A) GFP-CAAX, (B) NLS-mCherry, (C) Pax7, (D) Merge. Insets in (A–D) Magnifications of the regions indicated in (A–D) showing the cellular localisation of the markers. (E–H) E5 Transgenic embryo showing GFP-CAAX (E) and NLS-mCherry (F) in the transition zone (TZ, arrow) where progenitors from the dermomyotome translocate to elongate and differentiate. (I–K) E5 embryos showing strong and specific expression of the muscle-specific reporter in somites (arrowheads). In this quail line, transgenic embryos can be selected at hatching by tηε GFP expression in lens due to the CrystallGFP minigene (arrows). (H–J) E7 transgenic embryo showing muscle-specific expression of the transgene in the head, limbs and trunk.
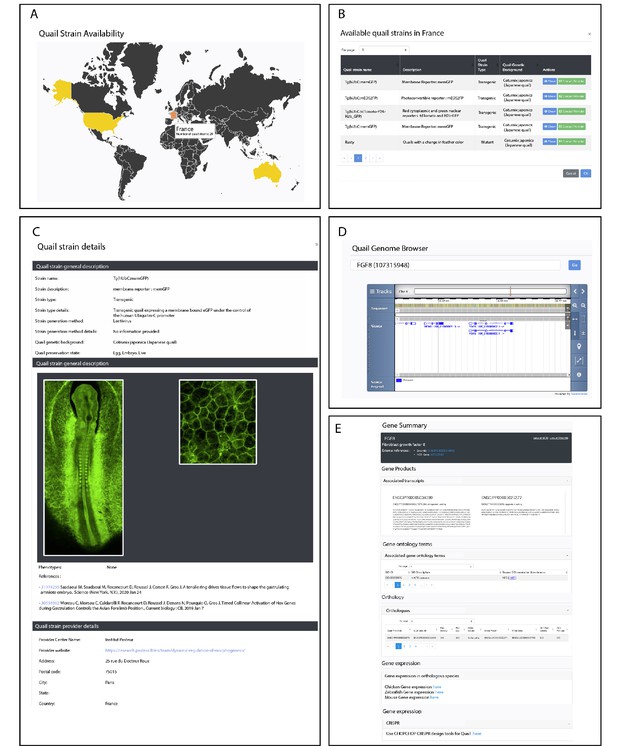
Description of QuailNet features.
(A) Interactive world map displaying the number of quail strains available by country. (B) List of quail strains together with a general description by country. (C) Detailed description of a specific quail strain (e.g. Tg(hUbC:memGFP)). (D) Quail genome browser displaying genomic information and location of a queried gene (e.g. FGF8). (E) Information associated with a queried gene (e.g. FGF8).
Videos
Time-lapse video of an E2 TgT2(CAG:GFP-CAAX-IRES-NLS-mCherry) embryo observed in ovo.
Embryo was maintained at 38°C and imaged every 10mn for 12 hr using Thunder Imager Model Organism Leica stereo microscope equipped with 1x lens.
Time-lapse video of an E2.5 TgT2(CAG:GFP-CAAX-IRES-NLS-mCherry) embryo.
Embryo was maintained at 38°C and imaged every 10mn for 12 hr using Thunder Imager Model Organism Leica stereo microscope equipped with 5x lens.
Time-lapse video of an E2.5 TgT2(CAG:Kaede) embryo.
Embryo was imaged using a Leica SP8 upright confocal microscope. A ROI was defined in half of the neural tube and exposed to UV light, photoconverting the Kaede protein from green to red. The area was imaged every 15mn for 7 hr showing neural crest cells migrating away from the neural tube.
Quail TgT2(Mmu.MLC1F/3F:GFP-CAAX-IRES-NLS-mCherry,Gga.CRYBB1:GFP) embryo at 3 days of development, immunostained for GFP (green) and mCherry (red) from the transgene and counterstained for neural crest (HNK1, blue), clarified by the ‘3DISCO’ technique and imaged with a light sheet microscope (LSFM Z1 Zeiss).
Image rendering and video obtained with an Arivis software suite.
Wing of a quail TgT2(Mmu.MLC1F/3F:GFP-CAAX-IRES-NLS-mCherry,Gga.CRYBB1:GFP) embryo at 6 days of development, immunostained for GFP (green) and mCherry (red), clarified by the ‘3DISCO’ technique and imaged with a light sheet microscope (LaVision Biotec).
Image rendering and video obtained with an Arivis software suite.





