Accelerating with FlyBrainLab the discovery of the functional logic of the Drosophila brain in the connectomic and synaptomic era
Figures
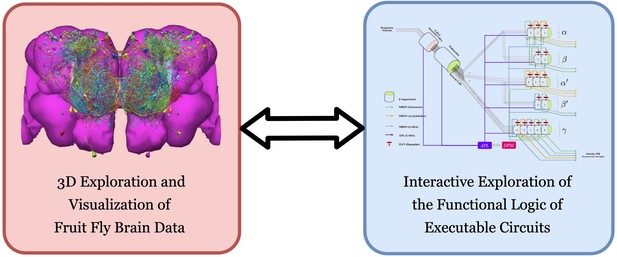
FlyBrainLab provides, within a single working environment, (left) 3D exploration and visualization of fruit fly brain data, and (right) creation of executable circuit diagrams from the explored and visualized circuit on the left followed by an interactive exploration of the functional logic of executable circuits.
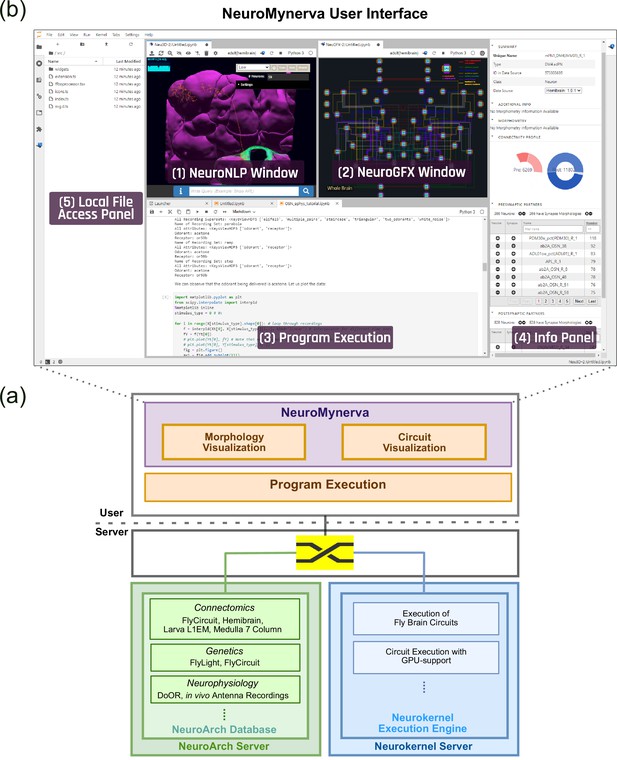
The software architecture and the user interface of FlyBrainLab.
(a) The main components of the architecture of FlyBrainLab: (top) NeuroMynerva user-side frontend, (bottom left) NeuroArch Database for storage of fruit fly brain data and executable circuits, (bottom right) Neurokernel Execution Engine for execution of fruit fly brain circuits on GPUs (see also Appendix 1—figure 1 for a schematic diagram of the FlyBrainLab software architecture). (b) NeuroMynerva User Interface. The UI typically consists five blocks, including a (1) NeuroNLP 3D Visualization Window with a search bar for NLP queries, providing capabilities for displaying and interacting with fly brain data such as the morphology of neurons and position of synapses. (2) NeuroGFX Executable Circuits Window, for exploring executable neural circuits with interactive circuit diagrams. (3) Program Execution Window with a built-in Jupyter notebook, executing any Python code including calls to the FlyBrainLab Client (see also Appendix 1.2), for direct access to database queries, visualization, and circuit execution, (4) Info Panel displaying details of highlighted neurons including the origin of data, genetic information, morphometric statistics and synaptic partners, etc. (5) Local File Access Panel with a built-in Jupyter file browser for accessing local files.

Building fly brain circuits with English queries.
