A motogenic GABAergic system of mononuclear phagocytes facilitates dissemination of coccidian parasites
Figures
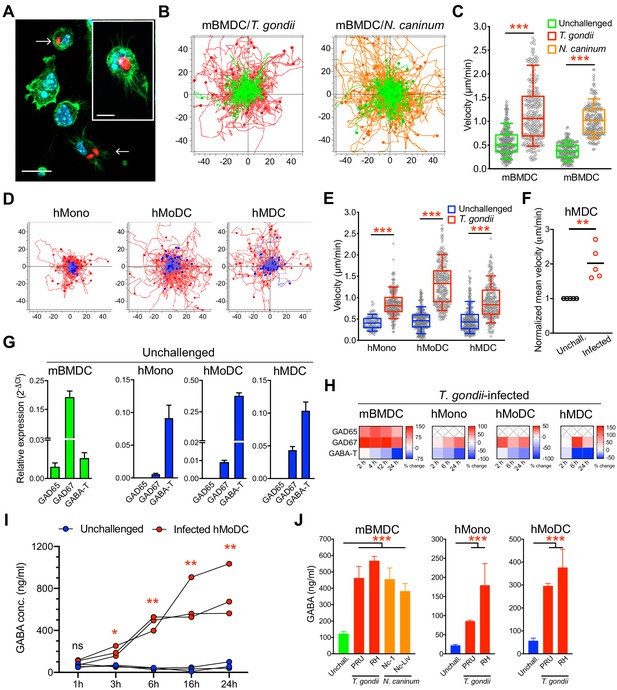
Migratory activation and GABAergic responses of mononuclear phagocytes challenged with T. gondii and N. caninum.
(A) Representative micrographs of mBMDCs infected by T. gondii tachyzoites (PRU-RFP, arrowheads) and by-stander non-infected BMDCs, stained for F-actin (Alexa Fluor 488 Phalloidin) and nuclei (DAPI). Scale bars, 20 μm, inset image 10 μm. (B) Representative motility plots of unchallenged mBMDCs (green) and T. gondii- or N. caninum-infected mBMDCs (red and orange, respectively). X- and y-axes show distances in μm. (C) Box-and-whisker dot plots show, for each condition in (B), median velocities (μm/min) of cells shown in motility plots (n = 3–6 independent experiments). (D) Motility plots of hMonocytes, hMoDCs and hMDCs, respectively, unchallenged (blue) or infected with T. gondii (PRU-RFP, red). (E) Quantifications of velocities (μm/min) in (D) are shown as box-and-whisker dot plots (n = 3–6 independent experiments). (F) Normalized mean velocities of unchallenged and T. gondii-infected hMDCs from human donors (n = 5 independent donors, 50–60 cells per donor). (G) Relative mRNA expression (2-ΔCt) of GABA synthesis enzymes (GAD65 and GAD67) and catabolic enzyme (GABA-T) in unchallenged mBMDCs, hMoDCs, hMDCs and hMonocytes, respectively, determined by real-time qPCR. (H) Corresponding heat maps for the indicated cell types show (%) transcriptional changes upon challenge with T. gondii relative to unchallenged cells at indicated time-points, as detailed in Materials and methods. (X) indicates no amplification (n = 3–6 independent experiments). (I) GABA (ng/ml) secreted in supernatants of hMoDCs challenged with T. gondii (ME49/PTG) at indicated time-points (n = 3 human donors). (J) GABA (ng/ml) secreted in supernatants of mBMDCs, hMonocytes and hMoDCs, respectively, challenged with different strains of T. gondii (PRU and RH) or N. caninum (Nc-1 and Nc-Liverpool) was quantified by ELISA (n = 6–8 independent experiments). Bar graphs show mean + SEM. Statistical significance was tested by Mann-Whitney test for (C, E, F), ordinary one-way ANOVA with Dunnett’s multiple comparison test for (J) and paired t-test for (I), *p<0.05, **p<0.01, ***p<0.001, ns p≥0.05.
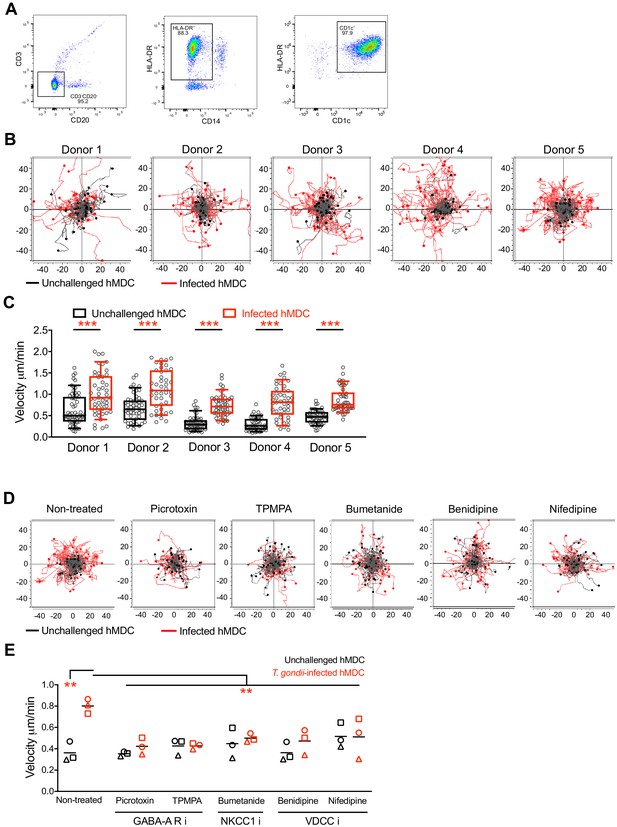
hMDCs exhibit a hypermotile phenotype upon T. gondii challenge.
