Cold-induced hyperphagia requires AgRP neuron activation in mice
Figures
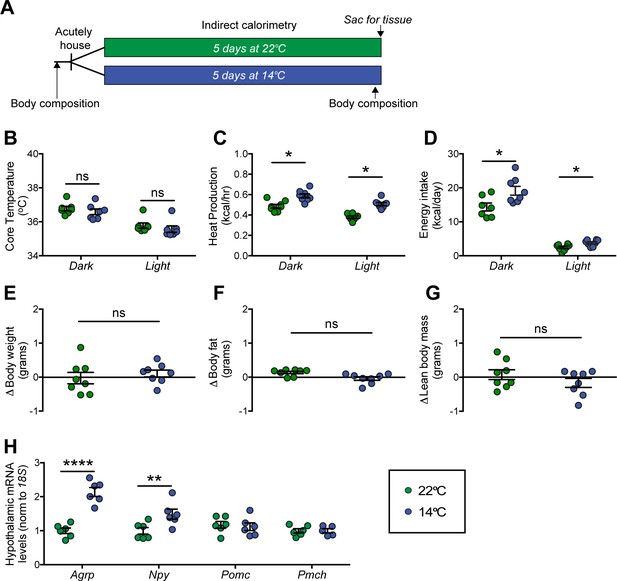
Effect of chronic cold exposure on determinants of energy balance and hypothalamic neuropeptide gene expression.
(A) Adult male, wild-type mice were housed at the start of the dark cycle following body-composition measures and were maintained in temperature-controlled chambers set to either mild cold (14°C) or, as control, room temperature (22°C) for 5 days. Following a final body-composition analysis, animals were sacrificed and hypothalamic punches rapidly dissected. Mean dark and light cycle (B) core body temperature, (C) heat production, and (D) energy intake over 5 days while housed at either 22°C or 14°C. Change in (E) body weight, (F) body fat, and (G) lean body mass at study end in the same mice, n = 7–8 per group, mean ± SEM. Student’s t-test, *p<0.05 vs. 22°C. (H) Hypothalamic mRNA levels of agouti-related peptide (Agrp), neuropeptide Y (Npy), pro-opiomelanocortin (Pomc), and pro-melanin concentrating hormone (Pmch) as determined by qRT-PCR, mean ± SEM. Two-way ANOVA with Holm-Sidak correction for multiple comparisons, *p<0.05, **p<0.01, ****p<0.0001 vs. 22°C.
-
Figure 1—source data 1
Wild-type chronic study.
- https://cdn.elifesciences.org/articles/58764/elife-58764-fig1-data1-v2.xlsx
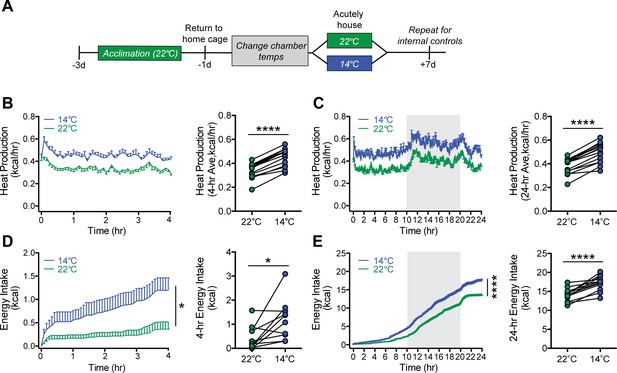
Acute mild cold exposure rapidly increases both energy expenditure and energy intake.
(A) Adult male wild-type mice were acutely housed in temperature-controlled chambers set to either mild cold (14°C) or, as a control, room temperature (22°C). Mean heat production (B and C) and energy intake (D and E) were measured over either 4 hr (B and D) or 24 hr (C and E), beginning at 10 AM, t = 0. n = 10 per group, mean ± SEM. ****p<0.0001, *p<0.05, based on mixed factorial-ANOVA for changes of food intake over time, and paired Student’s t-test to compare mean values of food intake and heat production.
-
Figure 2—source data 1
Wild-type acute study.
