SlitC-PlexinA1 mediates iterative inhibition for orderly passage of spinal commissural axons through the floor plate
Figures
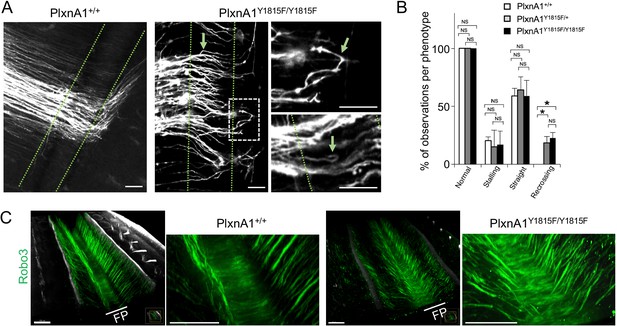
Y1815F mutation in PlxnA1 induces commissural axon recrossing and disorganized trajectories.
(A) Microphotographs illustrating commissural tracts of PlxnA1+/+ and PlxnA1Y1815F/Y1815F E12.5 open-books labeled with DiI. In PlxnA1+/+ embryos, axons extend straight toward the floor plate (FP), cross the FP, and turn rostrally at the FP exit. In PlxnA1Y1815F/Y1815F embryos, some axons turn back or are misdirected during the navigation of the FP (indicated by green arrows). The FP is delimited by dashed green lines. (B) Quantitative analysis of commissural axon phenotypes (PlxnA1+/+, N = 3 embryos; PlxnA1+/Y1815F, N = 5 embryos; PlxnA1Y1815F/Y1815F, N = 4 embryos). Data are shown as the mean ± s.e.m., Student's t-test has been applied, *: p<0.05. (C) Light sheet imaging of the spinal commissural tracts in PlxnA1+/+ and PlxnA1Y1815F/Y1815F embryos at E12.5, immunostained with anti-Robo3 antibody. Scale bar: 50 μm in (A), 150 μm in (C).
-
Figure 1—source data 1
Quantitative analysis of commissural axon phenotypes (B).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig1-data1-v2.xlsx
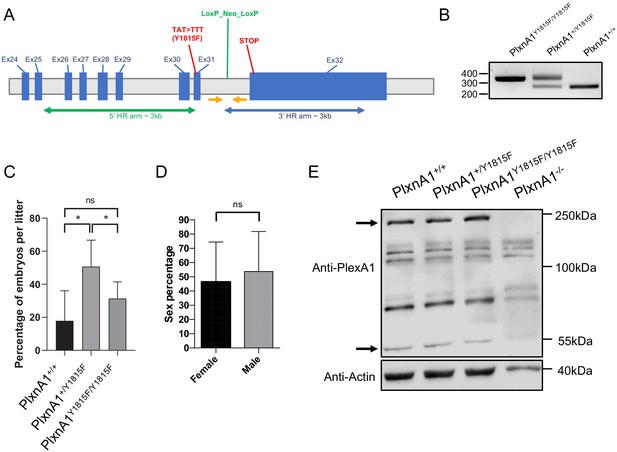
Generation of PlxnA1Y1815F mutant strain.
(A) Schematic representation of the PlxnA1Y1815F allele. The selection cassette encoding neo is inserted in the region spanning exons 25–32. The homolog arms are indicated in 5′ and 3′. TAT >TTT (Y1815F) mutation is inserted in introns 30 and 31. The genotyping primers are indicated as yellow arrows. (B) Genotyping PCR products: the genotyping primers indicated in (A) amplify a 341 bp fragment from the mutated allele (PlxnA1Y1815F/Y1815F) and a 262 bp fragment from the wild-type (PlxnA1+/+) allele. (C) Percentage of mice with each genotype coming from PlxnA1Y1815F/+× PlxnA1Y1815F/+ crossing (N = 8 litters, 46 mice total). (D) Overall percentage of female and male mice (N = 41 litters, 220 mice total). Data are shown as the mean ± s.d., Student's t-test has been applied, *: p<0.05. (E) Representative electrophoresis of spinal cord lysates prepared from E12.5 PlxnA1-/-, PlxnA1+/+, PlxnA1+/Y1815F, and PlxnA1Y1815F/Y1815F embryos, immunoblotted with anti-PlxnA1 and anti-actin antibodies. PlxnA1 is detected under two major forms, the integral form at 250 kDa and a short form at 55 kDa. Black arrows point the 250 kDa form present at higher rate and the 55 kDa form present at lower rate in the PlxnA1Y1815F condition, compared to the other genotypes.
