Introduction to the EQIPD quality system
Figures
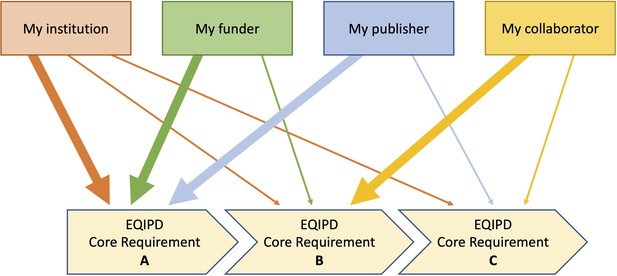
Flexible sequence of implementation of the EQIPD core requirements.
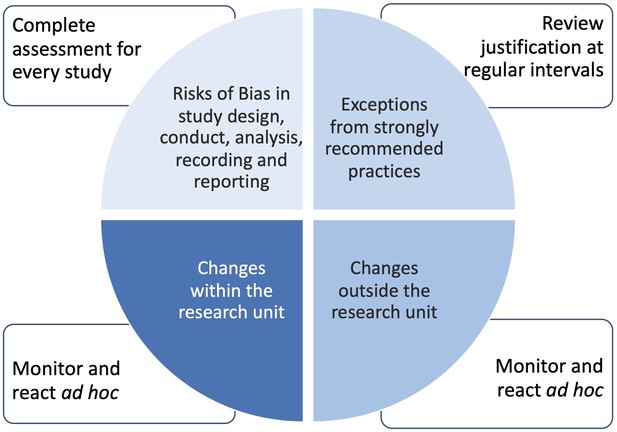
Depending on the current needs, a research unit may prioritize the implementation of one or another core requirement. For example, tasks related to core requirement ‘B’ are highly relevant for the research unit’s parent institution, the funding organization and a scientific journal where the research team plans to publish the results of their work. In contrast, core requirement ‘C’ is of lower importance and can, therefore, be addressed at a later timepoint.
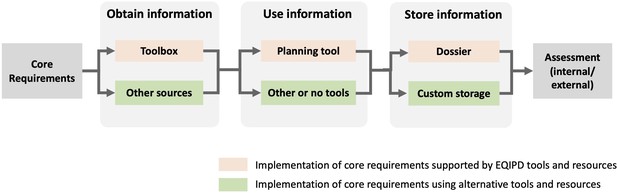
Implementation of the EQIPD Quality System (QS): From Core Requirements (CR) to assessment of a fully functional system.
The 18 CRs are the expectations formulated by EQIPD that serve as the starting point for implementing the QS. At any step during the implementation, the use of EQIPD tools is voluntary and serves only the purpose of making the implementation and maintenance of the QS easier. As the first step, unless such information is available from other sources, the research unit may consult with the Toolbox to obtain relevant research quality-related information. Once the necessary information is obtained, the research unit applies this knowledge and monitors the progress. This can be done using the Planning Tool, using alternative project management resources or even without any such tools. The Dossier is a repository of documents and information that are specific to the user’s research unit and that is organized according to a structure suggested by EQIPD (to keep all research quality-related information in one place and make it easily findable). However, the research unit may also opt to use its own way to store information. Finally, once the implementation is completed, the research unit may initiate an assessment to get feedback from experts outside of the research unit (either quality professionals within the same organization or a third party).
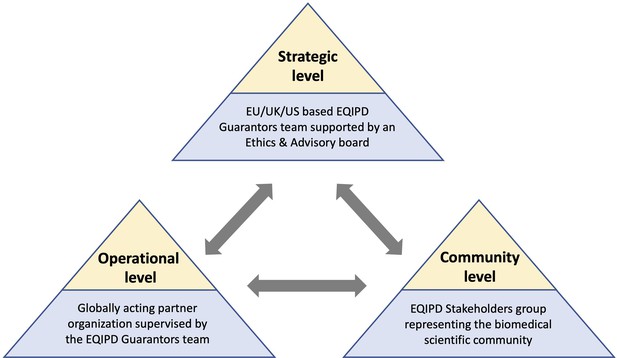
The proposed future governance model of EQIPD.
