Casein kinase 1G2 suppresses necroptosis-promoted testis aging by inhibiting receptor-interacting kinase 3
Figures
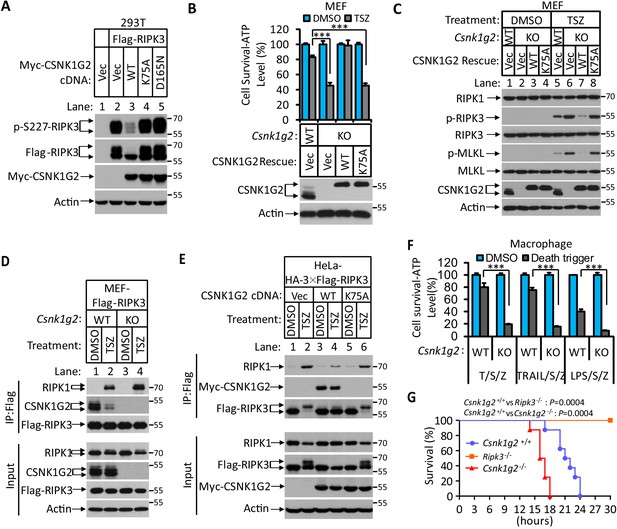
Knockout Csnk1g2 accelerates necroptosis.
(A) Western blotting analysis using antibodies against the indicated proteins. Cultured 293 T cells were transfected with Flag-tagged RIPK3 and the indicated versions of Myc-tagged CSNK1G2, including wild-type (WT) and two kinase-dead point mutants K75A and D165N for 20 hr. Cell extracts were then prepared and used for western blotting analysis. Vec, vector control. Numbers on the right indicate molecular weight markers (kDa). (B) Top: Cell viability as measured by Cell Titer-Glo. Cultured MEF with wild-type Csnk1g2 gene (WT) or with their Csnk1g2 gene knocked out (KO) followed by transfection with vector control (Vec) or indicated wild-type or a kinase-dead (K75A) mutant CSNK1G2 MEF. The cells were then treated with DMSO or TSZ as indicated for 12 hr before the intracellular ATP levels were measured by Cell Titer-Glo. T denotes 20 ng/ml TNF-α; S, denotes 100 nM Smac mimetic; Z denotes 20 μM Z-VAD-FMK. Data are mean ± SD of triplicate wells. ***p<0.001. p-values were determined by two-sided unpaired Student’s t-tests. Bottom: Aliquots of these treated cells were used for western blotting analysis using an antibody against CSNK1G2. (C) Western blotting of necroptosis activation markers phospho-RIPK3 (p-RIPK3) and phospho-MLKL (p-MLKL). Cultured MEF cells with indicated CSNK1G2 gene as in (B) were treated with indicated stimuli for 4 hr before the cell extracts were prepared and subjected to western blotting analysis as indicated. (D and E) Western blotting analysis of RIPK3-associated RIPK1 and CSNK1G2. Immunoprecipitates using an anti-Flag antibody from extracts of MEF-Flag-RIPK3 and MEF (Csnk1g2−/−)-Flag-RIPK3 cells (D) or HeLa-HA-3×Flag-RIPK3-Myc-CSNK1G2(WT and K75A) cells (E) treated with the indicated stimuli for 6 hr were subjected to western blotting analyzing using antibodies as indicated. (F) Cell viability measurement of bone marrow-derived macrophages from the wild-type (WT) or Csnk1g2 knockout mice. Macrophages were isolated from the WT or Csnk1g2 knockout mice (KO) and treated with the indicated necroptosis stimuli for 12 hr, and the cell viability was measured by Cell-Titer Glo. Trail: TNF-related apoptosis-inducing ligand. LPS: Lipopolysaccharide. Data are mean ± SD of triplicate wells. ***p<0.001. p-values were determined by two-sided unpaired Student’s t-tests. (G) Kaplan–Meier plot of survival of male Csnk1g2+/+(WT), Csnk1g2−/− (Csnk1g2 knockout littermates), or Ripk3−/− (Ripk3 gene knockout) mice (n = 10 for each genotype, age: 3 months) injected intraperitoneally with one dose of murine TNF-α (300 μg/kg). Body temperature was measured with a lubricated rectal thermometer. Mice with a temperature below 23°C were euthanized for ethical reasons. Generation of Csnk1g2−/− mice.
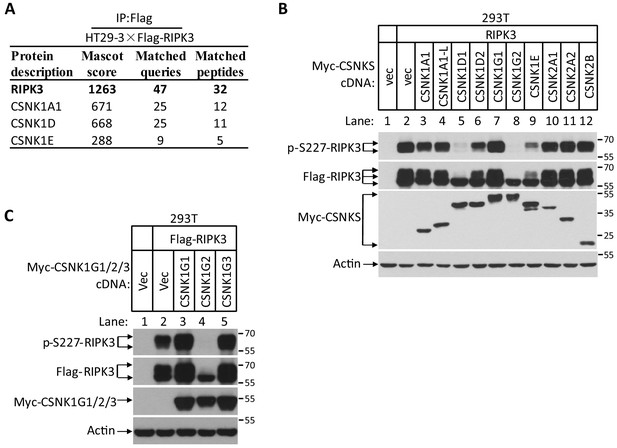
CSNK1G2 binds and inhibits RIPK3 kinase activity to prevent necroptosis.
(A) CSNK1s associate with RIPK3. The HT29-HA-3×Flag-RIPK3 cell lysates were immunoprecipitated using anti-Flag resin. The pull-down protein mixture was subjected to mass spectrometry analysis and the identified casein kinase was shown. (B) Measurement of the effect of co-expressed casein kinase members on RIPK3 kinase activity. Cultured 293T cells were co-transfected with Flag-tagged RIPK3 and indicated Myc-tagged CSNK1A1, CSNK1A1-L, CSNK1D1, CSNK1D2, CSNK1G1, CSNK1G2, CSNK1E, CSNK2A1, CSNK2A2, and CSNK2B for 20 hr. The cell extracts were then subjected to western blotting analysis using antibodies against Myc-tag, Flag-tag, β-actin, and phosphor-S227-RIPK3 as indicated. Numbers on the right indicate molecular weight markers (kDa). (C) Measurement of the effect of co-expression casein kinase 1G members on RIPK3 kinase activity. Cultured 293T cells were co-transfected with Flag-tagged RIPK3 and indicated Myc-tagged CSNK1G1, CSNK1G2, and CSNK1G3 for 20 hr. The cell extracts were then subjected to western blotting analysis using antibodies against Myc-tag, Flag-tag, β-actin, and phosphor-S227-RIPK3 as indicated. (A and B) The necroptotic effect of knockout Csnk1g2 in MEFs. Cultured parental MEF cells (WT) and MEF cells with their Csnk1g2 knocked out (KO) were treated with the indicated necroptotic stimuli for 12 hr. The cell viability of these necroptotic stimuli-treated cells was then measured using Cell-titer Glo in (A). Data are mean ± SD of triplicate wells. ***p<0.001. p-values were determined by two-sided unpaired Student’s t-tests. LPS 2 ng/ml, TRAIL 20 ng/ml. The cell extracts were then subjected to western blotting analysis using antibodies against RIP1, phosphor-S232-RIPK3 (p-RIPK3), β-actin, MLKL, and phosphor-S345-MLKL as indicated in (B).