-
Figure 1—figure supplement 1—source data 1
Percentage of mice with each genotype coming from PlxnA1Y1815F/+× PlxnA1Y1815F/+ crossing (C).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig1-figsupp1-data1-v2.xlsx
-
Figure 1—figure supplement 1—source data 2
Overall percentage of female and male mice (D).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig1-figsupp1-data2-v2.xlsx
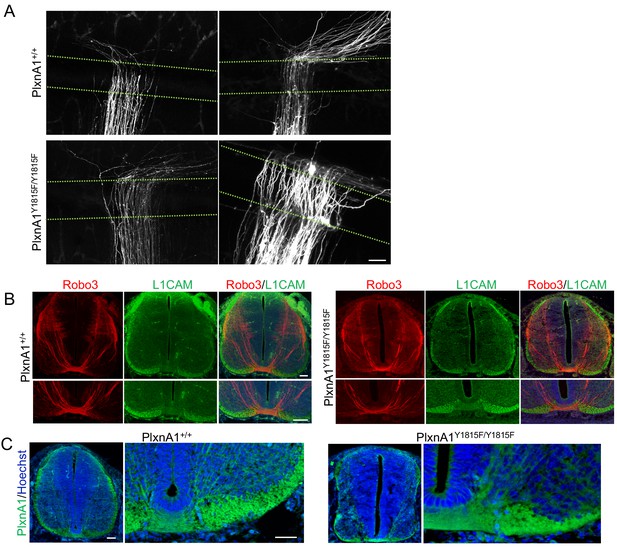
DiI traced commissural axon trajectories in PlxnA1Y1815F embryos are disorganized in the floor plate (FP) but not obviously prior to the crossing.
(A) Microphotographs illustrating commissural tracts of PlxnA1+/+ and PlxnA1Y1815F/Y1815F E12.5 open-books labeled with DiI, showing disorganized growth in the FP. The FP is delimited by dashed green lines. Scale bar: (B) Immunofluorescent labeling of Robo3 and L1CAM in transverse cryosections from E12.5 embryos in PlxnA1+/+ and PlxnA1Y1815F/Y1815F spinal cords. (C) PlxnA1 immunolabeling on E12.5 transverse sections of PlxnA1+/+ and PlxnA1Y1815F/Y1815F embryos at E12.5. Scale bar: 50 μm in (A–C).
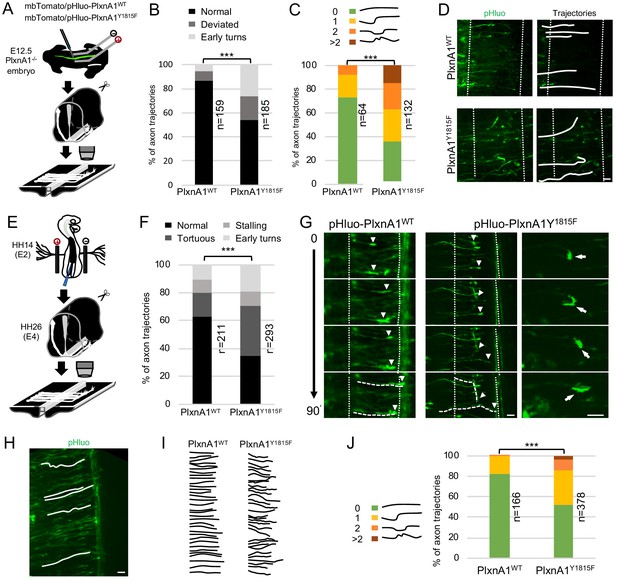
Commissural neurons expressing PlxnA1Y1815F at their surface fail to maintain straight growth during floor plate (FP) navigation.
(A) Schematic drawing of the paradigm of electroporation of pHluo-PlxnA1 receptor forms in PlxnA1-/- mouse embryonic spinal cord and open-book preparations for live imaging. (B) Histogram of axon trajectory phenotypes (PlxnA1WT, N = 8 embryos, 159 growth cones; PlxnA1Y1815F, N = 7 embryos, 185 growth cones). Chi-squared test has been applied, ***: p<0.001. (C) Histogram depicting the analysis of axon trajectories, classified by counting curvatures according to the indicated criteria (PlxnA1WT, N = 10 embryos, 64 growth cones; PlxnA1Y1815F, N = 7 embryos, 132 growth cones). (D) Microphotographs illustrating navigating commissural growth cones expressing the WT and the mutated pHluo-receptor at their surface, reported by the green pHluo fluorescent signal. (E) pHluo-receptor constructs were co-electroporated with the mbTomato in the chicken neural tube and spinal cords were mounted in open-books for time-lapse imaging. (F) Histogram quantifying axon trajectories reconstructed from time-lapse sequences of pHluo+ growth cones navigating the FP (PlxnA1WT, N = 3 embryos, 211 growth cones; PlxnA1Y1815F, N = 3 embryos, 293 growth cones). (G) Snapshots of the movies illustrating the commissural growth patterns (white triangles) and an illustration of a PlxnA1Y1815F+ growth cone turning back. The FP is delimited by dashed white lines. (H) Microphotograph illustrating traces of commissural axon trajectories. (I) Example of traces patterns from several snapshots. (J) Histogram depicting the analysis of axon trajectories, classified by counting curvatures according to the indicated criteria (PlxnA1WT, N = 3 embryos, 166 growth cones; PlxnA1Y1815F, N = 3 embryos, 378 growth cones). In (C, F, and J) Chi-squared test has been applied, ***: p<0.001. Scale bars: 10 μm in (D, G, and H).
