Improving emotional-action control by targeting long-range phase-amplitude neuronal coupling
Figures
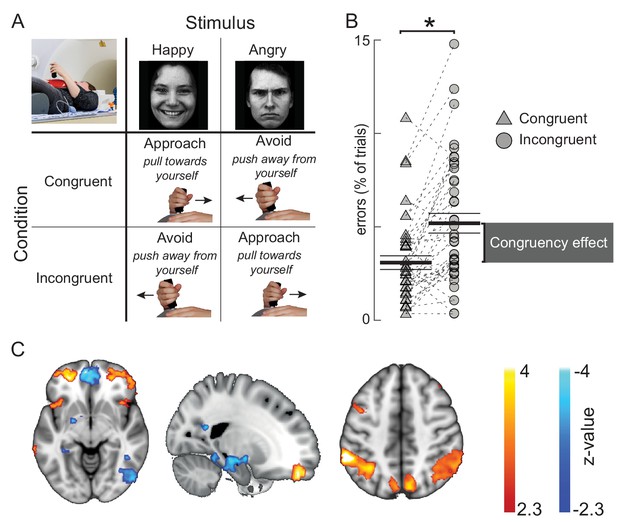
Behavioral and cerebral effects of the approach-avoidance task used to manipulate control over emotional action tendencies.
(A) Conceptual visualization of the approach-avoidance task. Participants pushed- or pulled a joystick away- or toward themselves to approach- or avoid happy and angry faces. Approaching angry- and avoiding happy faces is incongruent with action tendencies to approach appetitive and avoid aversive situations. (B) Behavioral results in the sham condition of the task. Participants make more errors in the incongruent trials (circles) than in the congruent trials (triangles). Black lines visualize the mean and standard error of the mean. Gray bar depicts the group average congruency effect. (C) Approach-avoidance congruency-related BOLD changes (p<0.01 cluster-level inferences corrected for multiple comparisons). Trials involving responses incongruent with automatic action tendencies showed stronger BOLD signal in anterior prefrontal- and parietal areas, and reduced signal in the left amygdala/hippocampus and medial prefrontal cortex.
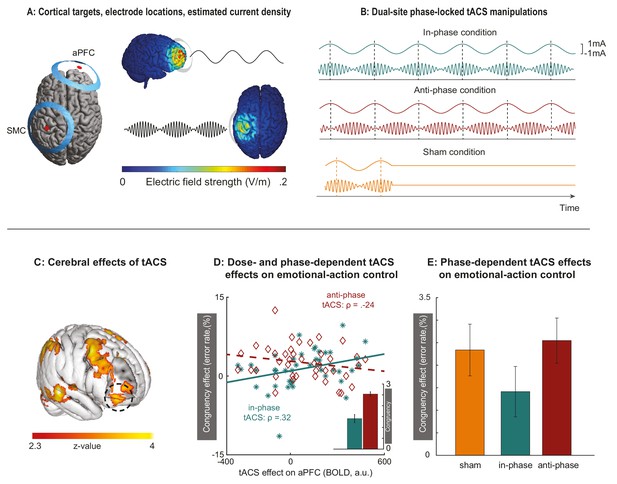
Behavioral effects of dual-site phase-coupled tACS on emotional-action control are dose- and phase-dependent.
(A)Two sets of ring electrodes were placed over the right aPFC and left SMC. Modeling of the current density showed that stimulation reached both regions of interest with intensities known to support phase entrainment when matched to the endogenous rhythms (Reato et al., 2010). (B) During the experiment, stimulation conditions were alternated between in-phase, anti-phase, and sham conditions in pseudo-random fashion. The 75 Hz stimulation over SMC was amplitude-modulated according to the 6 Hz stimulation over aPFC, either in-phase or anti-phase with the peaks of the 6 Hz aPFC stimulation. Sham consisted of an initial stimulation of 10 s, after which stimulation was terminated. (C) Concurrent tACS-fMRI quantified changes in BOLD signal evoked by the tACS intervention, across in-phase and anti-phase conditions, and independently from task performance. Online physiological effects of tACS are evident both under the aPFC electrode (black circle) and in other cortical regions. (D) Participants with stronger inhibitory responses to theta-band stimulation over aPFC (manifested as decreased BOLD signal; Scheeringa et al., 2011) improved their control over emotional actions (decreased congruency effect) during aPFC-SMC in-phase tACS (in green) but not during aPFC-SMC anti-phase tACS (in red), reflected in the interaction between emotional control (congruent, incongruent), stimulations phase (in-phase, anti-phase) and tACS-dose (BOLD signal in aPFC during stimulation versus sham [panel C]); F(1,39) = 9.3, p=0.004, partial eta2 = 0.19. Inset bar graphs illustrate group average parameter estimates of the congruency effects corrected for tACS-dose. The direction of the effect in the anti-phase condition tentatively suggests that stronger entrainment to the theta-band stimulation (decreased BOLD) increases congruency effects, as would be expected when aPFC-SMC communication is disrupted. (E) Without controlling for inter-participant variability in tACS-dose, the differential phase effect on emotional-actions control is less statistically reliable (p=0.06, partial eta2 = 0.088).