(A) Representative bivariate dot plots show characterization of hMDC preparations using flow cytometry. Cells were checked for contaminating T cells (CD3), B cells (CD20) and monocytes (CD14) and confirmed to be HLA-DR+ and CD1c+. (B) Representative motility plots of unchallenged and T. gondii-infected hMDCs from five independent human donors (1-5). X- and y-axes indicate distances in μm. (C) Box-and-whisker dot plots show, for each indicated condition, velocities (μm/min) of unchallenged and T. gondii-infected hMDCs, as in (B). (D) Representative motility plots of unchallenged and T. gondii-infected hMDCs treated with picrotoxin and TPMPA (inhibitors of GABA-A Rs), or bumetanide (NKCC1 inhibitor), or benidipine and nifedipine (inhibitors of VDCCs). (E) Dot plots show, for each indicated condition, median velocities (μm/min) of unchallenged and T. gondii-infected hMDCs, as in (D). Each dot indicates median velocity of 50–60 cells tracked from one individual donor (n = 3). Statistical significance was assessed by Mann-Whitney test for (C) and by ordinary one-way ANOVA with Tukey’s multiple comparison test for (E), **p<0.01, ***p<0.001.
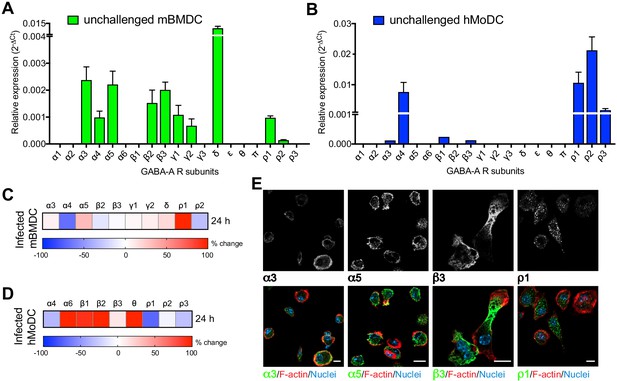
Modulated expression of GABA-A R subunits in DCs upon T. gondii infection.
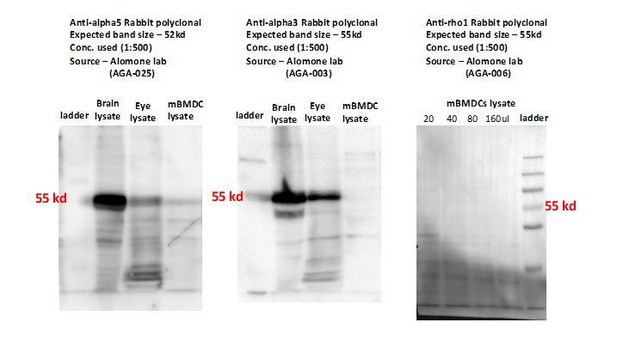
(A, B) Relative mRNA expression (Mean + SEM, 2-ΔCt) of GABA-A R subunits in unchallenged (A) mBMDCs (n = 7 independent experiments) and (B) hMoDCs (n = 3 independent experiments). (C, D) Heat maps show (%) transcriptional changes in the expression of GABA-A R subunits upon challenge of (C) mBMDCs and (D) hMoDCs with T. gondii (PRU-RFP) relative to unchallenged cells at indicated time-point (n = 3–4 independent experiments). (E) Representative micrographs of unchallenged mBMDCs stained with antibodies against GABA-A R α3, α5, β3 and ρ1 subunit (and Alexa Flour 488-conjugated secondary antibodies), respectively, and Alexa Fluor 647 Phalloidin (F-actin) and DAPI (nuclei). Scale bars, 10 μm, for β3 5 μm (n = 3 independent experiments).
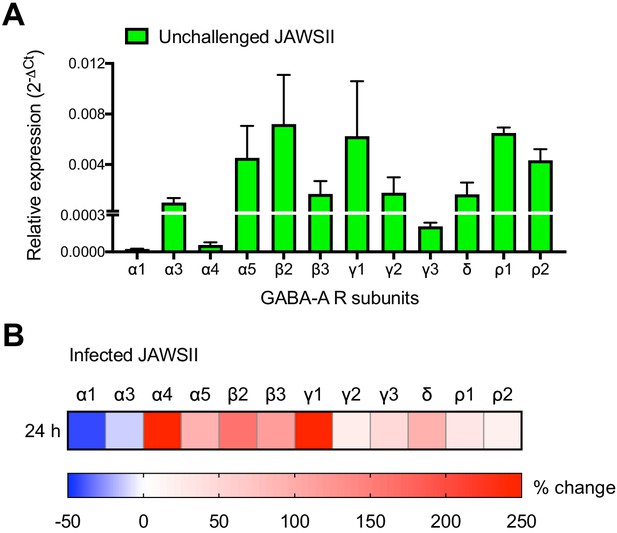
Expression of GABA-A receptor subunits in JAWSII is modulated upon T. gondii infection.
(A) Relative mRNA expression (mean + SEM, 2-ΔCt) of GABA-A receptor subunits in unchallenged JAWSII, a mouse DC line, (n = 3 independent experiments). (B) Heat map shows (%) transcriptional changes in the expression of GABA-A receptor subunits upon challenge of JAWSII with T. gondii (PRU-RFP) relative to unchallenged cells at indicated time-point (n = 3 independent experiments).
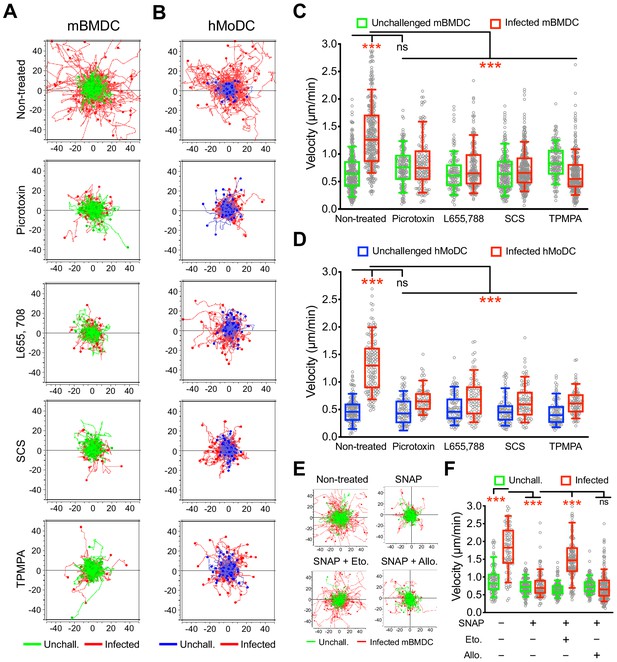
Impact of pharmacological modulation of GABA-A Rs on T. gondii-induced hypermotility of DCs.