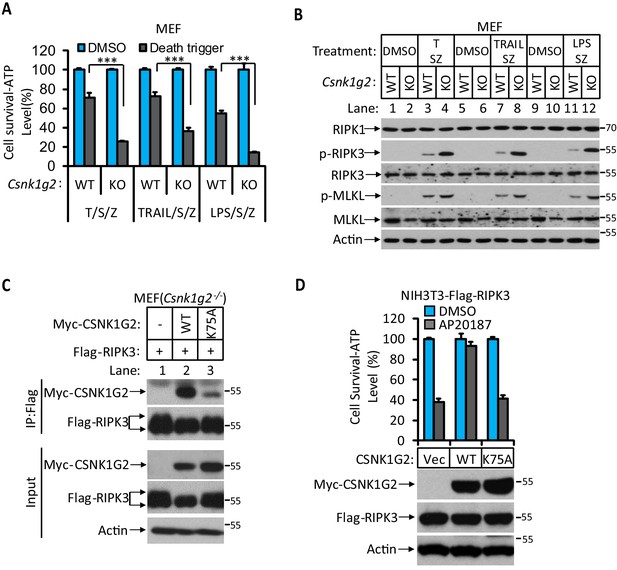
The kinase activity of CSNK1G2 is required for its binding to RIPK3.
(A and B) The necroptotic effect of knockout Csnk1g2 in MEFs. Cultured parental MEF cells (WT) and MEF cells with their Csnk1g2 knocked out (KO) were treated with the indicated necroptotic stimuli for 12 hr. The cell viability of these necroptotic stimuli-treated cells was then measured using Cell-titer Glo in (A). Data are mean ± SD of triplicate wells. ***p<0.001. p-values were determined by two-sided unpaired Student’s t-tests. LPS 2 ng/ml, TRAIL 20 ng/ml. The cell extracts were then subjected to western blotting analysis using antibodies against RIP1, phosphor-S232-RIPK3 (p-RIPK3), β-actin, MLKL, and phosphor-S345-MLKL as indicated in (B). (C) Cultured MEF cells with their Csnk1g2 gene knocked out (Csnk1g2−/−) were co-transfected with cDNAs encoding Myc-tagged wild-type (WT) CSNK1G2 or kinase-dead mutant (K75A) as indicated and Flag-RIP3 for 24 hr. The cell extracts were then subjected to western blotting analysis directly (Input), or immunoprecipitation with anti-Flag antibody and the precipitates were subjected to western blotting analysis using antibodies against Myc-tag, Flag-tag, and β-actin as indicated. (D) The effect of WT and kinase-dead mutant of CSNK1G2 on RIPK3 dimerization-induced necroptosis. Cultured NIH3T3-Flag-RIPK3-Myc-CSNK1G2(Vec, WT, and K75A) cells were treated with the FKBP dimerizer molecule AP20187 for 12 hr. The cell extracts were subsequently subjected to western blotting analysis using antibodies against Myc-tag, Flag-tag, and β-actin as indicated (lower panel). The cell viability was measured by Cell-titer Glo. Data are mean ± SD of triplicate wells.
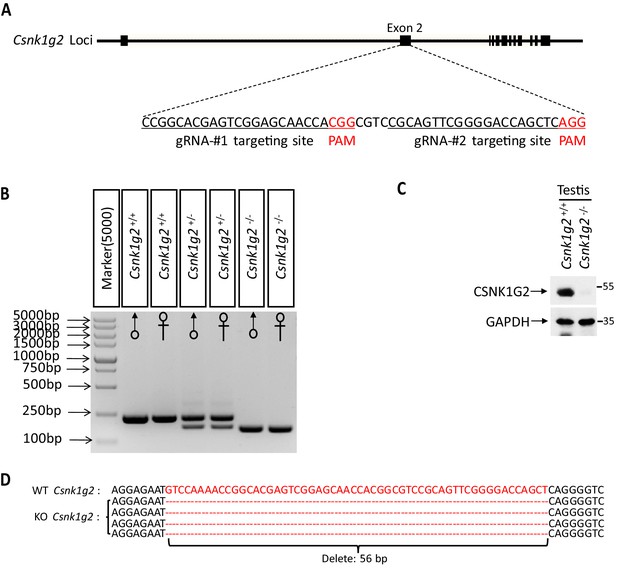
Generation of Csnk1g2−/− mice.
(A) Schematic of CRISPER-Cas9 strategy for the generation for Csnk1g2 knockout mice. The gene structure of CSNK1G2 and two guide RNA sequences targeting the exon 2 of Csnk1g2 were shown with the PAM sequences highlighted in red. (B) Genotyping results of genomic DNA from Csnk1g2+/+, Csnk1g2+/−, and Csnk1g2−/− mice with PCR primers covered the exon 2n targeted region. (C) Immunoblot of CSNK1G2 from testis extracts of 2-month old Csnk1g2+/+ and Csnk1g2−/− littermates using antibodies against CSNK1G2 and GAPDH as indicated (n = 3). (D) Sequence of the genomic of Csnk1g2 knockout mice covering the guide RNA targeted sites. The deleted 56 base pairs of DNA were highlighted in red.