(a1) Lateral feedback pathways in the visual motion detection circuit. (green) a T4a neuron, (gray) neurons presynaptic to the T4a neuron, (cyan) glutamatergic and GABAergic Dm neurons that are presynaptic to the neurons in gray, (yellow) Pm neurons that are presynaptic to the neurons in gray. (a2) Connectivity matrix of the pathways in (a1). (b1) Pathways between MBONs and neurons innervating FB layer 3. (yellow) MBONs that are presynaptic to neurons that have outputs in FB layer 3. (green) Neurons that have outputs in FB layer 3 that are postsynaptic to the MBONs in yellow. (red) MBONs postsynaptic to neurons in green. (b2) Connectivity matrix of the pathways in (b1). (c1) The pathways of the g compartment of the larva fruit fly. (cyan) g compartment MBONs, (yellow) KCs presynaptic to the g compartment MBONs, (green) a DAN presynaptic to the g compartment MBONs, (white) an OAN presynaptic to the g compartment MBONs, (c2) Connectivity matrix of the pathways in (c1). (d1) Pathways between LPTCs and a potential translational motion-sensitive neuron GLN. (yellow) LPTCs, (cyan) GLNs, (red) neurons that form the path between LPTCs to GLNs. (d2) Connectivity matrix of the pathways in (d1). color bar in a2, b2, c2, and d2: , where is the number of synapses between 2 neurons. (a1)–(d1) are screenshots downloaded from the NeuroNLP Window. The sequence of queries that generates these visualizations is listed in Materials and methods Use Case 1.
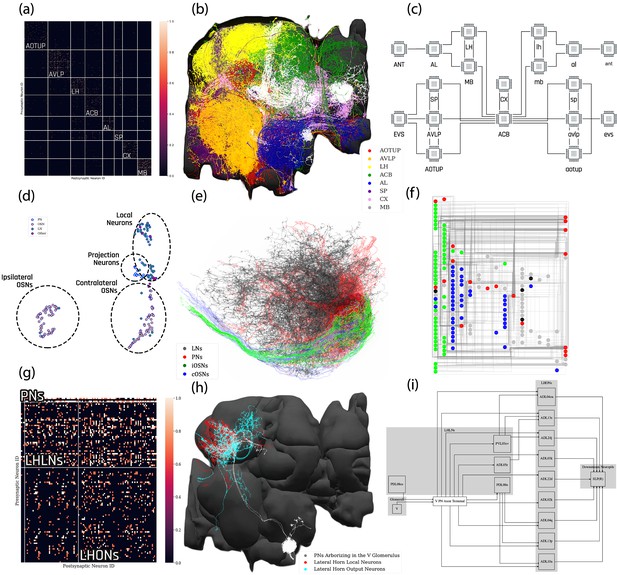
Exploratory analysis of the fly brain circuits.
(a) Louvain algorithm applied to all neurons in the Hemibrain dataset showing eight groups of densely connected neurons. Color indicates the value of , where is the number of synapses; values larger than one are shown in the same color as value 1. AOTUP: anterior optic tubercle with additional neurons of the posterior brain, AVLP: anterior ventrolateral protocerebrum, LH: lateral horn, ACB: neurons in the anterior central brain, AL: antennal lobe, SP: superior protocerebrum, CX: central complex, MB: mushroom body. Labels were added after visually inspecting the neurons in each group of neurons in (b). (b) A subset of neurons pseudo-colored according to the group they belong to in (a). (c) A brain-level circuit diagram created by hand according to the grouping of neurons and the inter-group edge information obtained in (a). Visual and olfactory inputs from, respectively, the early visual system (EVS) and antenna (ANT) were added. Groups in the left hemisphere were added by symmetry. (d) Adjacency Spectral Embedding algorithm applied to the VA1v connectome dataset using the NeuroGraph library. The color of each circle indicates the cell-type labeling from the original dataset. Groups of neurons labeled by dashed circles are based on validated cell types. (e) Visualization of neurons analyzed in (d). Neuron colors were assigned according to the groups in (d). (f) A circuit diagram of the VA1v circuit analyzed in (d) automatically generated by the NeuroGraph Library. (g) Connectivity matrix of the lateral horn neurons downstream the V glomerulus projection neurons of the antennal lobe. Colorbar configured in the same way as in (a). (h) Morphology of the neurons in (g). (white) PNs arborizing in the V glomerulus, (red) LHLNs, (cyan) LHONs. (i) A circuit diagram automatically generated by the circuit visualization utilities of NeuroGraph starting with the circuit in (g) and (h), and the superior lateral protocerebrum (SLP), the primary neuropil that the LHONs project to.