- https://cdn.elifesciences.org/articles/58764/elife-58764-fig2-data1-v2.xlsx
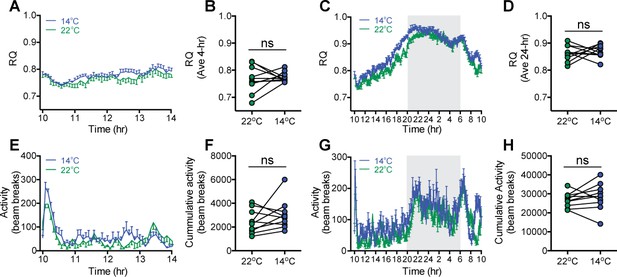
Effect of acute mild cold exposure on respiratory quotient (RQ) and ambulatory activity.
(A and C) Time-series and (B and D) mean RQ over 4 hr and 24 hr, respectively, and (E and G) time-series and (F and H) total ambulatory activity over 4 hr and 24 hr, respectively, in adult male wild-type mice moved into housing at either 14°C or 22°C beginning at 10 AM, n = 10 per group, mean ± SEM, paired Student’s t-test.
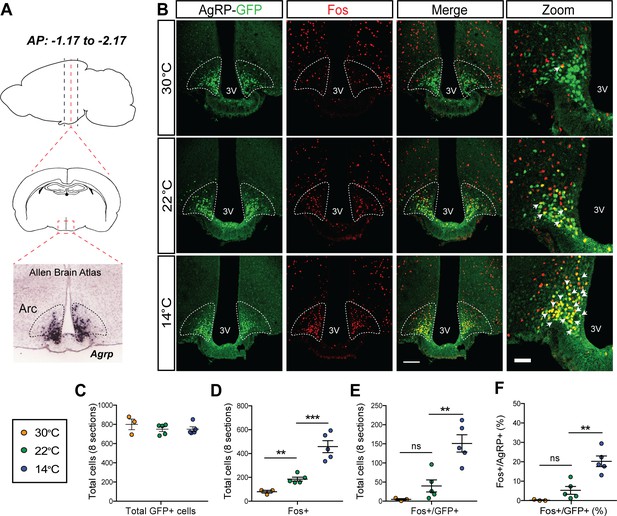
Acute mild cold exposure activates agouti-related peptide (AgRP) neurons.
(A) Representative sagittal and coronal images and Agrp hybridization in situ from Allen Brain Institute of the arcuate nucleus (ARC). (B) Immunohistochemical detection of AgRP-locus driven green fluorescent protein (GFP) (green), Fos (red), and colocalization of GFP and Fos (two right panels) in the ARC of AgRP-Cre:GFP mice 90 min after housing at either 14°C, 22°C, or 30°C. Quantitation of (C) total AgRP+ cells, (D) total Fos+ cells, and the total number (E) and percent (F) of AgRP neurons that co-express Fos across eight rostral to caudal sections of the ARC (AP: −1.17 to −2.17). Thin bar = 100 µm, thick bar = 50 µm, n = 3–5group, mean ± SEM. One-way ANOVA with Sidak correction for multiple comparisons, ***p<0.001, **p<0.01, *p<0.05 vs. 22°C.
-
Figure 3—source data 1
Arcuate nucleus Fos quantification.
- https://cdn.elifesciences.org/articles/58764/elife-58764-fig3-data1-v2.xlsx
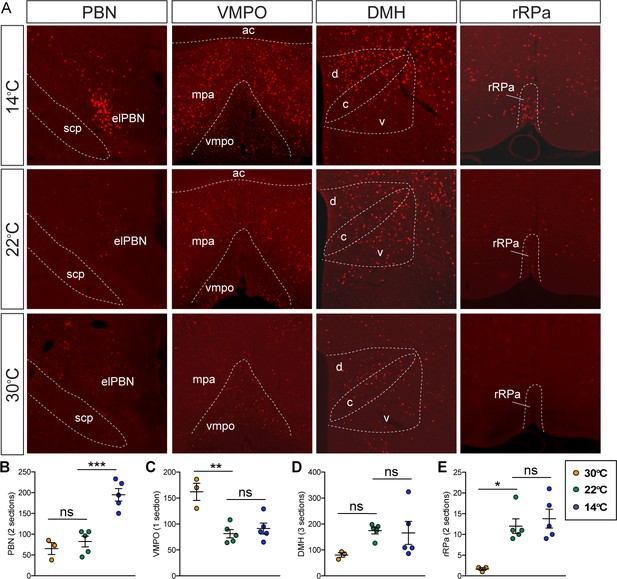
Fos induction in known thermoregulatory brain regions.