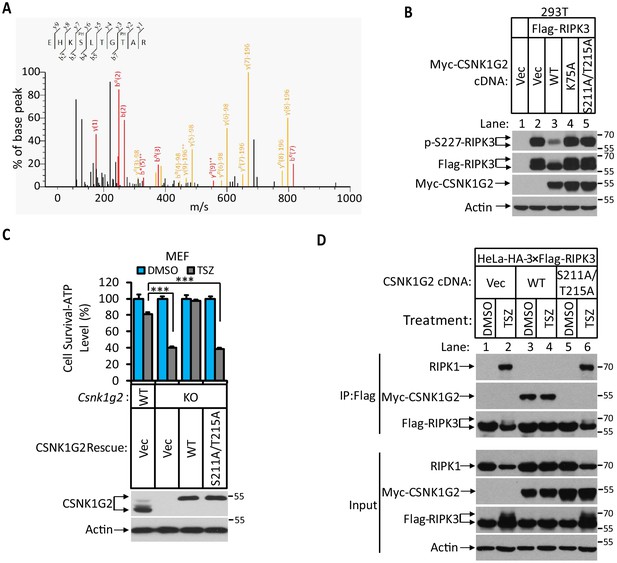
Identification and characterization of CSNK1G2 auto-phosphorylation sites.
(A) MS/MS spectrum of CSNK1G2 phosphorylation sites. The identified phosphorylated peptide to be EHKPSLTGPTAR with S211 and T215 as the phosphorylated amino acid residues. The b- and y-type product ions are indicated in the spectrum. Data are related to those in Figure 2—figure supplement 1. (B) Effect on RIPK3 auto-phosphorylation by co-expression of the indicated version of CSNK1G2. Cultured 293 T cells were transfected with Flag-tagged RIPK3 cDNA together with indicated Myc-tagged wild-type (WT) CSNK1G2, or kinase-dead mutant K75A, or phosphorylation site-resistant mutant S211A/T215A for 20 hr. The cell extracts were then subjected to western blotting analysis using antibodies against phospho-S227-RIPK3, Flag-RIPK3, and Myc-CSNK1G2 as indicated. (C) Top: Cell viability measurement of effect of phosphorylation sites mutant CSNK1G2 on necroptosis. Cultured WT MEF or MEF with their Csnk1g2 gene knocked out (KO) were transfected with either vector control (Vec) or cDNA encoding WT Csnk1g2 or phosphorylation sites mutant (S211A/T215A) followed by treatment of DMSO or necroptotic stimuli TSZ as indicated for 12 hr. The cell viability was measured by Cell-titer Glo. Data are mean ± SD of triplicate wells. ***p<0.001. p-values were determined by two-sided unpaired Student’s t-tests. Bottom: Aliquots of these treated cells were used to make cell extracts for western blotting analysis using an antibody against CSNK1G2 protein. The effect of CSNK1G2 on RIPK1/RIPK3 interaction as measured by co-IP. Cultured HeLa-HA-3×Flag-RIPK3 cells were transfected with either vector control (Vec) or WT or phosphorylation site mutant Myc-CSNK1G2 (S211A/T215A) as indicated. The cells were then treated with DMSO or necroptosis stimuli TSZ for 6 hr. The cell extracts were prepared and subjected to immunoprecipitation with an anti-Flag antibody. The extracts (Input) and the immunoprecipitates (IP: Flag) were then subjected to western blotting analysis using antibodies as indicated.
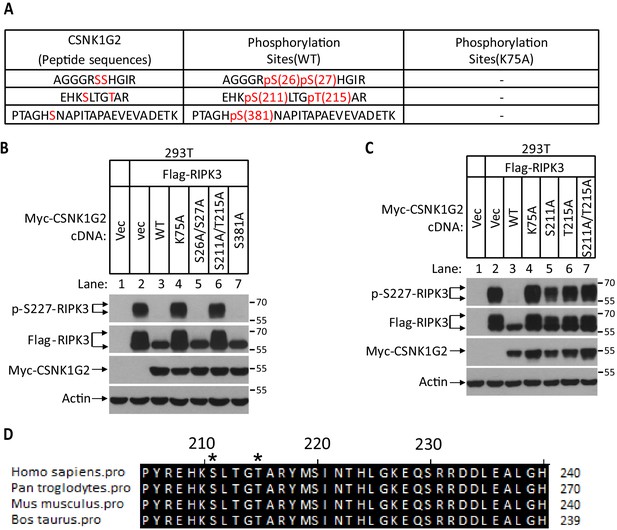
Auto-phosphorylation sites on CSNK1G2.
(A) Identification of auto-phosphorylation sites on CSNK1G2. Myc-tagged wild-type (WT) CSNK1G2 or its kinase-dead mutant (K75A) was co-transfected with Flag-RIP3 in 293 T cells for 24 hr. CSNK1G2 was immunoprecipitated using anti-Myc resins. The CSNK1G2 (WT, K75A) bands were excised and analyzed by MS/MS. The identified phosphorylated peptides were shown in the table with the phosphorylated amino acid residues highlighted in red. No phosphorylated peptide was identified in CSNK1G2 (K75A) sample. (B) The effect of phosphorylation sites mutants CSNK1G2 on RIPK3 kinase activity. Cultured 293 T cells were transfected with vector control (Vec) or Flag-tagged RIPK3 and Myc-tagged WT, or kinase-dead (K75A) or different phosphorylation site mutants as indicated (S26A/S27A, S211A/T215A, and S381A) for 20 hr. The cell extracts were then subjected to western blotting analysis using antibodies against Flag-tag, Myc-tag, phosphor-Serine227-RIPK3, and β-actin as indicated. (C) The effect of S211A and T215A mutants of CSNK1G2 on RIPK3 kinase activity. Cultured 293 T cells were transfected with vector control (Vec) or Flag-tagged RIPK3 and Myc-tagged WT, or a kinase-dead mutant (K75A), or S211A or T215A mutants for 20 hr. The cell extracts were then subjected to western blotting analysis using antibodies against Flag-tag, Myc-tag, phosphor-Serine227-RIPK3, and β-actin as indicated. (D) Alignment of amino acid sequences around the auto-phosphorylation sites of CSNK1G2 in four vertebrate species. The Serine 211 and Threonine 215 (human origin) residues are denoted by asterisks (*). The numbers on the right indicate the corresponding histidine in CSNK1G2 of indicated species.
