Lack of airway submucosal glands impairs respiratory host defenses
Figures
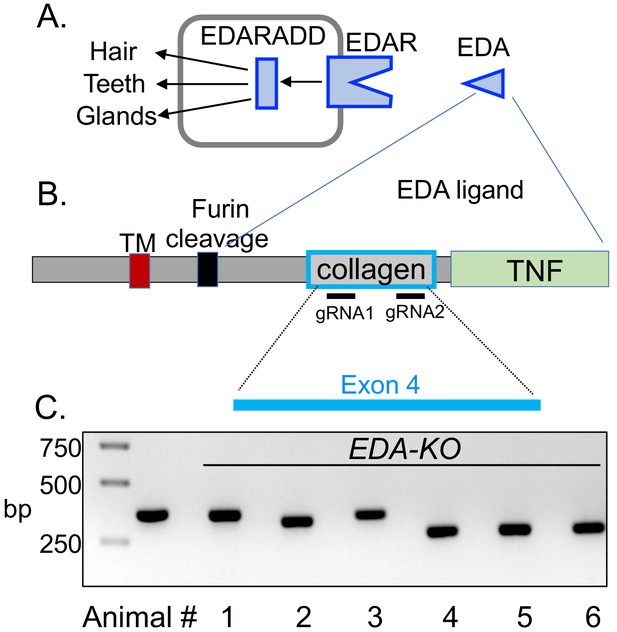
CRISPR/Cas9 editing produced pigs with a disrupted EDA gene (EDA-KO).
(A) Diagram of interaction of EDA, EDAR, and EDARADD at the epithelial placode. (B) Schematic of EDA protein domains, including the transmembrane domain (TM), furin cleavage site, collagen domain, and TNF domain. Exon 4 of the EDA gene encodes the collagen domain. Relative positions of guide RNAs are shown. (C) PCR fragments from six edited pigs from one litter. Lane two shows position of predicted wild-type product. Pigs 1–5 were male; pig six was female.

Nucleotide sequence of exon 4 of EDA gene and sequences of edited pigs.
Top shows wild-type DNA sequence with predicted amino acid sequence directly above. The glycines in the GXY motif are in red. Below that are DNA sequences of the pigs shown in Figure 1C. Deletions are shown as dashes. Pigs 1–5 were male, and pig 6 was female. Pigs 4 and 6 had two alleles.
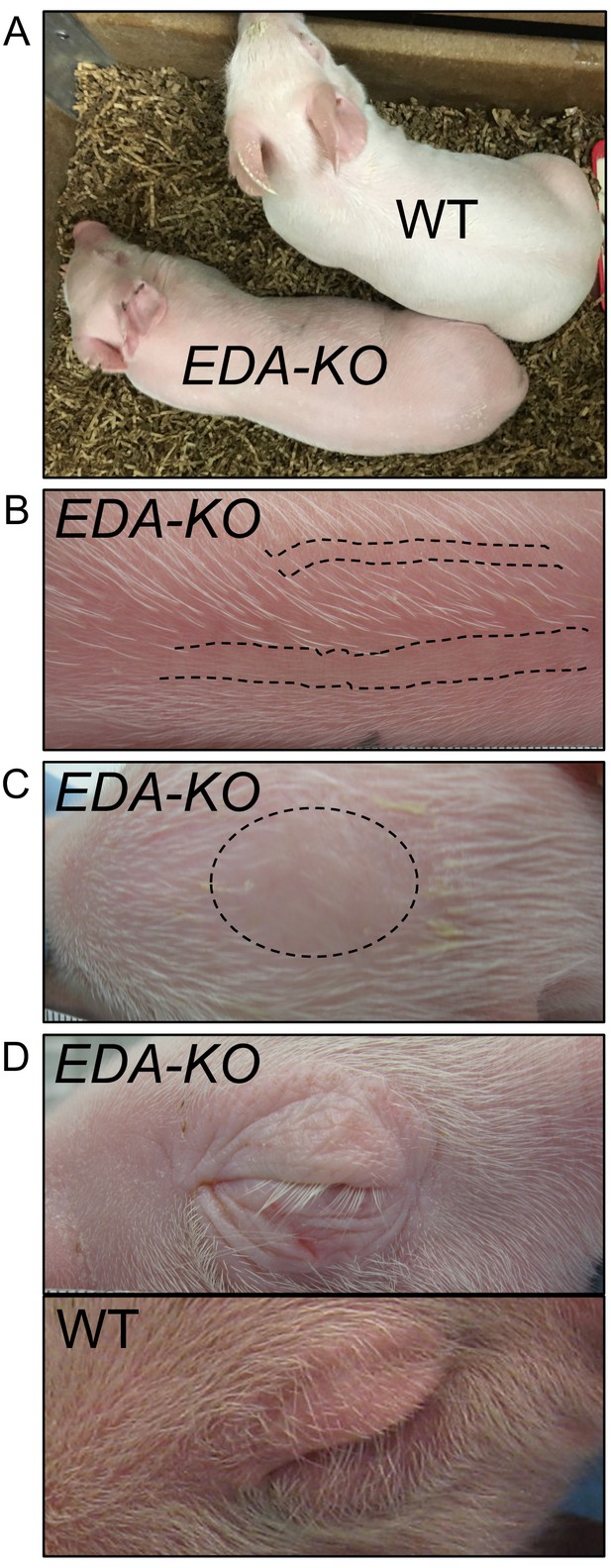
EDA-KO piglets had an appearance consistent with hypohidrotic ectodermal dysplasia.