Behavioral and neural effects in different task conditions.
(A) Average congruency-effects in percentage correct over all three conditions. Differences between in-phase and anti-phase were trend-significant (p=0.06) when not controlling for dose dependence. (B) Average reaction time congruency effects, visual inspection shows similar patterns as the error-rates but there are no statistical differences between conditions. Reaction time effects were not anticipated based on earlier brain-stimulation studies using this task (Volman et al., 2011a). (C) Increased activity in posterior parietal and premotor areas for incongruent versus congruent and (D) stimulation versus sham. (E) Contrasting in-phase with the anti-phase condition shows increased activity in the left aPFC and PPC, contralateral to the prefrontal stimulation site. Speculatively, these effects might suggest network compensatory effects when right aPFC is desynchronized from the control network.
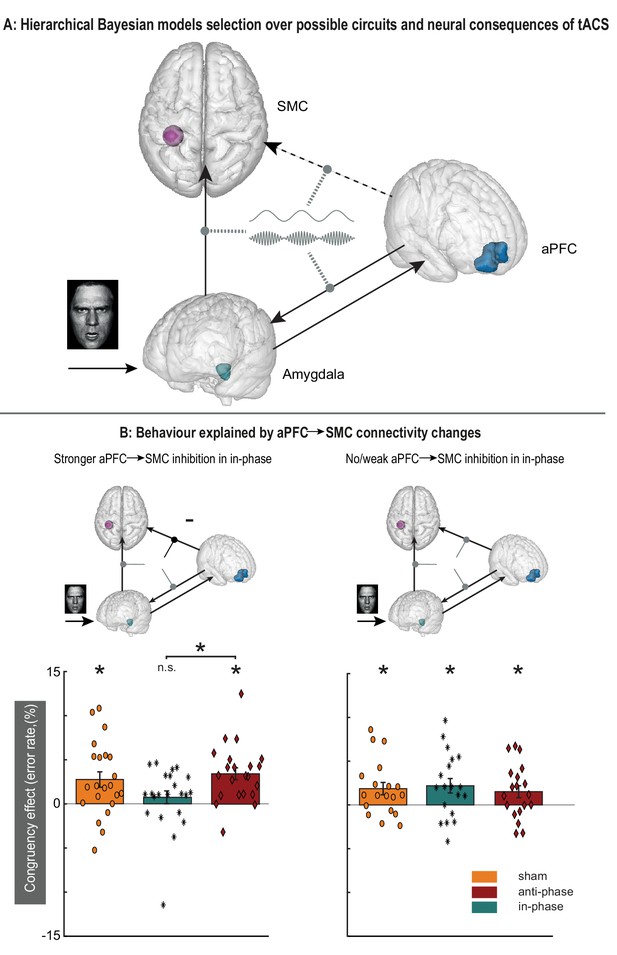
Modulatory effects of dual-site phase-coupled tACS on emotional-action control depend on effective connectivity between aPFC and SMC.
(A) Model selection compared models with and without a direct connection between aPFC→SMC (dashed arrow), and tACS modulations on different connections (gray dashed oval arrows). (B) Model selection indicates that tACS affects multiple connections in the network (top panels; supplementary materials), but only tACS-related changes in connectivity between aPFC→SMC predict behavioral effects of the stimulation (interaction between emotional control [congruent, incongruent], stimulations phase [in-phase, anti-phase] and p=0.019, np2 = 0.13; lower panels). Those participants with stronger inhibitory influence of aPFC over SMC in the in-phase condition (lower left panel, n = 22) showed decreases in congruency effects in the in-phase condition but not in the anti-phase and sham condition. The rest of the participants did not show a differential effect between stimulation conditions. Asterisks: p<0.01.
