Sensitizing Staphylococcus aureus to antibacterial agents by decoding and blocking the lipid flippase MprF
Figures
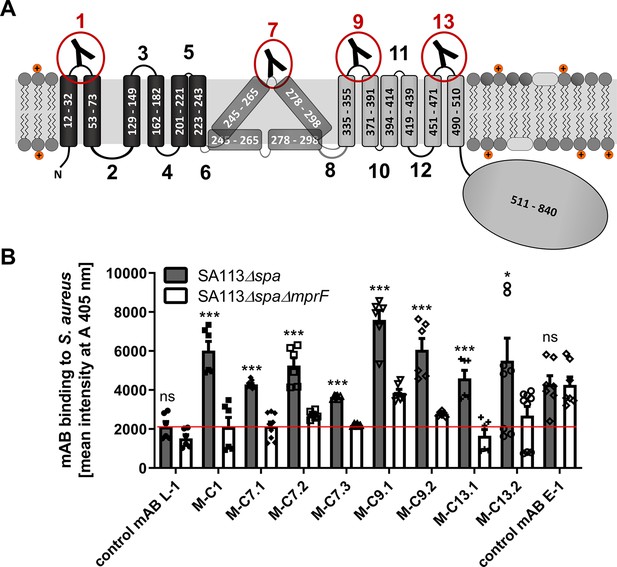
Multiple peptide resistance factor (MprF) topology and binding of monoclonal antibodies (mABs) to MprF-expressing S. aureus cells.
(A) MprF membrane topology is given according to our recent study (Ernst et al., 2015) showing synthase and flippase domains in gray and black, respectively. Amino acid (aa) positions of transmembrane segment (TMS) and the C-terminal hydrophilic domain are indicated. TMSs from aa 245–265 and aa 278–298 are shown in two alternative positions as computational and experimental results of transmembrane topology have been contradictory (Ernst et al., 2015). Localizations of MprF’s TMS-connecting loops are numbered starting from the N-terminus, antibody-targeted loops are indicated by red circles and antibody symbols. (B) Specific binding of mABs (100 nM) to S. aureus was analyzed by ELISA using SA113 strains deficient in the IgG-binding protein A (Spa) comparing the SA113 spa mutant (Δspa) and spa mprF double knockout mutant (ΔspaΔmprF). The red line indicates the mean intensity measured at A 405 nm (affinity) of the isotype control mAB L-1 bound to S. aureus SA113Δspa. Means and standard error of the mean (SEM) of at least three biological replicates are shown. Significant differences between SA113Δspa and SA113ΔspaΔmprF were calculated by Student’s paired t-test (ns, not significant; *p < 0.05; ***p < 0.0001).
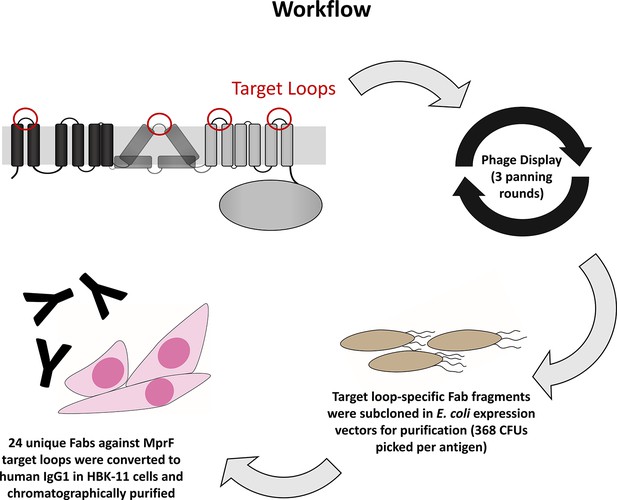
Workflow for the development of multiple peptide resistance factor (MprF)-specific monoclonal antibodies (mABs).