-
Figure 2—source data 1
Histogram of axon trajectory phenotypes (B).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig2-data1-v2.xlsx
-
Figure 2—source data 2
Histogram depicting the analysis of axon trajectories, classified by counting curvatures according to the indicated criteria (C).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig2-data2-v2.xlsx
-
Figure 2—source data 3
Histogram quantifying axon trajectories reconstructed from time-lapse sequences of pHluo+ growth cones navigating the FP (F).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig2-data3-v2.xlsx
-
Figure 2—source data 4
Histogram depicting the analysis of axon trajectories, classified by counting curvatures according to the indicated criteria (J).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig2-data4-v2.xlsx
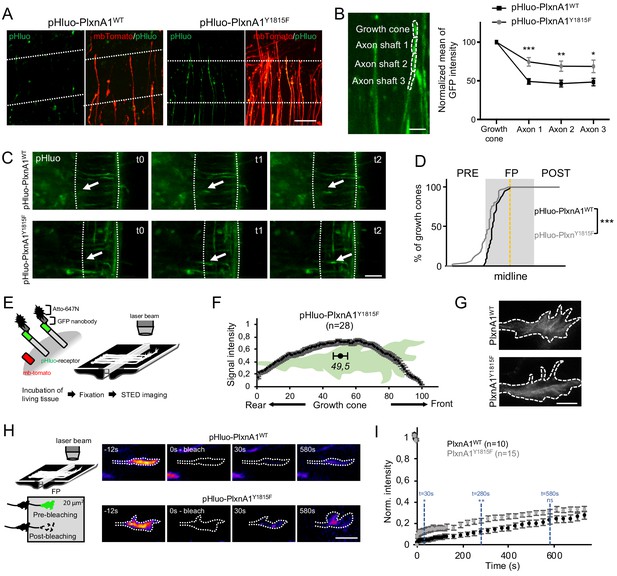
The Y1815F mutation alters the spatio-temporal pattern of cell surface PlxnA1 distribution and membrane mobility at the growth cone.
(A) Microphotographs of commissural axons navigating the floor plate (FP) in living open-books of electroporated PlxnA1-/- mice embryos. The FP is delimited by dashed white lines. (B) Method of quantification and quantification of the pHluo signal (PlxnA1WT, N = 6 embryos, 24 growth cones; PlxnA1Y1815F, N = 4 embryos, 18 growth cones). Data are shown as the mean ± s.e.m., Student's t-test has been applied, *: p<0.05, **: p<0.01, ***: p<0.001. (C) Microphotographs of live imaging movies illustrating the sorting of PlxnA1 receptor at the surface of commissural growth cones navigating the FP (delimited by dashed white lines). (D) Cumulative fractions of growth cones that sort PlxnA1 to their surface, reported by the pHluo fluorescence, in electroporated chick embryos (pHluo-PlxnA1WT, N = 2 embryos, 38 growth cones; pHluo-PlxnA1Y1815F, four embryos, 53 growth cones). KS test has been applied, ***: p<0.001. (E) Schematic representation of the paradigm of STED imaging in open-books. (F) Quantification of the center of mass of the signal (N = 28 growth cones from five embryos). Data are shown as the mean ± s.e.m. (G) STED microscopy images of pHluo-PlxnA1Y1815F cell surface distribution in commissural growth cones navigating the FP. (H) Schematic representation of the paradigm of fluorescence recovery after photobleaching (FRAP) experiments. Representative color-coded images from video-time lapse sequences illustrating photobleaching and fluorescence recovery. (I) Graphs of fluorescence recovery for pHluo-PlxnA1WT and pHluo-PlxnA1Y1815F (PlxnA1WT, N = 2 embryos, 10 growth cones; PlxnA1Y1815F, N = 2 embryos, 15 growth cones). Data are shown as the mean ± s.e.m., Student's t-test has been applied at t = 30 s, t = 280 s, and t = 580 s, ns: non-significant, *: p<0.05, **: p<0.01. Scale bars: 5 μm in (G), 10 μm in (B and H), 50 μm in (A and C).
-
Figure 3—source data 1
Quantification of the pHluo signal (B).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig3-data1-v2.xlsx
-
Figure 3—source data 2
Cumulative fractions of growth cones that sort PlxnA1 to their surface, reported by the pHluo fluorescence, in electroporated chick embryos (D).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig3-data2-v2.xlsx
-
Figure 3—source data 3
Quantification of the center of mass of the signal (F).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig3-data3-v2.xlsx
-
Figure 3—source data 4
Graphs of fluorescence recovery for pHluo-PlxnA1WT and pHluo-PlxnA1Y1815F (I).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig3-data4-v2.xlsx
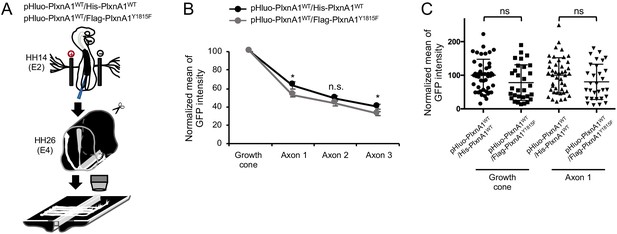
pHluo-PlxnA1WT has similar distribution pattern in commissural axons navigating the floor plate (FP) when co-expressed with non-fluorescent PlxnA1WT or PlxnA1Y1815F in chick open-books.
