Small molecule cognitive enhancer reverses age-related memory decline in mice
Figures
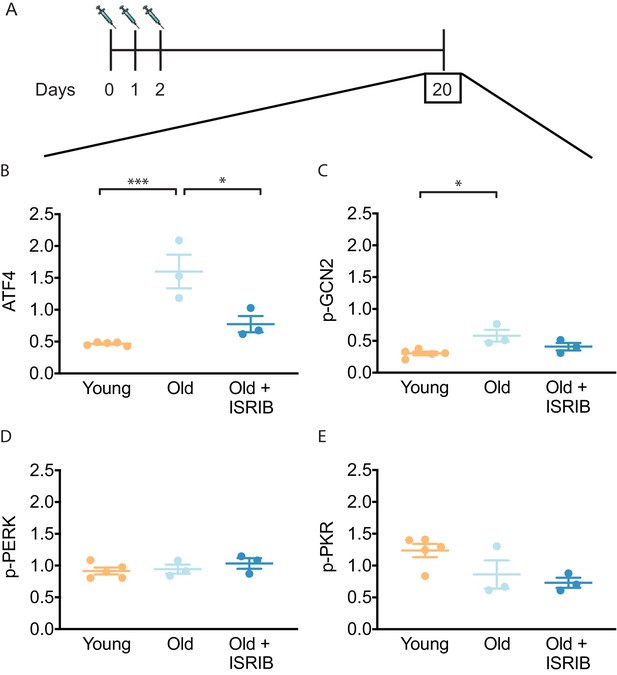
ISRIB resets the ISR in the brain of old mice.
(A) Experimental dosing scheme: ISRIB treatment denoted by syringes (three injections). (B) ISRIB treatment reduced ATF4 protein levels chronically 18 days after ISRIB treatment was complete. One-way ANOVA (F = 18.8, p<0.001); with Tukey post-hoc analysis. (C) Modest age-induced increases in p-GCN2 when comparing young and old male mice. One-way ANOVA (F = 6.6, p<0.05); with Tukey post-hoc analysis. (D, E) Age and ISRIB administration did not impact p-PERK or p-PKR protein levels. Brain lysates of specific protein levels listed normalized to actin. Young n = 5, Old = 3, Old + ISRIB = 3. Individual animal values represented by dots; lines depict group mean ± SEM. *p<0.05; ***p<0.001.
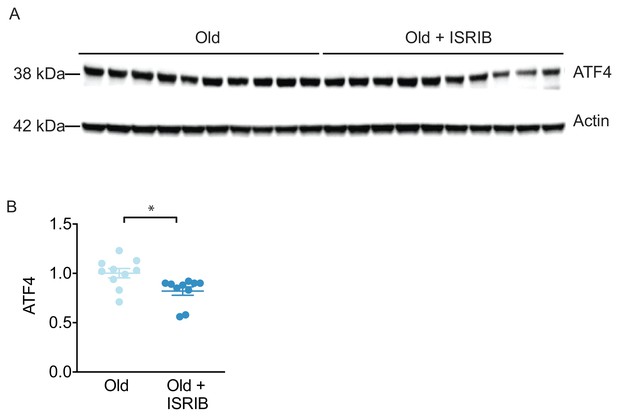
ISRIB downregulates ATF4 during administration.
The impact of ISRIB on known ISR activation pathways was investigated by western blot analysis of brain lysates after 3 ISRIB injections. (A) Raw western blot data. Each lane represents an individual animal brain extract. (B) ISRIB treatment reduced ATF4 protein levels during drug administration. Old males (Chou et al., 2018) and females (Anderson et al., 1998): Old n = 10; Old + ISRIB n = 10. Student’s t-test. *p<0.05. Individual animal values represented by dots; lines depict group mean ± SEM.