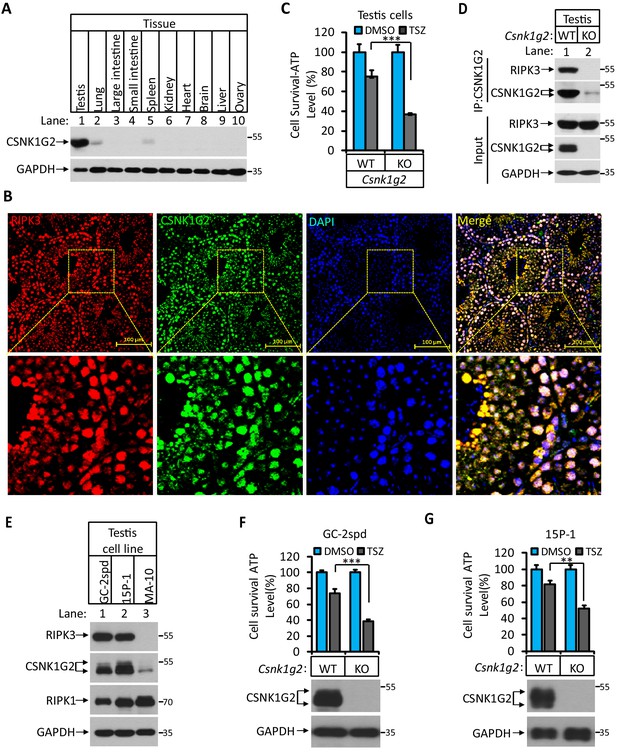
CSNK1G2 and RIPK3 are co-expressed in the seminiferous tubules of the mouse testis.
(A) The expression of CSNK1G2 in mouse testis, lung, large intestine, small intestine, spleen, kidney, heart, brain, liver, and ovary tissues (n = 3, 2 months). The indicated tissue extracts were subjected to western blotting analysis using antibodies against CSNK1G2 and GAPDH as indicated. (B) The expression of RIPK3 and CSNK1G2 in mouse testis. The testis sections of 2-month-old wild-type mice (n = 3) were stained sequentially with antibodies against RIPK3 and CSNK1G2 followed by fluorescent-conjugated secondary antibody. Counterstaining with DAPI, blue. Scale bar on the upper panel is 100 μm. The areas marked by the yellow boxes on the upper panel were shown in the lower panel. (C) The sensitivity of primary cells from the seminiferous tubules of Csnk1g2−/− and Csnk1g2+/+ testis to necroptosis induction. The cells from the seminiferous tubules of 2-month-old littermates with indicated genotype were isolated and cultured in vitro before treated with DMSO or TSZ as indicated for 12 hr. The cell viability was then measured by Cell-Titer Glo. Data are mean ± SD of triplicate wells. ***p<0.001. p-values were determined by two-sided unpaired Student’s t-tests. (D) Western blotting analysis of CSNK1G2 interaction with RIPK3 in mouse testis. The wild-type and Csnk1g2 knockout testis (3 months) were ground and resuspended in lysis buffer, homogenized for 30 s with a Paddle Blender. The supernatants were collected, and CSNK1G2 was immunoprecipitated using anti-CSNK1G2 antibody. The immunocomplexes and lysates were analyzed by western blotting using antibodies as indicated. Each group was from a pool of three mice. (E) The expression of RIPK3, CSNK1G2, and RIPK1 proteins in GC-2spd, 15 P-1, and MA-10 cells. The extracts from the indicated cultured cells were subjected to western blotting analysis using antibodies against RIPK3, CSNK1G2, RIPK1, and GAPDH as indicated. (F and G) The effect of CSNK1G2 on necroptosis of GC-2spd and 15 P-1 cells. Cultured parental GC-2spd (f) or 15 P-1 cells (g) (WT), and GC-2spd or 15 P-1 cells with their Csnk1g2 gene knocked out (Csnk1g2−/−) were treated with DMSO or TSZ as indicated for 4 hr. The cell viability was measured by Cell-titer Glo. Data are mean ± SD of triplicate wells. **p<0.01, ***p<0.001. p-values were determined by two-sided unpaired Student’s t-tests. Bottom, immunoblot of CSNK1G2. Cell extracts from aliquots of these cells were also subjected to western blotting analysis using antibodies against CSNK1G2 and GAPDH as indicated, and the results were shown at the bottom.
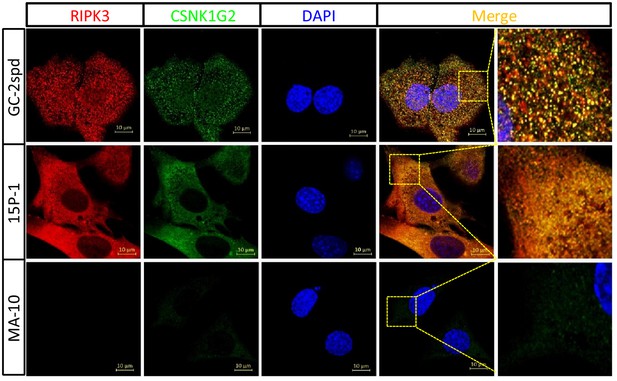
Immunofluorescent analysis of RIPK3 and CSNK1G2 expression in GC-2spd, 15 P-1, and MA-10 cells.
The GC-2spd, 15 P-1, and MA-10 cells cultured on cover slides were sequentially stained with antibodies against RIPK3 and CSNK1G2 followed by secondary antibodies conjugated with red (RIPK3) or green (CSNK1G2). Counterstaining with DAPI, blue. Scale bar, 10 μm.
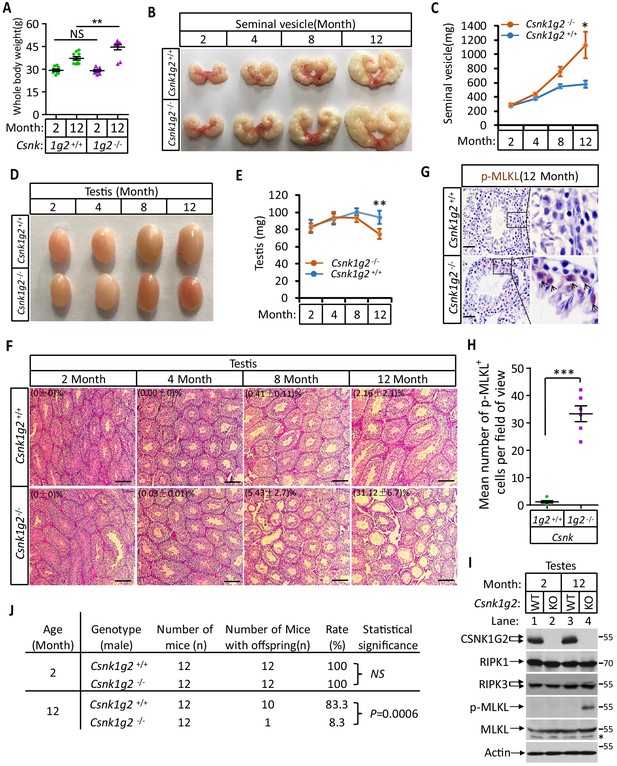
Accelerated reproductive system aging in Csnk1g2−/− male mice.
