An optimized CRISPR/Cas9 approach for precise genome editing in neurons
Figures
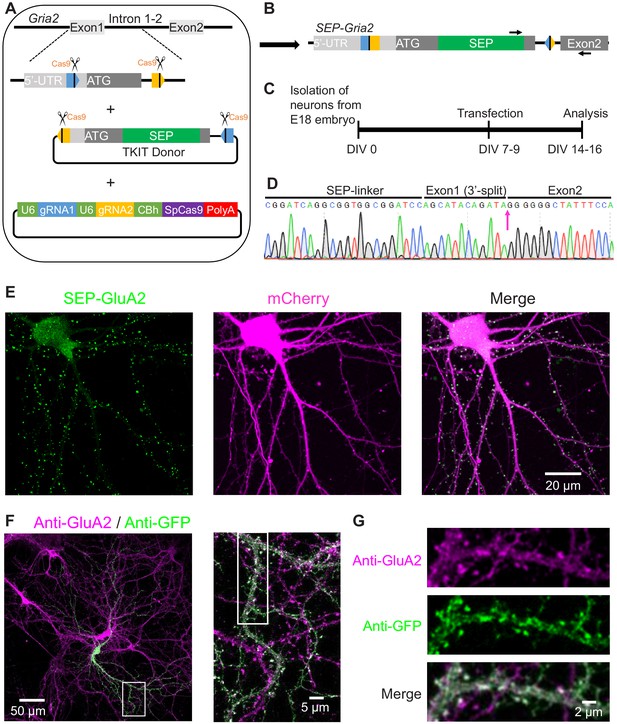
TKIT-mediated genome editing in neuron culture.
(A) Graphical representation of the mouse genomic loci of Gria2 and schematic of TKIT gene targeting. The blue and orange pentagons indicate gRNA target sequences. The black vertical line within pentagon indicates the Cas9 cleavage site. (B) Schematic of genomic DNA following SEP KI to Gria2 with TKIT. Arrows indicate primers for RT-PCR and cDNA sequencing. (C) Experimental scheme for SEP KI in primary cultured neurons. (D) Sequence analysis of the cDNA from mRNA extracted from electroplated neuronal culture of SEP-GluA2. (E) Representative images showing the endogenous SEP signal in live neurons following TKIT-mediated SEP-GluA2 knock-in. Scale bar, 20 μm. (F) Representative images of immunofluorescence showing that SEP-GluA2 (green) colocalized with GluA2 (magenta). Scale bars display 50 or 5 μm. (G) Enlarged dendrite and spine (white boxes in right panel of F). Scale bars display 2 μm.
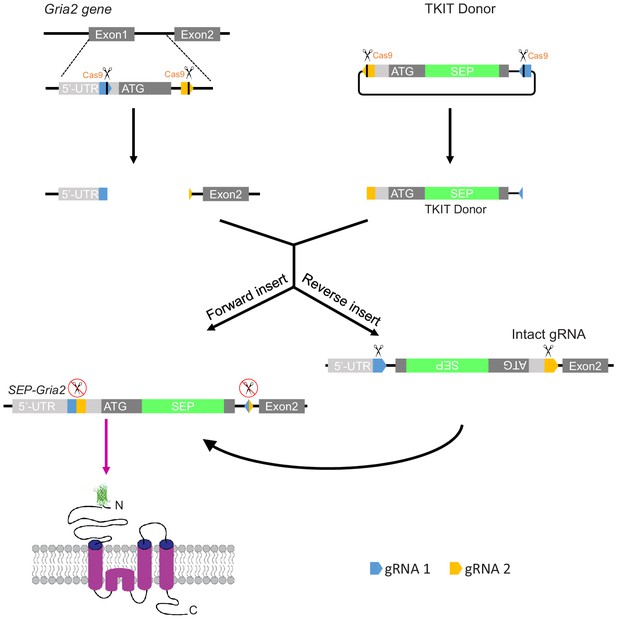
Strategy to promote donor DNA insertion in the correct orientation.
Graphical representation of TKIT-mediated genome editing with Streptococcus pyogenes Cas9. The blue and orange pentagons indicate guide RNA 1 and guide RNA 2 recognition sites, respectively. The black line within the pentagon indicates the Cas9 cleavage site. The light gray rectangle indicates the 5-UTR of gene of interest and the dark gray rectangle the coding sequence. Black arrows indicate the possible outcomes following genomic insertion of the donor DNA.
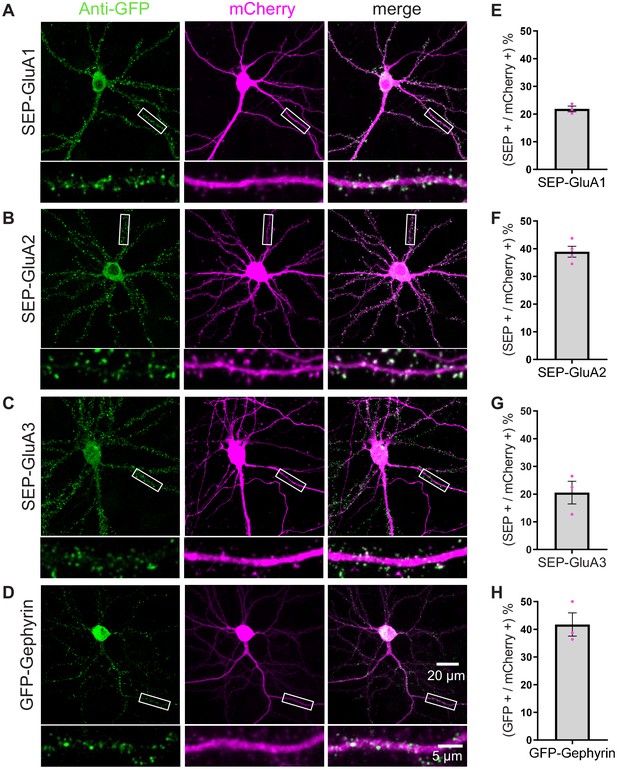
Efficient KI of SEP-GluA1-3 and GFP-gephyrin using TKIT.
(A) SEP-GluA1, (B) SEP-GluA2, (C) SEP-GluA3, and (D) GFP-gephyrin visualized with confocal microscopy after neurons were co-transfected with TKIT constructs and a mCherry cell fill. Segments of dendrite in the white boxed region are enlarged below. Scale bar, 20 μm or 5 μm. Average efficiency of KI for (E) SEP-GluA1, (F) SEP-GluA2, (G) SEP-GluA3, and (H) GFP-gephyrin. Error bars display SEM.