(A) Physical appearance of wild-type (WT) and EDA-KO piglets. (B) Image of skin on the back of an EDA-KO pig showing alternating bands of hair and bare skin (dotted lines). (C) Image of skin from the top of head of EDA-KO showing bald spot. (D) Eyelids in EDA-KO pig lacked hair, although eyelashes were intact. Eyelids of a wild-type pig are shown for comparison.
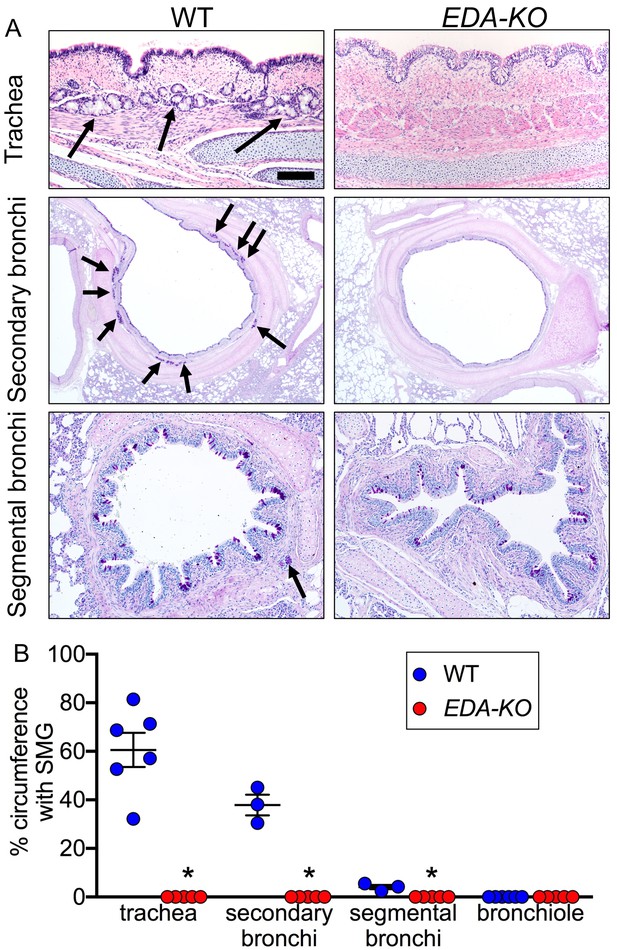
EDA-KO pigs lacked SMGs.
(A) Sections of wild-type and EDA-KO conducting airways, HE (top) and dPAS (middle and bottom) stains. Arrows point to SMGs. Scale bar = 125 mm (top and bottom panels) and 625 mm (middle panels). (B) Data are percentage of airway circumference containing SMG in indicated parts of the airway. Each data point is from a different pig. Trachea: n = 6 wild-type and 5 EDA-KO, *p=0.004. Secondary bronchi: n = 3 wild-type and 5 EDA-KO, p=0.018. Segmental bronchi: n = 3 wild-type and 5 EDA-KO, p=0.018. Bronchioles: n = 6 wild-type and 5 EDA-KO, p=1.00. Statistical analysis was by Mann-Whitney test.