(A) Paradigm of electroporation. (B) Histogram of the quantification of the pHluo signal (pHluo-PlxnA1WT/his-PlxnA1WT: N = 4 embryos, 14 growth cones; pHluo-PlxnA1WT/flag-PlxnA1Y1815F, N = 4 embryos, 22 growth cones). Data are shown as the mean ± s.e.m., Student's t-test has been applied, *: p<0.05. (C) Quantification of the pHluo signal in growth cones and proximal shafts of commissural axons navigating the FP. Data are shown as the mean ± s.d., Student's t-test has been applied. ns: non-significant.
-
Figure 3—figure supplement 1—source data 1
Histogram of the quantification of the pHluo signal (B).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig3-figsupp1-data1-v2.xlsx
-
Figure 3—figure supplement 1—source data 2
Quantification of the pHluo signal in growth cones and proximal shafts of commissural axons navigating the FP (C).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig3-figsupp1-data2-v2.xlsx
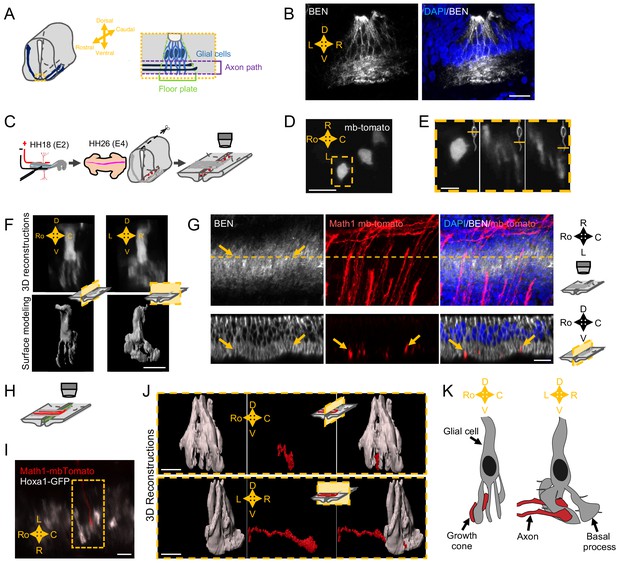
Spatial organization of the commissural axon navigation path.
(A) Schematic drawings of a spinal cord at E12.5 when commissural axons cross the floor plate (FP) (left) and close-up of the FP (right) with glial cells (light blue) and crossing axons (dark blue). (B) Immunostaining of an E4 FP transverse section with FP specific BEN antibody (left) and merged with DAPI staining (right). (C) In ovo FP electroporation procedure. Sparse electroporated mbTomato electroporated cells are visualized in red. (D) Open-book imaging of E4 chick FP with sparse mbTomato electroporation. The dashed rectangle highlights a glial cell. (E) Close-up of the single glial cell observed in (D), at three different positions along the dorso-ventral axis, as shown by the schematic representation on the upper right corner of each image. (F) 3D reconstruction (upper) and surface modeling (lower) of a single FP cell seen in a sagittal (left) or transverse (right) section. (G) 3D reconstruction of axons (electroporated with mbTomato, red) navigating through the FP stained with DAPI (blue) and BEN (white). The yellow dashed line corresponds to the cut plane resulting in the sagittal optic section shown in the lower panel. Yellow arrows point growth cones intercalated between BEN labeled glial cell processes when they navigate the FP (see also Videos 9–13). (H) Schematic drawings of an open-book with a sparse electroporation of commissural axons and a broad FP electroporation. Two plasmids are used: Math1-mbTomato in commissural neurons and HoxA1-GFP in the FP. (I) Ventral longitudinal view from an open-book electroporated as described in (H). The yellow dashed rectangle delineates a close-up of a crossing growth cone electroporated with Math1-mbT omato (red) navigating along the basal feet of glial cells electroporated with Hoxa1-GFP (white). (J) Surface reconstruction of a growth cone electroporated with Math1-mbTomato (red) navigating along the basal feet of glial cells electroporated with Hoxa1-GFP (white) as seen in a sagittal section (upper) or transversal section (lower). (K) Schematic drawing of a glial cell and two axons crossing through its basal end-feet, in a sagittal view (left) or transverse view (right). Scale bars: 5 μm in (D–G, I, and J), 20 μm in (B).