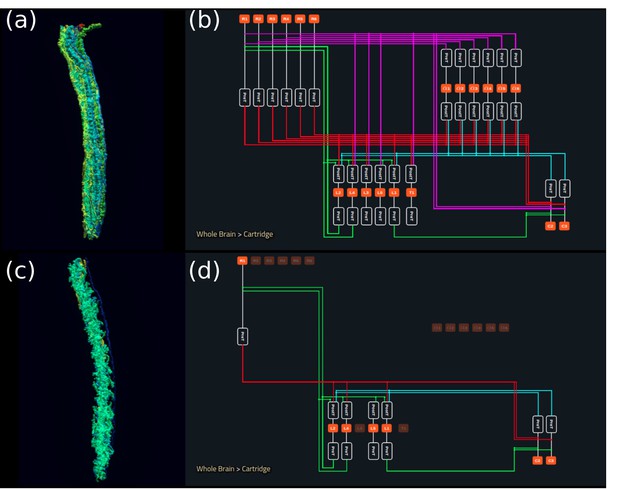
Interactive exploration of executable circuit models.
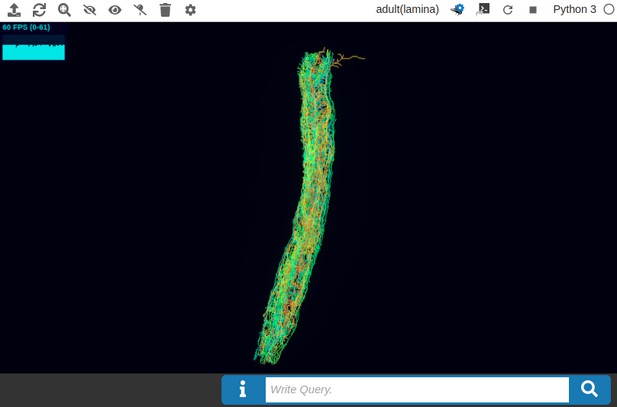
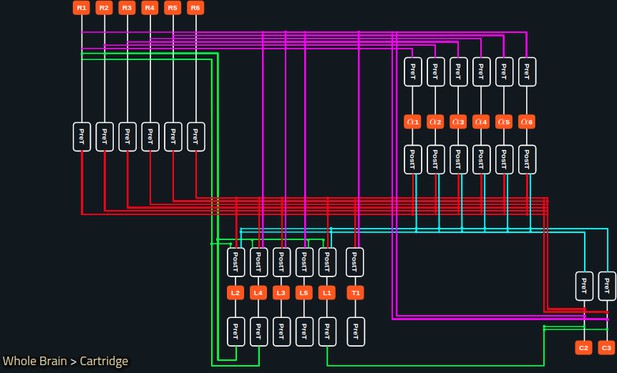
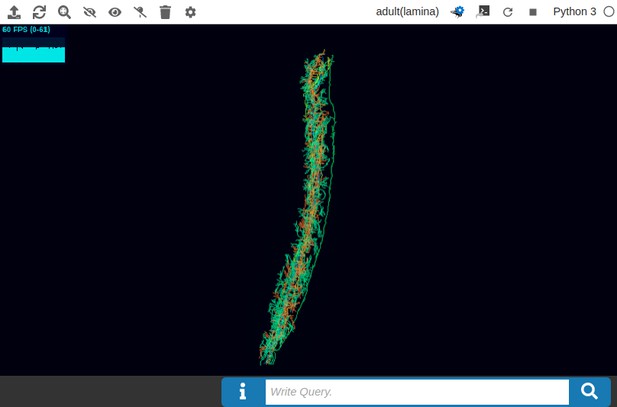
(a) The pathways of a Lamina cartridge visualized in the NeuroNLP window. (b) A circuit diagram of the cartridge in (a) displayed in NeuroGFX window. (c) The cartridge pathways modified interactively using the circuit diagram in (b) that results in the circuit diagram in (d). (d) The circuit diagram modeling the chosen pathways in (c).
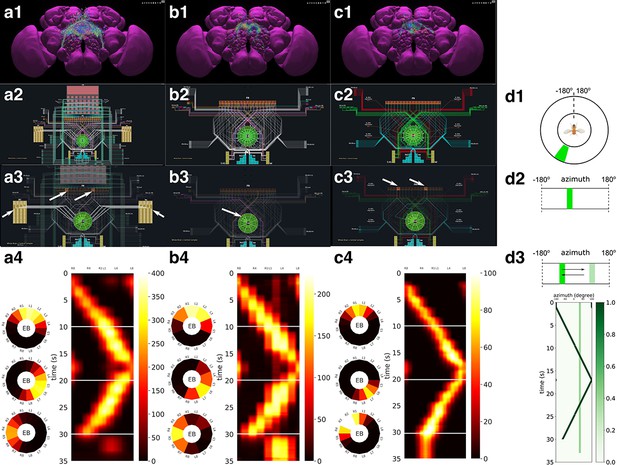
Analysis, evaluation and comparison of three models of CX published in the literature.