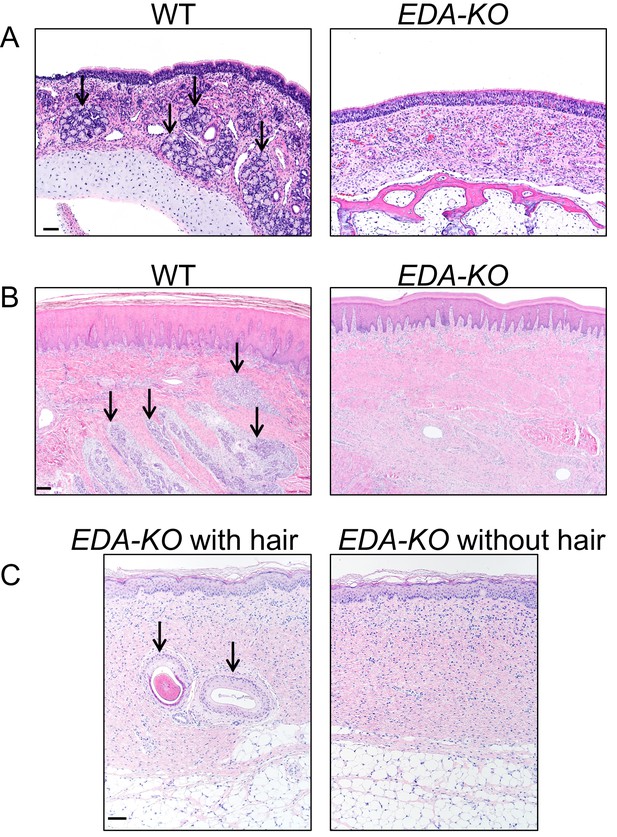
Histopathology of nasal mucosa, nasal planum, and skin of wild-type and EDA-KO piglets.
(A) Wild-type nasal mucosa had glands (arrows); these were absent in EDA-KO pigs. (B) Wild-type nasal planum had eccrine glands (arrows);these were absent in EDA-KO pigs. (C) EDA-KO pigs had sections of haired skin that had hair follicles (left, arrows); hairless regions of skin lacked hair follicles. HE stains. Scale bar = 60 µm (A), 150 µm (B), and 80 µm (C).
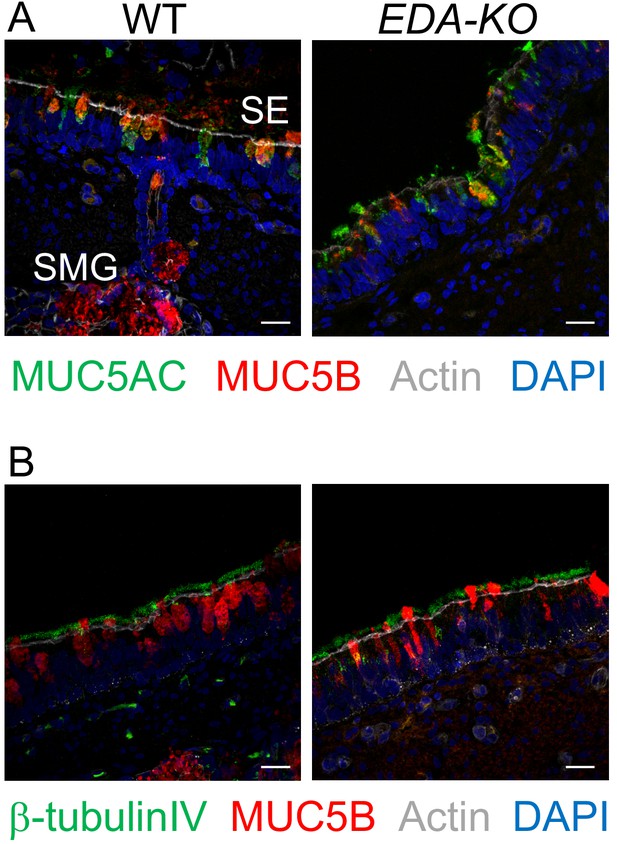
MUC5AC, MUC5B, and β-tubulin IV immunolocalization is similar in airway surface epithelia of wild-type and EDA-KO piglets.