(A) Representative images of Fos induction in known thermoregulatory brain regions. Quantification of Fos induction in (B) parabrachial nucleus (PBN), (C) ventromedial preoptic area (VMPO) and medial preoptic area (MPA), (D) dorsomedial hypothalamus (DMH), and (E) rostral raphe pallidus (rRPa) following 90 min of exposure to either 30°C, or 22°C or 14°C, n = 3–5 per group. Mean ± SEM, one-way ANOVA with Sidak correction for multiple comparisons, ***p<0.001, **p<0.01, *p<0.05 vs. 22°C.
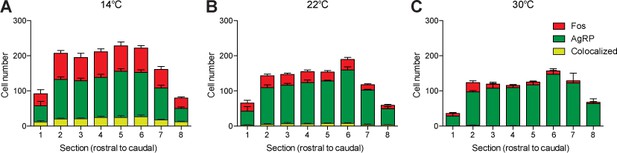
Fos induction in agouti-related peptide (AgRP) neurons across entire arcuate nucleus (ARC).
Fos induction in AgRP neurons across entire rostral to caudal axis of ARC after exposure to 90 min at (A) 14°C, (B) 22°C, or (C) 30°C.
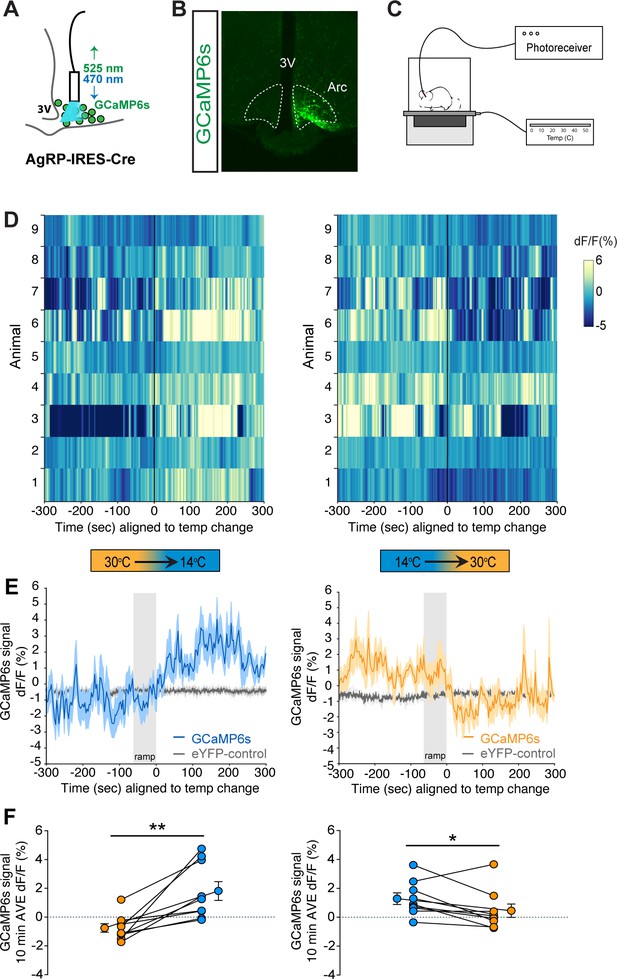
Cold sensation increases agouti-related peptide (AgRP)-neuron GCaMP activity in a rapidly reversible manner.
(A) Representative diagram of fiber photometry with fiber placement at the arcuate nucleus. (B) Unilateral GCaMP6s expression in AgRP neurons. (C) Representative diagram of experimental set up. (D) Heat maps of dF/F (%) for individual animals, (E) trace of averaged dF/F (%) GCaMP6s signal, and (F) quantification of mean dF/F (%) differences between 30°C and 14°C GCaMP6s activity during 10 min exposure. Gray bar signifies 60 s temperature-ramp transition from either 14°C to 30°C or 30°C to 14°C, denoted by ‘ramp’, n = 9 per group, mean ± SEM. Student’s paired t-test, **p<0.01, *p<0.05.