(a1–a4) Model A (Givon et al., 2017), (b1–b4) Model B (Kakaria and de Bivort, 2017), (c1–c4) Model C (Su et al., 2017). (a1, b1, c1) Morphology of the neurons visualized in the NeuroNLP window (see Figure 2b). Displayed number of neurons in: (a1) 366, (a2) 87, (a3) 54. (a2, b2, c2) Neuronal circuits in the NeuroNLP window depicted in the NeuroGFX window (see Figure 2b) as abstract interactive circuit diagrams. The naming of the ring neurons in (c2) follows Su et al., 2017. Number of neurons in the diagram: (b1) 348, (b2) 60, (b3) 56. As the FlyCircuit dataset contains duplicates, some neurons in the diagrams may correspond to multiple neurons in the dataset and some do not have correspondences due to the lack of morphology data. (a3, b3, c3) When a single vertical bar is presented in the visual field (d1/d2), different sets of neurons/subregions (highlighted) in each of the models, respectively, receive either current injections or external spike inputs. (a4, b4, c4) The mean firing rates of the EB-LAL-PB neurons innervating each of the EB wedges of the three models (see Materials and methods, Use Case 4), in response to the stimulus shown in (d3). Insets show the rates at 10, 20, and 30 s, respectively, overlaid onto the EB ring. (d1) A schematic of the visual field surrounding the fly. (d2) The visual field flattened. (d3) Input stimulus consisting of a bar moving back and forth across the screen, and a second fixed bar at and with lower brightness.
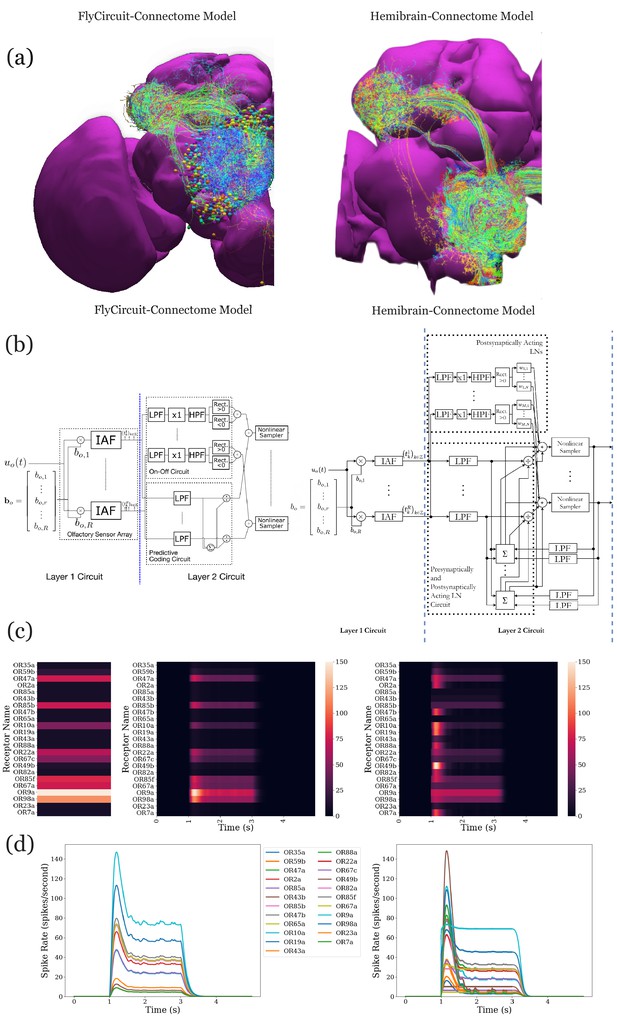
Analysis, evaluation, and comparison between two models of the antenna and antennal lobe circuit of the adult fly based on the FlyCircuit (left) dataset (Chiang et al., 2011) and an exploratory model based on the Hemibrain (right) dataset (Scheffer et al., 2020).