Biotinylated MprF peptide loops were incubated with the HuCal phage display library expressing single-chain human Fab fragments (Prassler et al., 2011), antigen-binding phages were enriched in three iterative panning rounds, bound antigen-specific phages were isolated, and respective Fab fragments were subcloned in E. coli expression vectors to yield His-tagged Fab fragments. Twenty-four unique Fabs against 4 MprF-derived peptides were converted to human IgG by cloning in an IgG1 expression vector system for production in human HKB11 cells, and IgGs were purified via protein A chromatography.
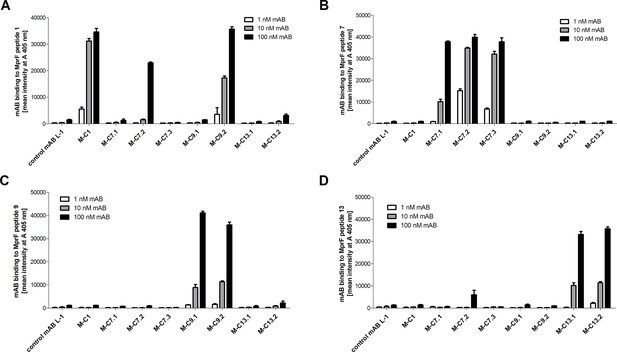
Specific binding of selected monoclonal antibodies (mABs) to cyclic multiple peptide resistance factor (MprF)-derived target peptides analyzed by ELISA.
Biotinylated cyclic peptides corresponding to the MprF loops 1, 7, 9, or 13 were incubated with eight anti-MprF IgGs or the control mAB L-1 in phosphate-buffered saline (PBS). (A–D) show binding of mABs at increasing concentrations to cyclic MprF loops 1, 7, 9, and 13, respectively. The means and standard error of the mean (SEM) of mean intensity measured at A 405 nm in three biological replicates are shown.
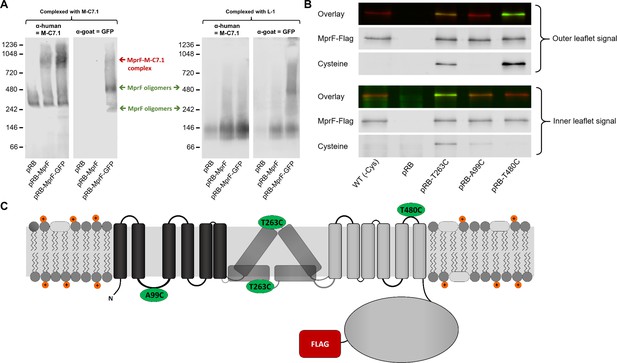
Binding of M-C7.1 to multiple peptide resistance factor (MprF) and membrane localization of the M-C7.1-targeted MprF loop 7.
(A) Detection of M-C7.1 binding to MprF. Plasmid-encoded native and green fluorescent protein(GFP)-tagged MprF variants were expressed in S. aureus SA113ΔspaΔmprF and living cells were preincubated with M-C7.1 or the isotype control monoclonal antibody (mAB) L-1 (in order to form MprF–mAB complexes). MprF variants complexed with M-C7.1 or control mAB L-1, respectively, were detected by blue native PAGE followed by Western blotting using two different primary (anti-GFP or M-C7.1) and corresponding secondary antibodies. SA113ΔspaΔmprF expressing the empty vector (pRB) served as negative control. Molecular masses in kDa of marker proteins are given on the left of the blot. Arrows mark both the MprF–M-C7.1 complex at 900 kDa and the MprF oligomers at 250 and 500 kDa, which were previously described (Ernst et al., 2015). (B) Cellular localization of the antigen epitope of M-C7.1 using the substituted cysteine accessibility method (SCAM) for specific loops between the MprF transmembrane segments (TMSs). The substituted cysteine T263C is localized in M-C7.1’s target peptide sequence in MprF. Substitution of A99C served as inside control, substitution of T480C served as outside control (see topology model, part C). S. aureus SA113ΔmprF expressing the empty vector (pRB) and an MprF variant lacking all native cysteines (wild-type [WT] (-Cys)) served as additional negative controls. All MprF variants were plasmid-encoded, FLAG tagged at the C-terminus to allow immunoprecipitation and detection, and were expressed in S. aureus SA113ΔmprF. Substituted extracellular cysteine residues were labeled with Na*-(3-maleimidylpropionyl)-biocytin (MPB) (outer leaflet signal, green in overlay), while labeling of substituted internal cysteine with MPB was performed after the blocking of external cysteines with 4-acetamino-4′-maleimidylstilbene-2,2′-disulfonic acid (AMS) (inner leaflet signal, green in overlay). MprF was detected via antibody staining by an anti-FLAG antibody (red in overlay). (C) MprF topology showing location and amino acid exchanges of artificial cysteine residues for SCAM detection in green.