The EQIPD Guarantors group and the EQIPD Ethics and Advisory Board are responsible for the overall guidance, administration of academic and educational programs, as well as dissemination of the EQIPD vision (Strategic level). An independent partner organization, commissioned by the EQIPD Guarantors, will provide the operational support and the day-to-day services for the EQIPD community (Operational level). The EQIPD Stakeholder group, composed of scientists, funders, quality professionals, manufacturers of research tools, and publishers, provides feedback on the practical aspects of the EQIPD Quality System and facilitates connections to a broader biomedical research community (Community level).
Tables
Comparison of quality systems.
| Quality system | ISO 9001 | GLP (FDA, OECD) | EQIPD |
|---|---|---|---|
| Year Launched | 1987, 2015 | 1976, 1981 | 2020 |
| Application area | A general QMS that can be applied to all aspects of organizations (not focused on biomedical research) | Non-clinical health and environmental safety studies upon which hazard assessments are based | Non-regulated preclinical (non-clinical) biomedical research |
| Initial stimulus to be developed | Procuring organizations needed a basis of contractual arrangements with their suppliers (i.e., basic requirements for a supplier to assure product quality) | Regulators such as FDA aimed to avoid poorly managed or fraudulent non-clinical studies on safety of new drugs | Biomedical research community (industry and academia) recognized the negative impact of lacking research rigor on the development of novel therapeutics, and the need for a comprehensive practical solution to help enhance preclinical data reliability |
| Customers | Typically outside of the organization (anyone who requires a product or service) | Typically outside of the organization (patients, regulators, sponsors, etc.) | In most cases, both inside (scientists themselves) and outside (patients, funders, collaboration partners, publishers, etc.) of the organization |
| Objectives | To certify that a product (which can be preclinical data) or a service is provided with consistent, good-quality characteristics, which satisfy the stated or implied needs of customers | To ensure the quality, integrity and reliability of data on the properties and/or safety of test items concerning human health and/or the environment | To facilitate generating robust and reliable preclinical data and thereby boost innovation |
| Main focus | Standardization of processes The organizational overall performance is continuously improved (process approach) to enhance customer satisfaction and development initiatives are done on a sound basis for sustainability | The organizational process and the conditions under which non-clinical health and environmental safety studies are planned, performed, monitored, recorded, archived and reported | The outcome of research activities that is robust, reliable, traceable, properly recorded, reconstructible, securely stored and trustworthy (generated under appropriately unbiased conditions) |
| Dedicated quality professionals | Not required (advisable for larger organizations) | Required | Not required (advisable for larger organizations) |
| Formal training on implementation and use | Not required | Required | Advisable, but not required |
| Assessments | External (ISO auditors) and internal (internal auditors) | External (health authorities/governmental inspectors) and internal (QA auditors) | Self-assessment (by Process Owner), external (by EQIPD)* |
-
*Additional internal assessments may be conducted by qualified colleagues (e.g. dedicated quality professionals) outside the research unit but within the same organization (advisable for larger organizations).
EQIPD Core Requirements.
| Categories | # | Item |
|---|---|---|
| Research team | 1 | Process Owner for the EQIPD Quality System must be identified |
| 2 | Communication process must be in place | |
| Quality culture | 3 | The research unit must have defined quality objectives |
| 4 | All activities must comply with relevant legislation and policies | |
| 5 | The research unit must have a procedure to act upon concerns of potential misconduct | |
| Data integrity | 6 | Generation, handling and changes to data records must be documented |
| 7 | Data storage must be secured at least for as long as required by legal, contractual or other obligations or business needs | |
| 8 | Reported research outcomes must be traceable to experimental data | |
| 9 | Reported data must disclose all repetitions of a study, an experiment, or a test regardless of the outcome | |
| Research processes | 10 | Investigator must declare in advance whether a study is intended to inform a formal knowledge claim |
| 11 | All personnel involved in research must have adequate training and competence to perform assigned tasks | |
| 12 | Protocols for experimental methods must be available | |
| 13 | Adequate handling and storage of samples and materials must be ensured | |
| 14 | Research equipment and tools must be suitable for intended use and ensure data integrity | |
| Continuous improvement | 15 | Risk assessment must be performed to identify factors affecting the generation, processing and reporting of research data |
| 16 | Critical incidents and errors during study conduct must be analyzed and appropriately managed | |
| 17 | An approach must be in place to monitor the performance of the EQIPD Quality System, and address identified issues | |
| Sustainability | 18 | Resources for sustaining the EQIPD Quality System must be available |
Key principles.