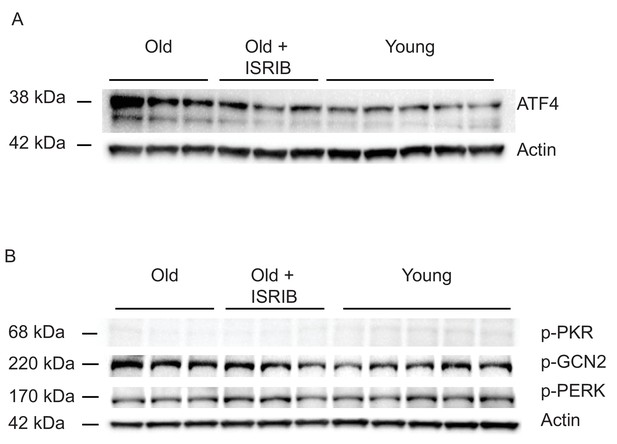
ISRIB down-regulates the ISR in the brain of old mice.
The impact of ISRIB on known ISR kinases and activation pathways was investigated by western blot analysis of brain lysates (raw western blot data) at day 20. (A) ATF4 (B) p-GCN2, p-PKR, p-PERK. Each lane represents an individual animal brain extract.
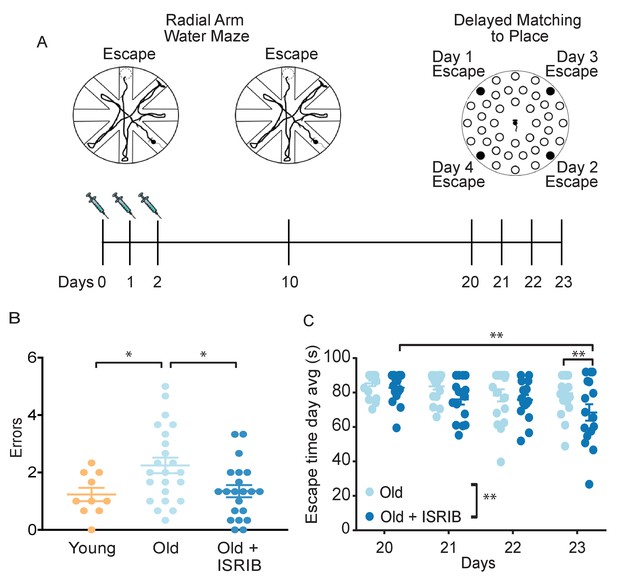
Inhibition of the ISR reverses age-induced decline in spatial, working, and episodic memory.
(A) Experimental Design: Old (~19 months) animals underwent behavioral analysis in a radial arm water maze (RAWM) and a delayed matching to place paradigm (DMP). ISRIB or vehicle administration (2.5 mg/kg intraperitoneal) occurred daily during the learning phase of RAWM denoted by syringes (days 0–2). (B) ISRIB treatment improved memory 1 week after administration in male rodents. One-way ANOVA (F = 4.8, p<0.05); with Tukey post-hoc analysis. Young n = 10; Old n = 23; Old + ISRIB n = 21. (C) Age-induced deficits in working and episodic learning and memory restored weeks after ISRIB administration. Animals performed the DMP from day 20 to day 23. Average of all trials per group for each day. Days 20, 21 = 4 trials/day. Days 22,23 = 3 trials/day. Two-way repeated measures ANOVA reveals a significant difference between groups p<0.01 (denoted in figure legend) and time effect p<0.01. *p<0.05, **p<0.01. Old n = 18; Old + ISRIB n = 16. Individual animal scores represented by dots; lines depict group mean ± SEM.