-
Figure 2—source data 1
Efficiency counts to assess TKIT knockin of SEP-GluA1, SEP-GluA2, SEP-GluA3, and GFP-Gephyrin in mouse neurons.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig2-data1-v1.xlsx
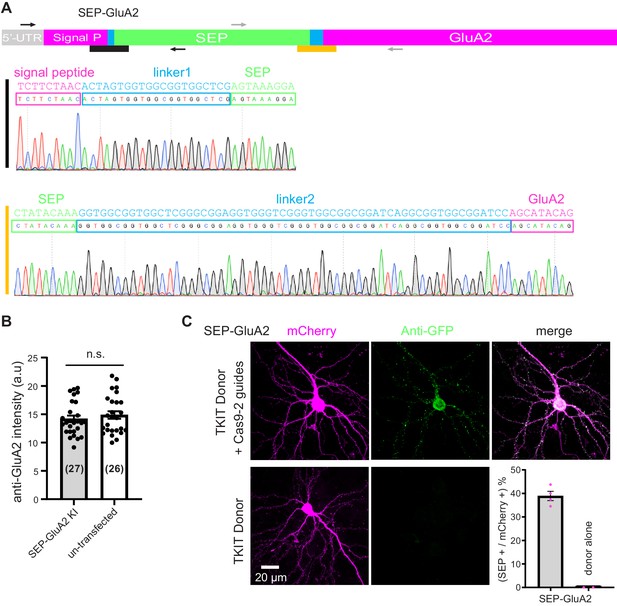
Correct splicing of SEP-GluA2 and comparable GluA2 levels in KI and WT surrounding neurons.
(A) Sequence analysis of cDNA reverse-transcribed from mRNA extracted from neuronal cultures electroporated with TKIT SEP-GluA2 constructs. The black line indicates the junction between the signal peptide and linker1-SEP and the orange line indicates the junction between SEP-linker2 and the mature protein. The magenta region indicates the endogenous protein sequence, the green sequence indicates SEP and the blue sequence indicates linkers. Black and gray arrows indicate the location of PCR primer pairs. (B) Quantification of synaptic GluA2 levels with immunofluorescence. GluA2 levels in SEP-GluA2 KI neurons are comparable with non-transfected (WT) neurons (data from 27 SEP-GluA2 KI neurons and 26 surrounding non-transfected neurons). Data presented as mean ± SEM. Individual data points are shown as black dots. Unpaired Student’s t-test for comparison (p=0.4495). (C) Transfection of TKIT donor alone does not produce KI. Representative images showing TKIT of SEP-GluA2 KI compared to TKIT donor-only control. Average efficiency of KI for SEP-GluA2 compared to donor-only control (bottom right corner). Scale bar indicates 20 μm.
-
Figure 2—figure supplement 1—source data 1
GluA2 expression levels in SEP-GluA2 knockin and untransfected neurons.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig2-figsupp1-data1-v1.xlsx
-
Figure 2—figure supplement 1—source data 2
SEP-GluA2 knockin efficiency compared to donor-only control.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig2-figsupp1-data2-v1.xlsx
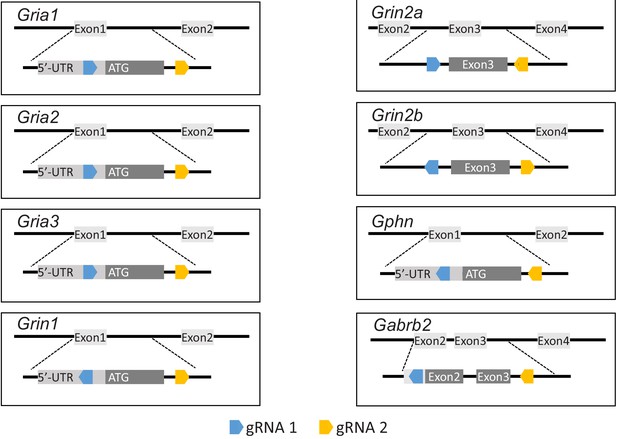
Location of TKIT guide pairs.
Schematic illustrating the location of all guide RNAs and their orientation in all genes targeted for TKIT. In each panel the top portion indicates the partial exons and introns being targeted. The bottom part shows an enlargement of the region to be edited. The light gray rectangle indicates the 5’-UTR. The dark gray rectangle indicates the coding sequence. The blue and orange pentagons indicate guide RNA (gRNA) 1 and 2, respectively. The genes Gria1, Gria2, Gria3, Grin1, Grin2a, Grin2b, Gepn, and Gabrb2 encode for GluA1, GluA2, GluA3, NR1, NR2A, NR2B, Gephyrin, and GABAAR Beta-2, respectively.
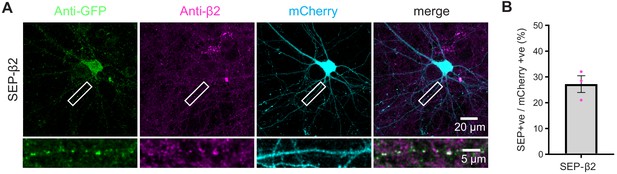
TKIT of SEP-GABAAR β2.
(A) Representative immunofluorescent images demonstrating TKIT of SEP-GABAAR β2 (SEP- β2). The dendritic region within the white box is enlarged below to better illustrate co-localization between SEP-β2 (green) and anti-β2 signal (magenta). Note: the cell-fill channel was removed from the enlarged overlay images for clarity. Scale bars display 20 μm or 5 μm. (B) Average efficiency of SEP-β2 KI was 27.22 ± 3.24%. Data presented as mean ± SEM.
-
Figure 2—figure supplement 3—source data 1
Efficiency counts to assess TKIT knockin of SEP-GABAAR β2 in mouse neurons.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig2-figsupp3-data1-v1.xlsx
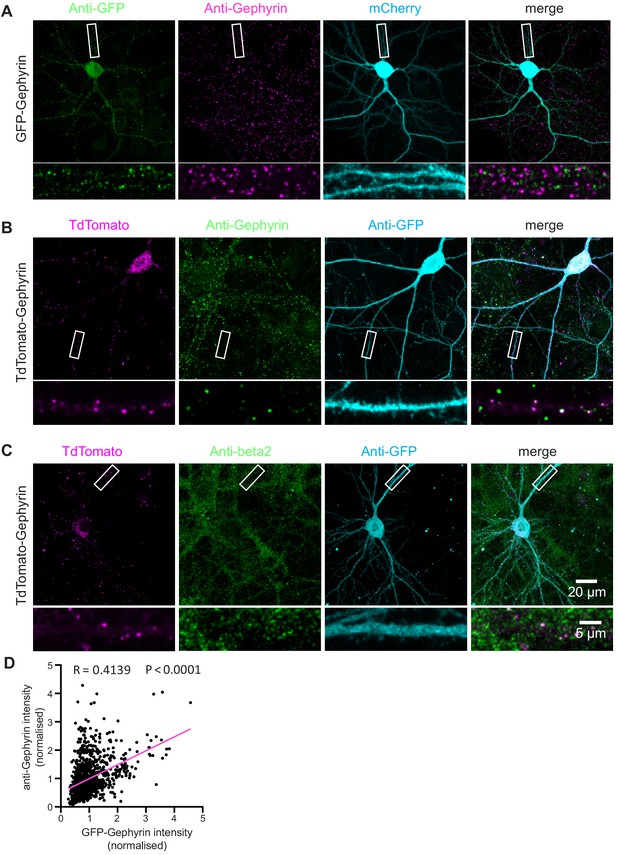
TKIT of GFP-Gephyrin and tdTomato-Gephyrin.