(A, B) Representative motility plots of unchallenged mBMDCs and T. gondii (PRU-RFP)-infected (A) mBMDCs and (B) hMoDCs treated with GABA-A R inhibitor picrotoxin (open channel blocker) or subunit specific inhibitors L655,708 (α-specific), SCS (β-specific) and TPMPA (ρ-specific) at concentrations stated in Materials and methods. X- and y-axes indicate distances in μm. (C, D) Box-and-whisker dot plots show, for each condition, median velocities (μm/min) as indicated in (A) and (B) (n = 3–6 independent experiments). (E) Representative motility plots and (F) velocities of unchallenged mBMDCs and T. gondii-infected mBMDCs treated with SNAP (GAT inhibitor) in presence of etomidate and allopregnanolone (allosteric modulators of β and δ subunit-containing GABA-A Rs, respectively). (n = 3–4 independent experiments). Statistical significance was tested by ordinary one-way ANOVA with Dunnett’s multiple comparison test for (C, D, F), ***p<0.001, ns p≥0.05.
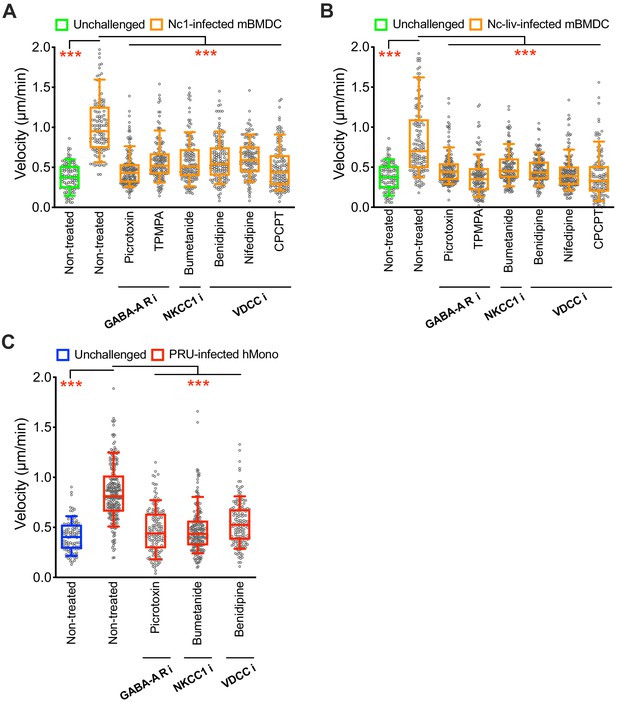
Effect of pharmacological inhibitors of GABA-A Rs, NKCC1 and VDCCs on hypermotility.
(A, B) Box-and-whisker dot plots show median velocities (μm/min) of mBMDCs challenged with N. caninum Nc-1 strain (A) and Nc-Liverpool strain (B) and treated with GABA-A R inhibitors, NKCC1 inhibitor or VDCC inhibitors as indicated, respectively (n = 3 independent experiments). (C) Box-and-whisker dot plots show median velocities (μm/min) of hMonocytes challenged with T. gondii (PRU) and treated with GABA-A R inhibitor, NKCC1 inhibitor or VDCC inhibitor, respectively (n = 3 independent experiments). Statistical significance was assessed by ANOVA with Dunnett’s multiple comparison test for (A, B, C), ***p<0.001.
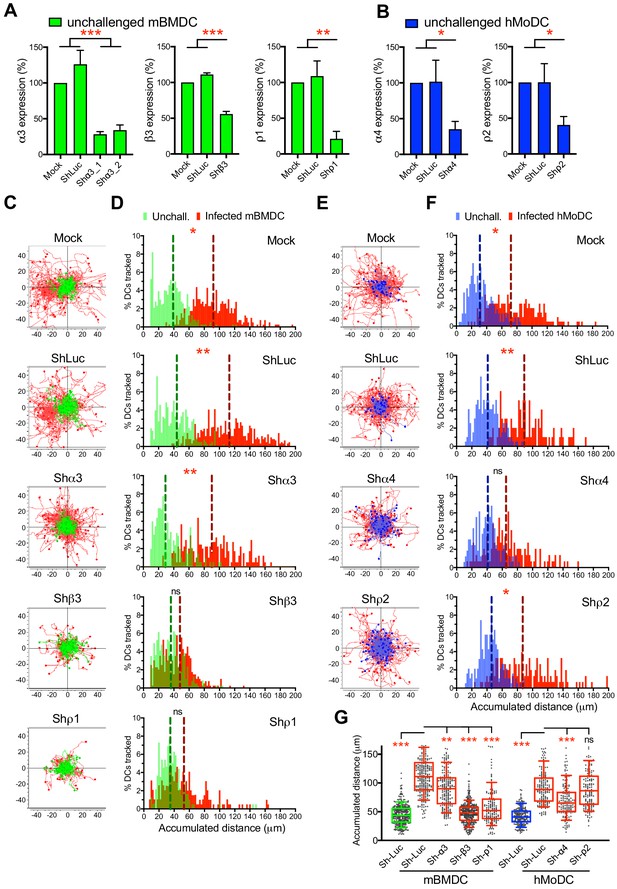
Targeted gene silencing of GABA-A R subunits impacts DC hypermotility.
(A, B) The mRNA expression in unchallenged (A) mBMDCs and (B) hMoDCs, treated with shRNA for control Luc and GABA-A R subunits, related to mock-treated cells (Mean + SEM, %). For shα3, two separate constructs were used (n = 3–5 independent experiments for mBMDCs and 6–7 for hMoDCs). (C, E) Representative motility plots of (C) mBMDCs and (E) hMoDCs, treated as in (A–B) and challenged with T. gondii (PRU-RFP). X- and y-axes indicate distances in μm. (D, F) Histograms of accumulated distances migrated (μm) by (D) mBMDCs and (F) hMoDCs as in (C and E), respectively. Dotted lines indicate median values (n = 3–4 independent experiments). (G) Box-and-whisker dot plots show, for each indicated condition, median velocities (μm/min) of unchallenged and T. gondii-infected mBMDCs and hMoDCs (n = 3–4 independent experiments). Statistical significance was tested by ordinary one-way ANOVA with Dunnett’s multiple comparison test for (A, B, G) and by Mann-Whitney test for (D, F), *p<0.05, **p<0.01, ***p<0.001, ns p≥0.05.