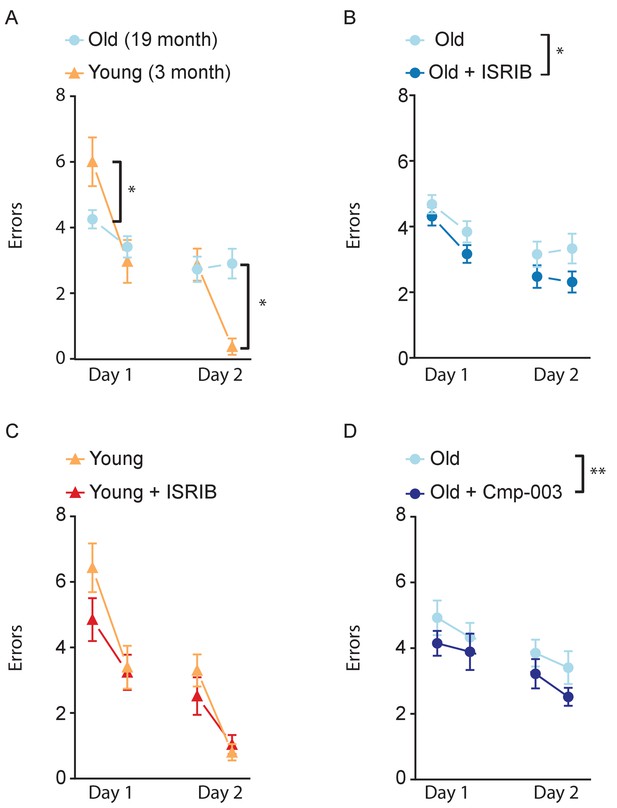
ISR inhibitors relieve age-induced deficits in spatial learning.
RAWM was used to measure age-induced deficits in spatial learning. Animals ran two blocks (three trials/block) on each learning day. (A) Old animals performed significantly worse that young animals. Two-way repeated measures ANOVA revealed a significant interaction (p<0.001). Bonferroni post-hoc to determine differences at various blocks. Old males n = 19, Young males n = 10. *p<0.05. (B) ISRIB or vehicle administration (2.5 mg/kg intraperitoneal) occurred days 0–2. Compared with the old group, ISRIB treated animals made significantly fewer errors over the course of learning. Two-way repeated measures ANOVA reveals a significant difference between groups p<0.05. Old males n = 19; Old + ISRIB males n = 15. (C) No differences were measured between young +/- ISRIB administration. Two-way repeated measures ANOVA revealed no significant differences. Young males n = 10; Young males + ISRIB n = 10 (D) Cmp-003 (5 mg/kg intraperitoneal) administration occurred days 0–2. Old mice that received Cmp-003 performed significantly better than old mice that received vehicle. Two-way repeated measures ANOVA revealed a significant group (p<0.01) and time effect (p<0.05). Old males n = 9, Old males + Cmp-003 n = 9. **p<0.01. Data are means ± SEM.
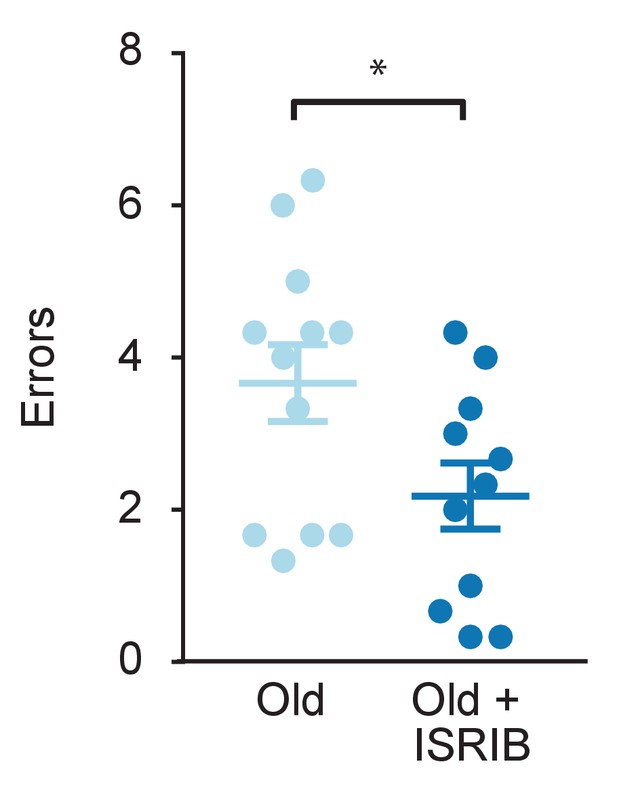
ISRIB reduces age-induced memory deficits in female mice.