(A) Representative immunofluorescent images demonstrating TKIT of GFP-Gephyrin. The dendritic region within the white box is enlarged below to better illustrate co-localization between GFP-Gephyrin (green) and anti-Gephyrin signal (magenta). (B) Representative immunofluorescent images demonstrating TKIT of tdTomato-Gephyrin. The dendritic region within the white box is enlarged below to better illustrate co-localization between tdTomato-Gephyrin (magenta) and anti-Gephyrin signal (green). (C) Representative immunofluorescent images demonstrating TKIT of tdTomato-Gephyrin. The dendritic region within the white box is enlarged below to better illustrate co-localization between tdTomato-Gephyrin (magenta) and the β2 GABAA receptor subunit (green). Note: the cell-fill channel was removed from the enlarged overlay images for clarity. Scale bars display 20 μm or 5 μm. (D) Scatter plot showing the correlation between GFP-Gephyrin (from KI) and anti-Gephyrin staining intensity (from immunofluorescence). Correlation (slope) between GFP and anti-Gephyrin fluorescent intensity was calculated by Simple Linear Regression (GraphPad Prism 7 or 8). Data from 20 neurons and 1007 GFP-Gephyrin puncta.
-
Figure 2—figure supplement 4—source data 1
Immunofluorescence data for correlation of GFP-Gephyrin knockin signal with gephyrin.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig2-figsupp4-data1-v1.xlsx
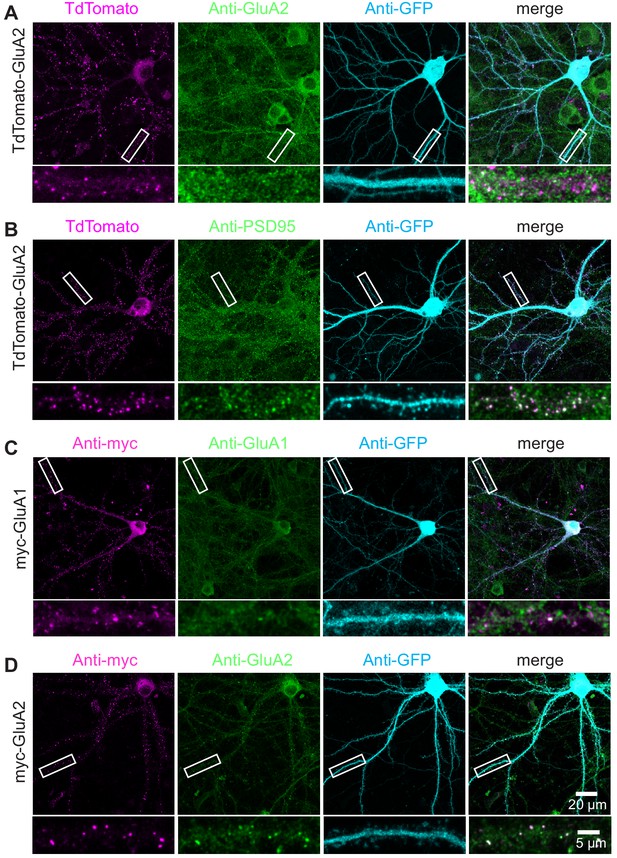
TKIT of alternative tags for AMPA receptors.
(A and B) Representative immunofluorescent images demonstrating TKIT of tdTomato-GluA2. The dendritic region within the white box is enlarged below to better illustrate co-localization between tdTomato-GluA2 (magenta) and GluA2 (green, A) or PSD95 (green, B). (C and D) Representative immunofluorescent images demonstrating TKIT of myc-GluA1 and myc-GluA2. The dendritic region within the white box is enlarged below to better illustrate co-localization between myc-GluA1 and myc-GluA2 (magenta) with GluA1 and GluA2 (green), respectively. Note: The cell-fill channel was removed from the enlarged merged images for clarity. Scale bars display 20 μm or 5 μm.
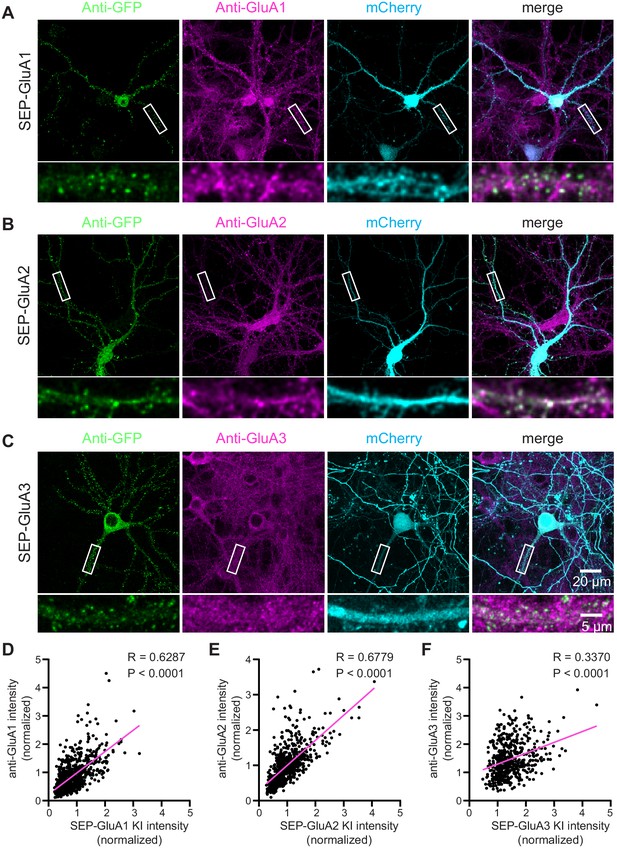
Correlation of SEP-GluA1-3 KI with endogenous AMPA receptors.
(A–C) Representative immunofluorescent images showing TKIT of SEP-GluA1 (A), SEP-GluA2 (B), and SEP-GluA3 (C). The dendritic region within the white box is enlarged below to better illustrate co-localization between SEP-GluA1 (green) and GluA1 (magenta, A), SEP-GluA2 (green) and GluA2 (magenta, B), and SEP-GluA3 (green) and GluA3 (magenta, C). Note: The cell-fill channel was removed from the enlarged overlay images for clarity. Scale bars display 20 μm or 5 μm. (D–F) Scatter plots showing the correlation between SEP-GluA1-3 and anti-GluA1-3 immunofluorescence intensity. Correlation (slope) between SEP and anti-GluAs fluorescent intensity was calculated by Simple Linear Regression (GraphPad Prism 7 or 8). Data from 14 neurons and 733 synapses for SEP-GluA1, 13 neurons and 668 synapses for SEP-GluA2, and 10 neurons and 508 synapses for SEP-GluA3.