| Principle | Explanation | Examples (related to the use of randomization) |
|---|---|---|
| Engage with autonomy | Decisions about specific needs and solutions are made by researchers, and not by EQIPD. EQIPD has formulated core requirements for the QS implementation and, as a partner in this process, EQIPD asks critical questions and provides recommendations that are voluntary to follow and are provided only to help the researchers throughout the implementation and use. | EQIPD recommends applying randomization to all studies but it is for the researcher to decide whether randomization is applied to a particular study or a particular study design |
| Grow through reflection | What it means to have the right quality level in place is suggested by your environment (collaborators, funders, institution, etc.). EQIPD does not ‘invent’ needs or requirements of your funders or your collaborators. As a partner in this process, EQIPD QS only allows you to see these requirements better and suggests ways of implementing them (Gilis, 2020). | EQIPD identifies overlapping requirements from different stakeholders toward the use and reporting of randomization. |
| Focus on goal | Focus on the outcome (performance standards), not on the path, timelines or the tools to get there (Guillén, 2010). | EQIPD highlights the importance of ‘randomness’ (lack of pattern or predictability) in the correctly developed randomization sequence but leaves it up to the user to select a specific method or tool. |
| Be transparent | Key research processes must be transparent. This principle applies specifically to retention and accessibility of information related to key decisions related to study design, conduct, or analysis (e.g. decisions to include or exclude certain data points in the analysis). | If one decides not to apply randomization, the decision must be stated and must be justified, recorded and reported. |
| Leave a trace | Key research processes must be traceable. Complementary to the principle above, this principle refers to retention and accessibility of all information that is necessary for a complete reconstruction of a key research process (e.g. raw data related to reported data are findable, and reported data are reconstructable from raw data). | If one does apply randomization, the way you apply randomization must be traceable and reported. |
Expectations toward rigor in study design.
| All research | Research informing a formal knowledge claim (i.e. research requiring maximal rigor) | |
|---|---|---|
| Study plan | Should be defined and documented before starting the experiments | Must be defined and documented before starting the experiments |
| Study hypothesis | Advised to define | Must be pre-specified |
| Blinding | Advised to implement | Should be implemented, exceptions must be justified and documented |
| Randomization | Advised to implement | Should be implemented, exceptions must be justified and documented |
| Sample size calculation | Advised to define and document before starting the experiments | Must be defined and documented before starting the experiments (e.g. included in the study plan) |
| Data analysis | Advised to define and document before starting the experiments | Must be defined and documented before starting the experiments (e.g. as a formal statistical analysis plan and/or included in the study plan) |
| Inclusion and exclusion criteria | Advised to define and document before starting the experiments | Must be defined and documented before starting the experiments (e.g. included in the study plan) |
| Deviations from study plan | Advised to document | Must be documented |
| Preregistration | - | Should be implemented |
Levels of use of the EQIPD framework.
| Levels of use: | Information only (incl. training) | Purpose-fit certification | Quality System |
|---|---|---|---|
| EQIPD guidance: | Recommendations on best practices, examples, templates | Basic set of core requirements | Full set of core requirements |
| Main users: | Research units, funding organizations | Research units | Research units |
| Expected use: | As necessary, follow specific recommendations or use provided tools to improve work processes (e.g. increase transparency or make raw data findable or improve reporting) As appropriate, use information provided by EQIPD in training programs; communicate to collaborators, grantees, etc. | Confirm that current quality practices are in line with the basic set of EQIPD core requirements (related to data integrity and rigor in study design, conduct, analysis, and reporting) | Align current research quality practices with the EQIPD expectations (implement full set of core requirements including those that define quality system – i.e. availability of resources, process owner, quality objectives, and continuous improvement mechanisms) |
| Dedicated efforts by the research unit (e.g., regular and sustained efforts, dedicated personnel): | None | Limited | Yes (proportional to quality objectives) |
| Context of use: | Research unit is informed about expectations by current or future collaborators, funders, sponsors, publishers, etc. | Flexible solution driven by the time- and resource-critical needs of specific collaboration(s) | Stable solution for long-term maintenance of research rigor standards |
| Assessment by the EQIPD team: | No | Yes | Yes |