RAWM was used to measure age-induced deficits in learning and memory. ISRIB treatment improved memory 1 week after administration in female rodents. Student t-test. Old female n = 12; Old female + ISRIB n = 11. *p<0.05. Individual animal scores represented by dots, lines depict group mean and SEM.
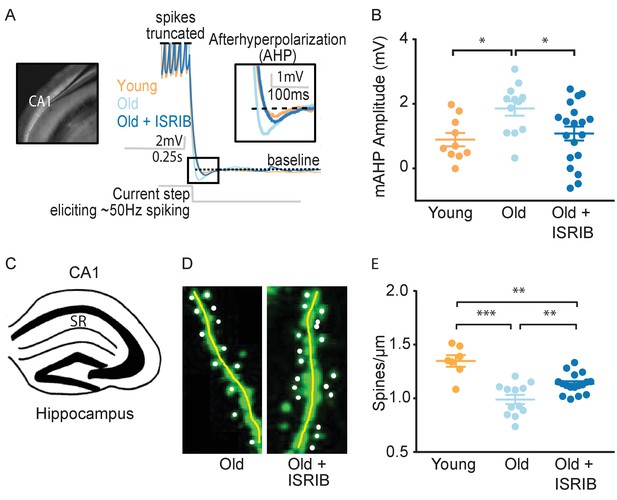
ISRIB treatment alleviates age-associated changes in CA1 pyramidal neuron function and structure.
(A) Left: Image of pipette patched onto CA1 neuron in sagittal slice of hippocampus. Right: Representative traces from hippocampal CA1 pyramidal neurons from old animals treated with either vehicle (light blue) or ISRIB (dark blue) or young animals treated with vehicle (orange) showing the response to a current injection eliciting ~50 Hz spiking activity. Spikes are truncated (dashed line), and the AHP is visualized immediately following cessation of current injection (yellow square) and quantified as the change in voltage from baseline (dotted line). (B) Age-induced increases in AHP were measured when comparing young and old animals. ISRIB treatment reversed increased AHP to levels indistinguishable from young animals. Animals were injected with ISRIB (2.5 mg/kg) or vehicle intraperitoneal 1 day prior to recordings. One-way ANOVA (F = 4.461, p<0.05); with Tukey post-hoc analysis. *p<0.05. Each neuron is represented with a symbol; lines indicate the mean ± SEM (Neurons: Young males n = 10 (5 animals); Old males n = 12 (5 animals), Old + ISRIB males n = 19 (7 animals)) with 1–5 neurons recorded per animal. (C–E) Spine density was quantified in the CA1 region of the dorsal hippocampus from young and old Thy1-YFP-H mice. (C) Diagram of hippocampal region analyzed. SR = stratum radiatum. (D) Representative images from Old and Old + ISRIB mice. (E) A decrease in dendritic spine density was measured when comparing old mice to young mice. ISRIB treatment significantly increased spine density levels of old mice when compared to vehicle-treated old mice. 63x magnification with a water immersion objective. Young males n = 7 slides (two animals); Old males + Vehicle n = 12 slides (three mice); Old males + ISRIB n = 17 slides (four mice). Individual slide scores (relative to old mice) represented in dots, lines depict group mean ± SEM. One-way ANOVA (F = 18.57, p<0.001) with Tukey post-hoc analysis. **p<0.01; ***p<0.001.
-
Figure 3—source data 1
List of electrophysiology reagents.
Column 1 – reagent name; Column 2 – Company of purchase; Column 3 – Catalog number.