-
Figure 2—figure supplement 6—source data 1
Immunofluorescence data for correlation of SEP-GluA1, SEP-GluA2, and SEP-GluA3 knockin signal with GluA1, GluA2, and GluA3, respectively.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig2-figsupp6-data1-v1.xlsx
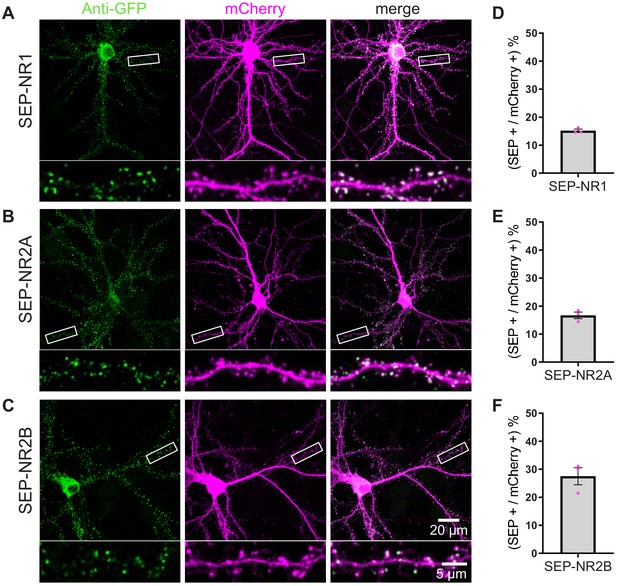
TKIT of SEP NMDA receptor subunits.
(A) SEP-NR1, (B) SEP-NR2A, and (C) SEP-NR2B KI were visualized with confocal microscopy after neurons were co-transfected with TKIT constructs and a mCherry cell fill. Segments of dendrite in the white boxed region are enlarged below. Scale bar, 20 μm or 5 μm. Average efficiency of KI for (D) SEP-NR1, (E) SEP-NR2A, and (F) SEP-NR2B. Error bars display SEM.
-
Figure 3—source data 1
Efficiency counts to assess TKIT knockin of SEP-NR1, SEP-NR2A, and SEP-NR2B in mouse neurons.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig3-data1-v1.xlsx
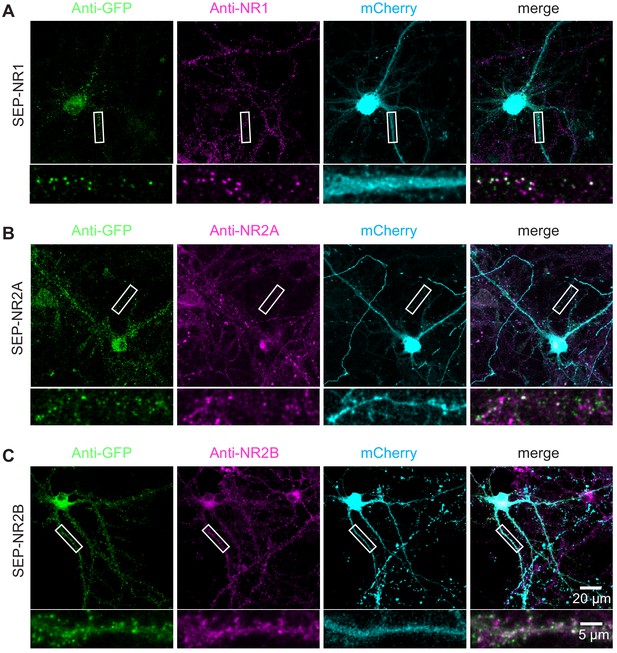
TKIT of SEP-NMDA receptors.
Representative immunofluorescent images demonstrating TKIT of SEP-NR1 (A), SEP-NR2A (B), and SEP-NR2B (C). The dendritic region within the white box is enlarged below to better illustrate co-localization between SEP-NR1 (green) and anti-NR1 (magenta, A), SEP-NR2A (green), and anti-NR2A (magenta, B), and SEP-NR2B (green) and anti-NR2B (magenta, C). Note: The cell-fill channel was removed from the enlarged overlay images for clarity. Scale bars display 20 μm or 5 μm.
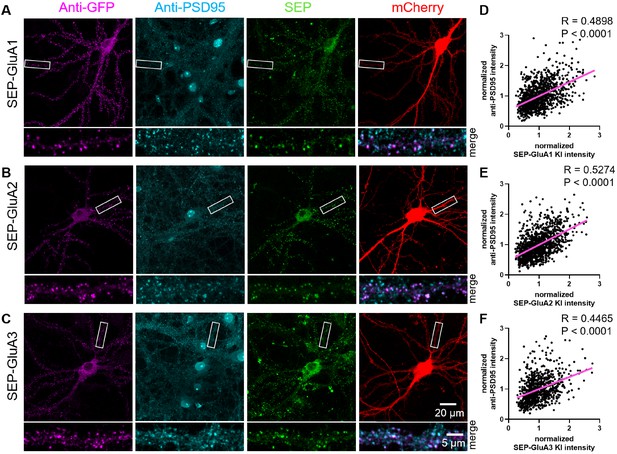
Validation of synaptic localization following SEP KI to AMPARs.
TKIT of (A) SEP-GluA1, (B) SEP-GluA2, and (C) SEP-GluA3 with endogenous SEP signal (green), PSD95 staining (cyan), GFP staining (magenta), and mCherry cell fill (red). Segments of dendrite in the white boxed region are enlarged below. Note: the endogenous SEP signal and cell-fill channel (mCherry) were removed from the enlarged overlay images for clarity. Scale bars display 20 or 5 μm. (D–F) Scatter plots showing the correlation between SEP-GluA1-3 signal and PSD95 immunofluorescence signal. Correlation (slope) between SEP and anti-PSD95 fluorescent intensity was calculated by Simple Linear Regression (GraphPad Prism 7 or 8). (D) SEP-GluA1 correlations with data from 22 neurons and 1114 synapses, (E) SEP-GluA2 correlations with data from 23 neurons and 1191 synapses, and (F) SEP-GluA3 correlations with data from 16 neurons and 810 synapses.
-
Figure 4—source data 1
Immunofluorescence data for correlation of PSD95 signal with SEP-GluA1, SEP-GluA2, and SEP-GluA3.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig4-data1-v1.xlsx
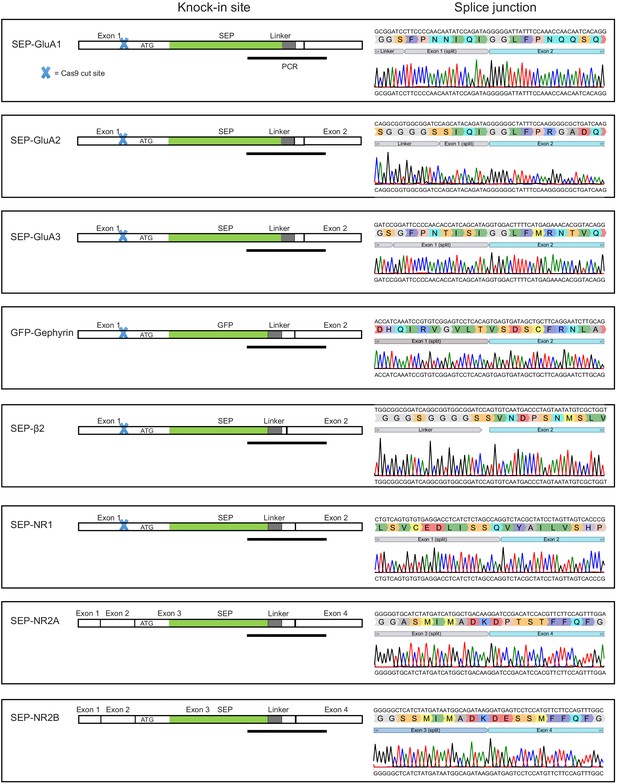
Intact splicing with TKIT.