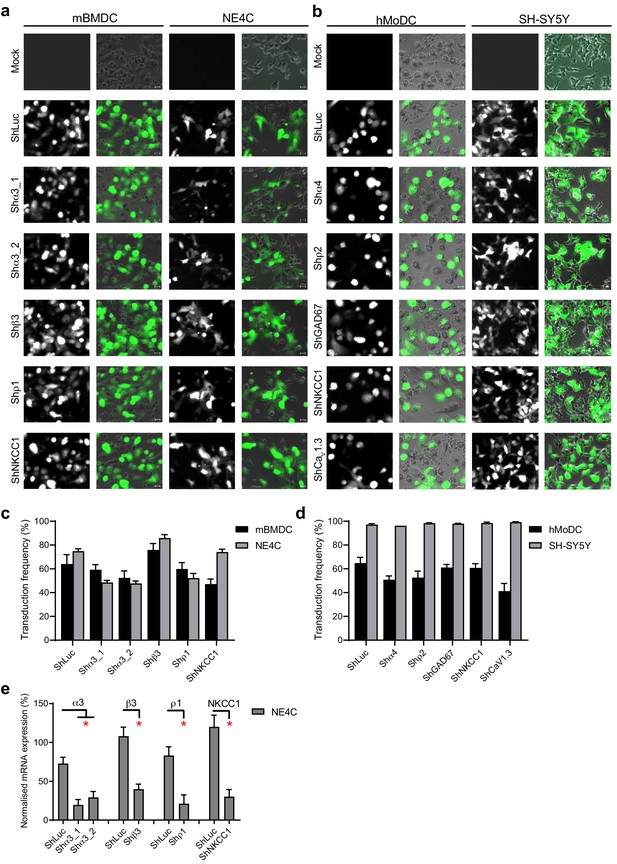
ShRNA transduction efficiency.
(A–B) Representative micrographs show (A) mBMDCs and the murine neuronal cell line NE4C, and (B) hMoDCs and the human neuronal cell line SH-SY5Y treated with control shRNA (shLuc) or shRNA to indicated target gene, respectively. (C, D) Bar graphs show mean % (+ SEM) GFP+ cells, representative of the lentiviral transduction efficiency quantified in mBMDCs and NE4Cs (C) and hMoDCs and SH-SY5Y (D). (E) Bar graph shows mean (+ SEM) mRNA knock-down (%) assessed in unchallenged transduced NE4Cs. Statistical significance was assessed by Student’s t-test, *p<0.05.
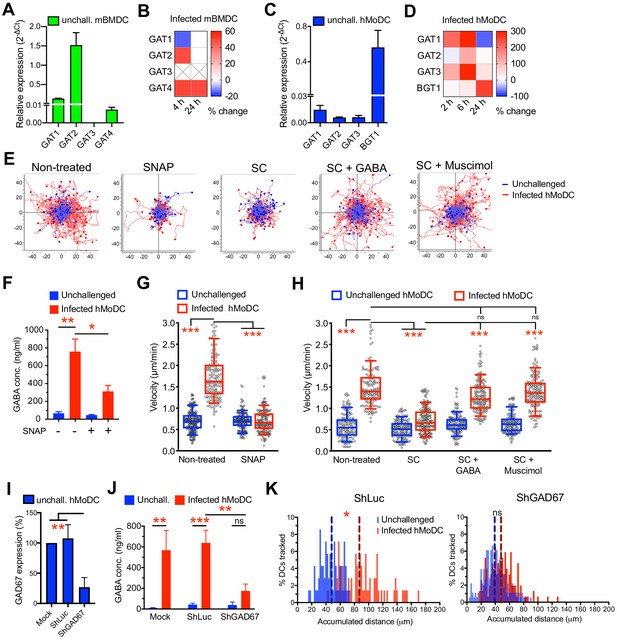
Impact of GABA synthesis enzymes and GABA transporters on hypermotility.
(A, C) Relative mRNA expression (2-ΔCt) of GABA transporters in unchallenged (A) mBMDCs and (C) hMoDCs (n = 3 independent experiments). (B, D) Heat maps show (%) transcriptional changes in the expression of GABA transporters upon challenge of (B) mBMDCs and (D) hMoDCs with T. gondii (PRU-RFP) relative to unchallenged cells at indicated time-points. (X) indicates no amplification (n = 3–4 independent experiments). (E) Representative motility plots of unchallenged and T. gondii-infected hMoDCs treated with GABA transporter inhibitor (SNAP) and GABA synthesis inhibitor (SC) in absence and presence of GABA or GABA analog (muscimol). X- and y-axes indicate distances in μm. (F) GABA (ng/ml) secreted in supernatants of hMoDCs challenged with T. gondii (ME49/PTG) in presence of SNAP was quantified by ELISA (n = 3 independent experiments). (G, H) Box-and-whisker dot plots show, for each indicated condition, velocities (μm/min) of unchallenged and T. gondii-infected hMoDCs, as in (E) (n = 3 independent experiments). (I) The mRNA expression (2-ΔCt) in control shLuc-treated and shGAD67-treated unchallenged hMoDCs related (%) to mock-treated cells (n = 5 independent experiments). (J) GABA secreted (ng/ml) in supernatants of mock-, shLuc- and shGAD67-treated hMoDCs challenged with T. gondii (PRU-RFP) (n = 5 independent experiments). (K) Histograms show accumulated distances migrated (μm) by control shLuc-treated and shGAD67-treated hMoDCs, respectively, challenged with T. gondii (PRU-RFP). Dotted lines indicate median values (n = 3–4 independent experiments). Bar graphs show mean + SEM. Statistical significance was tested by paired t-test for (F), ordinary one-way ANOVA with Dunnett’s multiple comparison test for (G, H, I, J) and Mann-Whitney test for (K), *p<0.05, **p<0.01, ***p<0.001, ns p>0.05.