- https://cdn.elifesciences.org/articles/62048/elife-62048-fig3-data1-v2.docx
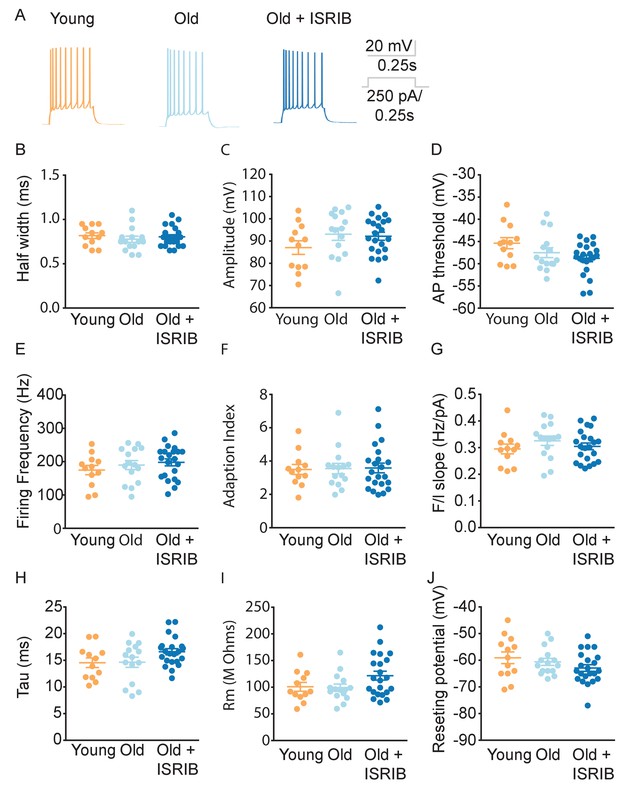
Age and ISRIB treatment do not modify other passive or active intrinsic membrane properties in CA1 pyramidal neurons.
(A) Representative traces from CA1 pyramidal neurons showing the membrane potential response to a 250 pA current injection in neurons from old animals treated with either vehicle (light blue) or ISRIB (dark blue) or young animals treated with vehicle (orange). Quantification of the action potential (AP) including the half width (B), amplitude (C), and threshold (D) did not show significant differences between CA1 pyramidal recordings from old, old + ISRIB-treated, or young mice. Likewise, evaluation of the maximum firing frequency (E) or how the frequency of spiking changes over time, quantified by the adaptation index (F) or with current injection, quantified by the slope of the relationship of firing frequency versus amplitude of current injection (F/I slope) (G) was also not significantly different between groups. Finally, passive membrane properties including the membrane time constant (tau) (H), membrane resistance (Rm) (I), and the resting membrane potential (J) were not significantly altered by age or ISRIB treatment. Each neuron is represented with a symbol; solid lines indicate the mean ± SEM. (One-way ANOVA for all comparisons; Neurons: and Young males n = 12 (5 animals); Old males n = 15 (5 animals), Old + ISRIB males n = 22 (7 animals)) with 1–5 neurons recorded per animal.
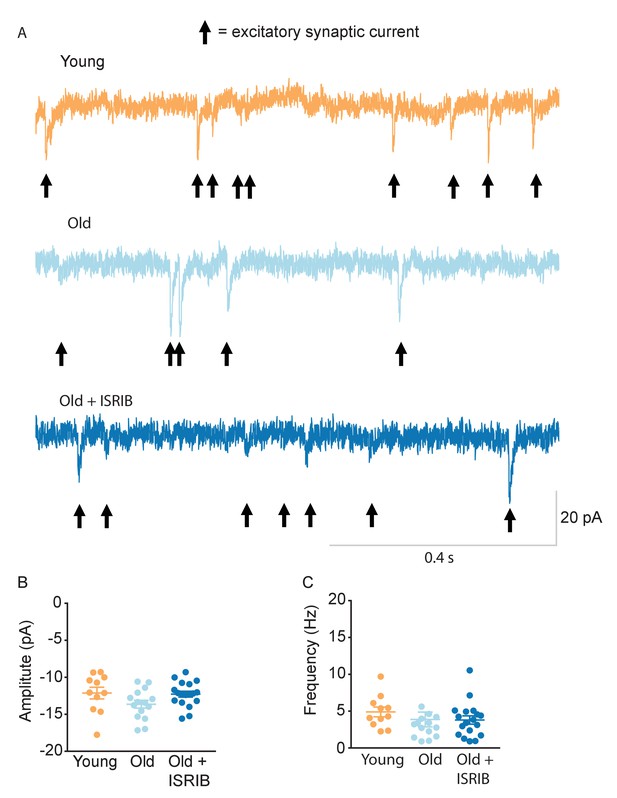
Age and ISRIB treatment do not affect spontaneous excitatory post-synaptic currents (sEPSC) in CA1 pyramidal neurons.