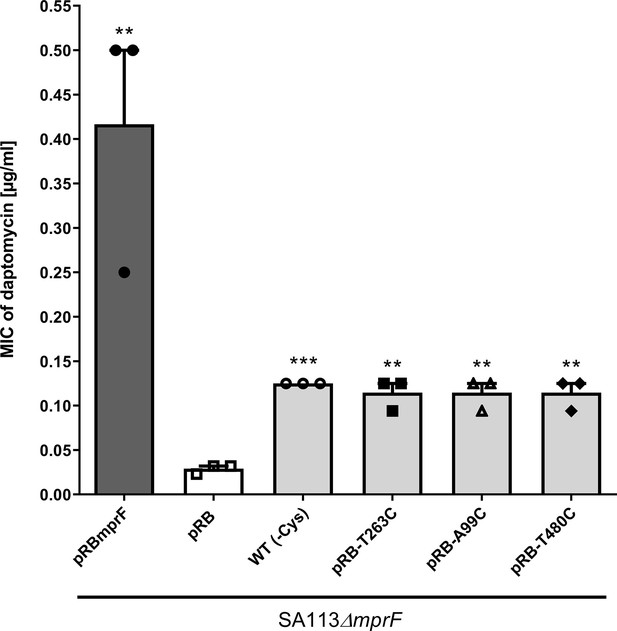
Effects of cysteine replacement and insertion on multiple peptide resistance factor (MprF) function, assessed by measuring daptomycin susceptibility.
Minimal inhibitory concentrations (MICs) of daptomycin against the indicated S. aureus strains are shown. The mprF deletion mutant with empty pRB474 plasmid served as a negative control, whereas cysteine depleted mprF-expressing variants show a fourfold increased daptomycin MIC while wild-type mprF-expressing positive control shows a tenfold increased MIC. The means + standard error of the mean (SEM) of results from three independent experiments are shown. Values that are significantly different from the values determined for S. aureus SA113ΔmprF bearing pRB474 (pRB), calculated by Student’s paired t-test, as indicated (**p < 0.01; ***p < 0.0001).
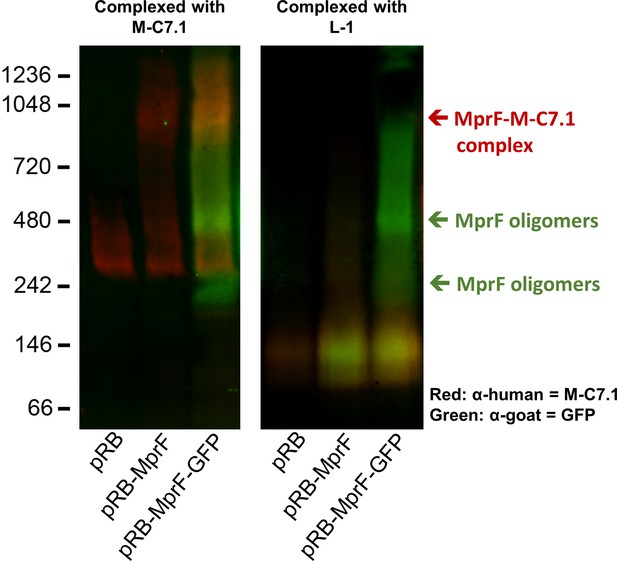
Detection of M-C7.1 binding to multiple peptide resistance factor (MprF).