Schematic of cDNAs indicating the KI site and neighboring exons for each SEP/GFP TKIT target. The blue cross indicates DNA cut sites. Sanger sequencing was used to analyze cDNA reverse-transcribed from mRNA extracted from neuronal cultures electroporated with each SEP/GFP TKIT target. Sequencing results indicate intact splicing of tagged proteins. The black line under the schematic indicate the region PCR amplified and sequenced. Note: The downstream cut site for SEP-GluA1, SEP-GluA2, SEP-GluA3, GFP-Gephyrin, SEP-β2, and SEP-NR1 is located within introns and therefore is not indicated within the schematic. Both cut sites for SEP-NR2A and SEP-NR2B are located within introns.
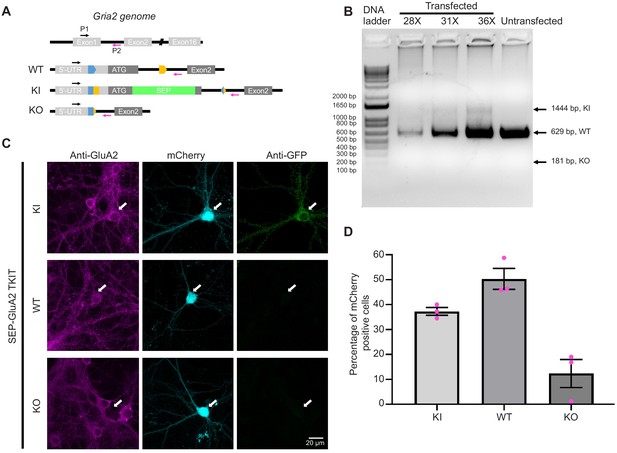
Low levels of GluA2 KO with TKIT.
(A) Top panel: cartoon showing Gria2 gene structure and the location of PCR primers (upstream P1, black arrow and downstream P2, magenta arrow). Bottom panel: three predicted genotypes (WT, KI, and KO). Arrows indicate the same primers showed in top panel. (B) Genomic DNA was extracted from neuronal cultures following electroporation of TKIT constructs. PCR was performed with one pair of PCR primers to detect KI/WT/KO outcomes with different numbers of cycles (28, 31, and 36). Following DNA electrophoresis of PCR products, there was clear KI and WT bands, and no detectable KO band. Note: proportion of WT was overestimated, because part of WT was from non-transfected cells. (C) Representative immunofluorescent images of TKIT mediated SEP-GluA2 KI, including KI, WT, and KO neurons. White arrows indicate transfected neurons. Scale bars represent 20 μm. (D) Quantification of different fractions of KI, WT, or KO cells based on data from 252 neurons (three experiments) following SEP-GluA2 TKIT mediated KI. Mean of KI: 37.3%, WT: 50.3%, KO: 12.4%. Results shown as mean ± SEM.
-
Figure 4—figure supplement 2—source data 1
GluA2 immunofluorescence data to assess the proportion of neurons that are wild type (unedited), knockin (SEP-GluA2), or knockout for GluA2.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig4-figsupp2-data1-v1.xlsx
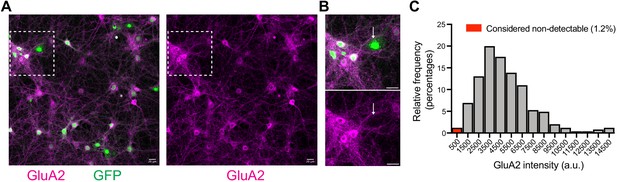
Occasional detection of GluA2-lacking neurons in the absence of TKIT construct transfection.
(A) Representative images of mouse cortical neurons at DIV15. Cells were transduced with AAV-GFP at DIV2, to serve as a morphology marker. Immunofluorescence for GluA2 was performed to assess the level of GluA2 expression. (B) Enlargement of white boxed region showing GFP (green) and GluA2 (magenta). The white arrow indicates an example of a cell lacking detectable levels of GluA2. (C) GluA2 levels were quantified at the cell body of 245 neurons, from three coverslips. We considered cells will levels of GluA1 lower than 1000 (a.u.) to be GluA2 lacking. Scale bars indicate 20 μm.
-
Figure 4—figure supplement 3—source data 1
GluA2 immunofluorescence data to assess the fraction of neurons that are GluA2-lacking under basal conditions.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig4-figsupp3-data1-v1.xlsx
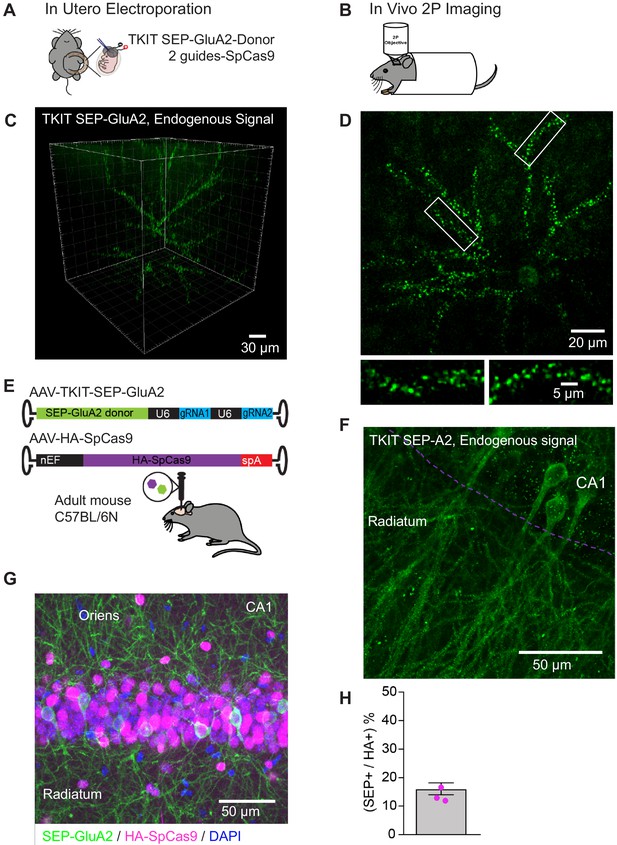
TKIT mediated KI in vivo.