Images are confocal immunofluorescence of wild-type and EDA-KO trachea. In all panels, actin is labeled with phalloidin in grey and nuclei are labeled with DAPI in blue. Scale bar = 20 µm. (A) Images show goblet cells expressing MUC5AC (green) and MUC5B (red). A portion of a SMG is indicated; SMGs were detected in wild-type only. (B) Images indicate ciliated cells (β-tubulin IV, green) and MUC5B (red).

ASL of EDA-KO pigs has impaired killing of S.aureus in vivo.
(A) Schematic showing S. aureus attached to gold grids by biotin-streptavidin linkages. The grids were placed on ASL of trachea for 1 min in vivo. Then the bacteria were counted and the percentage that were dead was determined. (B) Example of live (green)/dead (red) staining of bacteria after the grid was removed from the airway. (C) Percentage of dead bacteria. *p<0.0012 by Mann-Whitney test.
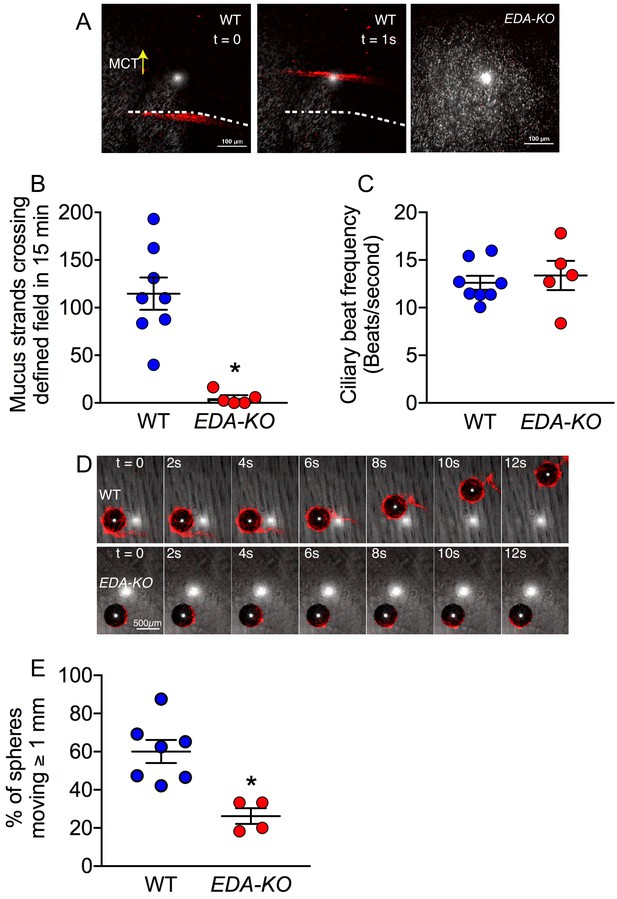
Loss of SMGs eliminates mucus strands and impairs MCT ex vivo.
(A) Data are confocal images of tracheal surface of wild-type and EDA-KO pigs. Mucus was labeled with fluorescent nanospheres (red). Left and middle panel show movement of mucus strand with time. Strands were not observed in EDA-KO airway in right panel. The white spots in the middle of the field are reflected light. (B) Number of mucus strands crossing the microscopy field in 15 min. N = 8 wild-type and 5 EDA-KO pigs. *p=0.0016. (C) Ciliary beat frequency on trachea under methacholine stimulation. N = 8 wild-type and 5 EDA-KO pigs. p=0.4351. (D) Metallic spheres were dropped onto the airway surface and movement was tracked with time. Mucus was labeled with fluorescent nanospheres (red). Images from wild-type airway show a mucus strand attached to a sphere and pulling it across the field. Images from EDA-KO airway show mucus attached to sphere that was rolling in place. (E) Fraction of metallic spheres that moved at least 1 mm during a 15-min observation period. N = 7 wild-type and 4 EDA-KO pigs. * indicates p=0.0061. For panels B, C, and E, each dot represents a different pig. Statistical significance was evaluated with a Mann-Whitney test.