(a) Morphology of olfactory sensory neurons, local neurons, and projection neurons in the antennal lobe for the two datasets. The axons of the projection neurons and their projections to the mushroom body and lateral horn are also visible. (b) Circuit diagrams depicting the antenna and antennal lobe circuit motifs derived from the two datasets. (c) Response of the antenna/antennal lobe circuit to a constant ammonium hydroxide step input applied between 1 s and 3 s of a 5 s simulation; (left) the interaction between the odorant and 23 olfactory receptors is captured as the vector of affinity values; (middle and right) a heatmap of the uniglomerular PN PSTH values (spikes/second) grouped by glomerulus for the two circuit models. (d) The PN response transients of the two circuit models for uniform noise input with a minimum of 0ppm and a maximum of 100 ppm preprocessed with a 30 Hz low-pass filter (Kim et al., 2011) and delivered between 1 s and 3 s.
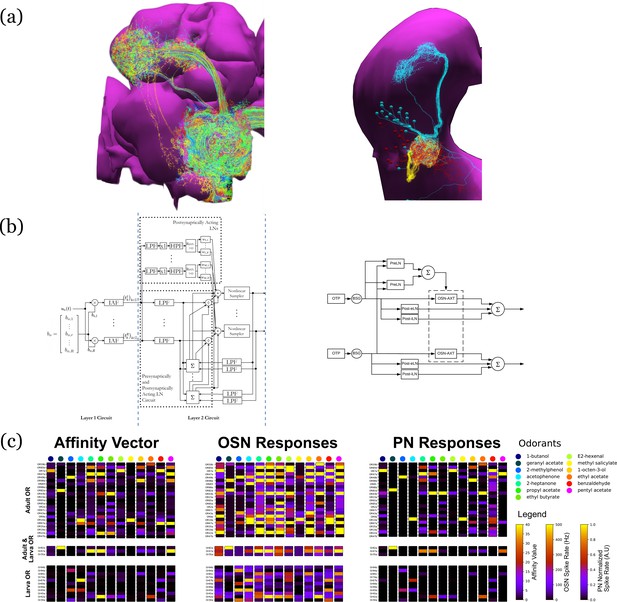
Evaluation and Comparison of two Drosophila Early Olfactory System (EOS) models describing adult (left, developed based on Hemibrain dataset) and larval (right, developed based on LarvaEM dataset) circuits.
(a) Morphology of Olfactory Sensory Neurons (OSNs) in the Antenna (ANT), Local Neurons (LNs) in the Antennal Lobe (AL) and Projection Neurons in the AL. (b) Circuit diagrams depicting the Antenna and Antennal Lobe circuit motifs. (c) (left) Interaction between 13 odorants and 37 odorant receptors (ORs) characterized by affinity values. The ORs expressed only in the adult fruit flies are grouped in the top panel; the ones that are expressed in both the adult and the larva are grouped in the middle panel; and those expressed only in the larva are shown in the bottom panel. Steady-state outputs of the EOS models to a step concentration waveform of 100 ppm are used to characterize combinatorial codes of odorant identities at the OSN level (middle) and the PN level (right).