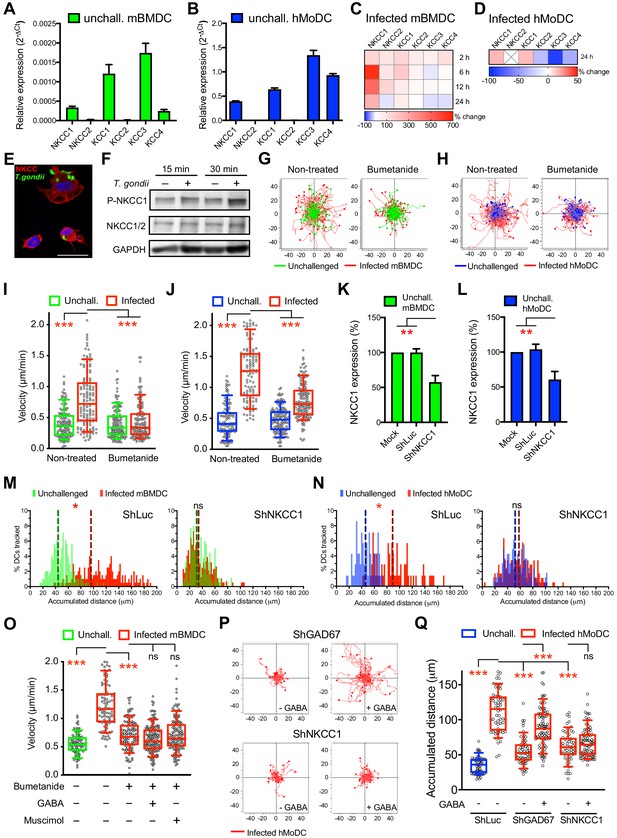
NKCC1 is a determinant of GABAergic hypermotility.
(A, B) Relative mRNA expression (2-ΔCt) of cation chloride transporters (CCCs) in unchallenged (A) mBMDCs and (B) hMoDCs (n = 3 independent experiments). (C, D) Heat map depicts (%) transcriptional expression changes of CCCs in (C) mBMDCs and (D) hMoDCs challenged with T. gondii (PRU-RFP) relative to unchallenged cells at indicated time points. (X) indicates no amplification (n = 3 independent experiments). (E) Immunostaining of mBMDCs challenged with T. gondii (ME49/PTG-GFP) stained with NKCC1/2 monoclonal antibody (Alexa Flour 594-conjugated anti-mouse secondary antibody) and DAPI (nuclei). Scale bars: 10 μm. (F) Representative Western blot of lysates from T. gondii-challenged mBMDCs for indicated time, immunoblotted with phospho-NKCC1 and total NKCC1/2 antibodies. GAPDH was used as loading reference. (n = 4 independent experiments). (G, H) Representative motility plots of unchallenged and T. gondii-infected (G) mBMDCs and (H) hMoDCs treated with NKCC1 inhibitor (bumetanide). X- and y-axes indicate distances in μm. (I, J) Box-and-whisker dot plots show, for each indicated condition, median velocities (μm/min) of unchallenged and T. gondii-infected (I) mBMDCs and (J) hMoDCs as in (G, H) (n = 3 independent experiments). (K, L) The mRNA expression (2-ΔCt) of control shLuc- and shNKCC1-treated unchallenged (K) mBMDCs and (L) hMoDCs related (%) to mock-treated cells (n = 7 independent experiments for mBMDCs and n = 4 for hMoDCs). Bar graphs show mean + SEM. (M, N) Histograms show accumulated distances migrated (μm) by control shLuc-treated and shNKCC1-treated (M) mBMDCs and (N) hMoDCs, respectively, challenged with T. gondii (PRU-RFP). Dotted lines indicate median values (n = 3 independent experiments). (O) Velocities of unchallenged and T. gondii-infected mBMDCs treated with GABA or muscimol in presence of bumetanide. (P) Representative motility plots of T. gondii-infected shGAD67- and shNKCC1-treated hMoDCs in presence of GABA. (Q) Velocities of unchallenged and T. gondii-infected shLuc-, shGAD67- and shNKCC1-treated hMoDCs with or without GABA. Statistical significance was tested by ordinary one-way ANOVA with Dunnett’s multiple comparison test for (I, J, K, L, O, Q) and by Mann-Whitney test for (M, N), *p<0.05, **p<0.01, ***p<0.001, ns p≥0.05.
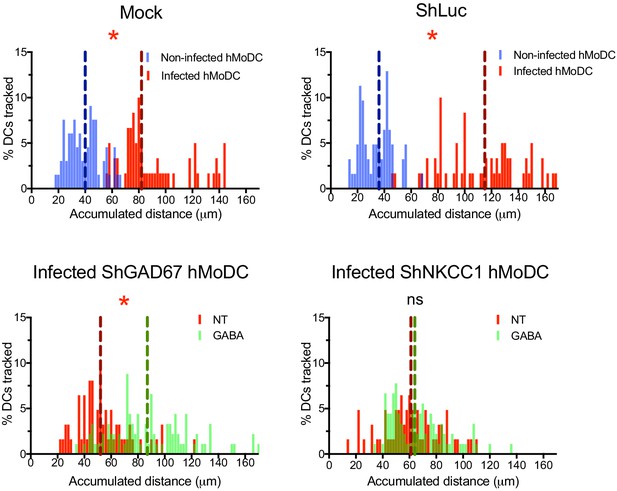
Histograms of accumulated distances migrated (μm) by mock- or shLuc-treated unchallenged and T. gondii-infected hMoDCs (upper panel), T. gondii-infected shGAD67- or shNKCC1-treated hMoDCs with or without GABA (lower panel).
Dotted lines indicate median values (n = 3 independent experiments). Statistical significance was tested by Mann-Whitney test, *p<0.05, ns p≥0.05.
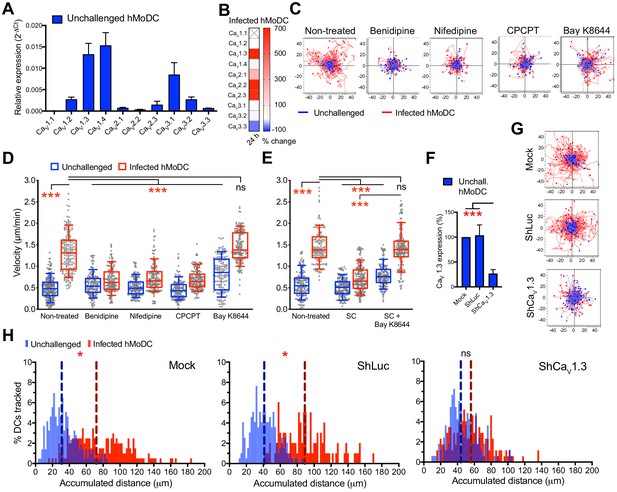
CaV1.3 is the mediator for downstream effects of activated GABA signaling.