(A) Body weights of Csnk1g2+/+ and Csnk1g2−/− male littermate mice when they were 2 and 12 months old (n = 10 for each genotype). (B and C) Macroscopic features (B) and weights (C) of seminal vesicles from Csnk1g2+/+ and Csnk1g2−/− male littermate mice (n = 10 for each genotype) at the indicated ages. (D and E) Macroscopic features (D) and weights (E) of testes from Csnk1g2+/+ and Csnk1g2−/− male littermate mice (n = 12 for each genotype) at the indicated ages. (F) H&E staining sections of testis from Csnk1g2+/+ and Csnk1g2−/− male littermate mice (n = 10 for each genotype) at the indicated ages. The number of empty seminiferous tubules was counted based on H&E staining, and the percentage of empty seminiferous tubules of each group is labeled in the upper left corner of the images. Scale bar, 200 μm. (G and H) Immunohistochemical staining (IHC) of testes from Csnk1g2+/+ and Csnk1g2−/− male littermate mice (n = 6 for each genotype) with phospho-MLKL (p-MLKL) antibody in (G). p-MLKL positive cells were counted in five fields per testis and quantified in (H). Scale bar, 100 μm. (I) Western blotting analysis of extracts from phosphate-buffered saline (PBS) perfused testes of Csnk1g2+/+ (WT) and Csnk1g2−/− (KO) male littermate mice of 2 and 12 months of age using antibodies against CSNK1G2, RIPK1, RIPK3, MLKL, and phospho-MLKL (p-MLKL) and β-actin as indicated. The number on the right is markers of molecular weight (kDa). Each group was from a pool of three mice. (J) Summary of fertility rates of Csnk1g2+/+ and Csnk1g2−/− male littermate mice (n = 12). Each male mice of 2 or 12 months of age was housed in the same cage with a pair of 10-week-old wild-type female mice for 2 months; females were replaced every 2 weeks. The number of male mice with reproduction capacity was counted. p-values were determined using Fisher’s exact tests (unpaired, two-tailed). All quantified data in the figure except (J) represent the mean ± SEM. *p<0.05, **p<0.01, ***p<0.001. p-values were determined by two-sided unpaired Student’s t-tests. NS, not significant.
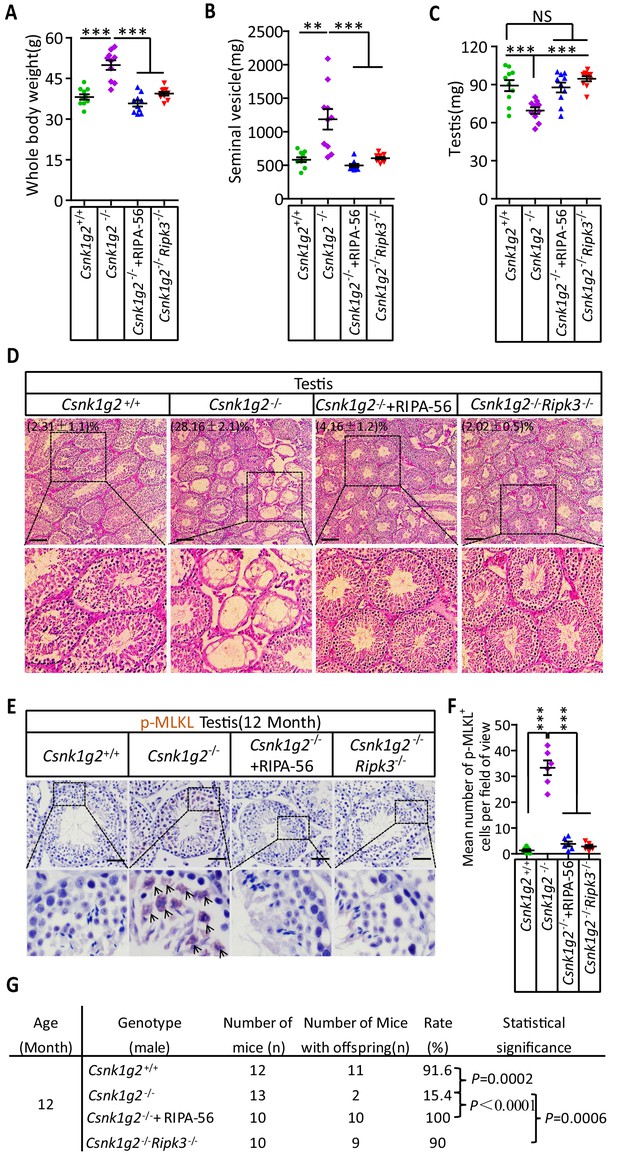
Rescuing the accelerated male reproductive system aging of Csnk1g2 knockout mice with a RIPK1 kinase inhibitor or Ripk3 knockout.
(A) The body weights of 12-month-old Csnk1g2+/+, Csnk1g2−/−, and Csnk1g2−/− fed with a RIPK1 kinase inhibitor (RIPA-56)-containing diet, and Csnk1g2−/−Ripk3−/− male littermate mice (n = 10 for each genotype). (B) The weights of seminal vesicles from 12-month-old Csnk1g2+/+, Csnk1g2−/−, Csnk1g2−/−+RIPA-56, and Csnk1g2−/−Ripk3−/− male littermate mice (n = 10 for each genotype). (C) The weights of testes from 12-month-old Csnk1g2+/+, Csnk1g2−/−, Csnk1g2−/−+RIPA-56, and Csnk1g2−/−Ripk3−/− male littermate mice (n = 10 for each genotype). (D) H&E staining of testis sections from 12-month-old Csnk1g2+/+, Csnk1g2−/−, Csnk1g2−/−+RIPA-56, and Csnk1g2−/−Ripk3−/− male littermate mice (n = 10 for each genotype). The number of empty seminiferous tubules was counted based on H&E staining, and the percentage of empty seminiferous tubules was labeled in the upper left corner of the images: scale bar, 200 μm. (E and F) IHC staining of testes from 12-month-old Csnk1g2+/+, Csnk1g2−/−, Csnk1g2−/−+RIPA-56, and Csnk1g2−/−Ripk3−/− male littermate mice (n = 8 for each genotype) with an anti-phospho-MLKL (p-MLKL) antibody (E). p-MLKL positive cells were counted in five fields per testis and quantified in (F). Scale bar, 100 μm. (G) Summary of the fertility rates of 12-month-old Csnk1g2+/+, Csnk1g2−/−, Csnk1g2−/−+RIPA-56, and Csnk1g2−/−Ripk3−/− male littermate mice (n = 10 for each genotype). Each male mouse was caged with a pair of 10-week-old wild-type female mice for 2 months; females were replaced every 2 weeks. The number of male mice with reproduction capacity was counted. p-values were determined using Fisher’s exact tests (unpaired, two-tailed). Csnk1g2−/−+RIPA-56 mice: Csnk1g2−/− male mice were fed with AIN93G or AING3G containing RIPA-56 (RIPA-56: 300 mg/kg) for 10 months started when they were 2 months old in an SPF facility. All quantified data in the figure except (G) represent the mean ± SEM. **p<0.01, ***p<0.001. p-values were determined by two-sided unpaired Student’s t-tests. NS, not significant.