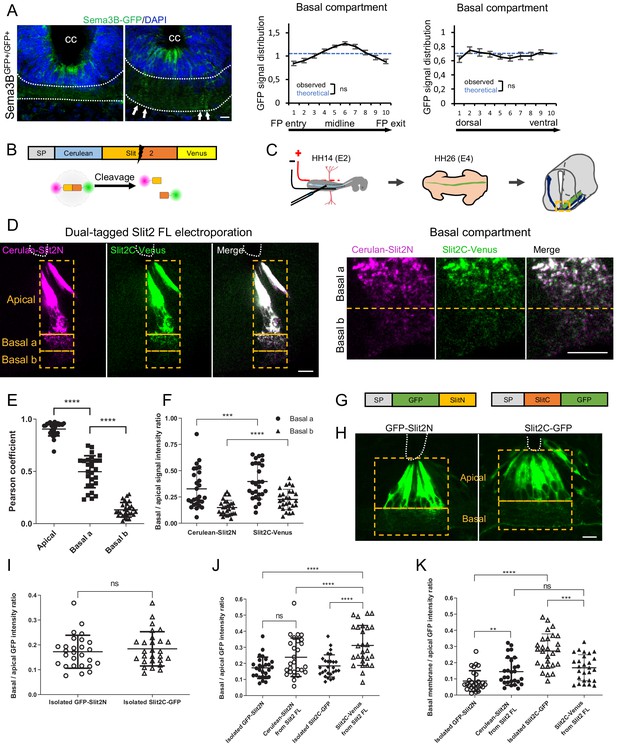
Slit2N and Slit2C decorate the floor plate (FP) glia basal processes and have distinct diffusion properties conditioned by Slit2-FL processing.
(A) Transverse section of Sema3B-GFP homozygous E12.5 embryo stained with DAPI. Endogenous green fluorescent protein (GFP) reports Sema3B expression and is quantified in the left–right axis of the FP (left) and in the dorso-ventral axis (right). The axon path is delineated by white dashed lines and the white arrows indicate the deposition of Sema3B-GFP along the glial cell processes. Data are shown as the mean ± s.e.m., KS test has been applied, n.s. (B) Schematic drawings of dual-tagged Slit2 construct and activity. (C) In ovo FP electroporation procedure. (D) 80 μm transverse section of E4 chick spinal cord FP electroporated with dual-tagged Slit2 (left) and close-up of the basal domain (right). (E) Pearson coefficients quantify the degree of colocalization of Cerulean Venus in FP electroporated with dual-tagged Slit2, through three compartments: apical, basal a, and basal b, as delimited by the dashed rectangle in (D) (N = 3 embryos, three sections per embryo, three images analyzed per section). (F) Intensity ratio of the basal compartment over the apical compartment for Cerulean and Venus in FP electroporated with dual-tagged Slit2. (G) Schematic drawings of Slit2 isolated fragments fused to GFP. (H) 80 μm transverse sections of E4 chick spinal cord FP electroporated with isolated Slit2 fragments fused to GFP. The apical and basal compartment are delineated with yellow dashed lines. (I) Intensity ratio of the basal compartment over the apical compartment for GFP in FP electroporated with either Slit2N-GFP or Slit2C-GFP (N = 3 embryos, three sections per embryo, three images analyzed per section). (J) Comparison between the basal/apical intensity ratio of Slit2 isolated fragments compared to the basal/apical intensity ratio of Slit2N and Slit2C fragments generated by the cleavage of dual-tagged Slit2-FL. (K) Comparison between the basal membrane/apical domain intensity ratio of Slit2 isolated fragments compared to the basal membrane/apical domain ratio of Slit2N and Slit2C fragments generated by the cleavage of dual-tagged Slit2-FL. Data are shown as the mean ± s.d. in (E, F, and I–K), Student's t-test has been applied, ns: non-significant, ***: p<0.001, ****: p<0.0001. Scale bars: 10 μm in (A, D, and H).