(A) Schematic of in utero electroporation and (B) subsequent in vivo two-photon microscopy setup. (C) Two-photon 3D reconstruction of a layer 2/3 pyramidal neuron with TKIT-mediated SEP-GluA2 KI. (D) Maximum intensity projection of the same example cell in (C) with magnified dendritic segments (white boxes). Scale bars are shown in the bottom right corner of each image. (E) Dual AAV-HA-SpCas9 and AAV-TKIT-SEP-GluA2 injections were made into the dorsal hippocampus of adult C57BL/6N mice. (F) Confocal image of native SEP-GluA2 fluorescence signal. The CA1 cell layer is illustrated with a magenta dotted line with dendrites in Stratum radiatum. The image is a maximum intensity projection. (G) A composite confocal image showing the expression of SEP-GluA2 (GFP antibody, green), Cas9 (HA antibody, magenta), and nuclei (DAPI, blue). The image is a maximum intensity projection and is centered on the CA1 pyramidal cell layer. (H) Quantification of the 16.1% of HA-SpCas9 expressing cells that are also GFP positive. Scale bars are indicated in the bottom right corner of images.
-
Figure 5—source data 1
Immunohistochemistry data for SEP-GluA2 knockin efficiency calculation with dual virus approach.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig5-data1-v1.xlsx
TKIT mediated SEP-GluA2 KI in vivo, punctate signal could be clearly observed along the dendrites of layer 2/3 pyramidal neurons.
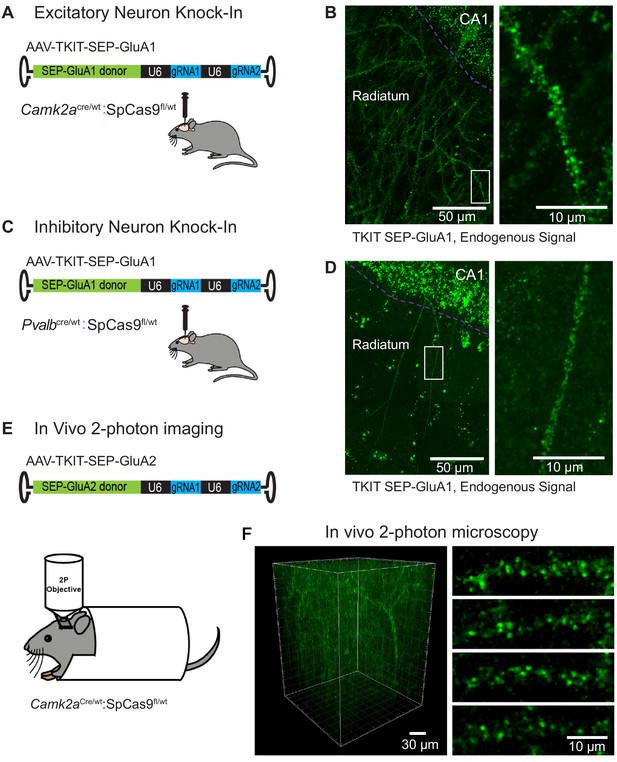
Cell-type specific KI with TKIT.
(A) Schematic of experimental design for targeting excitatory neurons using Camk2aCre:Cas9fl/wt animals (B) Super-resolution Airyscan confocal image of a CA1 pyramidal neuron with TKIT-mediated SEP-GluA1 KI illustrated as a maximum intensity projection. The CA1 cell layer is illustrated with a magenta dotted line with dendrites in Stratum radiatum. A zoomed in region (right) shows the dendritic segment indicated with a white box. (C) Schematic of experimental design for targeting PV positive interneurons using PvalbCre/wt:Cas9fl/wt animals. (D) Super-resolution Airyscan confocal image of a CA1 PV neuron with TKIT-mediated SEP-GluA1 KI illustrated as a maximum intensity projection. The CA1 cell layer is illustrated with a magenta dotted line with dendrites in Stratum radiatum. A zoomed in region (right) shows the dendritic segment indicated with a white box. (E) Schematic of experimental setup for two-photon image of virus-mediated TKIT GluA2 KI. (F) In vivo two-photon 3D reconstruction of a layer 2/3 pyramidal neuron in barrel cortex with TKIT-mediated SEP-GluA2 KI. High-magnification images of dendritic segments (right) from a maximum intensity projection of the same cell. Scale bars are indicated in the bottom right corner of images.
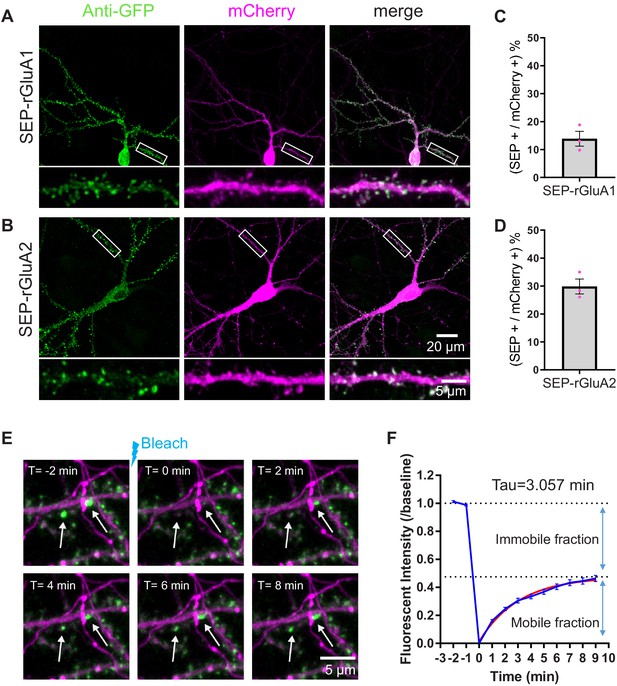
TKIT KI and FRAP in rat neurons.
TKIT-mediated KI of SEP-GluA1 (A) and SEP-GluA2 (B) in primary cultured rat neurons. Efficiency of KI for SEP-GluA1 (C) and SEP-GluA2 (D). (E) Representative example of a SEP-GluA1 containing spine undergoing bleaching and fluorescence recovery. The white arrow indicates the spine that will be bleached. Images are separated by 2 min intervals. Scale bar indicates 5 μm. (F) Summary of SEP-GluA1 FRAP data with estimated recovery time and immobile fraction of receptors. Red trace is from exponential (one-phase decay) fitting, GraphPad Prism 7.
-
Figure 7—source data 1
Efficiency counts to assess TKIT knockin of SEP-GluA1 and SEP-GluA2 in rat neurons.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig7-data1-v1.xlsx
-
Figure 7—source data 2
Fluorescence recovery after photobleaching data to assess SEP-GluA1 mobility in rat neurons.
Source Data for figure supplements.