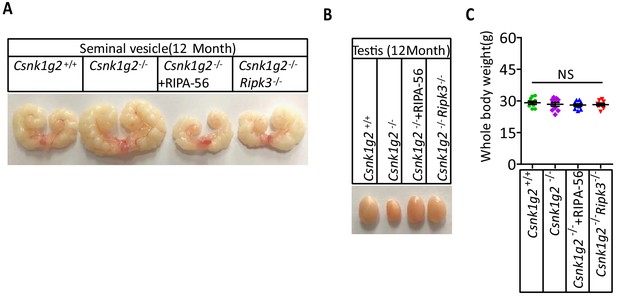
RIPK1 inhibitor-containing diet or double knockout Ripk3 prevents the appearance of accelerated male reproduction organ aging feature in Csnk1g2 knockout mice.
(A) Macroscopic features of a typical 12-month-old seminal vesicle from Csnk1g2+/+, Csnk1g2−/−, Csnk1g2−/−+RIPA-56, and Csnk1g2−/−Ripk3−/− male littermate mice (n = 10 for each genotype examined). (B) Macroscopic features of a typical 12-month testes from Csnk1g2+/+, Csnk1g2−/−, Csnk1g2−/−+RIPA-56, and Csnk1g2−/−Ripk3−/− male littermate mice (n = 10 for each genotype examined). (C) Body weights of 3-month-old Csnk1g2+/+, Csnk1g2−/−, Csnk1g2−/−+RIPA-56, and Csnk1g2−/−Ripk3−/− male mice (n = 10 for each genotype). p-values were determined by two-sided unpaired Student’s t-tests. NS, not significant.
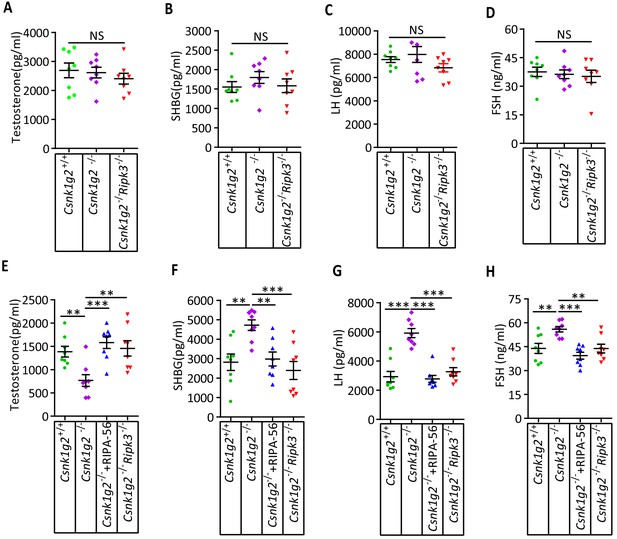
RIPK1 inhibitor-containing diet or double knockout Ripk3 prevents the hormonal changes associated with the accelerated male reproduction system aging in Csnk1g2 knockout mice.
(A–D) Serum hormonal levels of 3-month-old Csnk1g2+/+, Csnk1g2−/−, Csnk1g2−/−+RIPA-56, and Csnk1g2−/−Ripk3−/− male littermate mice. Littermates of male mice with the indicated genotype were killed, and the indicated hormone levels in serum were measured using ELISA kit for each hormone (n = 8 for each genotype). Data represent the mean ± SEM. **p<0.01, ***p<0.001. p-values were determined by two-sided unpaired Student’s t-tests. Csnk1g2−/−+RIPA-56 mice: 2-month-old Csnk1g2−/− male mice were fed with AIN93G or AING3G containing RIPA-56 (RIPA-56: 300 mg/kg) for 1 month in an SPF facility before used. (E–H) Serum hormonal levels of 12-month-old Csnk1g2+/+, Csnk1g2−/−, Csnk1g2−/−+RIPA-56, and Csnk1g2−/−Ripk3−/− male littermate mice. Littermates of 12-month-old male mice with the indicated genotype were killed, and the indicated hormone levels in serum were measured using ELISA kit for each hormone (n = 8 for each genotype). Data represent the mean ± SEM. **p<0.01, ***p<0.001. p-values were determined by two-sided unpaired Student’s t-tests. Csnk1g2−/−+RIPA-56 mice: 2-month-old Csnk1g2−/− male mice were fed with AIN93G or AING3G containing RIPA-56 (RIPA-56: 300 mg/kg) for 10 months in an SPF facility.
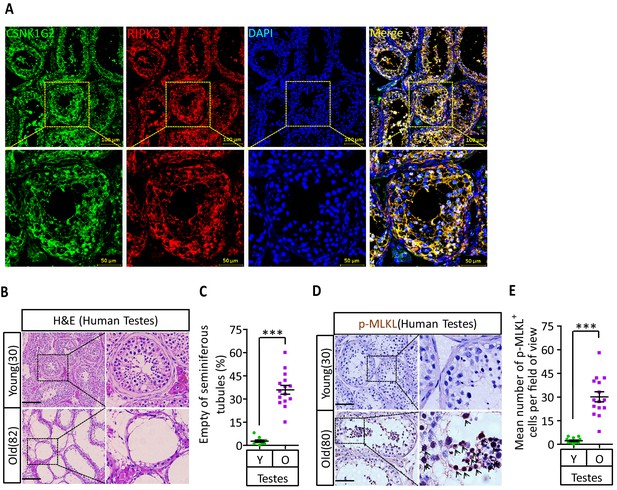
CSNK1G2 expression and necroptosis activation marker phosphor-Serine358-MLKL in human testes.