(A) Representative whole cell voltage-clamp recordings showing sEPSCs from CA1 pyramidal neurons from old animals treated with either vehicle (light blue) or ISRIB (dark blue) or young animals treated with vehicle (orange). Arrows denote synaptic currents. (B) The sEPSC amplitude was not significantly different between groups (one-way ANOVA). (C) The sEPSC frequency was unchanged after ISRIB treatment or compared to young mice (Kruskal-Wallis test). The median amplitude or frequency for each neuron is represented with a symbol; solid lines indicate the mean ± SEM. (Neurons: Young males n = 11 (5 animals); Old males n = 15 (5 animals), Old + ISRIB males n = 18 (7 animals) with 1–5 neurons recorded per animal.).
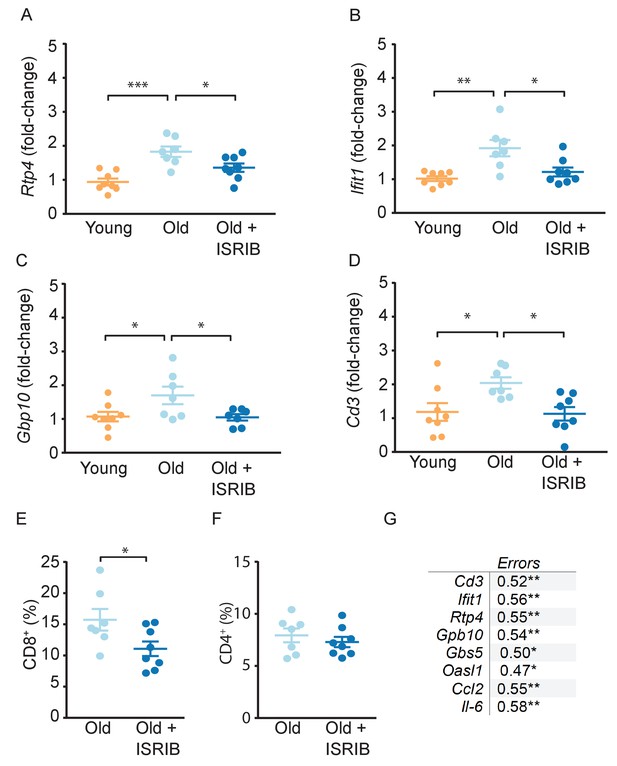
Age-induced inflammatory tone is reduced following ISRIB treatment.