- https://cdn.elifesciences.org/articles/65202/elife-65202-fig7-data2-v1.xlsx
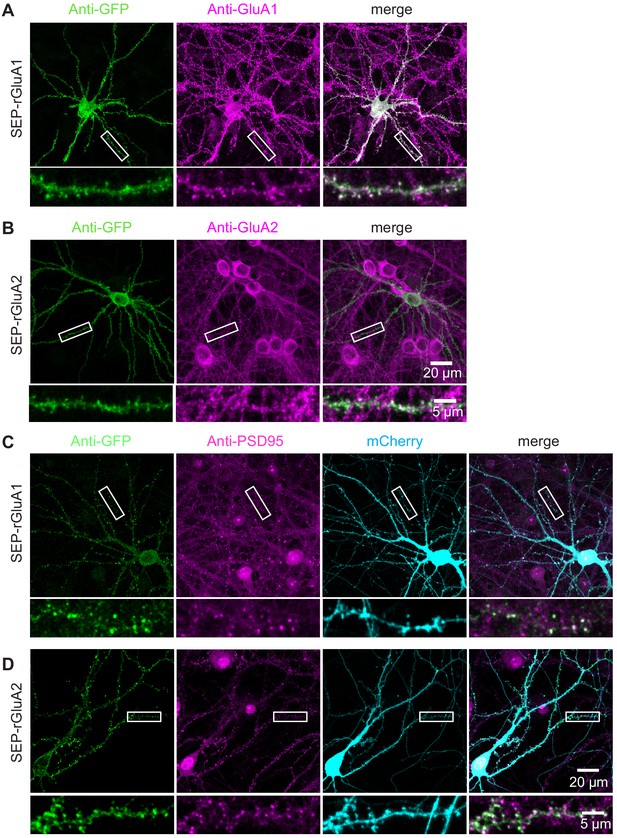
TKIT of SEP-GluA1 and SEP-GluA2 KI in rat neurons.
Representative immunofluorescent images demonstrating TKIT of SEP-GluA1 (A) and SEP-GluA2 (B) in rat primary cultured neurons. The dendritic region within the white box is enlarged below to better illustrate co-localization between (A) SEP-GluA1 (green) and GluA1 (magenta), or (B) SEP-GluA2 (green) and GluA2 (magenta). (C and D) Co-localization of SEP-GluA1 and SEP-GluA2 with PSD95. The dendritic region within the white box is enlarged below to better illustrate co-localization between (C) SEP-GluA1 (green) and PSD95 (magenta), or (D) SEP-GluA2 (green) with PSD95 (magenta). Note: The cell-fill channel was removed from the enlarged overlay images for clarity. Scale bars display 20 μm or 5 μm.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (Mus musculus) | GluA1 | GenBank; Ensembl | ID: 14799; ENSMUSG00000020524 | |
| Gene (Mus musculus) | GluA2 | GenBank; Ensembl | ID: 14800; ENSMUSG00000033981 | |
| Gene (Mus musculus) | GluA3 | GenBank; Ensembl | ID: 53623; ENSMUSG00000001986 | |
| Gene (Mus musculus) | GluN1 | GenBank; Ensembl | ID: 14810; ENSMUSG00000026959 | |
| Gene (Mus musculus) | GluN2A | GenBank; Ensembl | ID: 14811; ENSMUSG00000059003 | |
| Gene (Mus musculus) | GluN2B | GenBank; Ensembl | ID: 14812; ENSMUSG00000030209 | |
| Gene (Mus musculus) | Gephyrin | GenBank; Ensembl | ID: 268566; ENSMUSG00000047454 | |
| Gene (Mus musculus) | beta2 | GenBank; Ensembl | ID: 14401; ENSMUSG00000007653 | |
| Gene (Rattus norvegicus) | rGluA1 | GenBank | ID: 50592 | |
| Gene (Rattus norvegicus) | rGluA2 | GenBank | ID: 29627 | |
| Strain, strain background (Mus musculus, male and female) | Camk2a-Cre mouse | Jackson Laboratory strain 005359 | IMSR Cat# JAX:005359, RRID:IMSR_JAX:005359 | |
| Strain, strain background (Mus musculus, male and female) | Pvalb-Cre mouse | Jackson Laboratory strain 008069 | IMSR Cat# JAX:008069, RRID:IMSR_JAX:008069 | |
| Strain, strain background (Mus musculus, male and female) | Cas9fl/fl mouse | Jackson Laboratory strain 027632 | IMSR Cat# JAX:027632, RRID:IMSR_JAX:027632 | |
| Genetic reagent (Streptococcus pyogenes) | px330 | RRID:Addgene_42230 | RRID:Addgene_42230 | |
| Genetic reagent (Streptococcus pyogenes) | pAAV-nEF-SpCas9 | RRID:addgene_87115 | RRID:Addgene_87115 | |
| Genetic reagent (Escherichia coli) | pMiniT Vector | NEB | E1202 | NEB replaced this vector with pMiniT 2.0 Vector |
| Genetic reagent (Mus musculus) | AAV2/9-TKIT-SEP-GluA1 | Viral Core at the Janelia Research Campus | AAV for in vivo delivery TKITdonor and two guides | |
| Genetic reagent (Mus musculus) | AAV2/9-TKIT-SEP-GluA2 | Viral Core at the Janelia Research Campus | AAV for in vivo delivery TKITdonor and two guides | |
| Genetic reagent (Streptococcus pyogenes) | AAV2/9-nEF-HA-spCas9 | Viral Core at the Janelia Research Campus | AAV for in vivo delivery TKITdonor and two guides | |
| Genetic reagent (Aequorea victoria) | AAV2/9.CMV.PI.EGFP.WPRE.bGH | RRID:addgene_105530 | RRID:Addgene_105530 | |
| Biological sample (M. musculus) | Primary neuron culture | isolated from E18 mouse embryo and cultured | ||
| Biological sample (R. norvegicus) | Primary neuron culture | isolated from E18 rat embryo and cultured | ||
| Antibody | Anti-GluA1 (Rabbit polyclonal) | Homemade | JH4294 | IF (1:200) |
| Antibody | Anti-GluA2/3 (Rabbit polyclonal) | Homemade | JH4854 | IF (1:250) |
| Antibody | Anti-GluA2 (mouse monoclonal) | Eric Gouaux Lab | clone 15F1 | IF (1:5000) |
| Antibody | Anti-GluN1 (mouse monoclonal) | NeuroMab, clone N308/48 | UC Davis/NIH NeuroMab Facility Cat# N308/48, RRID:AB_2877408 | IF (1:200) |
| Antibody | Anti-GluN2a (Rabbit polyclonal) | Homemade | JH6097 | IF (1:200) |
| Antibody | Anti-GluN2b (Rabbit polyclonal) | Millipore, AB1548 | Millipore Cat# AB1548, RRID:AB_90759 | IF (1:100) |
| Antibody | Anti-beta2 (mouse monoclonal) | Millipore, MAB341 | Millipore Cat# MAB341, RRID:AB_2109419 | IF (1:200) |
| Antibody | Anti-PSD95 (mouse monoclonal) | NeuroMab, clone K28/43 | UC Davis/NIH NeuroMab Facility Cat# K28/43, RRID:AB_2877189 | IF (1:2500) |
| Antibody | Anti-Gephyrin (mouse monoclonal) | Synaptic systems, cat# 147011 | Synaptic Systems Cat# 147 011, RRID:AB_887717 | IF (1:2500) |
| Antibody | Anti-GFP (chicken polyclonal) | Abcam, cat # ab13970 | (Abcam Cat# ab13970, RRID:AB_300798) | IHC (1:5000) |
| Antibody | Anti-GFP (chicken polyclonal) | AVES, cat# GFP-1020 | Aves Labs Cat# GFP-1020, RRID:AB_10000240 | IF (1:5000) |
| Antibody | Anti-myc (mouse monoclonal) | Homemade | Clone 9E10 | IF (1:1000) |