(A) Expression of CSNK1G2 and RIPK3 in human testes. Sections from a testis sample of a 30-year-old human patient were stained sequentially with antibodies against CSNK1G2 and RIPK3 as indicated followed by green or red fluorescent-conjugated secondary antibodies as indicated. Counterstaining with DAPI, blue. Scale bar, 50/100 μm. Yellow boxes in the upper panels were shown in the lower panels. The experiment was repeated three times with three different patients. (B and C) H&E staining of testes from young and old men. Young man testes (25–30 years, n = 10; from testicular torsion necrosis patients) and old man testes (80–89 years, n = 15; from prostate cancer patients) were sectioned and stained with H&E in (B). The number of empty seminiferous tubules was counted based on H&E staining and quantification in (C); empty seminiferous tubules were counted in five fields per testis. Scale bar, 100 μm. (D and E) IHC of testes from young and old men with phosphor-Serine358-MLKL antibody (p-MLKL). Young man testes (25–30 years, n = 10; from testicular torsion necrosis patients) and old man testes (80–89 years, n = 15; from prostate cancer patients) were sectioned and stained with an antibody against phosphor-Serine358-MLKL antibody (D). p-MLKL+ cells were counted in five fields per testis and quantification in (E). Scale bar, 100 μm. All quantified data in the figure represent the mean ± SEM. ***p<0.001. p-values were determined by two-sided unpaired Student’s t-tests.
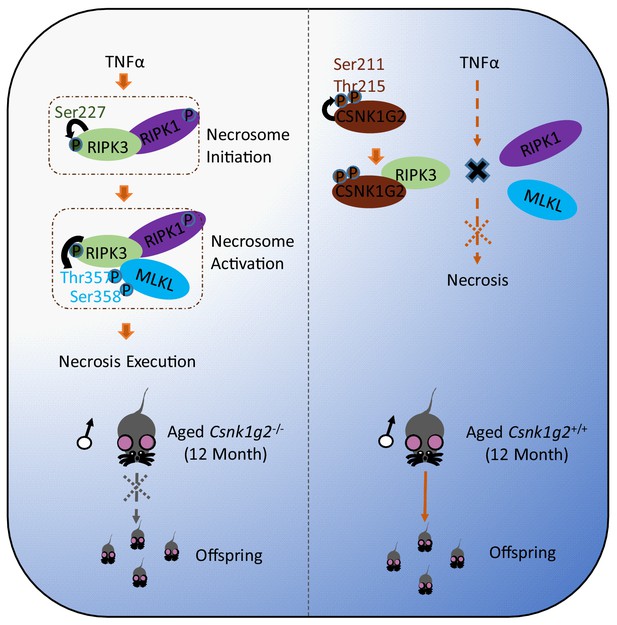
CSNK1G2 suppresses necroptosis-promoted testis aging by binding and inhibiting RIPK3.
Necroptosis induced by TNF-α is initiated by the necrosome formation, a protein complex containing both RIPK1 and RIPK3. CSNK1G2 blocks necrosome formation through binding RIPK3 and preventing RIPK3 activation. CSNK1G2 binding to RIPK3 is triggered by an autophosphorylation at serine211/threonine 215 sites in its kinase domain. Csnk1g2 knockout mice showed enhanced necroptosis response and premature aging of their testis.
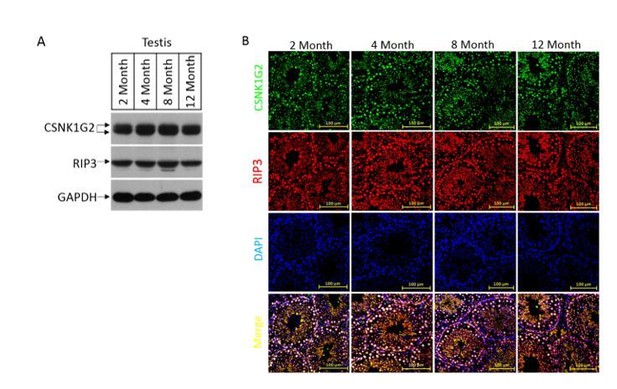
The expression, localization, and activity change of CSNK1G2 in wild type mouse testis.
(A) Western blotting analysis of extracts from phosphate-buffered saline(PBS) perfused testes of CSNK1G2+/+ (WT) mice of 2, 4, 8 and 12-month of age using antibodies against CSNK1G2, RIP3, and GAPDH as indicated.Each group was from a pool of three mice. (B) The expression of RIP3 and CSNK1G2 in mouse testis. The testis sections of 2, 4, 8 and 12-month old wild type mice (n=3) were stained sequentially with antibodies against RIP3 and CSNK1G2 followed by fluorescent-conjugated secondary antibody. Counterstaining with DAPI, blue. Scale bar on the upper panel is 100 μm.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Cell line (Homo sapiens) | HEK293T | ATCC | CRL-11268 | Female |
| Cell line (Homo sapiens) | HeLa | ATCC | CCL-2 | Female |
| Cell line (Mus musculus) | GC-2spd(ts) | ATCC | CRL-2196TM | Male |
| Cell line (Mus musculus) | 15 P-1 | ATCC | CRL-2618TM | Male |
| Cell line (Mus musculus) | MA-10 | ATCC | CRL-3050TM | Male |
| Cell line (Homo sapiens) | HeLa-HA-3×Flag-RIPK3 | Dr. Xiaodong Wang lab at National Institute of Biological Sciences, Beijing | N/A | |
| Cell line (Homo sapiens) | HeLa-HA-3×Flag-RIPK3-Myc-CSNK1G2 | Dr. Xiaodong Wang lab at National Institute of Biological Sciences, Beijing | N/A | |
| Cell line (Homo sapiens) | HeLa-HA-3×Flag-RIPK3-Myc-CSNK1G2(K75A) | Dr. Xiaodong Wang lab at National Institute of Biological Sciences, Beijing | N/A | |
| Cell line (Homo sapiens) | HeLa-HA-3×Flag-RIPK3-Myc-CSNK1G2 (S211A/T215A) | Dr. Xiaodong Wang lab at National Institute of Biological Sciences, Beijing | N/A | |
| Cell line (Mus musculus) | NIH3T3-Flag-mRIPK3 | Dr. Xiaodong Wang lab at National Institute of Biological Sciences, Beijing | N/A | |
| Cell line (Mus musculus) | NIH3T3-Flag-mRIPK3-Myc-mCSNK1G2 | Dr. Xiaodong Wang lab at National Institute of Biological Sciences, Beijing | N/A | |
| Cell line (Mus musculus) | NIH3T3-Flag-mRIPK3-Myc-mCSNK1G2(K75A) | Dr. Xiaodong Wang lab at National Institute of Biological Sciences, Beijing | N/A | |
| Cell line (Mus musculus) | MEF(Csnk1g2−/−) | Dr. Xiaodong Wang lab at National Institute of Biological Sciences, Beijing | N/A | |
| Cell line (Mus musculus) | MEF(Csnk1g2−/−)-Myc-mCSNK1G2 | Dr. Xiaodong Wang lab at National Institute of Biological Sciences, Beijing | N/A | |
| Cell line (Mus musculus) | MEF(Csnk1g2−/−)-Myc-mCSNK1G2(K75A) | Dr. Xiaodong Wang lab at National Institute of Biological Sciences, Beijing | N/A | |