(A) Relative mRNA expression (2-ΔCt) of voltage-dependent calcium channels (VDCCs) in unchallenged hMoDCs (n = 3 independent experiments). (B) Heat map shows (%) transcriptional changes in the expression of VDCCs subunits upon challenge of hMoDCs with T. gondii (PRU-RFP) relative to unchallenged cells at indicated time-point. (X) indicates no amplification (n = 3 independent experiments). (C) Representative motility plots of unchallenged and T. gondii-infected hMoDCs treated with benidipine (VDCC broad inhibitor), nifedipine (L-type inhibitor), CPCPT (CaV1.3 specific inhibitor) and bay K8644 (VDCC activator). X- and y-axis are distances in μm. (D) Box-and-whisker dot plots show, for each indicated condition, velocities (μm/min) of unchallenged and T. gondii-infected hMoDCs, as in (C), and (E) treated with SC (GABA synthesis inhibitor) in presence of bay K8644 (n = 3 independent experiments). (F) The mRNA expression in control shLuc- and shCaV1.3-treated unchallenged hMoDCs related (%) to mock-treated cells (n = 10 independent experiments). (G) Representative motility plots of hMoDCs, treated as in (F) and challenged with T. gondii (PRU-RFP). (H) Histograms of accumulated distances migrated (μm) by hMoDCs as in (G). Dotted lines indicate median values (n = 3 independent experiments). Bar graphs show mean + SEM. Statistical significance was tested by ordinary one-way ANOVA with Dunnett’s multiple comparison test for (D, E, F) and by Mann-Whitney test for (H), *p<0.05, ***p<0.001, ns p≥0.05.
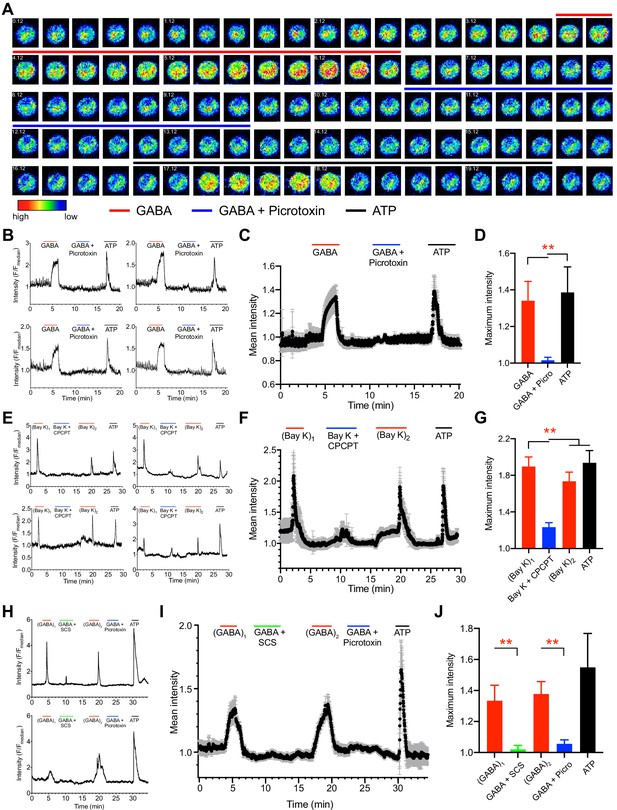
Ca2+ responses in unchallenged and T. gondii-infected DCs upon agonism and antagonism of GABA-A Rs and VDCCs.
(A) Representative time-lapse micrographs show live cell Ca2+ imaging of one single mBMDC preloaded with Ca2+ indicator dye (Fluo-8H AM) and sequentially perfused with GABA (1 mM), GABA (1 mM) + picrotoxin (100 μM) and ATP (150 μM). Colored lines indicate perfusion times (min:s) of treatments. Color scale depicts relative fluorescence intensity. (B) Representative live cell Ca2+ recording traces (relative fluorescence intensity, F/Fmedian) from four cells plotted against time (min) as in (A). Lines indicate respective perfusions times. (C) Mean Ca2+ response intensity (black dots) and SEM (gray whiskers) from one recording with 105 cells, plotted against time as in (B), and (D) maximum Ca2+ response intensity of 294 cells from three independent experiments. (E) Representative live cell Ca2+ recording traces from four cells sequentially perfused with bay K 8644 (40μM), bay K 8644 (40μM) + CPCPT (100 μM), 2nd application of bay K 8644 (40μM) and ATP (150 μM) at times indicated by lines. (F) Mean Ca2+ response intensity (black dots) and SEM (gray whiskers), from 72 cells plotted against time as in (E), and (G) maximum Ca2+ response intensity of 233 cells from three independent experiments. (H) Representative live cell Ca2+ recording traces from T. gondii (ME49-RFP)-infected cells sequentially perfused with GABA (1 mM), GABA (1 mM) + SCS (100 μM), GABA (1 mM), GABA (1 mM) + picrotoxin (100 μM) and ATP (150 μM) at times indicated by lines. (I) Mean Ca2+ response intensity (black dots) and SEM (gray whiskers) from 44 cells, plotted against time as in (H), and (J) maximum Ca2+ response intensity of 115 cells from three independent experiments. Bar graphs show mean + SEM. Statistical significance was assessed by ordinary one-way ANOVA with Dunnett’s multiple comparison test for (D, G, J), **p<0.01.
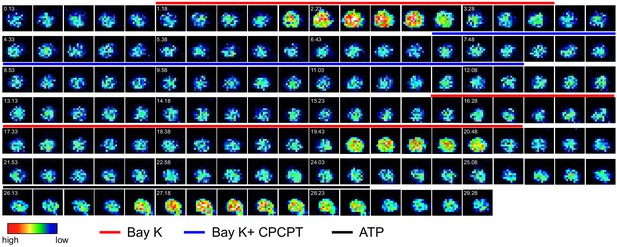
Single-cell micrographs of Ca2+ influx show VDCC activation.