-
Figure 4—source data 1
Averaged photometry data.
- https://cdn.elifesciences.org/articles/58764/elife-58764-fig4-data1-v2.xlsx
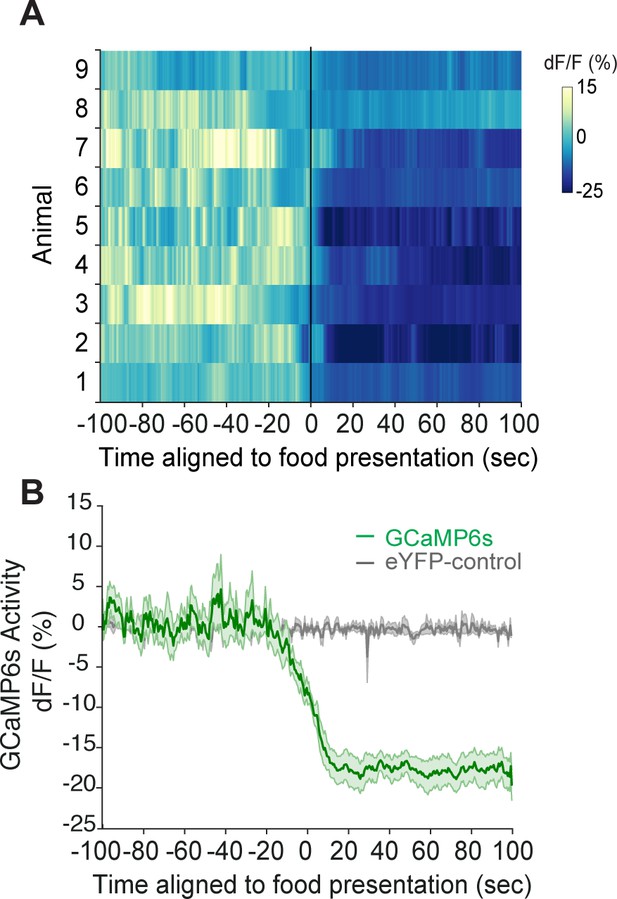
Post-fast refeeding inhibits agouti-related peptide (AgRP) neurons.
(A) Heat map showing GCaMP6s activity (dF/F (%)) in AgRP neurons from individual mice and (B) averaged GCaMP6s activity (dF/F (%)) (n = 9) or eYFP controls (n = 3), aligned to food pellet presentation following overnight fast. Mean ± SEM.
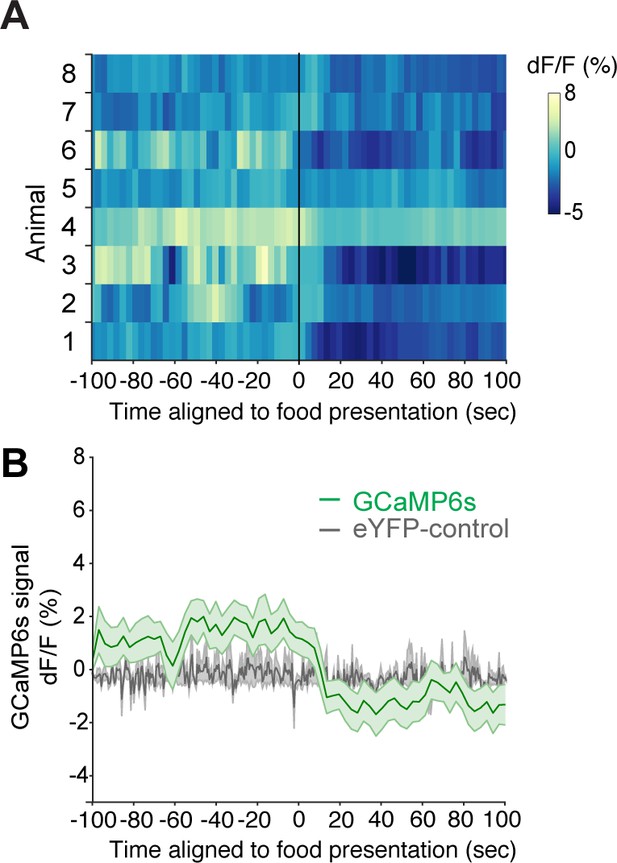
Agouti-related peptide (AgRP) neuron GCaMP6s activity is reduced by food presentation in ad lib fed mice following thermal challenge.