-
Figure 5—source data 1
Quantification of Sema3B expression in the left-right axis of the FP and in the dorso-ventral axis (A).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig5-data1-v2.xlsx
-
Figure 5—source data 2
Pearson coefficients quantify the degree of colocalization of Cerulean Venus in FP electroporated with dual-tagged Slit2 (E).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig5-data2-v2.xlsx
-
Figure 5—source data 3
Intensity ratio of the basal compartment over the apical compartment for Cerulean and Venus in FP electroporated with dual-tagged Slit2 (F).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig5-data3-v2.xlsx
-
Figure 5—source data 4
Intensity ratio of the basal compartment over the apical compartment for GFP in FP electroporated with either Slit2N-GFP or Slit2C-GFP (I).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig5-data4-v2.xlsx
-
Figure 5—source data 5
Comparison between the basal/apical intensity ratio of Slit2 isolated fragments compared to the basal/apical intensity ratio of Slit2N and Slit2C fragments generated by the cleavage of dual-tagged Slit2-FL (J).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig5-data5-v2.xlsx
-
Figure 5—source data 6
Comparison between the basal membrane/apical domain intensity ratio of Slit2 isolated fragments compared to the basal membrane/apical domain ratio of Slit2N and Slit2C fragments generated by the cleavage of dual-tagged Slit2-FL (K).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig5-data6-v2.xlsx
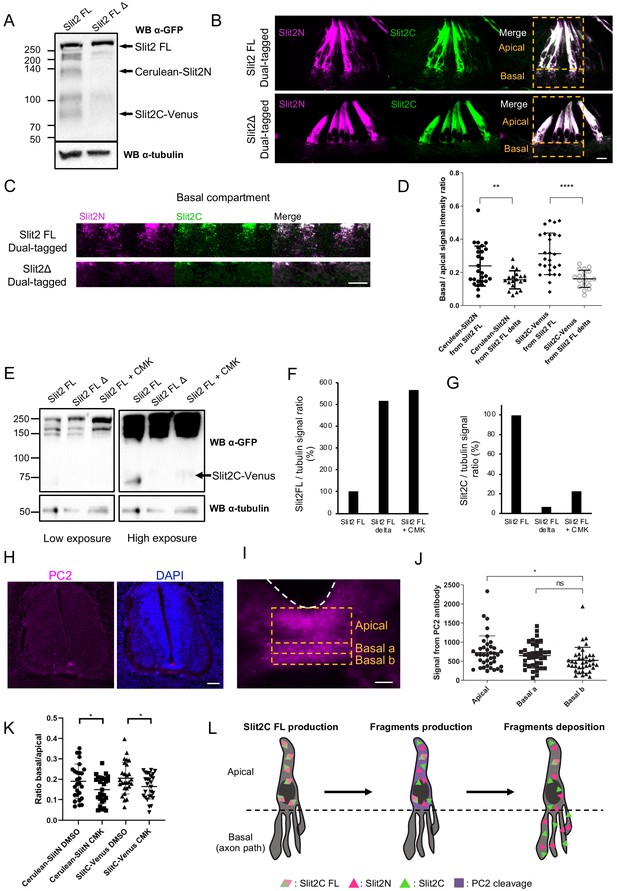
Slit2-FL cleavage plays a role in the proper diffusion of the protein and is dependent on PC2.
(A) Western blot detection of Cerulean and Venus in N2a cells transfected with either dual-tagged Slit2-FL or the uncleavable dual-tagged Slit2-FL Δ. An anti-green fluorescent protein (GFP) antibody was used, which recognizes Cerulean and Venus, two GFP derived fluorescent proteins. Protein sizes are in kDa. (B) Thick transverse sections of E4 chick spinal cord floor plate (FP) electroporated with dual-tagged Slit2-FL or dual-tagged Slit2-FL Δ (uncleavable form deprived from the cleavage site generating Slit2N and Slit2C fragments). (C) Close-up of the basal domains from (B). (D) Intensity ratio of the basal compartment over the apical compartment for Cerulean and Venus in FP electroporated with dual-tagged Slit2-FL or dual-tagged Slit2-FL Δ. (E) Western blot detection of Cerulean and Venus in N2a cells transfected with either dual-tagged Slit2-FL or the uncleavable dual-tagged Slit2-FL Δ and treated with PC2 inhibitor CMK. Tubulin is used as a loading control. (F and G) Ratio of the intensity of the Slit2-FL band (F) or the Slit2C band (G) over the intensity of tubulin band. (H) Immunofluorescent labeling of E4 chick spinal cord transverse sections using an antibody targeting PC2 (left panel) and DAPI (right panel). (I) Close-up of the PC2 labeling in the FP. (J) Quantification of the PC2 labeling in the three compartments delineated by yellow dashed lines. (K) Quantification of Slit2C and Slit2N basal/apical signal in control DMSO and CMK injected embryos after dual-tagged Slit2-FL electroporation. (L) Schematic representation of Slit2 fragments generation and deposition from the cleavage of Slit2-FL by PC2 in FP glial cells. Data are shown as the mean ± s.d. in (D and J) and Student's t-test has been applied. ns: non-significant, *: p<0.05, ***: p<0.001. Scale bars = 10 μm in (B, C, and I), 80 μm in (H).