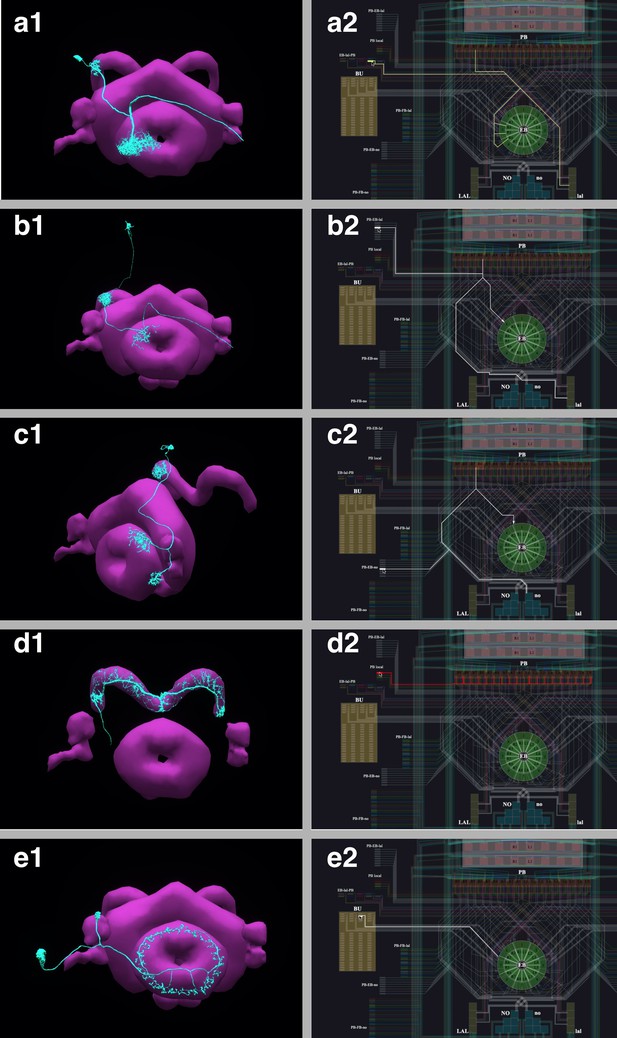
The correspondence between the morphology and the circuit diagram representation of 5 classes of neurons that determine the PB-EB interaction.
(a1, a2) EB-LAL-PB neuron and its wiring in the circuit diagram. (b1, b2) PB-EB-LAL neuron and its wiring in the circuit diagram. (c1, c2) PB-EB-NO neuron and its wiring in the circuit diagram. (d1, d2) PB local neuron and its wiring in the circuit diagram. (e1, e2) Ring neuron and its wiring in the circuit diagram.
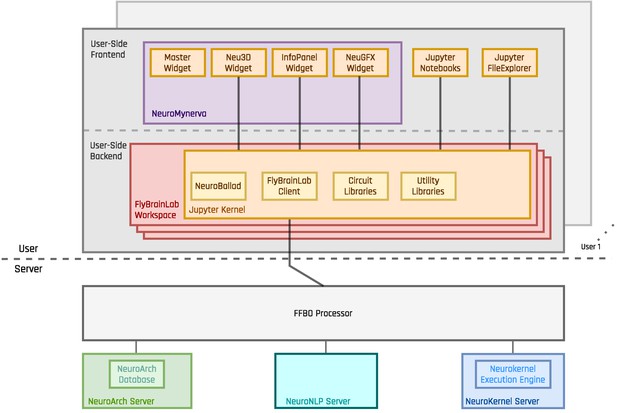
The architecture of FlyBrainLab.
The server-side architecture (Ukani et al., 2019) consists of the FFBO Processor, the NeuroNLP Server, the NeuroArch Server and the Neurokernel Server. The user-side provides the local execution environment as well as an easy-to-use GUI for multi-user access to the services provided by the server-side. The FlyBrainLab Utility Libraries and Circuit Libraries (see Sections 2 and 3 for details) can be loaded into the FlyBrainLab workspace of the user-side backend.
A screenshot of the model library in NeuroGFX for selecting the neuron model and specifying the parameters.
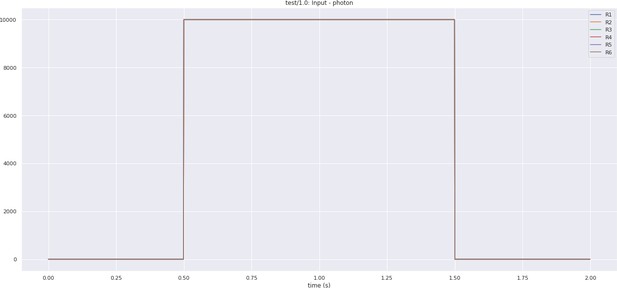
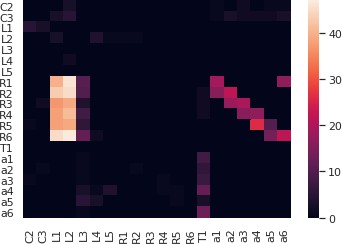
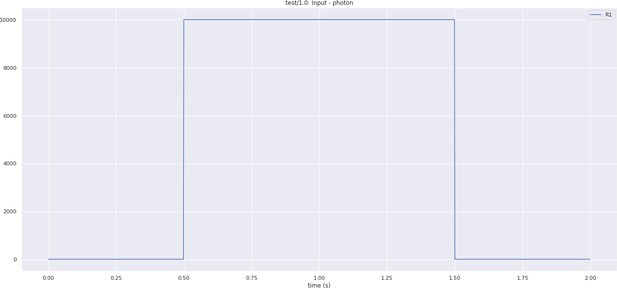
Inputs to the photoreceptors used during the execution of a full lamina cartridge circuit.