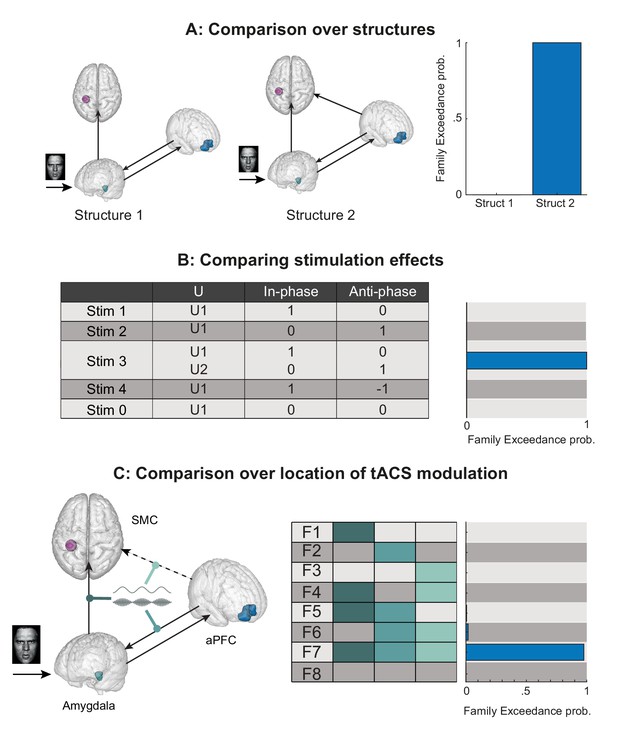
Comparing models of tACS modulation.
(A) Comparison over structure families. Structure one does not contain a direct connection between aPFC and SMC, tACS modulation of aPFC→ SMC connectivity is therefore forced through the amygdala. Structure two does contain a direct connection. Model comparison shows that structure two is more likely, suggesting that not all modulation of aPFC→ SMC is gated by the amygdala. (B) Comparison over stimulation effects. There were five ways in which the tACS manipulation could modulate connectivity in our models. ‘Stim 1’ and 'Stim 2' model effects of either in-phase or anti-phase effects, respectively. ‘Stim 3’ models in- and anti-phase effects to modulate connectivity independently of one another. ‘Stim 4’ models in-phase and anti-phase as being opposite in sign but similar in amplitude. ‘Stim 5’ models no effect of stimulation in either condition. The data is best described by Stim 3, showing independent effects of both stimulation conditions on connectivity. (C) Comparison over stimulation locations. tACS was allowed to modulate three different connections; (i) connection from aPFC → amygdala, (ii) connection from amygdala → SMC and (iii) connection from aPFC → SMC. We also included models having two, or all connections modulated by tACS. Model comparison shows that all three connections are influenced by tACS, suggesting network wide effects of stimulation. Only parameters extracted from aPFC→SMC connections predicted behavioral effects.
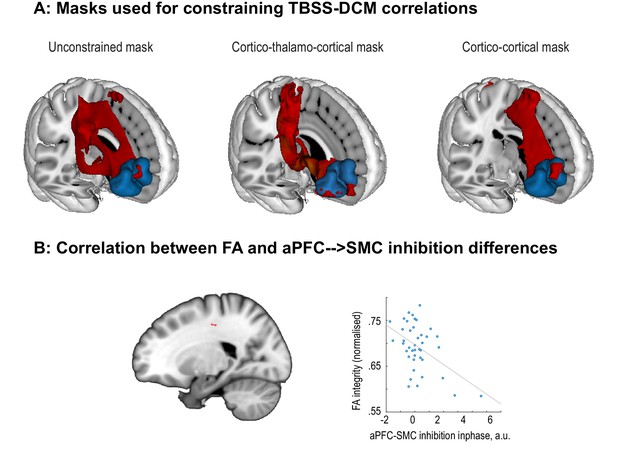
Exploratory correlations between tACS modulation and FA values.
(A) Masks (in red) that are used as region of interest for correlating aPFC→SMC connection estimates from DCM with FA values. The first mask was created by unconstrained probabilistic tractography between FPl (aPFC, in blue) and BA6. The second mask was constrained to go through the thalamus. The third mask was created by constraining tractography to cortico-cortical connections. (B) Voxels that survived TFCE correction for multiple comparisons when correlating FA values with DCM parameter estimates. Left panel shows surviving voxels when correcting for multiple comparison over voxels connecting FPl (aPFC) and BA6 via the thalamus (middle mask in A). Right panel shows the correlation (r(39) = −0.48; Rho (39)=−0.28) between FA integrity and contrast between DCM aPFC→ SMC parameters between in- and anti-phase. Participants with higher FA values seemed to show more inhibition in the in-phase relative to anti-phase condition. However, the correlation seems to be driven by only a small group of participants and warrants replication.