(A) Heat map showing GCaMP6s activity (dF/F (%)) in AgRP neurons from individual mice and (B) averaged GCaMP6s activity (dF/F (%)) (n = 9) or eYFP controls (n = 3), aligned to food pellet presentation at 22°C following thermal challenge. Mean ± SEM.
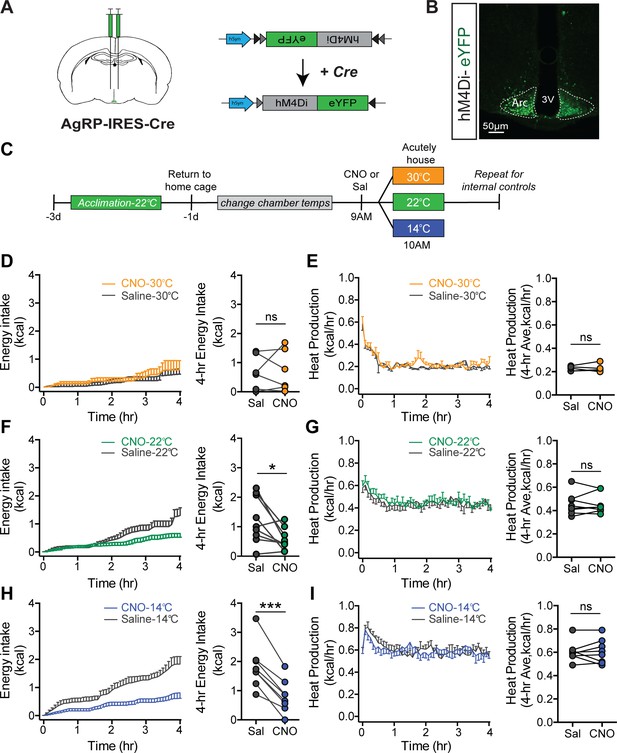
Cold-induced hyperphagia, but not thermogenesis, requires activation of agouti-related peptide (AgRP) neurons.
(A) Schematic depicting strategy for bilateral microinjection of the Cre-dependent inhibitory DREADD (hM4Di) virus into the arcuate nucleus (ARC) of AgRP-IRES-Cre mice. (B) Detection of bilateral hM4Di-eYFP in transduced AgRP neurons in the ARC. (C) Adult male hM4Di-eYFP AgRP-IRES-Cre mice were acclimated to temperature-controlled chambers set to 22°C. Animals were returned to their home cages and chamber temperatures were adjusted overnight. In the morning, animals were dosed i.p. with either CNO or saline 1 hr prior to being acutely housed at either mild cold (14°C), room temperature (22°C), or thermoneutrality (30°C) in a randomized, crossover manner. (D, F, and H) 4 hr time-series and mean values for energy intake, and (E, G, and I) 4 hr time-series and mean values of heat production in hM4Di-eYFP AgRP-IRES-Cre mice that were housed at either 30°C, 22°C, or 14°C after receiving an i.p. injection of either saline or CNO n = 6–8 per group, mean ± SEM. RM-ANOVA and Student’s t-test, ***p<0.001, *p<0.05 vs. saline.
-
Figure 5—source data 1
Inhibition of agouti-related peptide neurons.
- https://cdn.elifesciences.org/articles/58764/elife-58764-fig5-data1-v2.xlsx
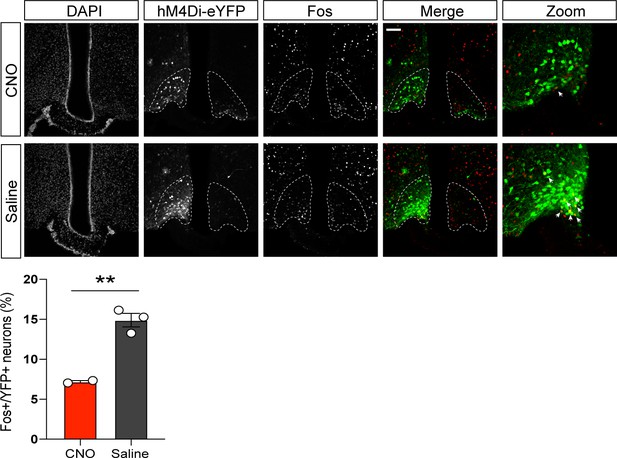
Inhibition of cold-induced Fos in hM4Di-expressing agouti-related peptide (AgRP) neuron.