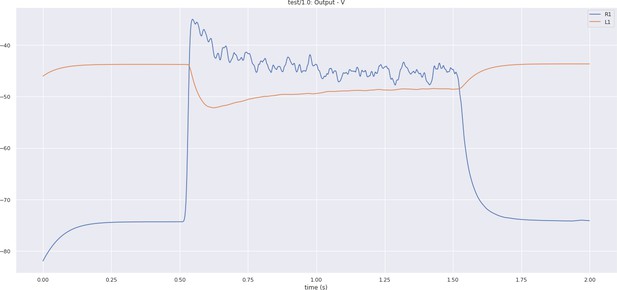
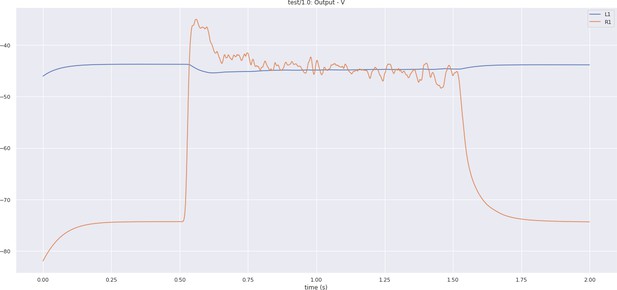
The output voltage of the R1 photoreceptor and L1 neuron of the lamina cartridge circuit.
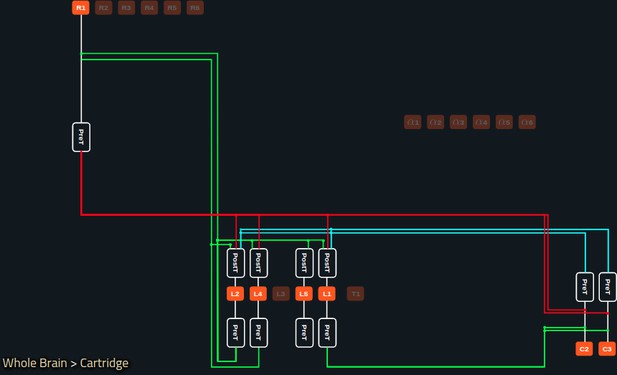
A reconfigured lamina cartridge obtained by disabling a number of neurons in the interactive circuit diagram.
Videos
Running three CX executable circuits in the FlyBrainLab.
Tables
Neurons and neuron types used for visualization and simulation in Figure 8.
| Neuropil | Neuron Type | Organism | Number (Model in Figure 8b) | Number (Visualization in Figure 8a) |
|---|---|---|---|---|
| Antenna | Olfactory Sensory Neuron | Adult | 51 receptor types (channels), 1357 total olfactory sensory neurons | 1357 |
| Larva | 14 receptor types (channels), 1 neuron expressing the same receptor type (14 neurons in total) | 21 | ||
| Antennal Lobe | Uniglomerular Projection Neuron | Adult | 1 neuron per channel, 51 total | 141 (Different number per glomerulus) |
| Larva | 1 neuron per channel (14 neurons in total) | 21 | ||
| Presynaptic Local Neuron | Adult | 97 neurons | 97 | |
| Larva | 6 pan-glomerular neurons | 5 | ||
| Postsynaptic Inhibitory Local Neuron | Adult | 77 neurons | 77 | |
| Larva | 0-1 neuron per channel (11 neurons in total) | 7 | ||
| Postsynaptic Excitatory Local Neuron | Adult | 77 (assumed to be the same as the Postsynaptic Inhibitory Local Neuron population) | 77 | |
| Larva | 0-1 neuron per channel (11 neurons in total) | 7 |
Additional files
-
Supplementary file 1
Full list of neurons used in the three CX models in Use Case 4 and their correspondence in the FlyCircuit dataset.
- https://cdn.elifesciences.org/articles/62362/elife-62362-supp1-v2.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/62362/elife-62362-transrepform-v2.pdf