-
Figure 6—source data 1
Intensity ratio of the basal compartment over the apical compartment for Cerulean and Venus in FP electroporated with dual-tagged Slit2-FL or dual-tagged Slit2-FL Δ (D).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig6-data1-v2.xlsx
-
Figure 6—source data 2
Ratio of the intensity of the Slit2-FL band over the intensity of tubulin band (F).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig6-data2-v2.xlsx
-
Figure 6—source data 3
Ratio of the intensity of the Slit2C band over the intensity of tubulin band (G).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig6-data3-v2.xlsx
-
Figure 6—source data 4
Quantification of the PC2 labeling in the three compartments delineated by yellow dashed lines (J).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig6-data4-v2.xlsx
-
Figure 6—source data 5
Quantification of Slit2C and Slit2N basal/apical signal in control DMSO and CMK injected embryos after dual-tagged Slit2-FL electroporation (K).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig6-data5-v2.xlsx
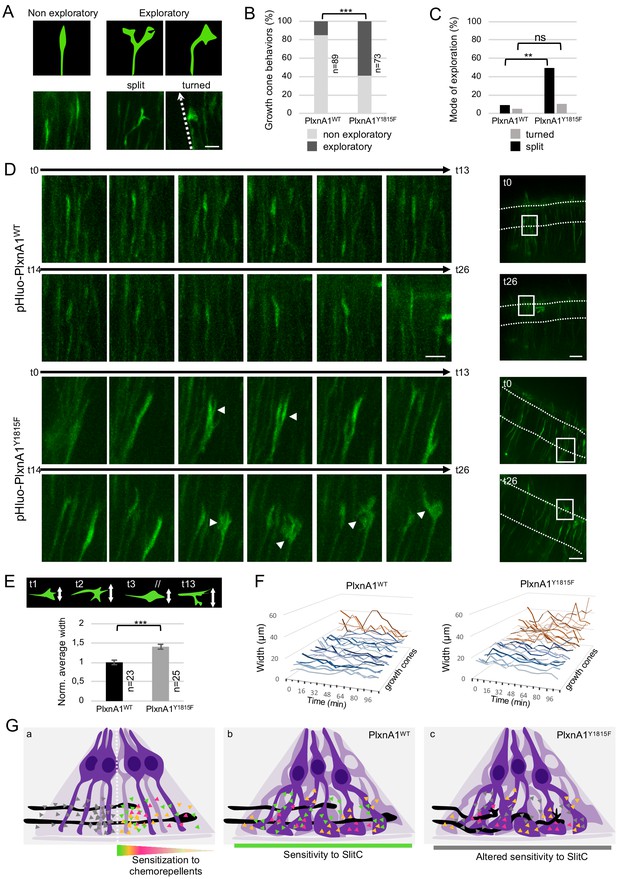
PlxnA1Y1815F commissural growth cones have an increased exploratory behavior exerted through morphological split.
(A) Analysis of growth cone behaviors with fast time-lapse sequences. Schematic representation of growth cone categories and microphotographs of representative growth cones. (B) Histogram of the quantification of exploratory growth cones in PlxnA1WT and PlxnA1Y1815F open-books (pHluo-PlxnA1WT, N = 5 electroporated embryos, 89 growth cones; pHluo-PlxnA1Y1815F, N = 3 electroporated embryos, 73 growth cones). Chi-squared test has been applied, ***: p<0.001. (C) Histogram depicting the mode of exploration adopted by PlxnA1WT and PlxnA1Y1815F growth cones. The percentage was calculated over the total growth cone population (pHluo-PlxnA1WT, N = 5 electroporated embryos, 89 growth cones; pHluo-PlxnA1Y1815F, N = 3 electroporated embryos, 73 growth cones). Chi-squared test has been applied between non-exploratory and split populations (**: p<0.01), and between non-exploratory and turned (ns: non-significant). (D) Time-lapse sequences of individual growth cones navigating the floor plate (FP). The right panels show growth cone positions at time 0 and time 26. Time interval: 8 min. (E) Quantification of growth cone width reporting their exploratory behavior during the FP navigation in PlxnA1WT and PlxnA1Y1815F open-books (pHluo-PlxnA1WT, N = 4 electroporated embryos, 23 growth cones; pHluo-PlxnA1Y1815F, N = 3 electroporated embryos, 25 growth cones). Data are shown as the mean ± s.e.m., Student's t-test has been applied, ***: p<0.001. (F) Histograms of individual growth cone width (μm) from t = 0 min to t = 122 min (pHluo-PlxnA1WT, N = 4 electroporated embryos, 23 growth cones; pHluo-PlxnA1Y1815F, N = 3 electroporated embryos, 25 growth cones). (G) Current and proposed novel model of the mechanisms ensuring proper midline crossing of spinal cord commissural axons. Scale bars: 10 μm in (A and D, left), 50 μm in (D, right).
-
Figure 7—source data 1
Quantification of exploratory growth cones in PlxnA1WT and PlxnA1Y1815F open-books (B and C).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig7-data1-v2.xlsx
-
Figure 7—source data 2
Quantification of individual growth cone width (F).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig7-data2-v2.xlsx
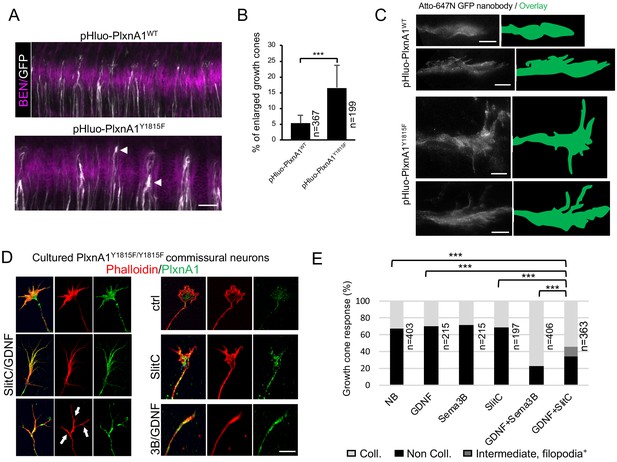
PlxnA1Y1815F mutation confers to commissural growth cones navigating the floor plate (FP) a complex morphology.