AgRP-IRES-Cre animals received unilateral microinjections of an AAV containing a Cre-dependent construct for the inhibitory DREADD, hM4Di-eYFP. Animals were then exposed to 14°C for 90 min following an injection of either CNO or saline. n = 3–5 per group. Mean ± SEM, Student’s unpaired t-test, **p<0.01.
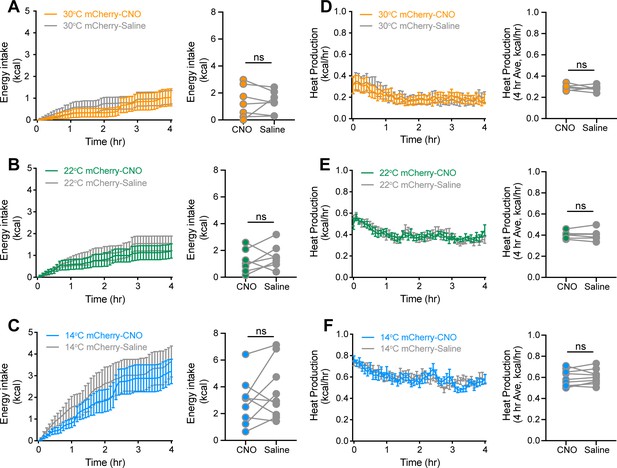
CNO controls associated with inhibition of agouti-related peptide (AgRP) neurons during acute exposure to three ambient temperatures.
Adult male AgRP-IRES-Cre bilaterally injected with a Cre-dependent control fluorophore (mCherry) were acclimated to temperature-controlled chambers set to 22°C. Animals were returned to their home cages and chamber temperatures were adjusted overnight. In the morning, animals were dosed i.p. with either CNO or saline 1 hr prior to being acutely housed at either mild cold (14°C), room temperature (22°C), or thermoneutrality (30°C) in a randomized, crossover manner. (A–C) 4 hr time-series and mean values for energy intake, and (D–F) 4 hr time-series and mean values of heat production in acutely housed animals at either 30°C, 22°C, or 14°C after receiving an i.p. injection of either saline or CNO, n = 6–8 per group, mean ± SEM. RM-ANOVA and paired Student’s t-test.
-
Figure 5—figure supplement 2—source data 1
mCherry control food intake and heat production.
- https://cdn.elifesciences.org/articles/58764/elife-58764-fig5-figsupp2-data1-v2.xlsx
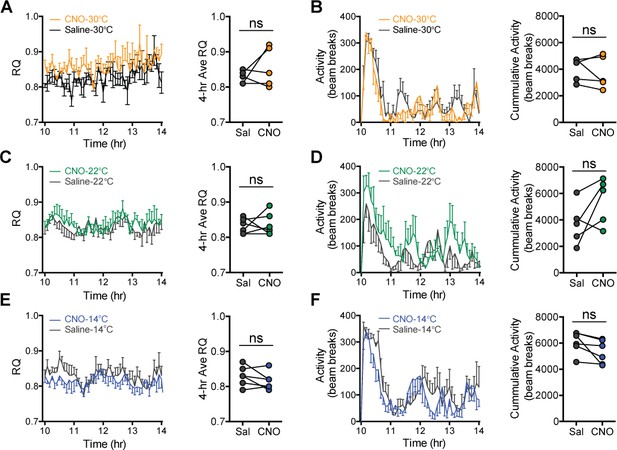
Respiratory quotient (RQ) and ambulatory activity after agouti-related peptide (AgRP) neuron inhibition at three ambient temperatures.