Representative time-lapse micrographs show live cell Ca2+ imaging of unchallenged single mBMDCs preloaded with Ca2+ indicator (Fluo-8H AM) and sequentially perfused with bay K 8644 (40μM), bay K 8644 (40μM) + CPCPT (100 μM), 2nd application of bay K 8644 (40μM) and ATP (150 μM). Colored lines indicate perfusion times (min:s) of treatments. Color scale depicts relative fluorescence intensity.
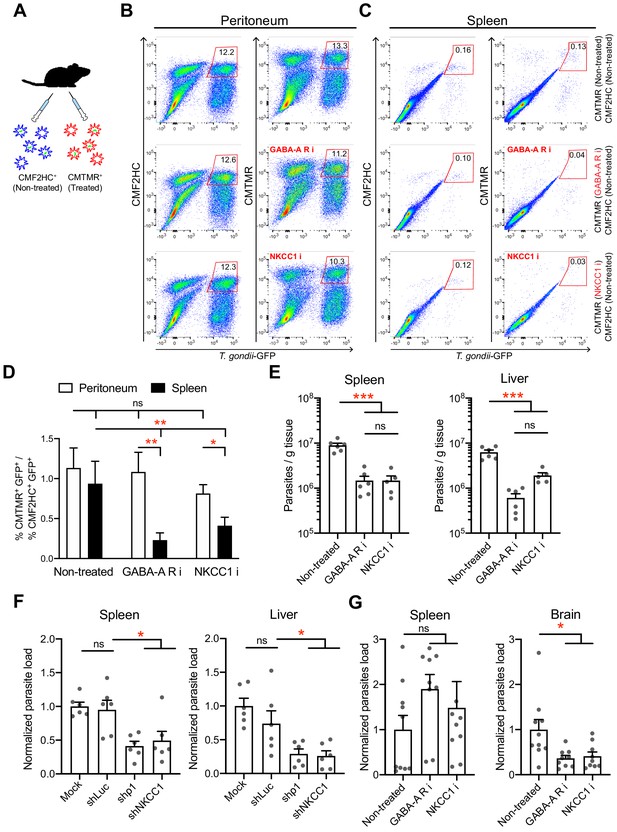
In vivo impact of GABAergic inhibition on DC migration and parasite loads.
(A) Schematic illustration of simultaneous adoptive transfers of T. gondii (ME49/PTG-GFP)-challenged mBMDCs prelabeled with CMF2HC or CMTMR dye and pretreated as described in Materials and methods, respectively. (B, C) Representative bivariate plots show CD11c+ cells from (B) peritoneal cavity and (C) spleen of C57BL/6 mice inoculated with T. gondii-challenged mBMDCs prelabeled with CMF2HC or CMTMR dye. Cells were analyzed by flow cytometry at 14–18 h post-inoculation (gating strategy in Figure 9—figure supplement 2A, B,C). Plots in upper, center and lower rows show, respectively, non-treated cells, cells pretreated with GABA-A R inhibitors (GABA-A R i) and NKCC1 inhibitor (NKCC1 i). (D) Bar graph shows the ratio of treated cells (CD11c+GFP+CMTMR+) to non-treated cells (CD11c+GFP+CMF2HC+) in peritoneal lavage and spleen after adoptive transfer of T. gondii-challenged mBMDCs (n = 7–10 mice per group). (E) Parasite loads in spleen and liver of C57BL/6 mice at day 4 post-inoculation of pharmacologically treated cells (GABA-A R i, NKCC1 i) related to non-treated cells and measured by plaquing assays (n = 5–6 mice per group). (F) Parasite loads in spleen and liver of C57BL/6 mice at day 5 post-inoculation of gene-silenced cells (shρ1, shNKCC1) related to mock- and control shLuc-transduced cells and measured by plaquing assays (n = 6 mice per group). (G) Parasite loads in spleen and brain of CD1 mice at day 7 post-inoculation of pharmacologically treated cells (GABA-A R i, NKCC1 i) related to non-treated cells and measured by plaquing assays (n = 9–10 mice per group). Bar graphs show mean + SEM. Statistical significance was assessed by ordinary one-way ANOVA with Tukey’s multiple comparison test, *p<0.05, **p<0.01, ***p<0.001, ns p≥0.05.
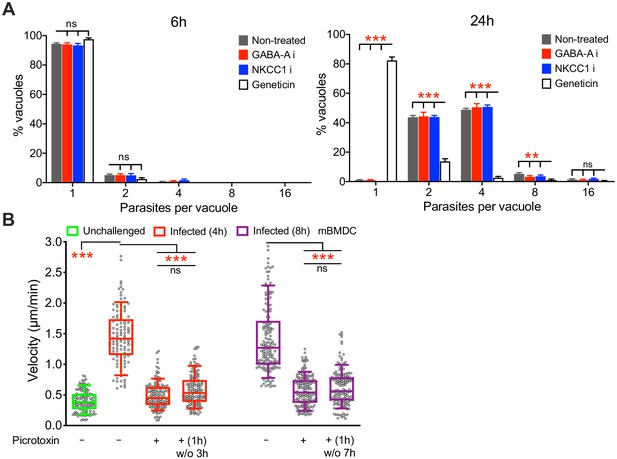
Effect of GABAergic inhibition on parasite replication in DCs.
(A) Bar graphs show parasite replication assessed in T. gondii-infected mBMDCs in presence of picrotoxin (50 μM), bumetanide (40 μM) and geneticin (10 μM), a blocker of polypeptide synthesis in eukaryotic cells, as a positive control. Number of parasites per vacuole is plotted against % of total vacuoles counted at indicated time points, as described in Materials and methods, (n = 3 independent experiments). (B) Box-and-whisker dot plots show, for each indicated condition, velocities (μm/min) of unchallenged and T. gondii-infected mBMDCs treated with picrotoxin for 4 h and 8 h or treated for 1 h, washed and recorded 3 h and 7 h post washing (n = 3 independent experiments). Bar graphs show mean + SEM. Statistical significance was assessed by ordinary one-way ANOVA with Tukey’s multiple comparison test, **p<0.01, ***p<0.001, ns p≥0.05.