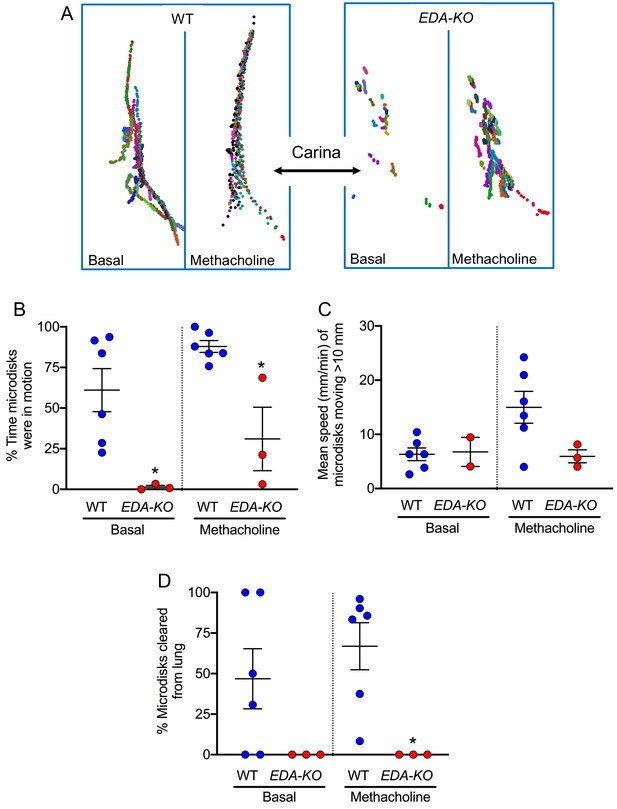
EDA-KO pigs have impaired MCT in vivo.
MCT was assessed by insufflating tantalum microdisks in sedated, spontaneously breathing pigs followed by acquisition of high-resolution CT scans every 9 s for 6.3 min (total 44 scans). Positions of individual microdisks were tracked. Pigs were studied under basal conditions and after stimulating submucosal gland secretion with intravenous methacholine. N = 6 wild-type and 3 EDA-KO pigs. Statistical significance between data from wild-type and EDA-KO was evaluated with a Mann-Whitney test. (A) Examples of individual microdisks (different colors) tracked in wild-type (left) and EDA-KO (right) pigs. Position of carina is indicated. (B) Percentage of time microdisks were moving. * indicates p=0.0238 under basal conditions and p=0.0238 under methacholine-stimulated conditions. (C) Mean speed (mm/min) of microdisks that moved more than 10 mm. One EDA-KO pig had no microdisks moving >10 mm under basal conditions; therefore, only two data points and the range are shown in that case. * indicates p=0.8571 under basal conditions and p=0.1667 under methacholine-stimulated conditions. (D) Percentage of microdisks that reached the larynx during the study. p=0.1667 under basal conditions. * indicates p=0.0238 under methacholine-stimulated conditions.
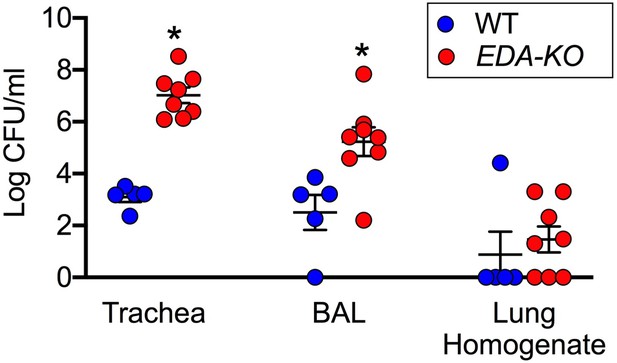
EDA-KO pigs have decreased eradication of Staphylococcus aureus from the lung.
S. aureus were aerosolized into the airways and 4 hr later samples were obtained by tracheal washes, bronchoalveolar lavage (BAL), and lung homogenates. Data are the log colony-forming units recovered. N = 5 wild-type and 8 EDA-KO pigs. * indicates p=0.0016 for trachea washes, * indicates p=0.0186 for BAL, and p=0.3908 for distal lung homogenates. Statistical analysis was with a Mann-Whitney test.
Videos
Mucus strands moving on wild-type trachea stimulated with methacholine.
Mucus strands were labeled with fluorescent nanospheres (red). Video is real time. Scale bar 100 μm. White dot in center is from reflected light.
Surface of EDA-KO trachea imaged as in Video 1.