(A) Microphotograph of spinal cord open-books from chick embryo electroporated with pHluo-PlxnA1WT or pHluo-PlxnA1Y1815F, and immunolabeled with anti-BEN antibody and Atto-647 GFP nanobodies. BEN labeling delineates the FP. (B) Comparative analysis of the proportion of complex growth cones observed after PlxnA1WT and PlxnA1Y1815F electroporation (PlxnA1WT, N = 5 embryos, 367 growth cones; PlxnA1Y1815F, N = 4 embryos, 199 growth cones). Spinal-cord open-books were subdivided in three fragments and observed by confocal microscopy as a dorso-ventral stack (27 stacks). A representative image for each condition is shown. Data are shown as the mean ± s.e.m., Student's t-test has been applied, ***: p<0.001. (C) Microphotographs of representative growth cones from STEP microscopy of atto-647N-GFP nanobodies-labeled chick open-books. Scale bar: 5 μm. (D) Microphotographs of cultured PlxnA1Y1815F/Y1815F commissural neuron illustrating representative growth cone morphologies exposed to different treatments. (E) Histogram depicting the proportion of collapsed growth cones in the different experimental conditions (NB, N = 403; GDNF, N = 215; Sema3B, N = 215; SlitC, N = 197; GDNF+Sema3B, N = 406; GDNF+SlitC, N = 363). The intermediate condition represents growth cones with a shrinked central domain but still having filopodia as shown in (D) with white arrows. Chi-squared test has been applied, ***: p<0.001. Scale bars: 5 μm in (C), 15 μm in (D), 50 μm in (A).
-
Figure 7—figure supplement 1—source data 1
Comparative analysis of the proportion of complex growth cones observed after PlxnA1WT and PlxnA1Y1815F electroporation (B).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig7-figsupp1-data1-v2.xlsx
-
Figure 7—figure supplement 1—source data 2
Histogram depicting the proportion of collapsed growth cones in the different experimental conditions (E).
- https://cdn.elifesciences.org/articles/63205/elife-63205-fig7-figsupp1-data2-v2.xlsx
Videos
pHluo-PlxnA1WT (Videos 1 and 2) and pHluo-PlxnA1Y1815F (Videos 3 and 4) are addressed to the cell surface of commissural growth cones during the FP navigation.
White arrows point the growth cones during FP navigation. FP: floor plate.
pHluo-PlxnA1WT (Videos 1 and 2) and pHluo-PlxnA1Y1815F (Videos 3 and 4) are addressed to the cell surface of commissural growth cones during the FP navigation.
White arrows point the growth cones during FP navigation. FP: floor plate.
pHluo-PlxnA1WT (Videos 1 and 2) and pHluo-PlxnA1Y1815F (Videos 3 and 4) are addressed to the cell surface of commissural growth cones during the FP navigation.
White arrows point the growth cones during FP navigation. FP: floor plate.
pHluo-PlxnA1WT (Videos 1 and 2) and pHluo-PlxnA1Y1815F (Videos 3 and 4) are addressed to the cell surface of commissural growth cones during the FP navigation.
White arrows point the growth cones during FP navigation. FP: floor plate.
Fluorescence recovery after photobleaching (FRAP) sequences of commissural growth cones in spinal cord open-books.
The pHluo-receptor fluorescence in an area of 15–20 μm2 covering the entire growth cone surface was bleached at 80–90%. The recovery was measured over a period of 17 min.
Fluorescence recovery after photobleaching (FRAP) sequences of commissural growth cones in spinal cord open-books.
The pHluo-receptor fluorescence in an area of 15–20 μm2 covering the entire growth cone surface was bleached at 80–90%. The recovery was measured over a period of 17 min.
Fluorescence recovery after photobleaching (FRAP) sequences of commissural growth cones in spinal cord open-books.
The pHluo-receptor fluorescence in an area of 15–20 μm2 covering the entire growth cone surface was bleached at 80–90%. The recovery was measured over a period of 17 min.
Fluorescence recovery after photobleaching (FRAP) sequences of commissural growth cones in spinal cord open-books.
The pHluo-receptor fluorescence in an area of 15–20 μm2 covering the entire growth cone surface was bleached at 80–90%. The recovery was measured over a period of 17 min.
3D reconstruction with IMARIS software of axons navigating through the floor plate stained with DAPI (in blue) and BEN (in white) and with mbTomato electroporated in axons (in red).
Same reconstitution as in Video 9 but on a limited slice of spinal cord cut in the rostro-caudal axis.
The surface of a single axon from Video 10 has been reconstructed.
3D reconstruction from a Math1-mbTomato and Hoxa1-GFP electroporation.
A single Math1-mbTomato (in red) electroporated axon is navigating through the basal end-feet of a Hoxa1-GFP (in white) electroporated floor plate cell.