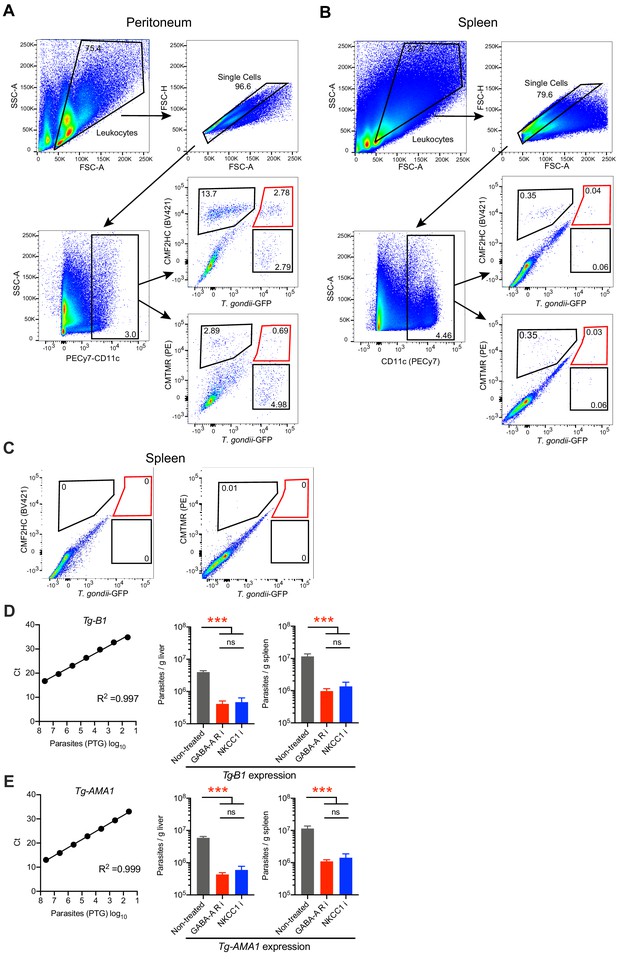
Gating strategy for flow cytometry analysis.
(A, B) Cells collected at 14–18 h from (A) peritoneal cavity and (B) spleen were first gated for leukocyte populations (SSC-A vs. FSC-A), singlets (FSC-H vs. FSC-A) and further gated for expression of PECy7-CD11c. Further, this CD11c+ population was gated to identify adoptively transferred T. gondii-challenged mBMDCs (GFP+) pre-labeled with CMF2HC (non-treated cells) and CMTMR (treated with inhibitors of GABA-A Rs or NKCC1). (C) Representative bivariate plots show CD11c+ cells from spleen of non-infetced C57BL/6 mice gated to identify CMF2HC- or CMTMR-labeled cells. (D, E) Standard curves for B1 and AMA-1 genes of T. gondii, used to extrapolate parasite numbers in tissues by qPCR. The number of parasites per gram tissue, as in Figure 9D, was quantified by qPCR expression of B1 and AMA-1 (n = 5–6 mice per group). Bar graphs show mean + SEM. Statistical significance was assessed by ordinary one-way ANOVA with Tukey’s multiple comparison test, ***p<0.001, ns p≥0.05.
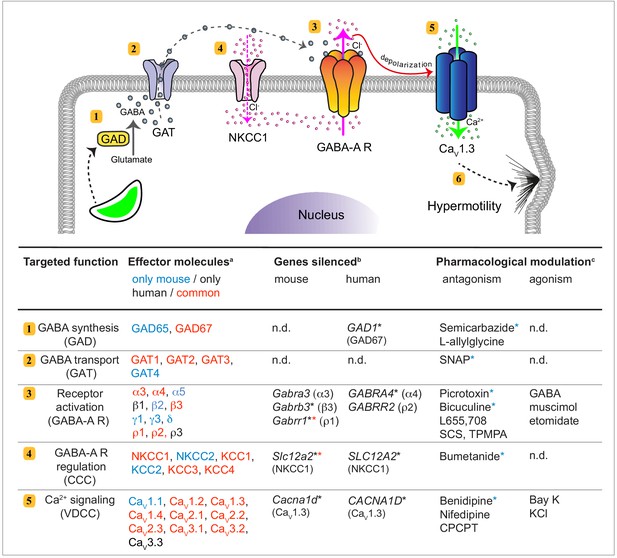
GABAergic signaling in mononuclear phagocytes with an impact on cell migration and coccidian parasite dissemination.
Schematic representation illustrates the molecular GABAergic signaling components (1-5) identified in human and murine mononuclear phagocytes. Their functions and experimental targeting approaches are detailed in the tabular representation, respectively. (6) Ca2+ influx sets the cell in a hypermotile state by activation of MAP kinases and cytoskeletal rearrangements (Ólafsson et al., 2020). In the tabular representation, (a)Red colored text indicates molecular components commonly expressed by mouse and human phagocytes. Blue and black color indicates components only detected in mouse and human cells, respectively. (b)Indicates genes targeted by shRNA with an impact on cell migration in vitro (*), as described under Materials and methods. (*) Red asterisks indicate conditions additionally tested in vivo in mice. (c)Indicates pharmacological agonists and antagonists with an impact on phagocyte motility in vitro tested on both mouse and human phagocytes. (*) Blue asterisks indicate conditions additionally tested in vivo in mice. n.d.: not determined.
Videos
Representative time-lapsed micrographs from a live cell.
Ca2+ imaging recording of mBMDC preloaded with Ca2+ indicator dye (Fluo-8H AM) and sequentially perfused with GABA (1 mM), GABA (1 mM) + picrotoxin (100 μM) and ATP (150 μM) at 3.42–6.46, 10.35–13.32 and 17.08–19.52 min, respectively. The Fluo-8H AM fluorescence signal (green) is converted to rainbow color scale depicting relative fluorescence intensity ranging from blue (lowest) to red (highest). Scale bar 20 μm.
Additional files
-
Supplementary file 1
Primer pair sequences used in real-time quantitative PCR.
- https://cdn.elifesciences.org/articles/60528/elife-60528-supp1-v2.docx
-
Supplementary file 2
GABA-A R subunits and VDCCs transcribed by phagocytes.
- https://cdn.elifesciences.org/articles/60528/elife-60528-supp2-v2.docx
-
Supplementary file 3
Sh-RNA construct sequences.
- https://cdn.elifesciences.org/articles/60528/elife-60528-supp3-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/60528/elife-60528-transrepform-v2.pdf