Inflammatory genes were investigated in the hippocampus of young and old mice by qPCR analysis. (A-C) ISRIB administration reversed age-induced increases in Rtp4, Ifit1, and Gbp10. (A) Rtp4, One-way ANOVA (F = 12.23, p<0.001) with a Tukey-post analysis. Young males n = 8; Old males n = 7; Old + ISRIB males n = 8. (B) Ifit1, One-way ANOVA (F = 8.8; p<0.01) with a Tukey-post analysis. Young males n = 8; Old males n = 7; Old + ISRIB males n = 8. (C) Gbp10, One-way ANOVA (F = F4.2, p<0.05) with a Tukey-post analysis. Young males n = 8; Old males n = 7; Old + ISRIB males n = 7. (D) Cd3 gene-expression (a marker for T cells) changes in the hippocampus of young and old animals were measured by qPCR analysis. Cd3 was significantly increased with age. ISRIB administration returned Cd3 expression levels to those comparable to young animals. One-way ANOVA (F = 5.2; p<0.05). Tukey-post hoc analysis. Young males n = 8; Old males n = 7; Old + ISRIB males n = 8. (E, F) Peripheral T cell levels were measured by flow cytometric analysis of whole blood. (E) ISRIB treatment reduced CD8+ T cell percentages (of CD45+ cells) in the peripheral blood. Student t-test. Old males n = 7; Old + ISRIB males n = 8. (F) CD4+ T cell percentages (of CD45+ cells) were not impacted. Individual animal scores represented by dots; lines depict group mean ± SEM. (G) A significant positive correlation was measured between cognitive performance on day 2 of the RAWM (errors) and multiple inflammatory markers (Cd3, Ifit1, Rtp4, Gbp10, Gbp5, Oasl1, Ccl2, Il-6). Linear regression was measured by Pearson R correlation, R value denoted with significance. *p<0.05; **p<0.01; ***p<0.001.
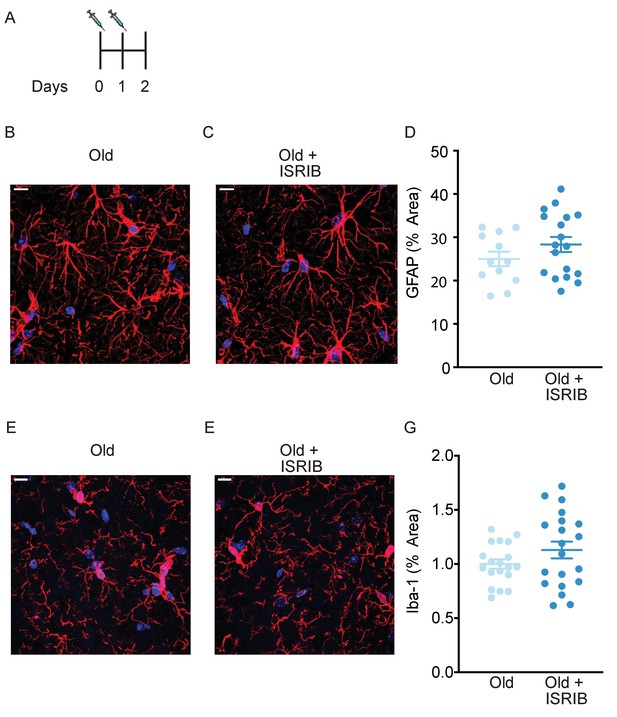
ISRIB administration does not impact glial cell activation.
(A) ISRIB administration scheme. (B–G) Glial cell was quantified in the stratum radiatum of the CA1 region of the dorsal hippocampus from old Thy1-YFP-H mice. GFAP was used to measure astrocyte activity. Representative images for GFAP staining of (B) old and (C) old + ISRIB mouse. (D) No differences in GFAP percent area were measured when comparing old and old + ISRIB animals. Iba-1 was used to measure microglia activity. Representative images for Iba-1 staining of (E) old and (F) old + ISRIB mouse. (G) No differences in Iba-1 percent area were measured when comparing old and old + ISRIB animals. 63x magnification with a water immersion objective. Old males n = 12–19 slides (three mice); Old males + ISRIB n = 18–20 slides (four mice). Individual slide scores (relative to old mice) represented in dots, lines depict group mean ± SEM.
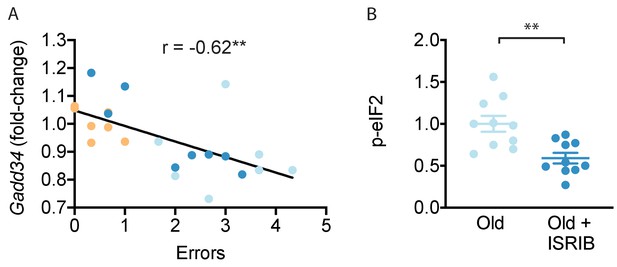
ISRIB treatment resets age-related ISR activation.