A 500 µm metallic sphere was placed on a wild-type pig trachea.
Mucus strands were labeled with fluorescent nanospheres (red). Mucus strand attaches to and initiates movement of the sphere, pulling it to the edge of the airway segment. Duration of video clip was 30 s and is compressed to 6 s here. Scale bar 500 μm.
Imaging as in Video 3 on EDA-KO pig trachea.
A sphere is shown spinning backwards and failing to move over the surface of the airway. Mucus attaches to the sphere’s surface allowing ready detection of sphere rotation. Duration of video clip was 900 s and is compressed to 32 s.
Transport of microdisks in wild-type pig airways under basal conditions.
The head is above the top of the image and tail below the bottom. Microdisks were insufflated into the airways and tracked by sequential CT scanning. Each microdisk is represented by a different colored circle; circles are ~280 times the area of microdisks to aid visualization. When microdisks reach the larynx, they disappear. Video is compressed from original duration of 6.3 min.
Transport of microdisks in EDA-KO pig airways under basal conditions.
Procedures are as described in legend of Video 5.
Transport of microdisks in wild-type pig treated with IV methacholine.
Transport of microdisks in EDA-KO pig treated with IV methacholine.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Antibody | Anti- MUC5B (rabbit polyclonal) | Santa Cruz | Cat# Sc-20119 | IF (1:2000) |
| Antibody | Anti-MUC5AC (mouse monoclonal) | Novus Biologicals | Cat # NBP2-15196 | IF (1:5000) |
| Antibody | anti-β-tubulinIV (mouse monoclonal) | Biogenex | Cat# Mu178-5UC | IF(1:300) |
| Chemical compound, drug | Methacholine (acetyl-β-methylcholine chloride) | Sigma | Cat# A2251 | |
| Chemical compound, drug | CleanCap Cas9 mRNA | TriLink Biotechnologies | Cat # L-7606 | 20 ng/ul |
| Commercial assay or kit | MEGAshort script T7 Transcription kit | Thermo-fisher | Cat# AM1354 | |
| Commercial assay or kit | KAPA Express Extract Kit Plus amplification module | KAPA Biosystems | Cat# KK7152 | |
| Commercial assay or kit | MEGAclear Transcription Clean-up kit | Thermo-fisher | Cat# AM1908 | |
| Commercial assay or kit | QIAquick PCR Purification Kit | Qiagen | Cat# 28104 | |
| Commercial assay or kit | Live/Dead Bacterial Viability Assay | Thermofisher | Cat # L13152 | |
| Commercial assay or kit | TOPO TA Cloning kit (with PCR 2.1-TOPO vector) | Invitrogen | Cat# K45-0001 | |
| Gene (Sus scrofa) | EDA1 | Ensembl | Ensembl gene link: ENSSSCG00000021647 | |
| Other | Visualizing media | Invitrogen | 4 nm Nano spheres | 1:10000 |
| Other | Visualizing media | BalTec | Tantalum spheres | 500 μm |
| Other | Visualizing media | Sigma | Tantalum disks | 350 μm |
| Sequence-based reagent | EDA guide 1 | This paper | Guide RNA | GGAATCCCTGGAATCCCTGG |
| Sequence-based reagent | EDA guide 2 | This paper | Guide RNA | GCCCGGTGGTCCCATAACAG |
| Sequence-based reagent | Forward primer | This paper | Primer | gcctgactttgtgttgttagaagtccata |
| Sequence-based reagent | Reverse primer | This paper | Primer | ctgctcttggtatcatgtactcctgatct |
| Software, algorithm | Imaging software | Olympus | CellSens | |
| Software, algorithm | Imaging software | NIH | ImageJ | |
| Software, algorithm | Imaging software | Nikon | NIS Elements | |
| Software, algorithm | Imaging software | Siemens | SOMATON Force | |
| Software, algorithm | Data analysis | GRAPHPAD Software | GRAPHPAD PRISM | |
| Software, algorithm | Data analysis | ITK-SNAP | ITK-SNAP | |
| Software, algorithm | Primer-Blast | NCBI | https://www.ncbi.nlm.nih.gov/tools/primer-blast/index.cgi | |
| Strain, strain background | S. aureus | Pezzulo et al., 2012 PMID:22763554 | S. aureus isolate 43SA |