(A, C, and E) 4 hr time-series and mean value for RQ, and (B, D, and F) 4 hr time-series and mean value of cumulative activity in hM4Di-eYFP AgRP-IRES-Cre mice that were housed at either 30°C, 22°C, or 14°C after receiving an i.p. injection of either saline or CNO in a randomized, crossover manner, n = 6–8 per group, mean ± SEM, paired Student’s t-test.
-
Figure 5—figure supplement 3—source data 1
Respiratory quotient and activity.
- https://cdn.elifesciences.org/articles/58764/elife-58764-fig5-figsupp3-data1-v2.xlsx
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent (Mus musculus, males) | C57/Bl6/J | Jackson Labs | RRID:IMSR_JAX:000664 | |
| Genetic reagent (Mus musculus, males) | B6; Agrptm1(cre)Lowl/J | Jackson Labs | RRID:IMSR_JAX:012899 | Gift from Steamson Chua, Jr |
| Genetic reagent (Mus musculus, males) | Agrp-Cre:GFP | Laboratory of Dr. Richard Palmiter | N/A | |
| Antibody | Anti-Fos (rabbit polyclonal) | Millipore | RRID:AB_2106755 | IF (1:10,000) |
| Antibody | Anti-GFP (Chicken polyclonal) | Abcam ab13970 | RRID:AB_300798 | IF (1:10,000) |
| Recombinant DNA reagent | AAV1-CAG-DIO-hM4Di-YFP-WPRE-bGHpA | A gift from Dr. Larry Zweifel (Sanford et al., 2017) | NA | |
| Recombinant DNA reagent | AAVDJ-EF1a-DIO-GCaMP6s-WPRE | UNC Viral Core | NA | |
| Recombinant DNA reagent | AAV-Ef1a-DIO-eYFP-WPRE-pA | UNC Viral Core | NA | |
| Recombinant DNA reagent | pAAV-hSyn-DIO-mCherry | Addgene | RRID:Addgene_50459 | |
| Sequence-based reagent | Agrp_Forward primer | Agrp_F | PCR primers | For: 5’-ATGCTGACTCG AATGTTGCTG-3’, |
| Sequence-based reagent | Agrp_Reverse primer | Agrp_R | PCR primers | Rev: 5’-CAGACTTAGACC TGGGAACTCT-3’ |
| Sequence-based reagent | Pomc_Forward primer | Pomc_F | PCR primers | For: 5’-CAGTGCCA GGACCTCAC-3’ |
| Sequence-based reagent | Pomc_Reverse primer | Pomc_R | PCR primers | Rev: 5’-CAGCGAGAGG TCGAGTTTG-3’ |
| Sequence-based reagent | Npy_Forward primer | Npy_F | PCR primers | For: 5’-CTCCGCTCTG CGACACTAC-3’ |
| Sequence-based reagent | Npy_Reverse primer | Npy_R | PCR primers | Rev: 5’-AGGGTCTTCAA GCCTTGTTCT-3’ |
| Sequence-based reagent | Pmch_Forward primer | Pmch_F | PCR primers | For: 5’-GAATTTGGAAGA TGACATAGTAT-3’ |
| Sequence-based reagent | Pmch_Reverse primer | Pmch_R | PCR primers | Rev: 5’-CCTGAGCATGTC AAAATCTCTCC-3’ |
| Sequence-based reagent | 18S Ribosomal protein_Forward primer | 18S_F | PCR primers | 5’-CGGACAGGATT GACAGATTG-3’ |
| Sequence-based reagent | 18S Ribosomal protein_Reverse primer | 18S_R | PCR primers | Rev: 5’-CAAATCGCTCCA CCAACTAA-3’ |
| Chemical compound, drug | Clozapine-N-oxide (CNO) | Sigma Aldrich, Inc | Cat #: C0832 | |
| Software, algorithm | Prism 7 and 8 | GraphPad | RRID:SCR_002798 | |
| Software, algorithm | R Software | R Project for Statistical Computing | RRID:SCR_001905 | |
| Software, algorithm | Matlab | Mathworks | RRID:SCR_001622 | Code provided on GitHub as: Cold_Hyperphagia_Requires_AGRP |
| Other | DAPI stain | Invitrogen | Cat #: D1306 | |
| Other | Fluoromount-G | Thermo Fisher Scientific | Cat #: 004958–02 |