(A) A significant negative correlation was measured between cognitive performance on day 2 of the RAWM (errors) and Ppp1r15a mRNA expression. Linear regression was measured by Pearson R correlation, R value denoted with significance. (B) ISRIB treatment reduced p-eIF2α protein levels. Brain lysates of p-eIF2α protein levels normalized to actin. Old males n = 10; ISRIB males n = 10. Student’s t-test. **p<0.01. Data are means ± SEM.
Tables
Impact of age and ISRIB on mRNA expression in the hippocampus.
Inflammatory, ISR mediators and neuronal health targets were investigated by qPCR analysis of hippocampal lysates after two ISRIB injections. Columns: (i) mRNA targets (ii) Young group mean ± SEM (iii) Old group mean ± SEM (iv) Old + ISRIB group mean ± SEM (v) ANOVA F value (vi) Significant denotation between groups (vii) n/group.
| TARGET | Young | Old | Old + ISRIB | ANOVA | Btw Groups | n: Yg/Old/Old + ISRIB |
|---|---|---|---|---|---|---|
| Ccl2 | 1.0 ± 0.1 | 2.3 ± 0.4 | 1.6 ± 0.1 | F = 5.6* | Yg v Old** | 8/7/7 |
| Cd11b | 1.0 ± 0.0 | 1.0 ± 0.0 | 1.2 ± 0.0 | F = 6.6** | Yg v Old + ISRIB** Old v Old + ISRIB* | 8/7/8 |
| Il1-β | 1.1 ± 0.1 | 1.4 ± 0.1 | 1.7 ± 0.1 | F = 3.6* | Yg v Old + ISRIB* | 8/7/8 |
| Tnf α | 1.0 ± 0.1 | 1.6 ± 0.3 | 2.1 ± 0.4 | F = 2.7 | 7/7/8 | |
| Il-6 | 1.1 ± 0.2 | 1.8 ± 0.1 | 1.8 ± 0.2 | F = 4.1* | Yg v Old* | 8/7/8 |
| Il-10 | 1.0 ± 0.1 | 1.4 ± 0.2 | 1.5 ± 0.2 | F = 1.1 | 7/7/7 | |
| Irf7 | 1.0 ± 0.1 | 1.4 ± 0.1 | 1.2 ± 0.1 | F = 1.5 | 8/7/8 | |
| Ifitm3 | 1.0 ± 0.0 | 1.2 ± 0.1 | 1.0 ± 0.0 | F = 0.8 | 8/7/8 | |
| Isg15 | 1.0 ± 0.0 | 1.3 ± 0.1 | 1.2 ± 0.0 | F = 2.0 | 8/7/7 | |
| Ifi204 | 1.1 ± 0.2 | 1.4 ± 0.2 | 1.5 ± 0.1 | F = 1.2 | 6/7/7 | |
| Gbp5 | 1.0 ± 0.0 | 1.2 ± 0.1 | 1.1 ± 0.0 | F = 2.6 | 8/7/8 | |
| Oasl1 | 1.0 ± 0.0 | 1.8 ± 0.3 | 0.9 ± 0.1 | F = 4.9* | Yg v Old*; Old v Old + ISRIB* | 8/7/8 |
| Ophn1 | 1.0 ± 0.0 | 0.9 ± 0.0 | 1.1 ± 0.0 | F = 1.8 | 8/7/8 | |
| Bdnf | 1.0 ± 0.0 | 0.9 ± 0.0 | 1.0 ± 0.0 | F = 2.7 | 8/7/8 | |
| Gadd34 | 1.0 ± 0.0 | 0.8 ± 0.0 | 0.9 ± 0.0 | F = 1.8 | 7/7/8 | |
| Pkr | 1.0 ± 0.0 | 1.0 ± 0.0 | 1.1 ± 0.0 | F = 1.0 | 8/7/8 |