| Cell line (Mus musculus) | MEF(Csnk1g2−/−)-Myc-mCSNK1G2(S211A/T215A) | Dr. Xiaodong Wang lab at National Institute of Biological Sciences, Beijing | N/A | |
| Antibody | Anti-RIPK3 (rabbit polyclonal) | ProSci | Cat# 2283; RRID:AB_203256 | WB (1:1000) |
| Antibody | Anti-CSNK1G2 (rabbit monoclonal) | Abcam | Cat# ab238121 | First described in this paper;WB (1:1000) |
| Antibody | Anti-GAPDH-HRP (mouse monoclonal) | MBL | Cat# M171-1; RRID:AB_10699462 | WB (1:20,000) |
| Antibody | Anti-β-Actin-HRP (rabbit polyclonal) | MBL | Cat# PM053-7; RRID:AB_10697035 | WB (1:20,000) |
| Antibody | Anti-Flag-HRP (mouse monoclonal) | Sigma-Aldrich | Cat# A8592; RRID:AB_439702 | WB (1:10,000) |
| Antibody | Anti-RIPK (rabbit monoclonal) | Cell Signaling | Cat# 3493S; RRID:AB_2305314 | WB (1:1000) |
| Antibody | Anti-Myc-HRP (mouse monoclonal) | MBL | Cat# M192-7 | WB (1:2000) |
| Antibody | Anti-Mouse-MLKL (rabbit polyclonal) | ABGENT | Cat# AP14272b; RRID:AB_11134649 | WB (1:1000) |
| Antibody | Anti-Mouse-p-MLKL (rabbit monoclonal) | Abcam | Cat# ab196436; RRID:AB_2687465 | WB (1:1000) |
| Antibody | Anti-Human-p-MLKL (rabbit monoclonal) | Abcam | Cat# ab187091; RRID:AB_2619685 | WB (1:1000) |
| Antibody | Anti-p-S227-RIPK3 (rabbit monoclonal) | Abcam | Cat# ab209384; RRID:AB_2714035 | WB (1:1000) |
| Antibody | Anti-p-S232-RIPK3 (rabbit monoclonal) | Abcam | Cat# ab222302 | WB (1:1000) |
| Antibody | Mouse anti-rabbit IgG (mouse monoclonal) | Cell Signaling | Cat# 5127S; RRID:AB_10892860 | WB (1:2000) |
| Antibody | Donkey anti-mouse, Alexa Fluor 488 (mouse polyclonal) | Thermo Fisher | Cat# A-21202; RRID:AB_141607 | IF (1:500) |
| Antibody | Donkey anti-mouse, Alexa Fluor 555 (mouse polyclonal) | Thermo Fisher | Cat# A-31570; RRID:AB_2536180 | IF (1:500) |
| Antibody | Donkey anti-rabbit, Alexa Fluor 488 (rabbit polyclonal) | Thermo Fisher | Cat# A-21206; RRID:AB_141708 | IF (1:500) |
| Antibody | Donkey anti-rabbit, Alexa Fluor 555 (rabbit polyclonal) | Thermo Fisher | Cat# A-31572 RRID:AB_162543 | IF (1:500) |
| Antibody | Anti-Flag M2 affinity gel | Sigma-Aldrich | A2220 | |
| Antibody | Anti-c-Myc Agarose | Sigma-Aldrich | 20168 | |
| Recombinant DNA reagent (plasmid) | pWPI-Flag-RIPK3 | This paper | N/A | Described in Materials and methods; available upon request |
| Recombinant DNA reagent (plasmid) | pWPI-Flag-mRIPK3 | This paper | N/A | Described in Materials and methods; available upon request |
| Recombinant DNA reagent (plasmid) | pCDNA3.1-Myc-CSNK1G2 | This paper | N/A | Described in Materials and methods; available upon request |
| Recombinant DNA reagent (plasmid) | pCDNA3.1-Myc-CSNK1G2(K75A) | This paper | N/A | Described in Materials and methods; available upon request |
| Recombinant DNA reagent (plasmid) | pCDNA3.1-Myc-CSNK1G2(D165N) | This paper | N/A | Described in Materials and methods; available upon request |
| Recombinant DNA reagent (plasmid) | pCDNA3.1-Myc-CSNK1G2(S211A) | This paper | N/A | Described in Materials and methods; available upon request |
| Recombinant DNA reagent (plasmid) | pCDNA3.1-Myc-CSNK1G2(T215A) | This paper | N/A | Described in Materials and methods; available upon request |
| Recombinant DNA reagent (plasmid) | pCDNA3.1-Myc-CSNK1G2(S211A/T215A) | This paper | N/A | Described in Materials and methods; available upon request |
| Recombinant DNA reagent (plasmid) | pWPI-Myc-CSNK1G2 | This paper | N/A | Described in Materials and methods; available upon request |
| Recombinant DNA reagent (plasmid) | pWPI-Myc-CSNK1G2(K75A) | This paper | N/A | Described in Materials and methods; available upon request |
| Recombinant DNA reagent (plasmid) | pWPI-Myc-CSNK1G2(S211A/T215A) | This paper | N/A | Described in Materials and methods; available upon request |
| Recombinant DNA reagent (plasmid) | pWPI-Myc-mCSNK1G2 | This paper | N/A | Described in Materials and methods; available upon request |
| Recombinant DNA reagent (plasmid) | pWPI-Myc-mCSNK1G2(K75A) | This paper | N/A | Described in Materials and methods; available upon request |
| Peptide, recombinant protein | Myc peptide | Sigma-Aldrich | 20170 | 1 mg/ml |
| Peptide, recombinant protein | 3xFlag peptide | ChinaPeptides | DYKDHDGDYKDHDIDYKDDDDK | 1 mg/ml |
| Software, algorithm | ImageJ | NIH | N/A | |
| Software, algorithm | GraphPad | Graphpad Software | N/A | |
| Software, algorithm | Prism | Graphpad Software | N/A | |
| Software, algorithm | Nikon A1-R | Nikon | https://www.nikoninstruments.com/Products/Confocal-Microscopes/A1R-HD |