| Antibody | Anti-HA (Rabbit monoclonal) | Cell Signaling, clone C29F4 | Cell Signaling Technology Cat# 3724, RRID:AB_1549585 | IF (1:500) |
| Recombinant DNA reagent (Escherichia coli) | pSG | This paper | For the guide2 cloning | |
| Recombinant DNA reagent (Streptococcus pyogenes) | GluA1 (px330:2-guide-Cas9) | This paper | Twoguides for GluA1 (mouse) in px330 | |
| Recombinant DNA reagent (Mus musculus) | pMiniT:SEP-GluA1 TKIT donor | This paper | TKIT donor for SEP-GluA1 (mouse) in pMiniT | |
| Recombinant DNA reagent (Mus musculus) | pMiniT:myc-GluA1 TKIT donor | This paper | TKIT donor for myc-GluA1 (mouse) in pMiniT | |
| Recombinant DNA reagent (Streptococcus pyogenes) | GluA2 (px330:2-guide-Cas9) | This paper | Two guides for GluA2 (mouse) in px330 | |
| Recombinant DNA reagent (Mus musculus) | pMiniT:SEP-GluA2 TKIT donor | This paper | TKIT donor for SEP-GluA2 (mouse) in pMiniT | |
| Recombinant DNA reagent (Mus musculus) | pMiniT:tdTomato-GluA2 TKIT donor | This paper | TKIT donor for tdTomato-GluA2 (mouse) in pMiniT | |
| Recombinant DNA reagent (Mus musculus) | pMiniT:myc-GluA2 TKIT donor | This paper | TKIT donor for myc-GluA2 (mouse) in pMiniT | |
| Recombinant DNA reagent (Streptococcus pyogenes) | GluA3 (px330:2-guide-Cas9) | This paper | Two guides for GluA3 (mouse) in px330 | |
| Recombinant DNA reagent (Mus musculus) | pMiniT:SEP-GluA3 TKIT donor | This paper | TKIT donor for SEP-GluA3 (mouse) in pMiniT | |
| Recombinant DNA reagent (Streptococcus pyogenes) | GluN1 (px330:2-guide-Cas9) | This paper | Two guides for GluN1 (mouse) in px330 | |
| Recombinant DNA reagent (Mus musculus) | pMiniT:SEP-GluN1 TKIT donor | This paper | TKIT donor for SEP-GluN1 (mouse) in pMiniT | |
| Recombinant DNA reagent (Streptococcus pyogenes) | GluN2A (px330:2-guide-Cas9) | This paper | Two guides for GluN2A (mouse) in px330 | |
| Recombinant DNA reagent (Mus musculus) | pMiniT:SEP-GluN2A TKIT donor | This paper | TKIT donor for SEP-GluN2A (mouse) in pMiniT | |
| Recombinant DNA reagent (Streptococcus pyogenes) | GluN2B (px330:2-guide-Cas9) | This paper | Two guides for GluN2B (mouse) in px330 | |
| Recombinant DNA reagent (Mus musculus) | pMiniT:SEP-GluN2B TKIT donor | This paper | TKIT donor for SEP-GluN2B (mouse) in pMiniT | |
| Recombinant DNA reagent (Streptococcus pyogenes) | Gephyrin (px330:2-guide-Cas9) | This paper | Two guides for Gephyrin (mouse) in px330 | |
| Recombinant DNA reagent (Mus musculus) | pMiniT:GFP-Gephyrin TKIT donor | This paper | TKIT donor for GFP-Gephyrin (mouse) in pMiniT | |
| Recombinant DNA reagent (Mus musculus) | pMiniT:tdTomato-Gephyrin TKIT donor | This paper | TKIT donor for tdTomato-Gephyrin (mouse) in pMiniT | |
| Recombinant DNA reagent (Streptococcus pyogenes) | beta2 (px330:2-guide-Cas9) | This paper | Two guides for beta2 (mouse) in px330 | |
| Recombinant DNA reagent (Mus musculus) | pMiniT:SEP-beta2 TKIT donor | This paper | TKIT donor for SEP-beta2 (mouse) in pMiniT | |
| Recombinant DNA reagent (Streptococcus pyogenes) | rGluA1 (px330:2-guide-Cas9) | This paper | Two guides for rGluA1 (mouse) in px330 | |
| Recombinant DNA reagent (Rattus norvegicus) | pMiniT:SEP-rGluA1 TKIT donor | This paper | TKIT donor for SEP-rGluA1 (mouse) in pMiniT | |
| Recombinant DNA reagent (Streptococcus pyogenes) | rGluA2 (px330:2-guide-Cas9) | This paper | Two guides for rGluA2 (mouse) in px330 | |
| Recombinant DNA reagent (Rattus norvegicus) | pMiniT:SEP-rGluA2 TKIT donor | This paper | TKIT donor for SEP-rGluA2 (mouse) in pMiniT | |
| Recombinant DNA reagent (Mus musculus) | pAAV-SEP-GluA1 TKIT | This paper | TKIT donor and two guides for SEP-GluA1 (mouse) in one pAAV vector | |
| Recombinant DNA reagent (Mus musculus) | pAAV-SEP-GluA2 TKIT | This paper | TKIT donor and two guides for SEP-GluA2 (mouse) in one pAAV vector | |
| Commercial assay or kit | NEBuilder HiFi DNA Assembly Cloning Kit | NEB | cat#: E5520 | |
| Software, algorithm | Fiji/ImageJ | Fiji | Fiji, RRID:SCR_002285 | |
| Software, algorithm | GraphPad Prism 7.0 | GraphPad Software | GraphPad Prism, RRID:SCR_002798 | |
| Software, algorithm | Benchling CRISPR tool | http://www.benchling.com | Benchling, RRID:SCR_013955 | |
| Software, algorithm | IMARIS 9.6.0 (Bitplane) | BitPlane | Imaris, RRID:SCR_007370 | |
| Other | Prolong Glass Antifade mounting media | Thermo Scientific | P36984 |
Additional files
-
Supplementary file 1
Location and sequences of TKIT guides.
- https://cdn.elifesciences.org/articles/65202/elife-65202-supp1-v1.docx
-
Supplementary file 2
PCR primers for TKIT donors or AAV constructs assembly.
- https://cdn.elifesciences.org/articles/65202/elife-65202-supp2-v1.docx