An overlay of single channels from Figure 2A showing antihuman IgG binding to M-C7.1 in red and an antigoat IgG binding to the primary anti-GFP IgG in green. SA113ΔspaΔmprF expressing the empty vector (pRB) served as negative control. Molecular masses in kDa of marker proteins are given on the left of the blot. Arrows mark the MprF–M-C7.1 complex at 900 kDa and the MprF oligomers at 250 and 500 kDa as previously described (Ernst et al., 2015). Further explanations are found in figure legend of Figure 2A.
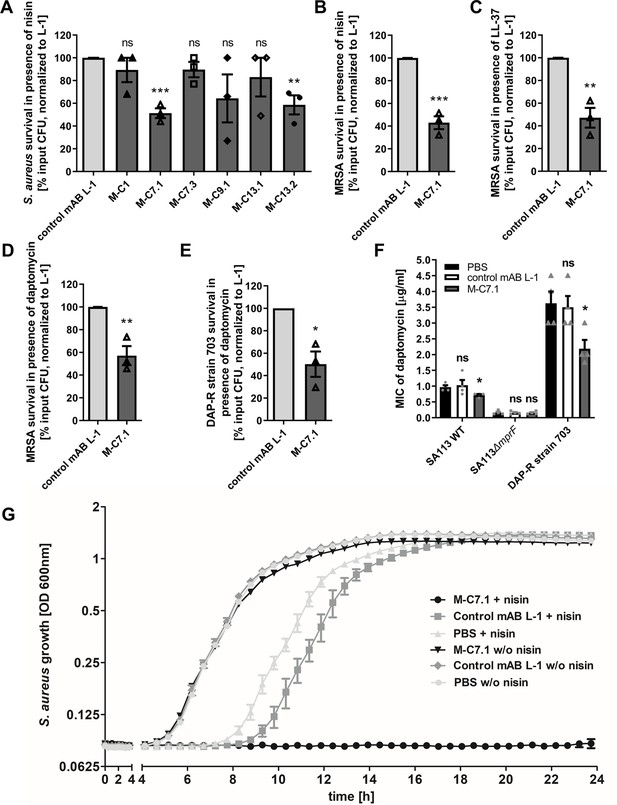
Killing and growth inhibition of S. aureus by antimicrobial peptides and antibiotics in the presence of M-C7.1.
(A) Survival of S. aureus SA113Δspa in the presence of nisin and 100 µg/ml anti-multiple peptide resistance factor (MprF) antibodies compared to 100 µg/ml control monoclonal antibody (mAB) L-1. Surviving colony-forming units (CFU) of S. aureus incubated with one of the antibodies and nisin were analyzed after 2 hr incubation and the negative control (isotype mAB L-1) was set to 100% survival. (B) Survival of community-associated methicillin-resistant S. aureus (CA-MRSA) wild-type (WT) strain USA300 in the presence of nisin and 100 µg/ml M-C7.1 compared to the isotype control mAB L-1. (C) Survival of USA300 WT in the presence of LL-37 and 100 µg/ml M-C7.1 compared to the isotype control mAB L-1. (D) Survival of USA300 WT in the presence of daptomycin and 100 µg/ml M-C7.1 compared to the isotype control mAB L-1. (E) Survival of daptomycin-resistant (DAP-R) CA-MRSA strain 703 (Jones et al., 2008) in the presence of daptomycin and 100 µg/ml M-C7.1 compared to the isotype control mAB L-1. (F) Daptomycin MICs of SA113 WT, SA113ΔmprF and DAP-R strain 703 when pretreated with PBS compared to 100 µg/ml control mAB L-1 and to 100 µg/ml M-C7.1. (G) Growth inhibition of USA300 WT in the presence of 4 µg/ml nisin and 1 µM M-C7.1 compared to 1 µM isotype control mAB L-1. Wells without nisin and/or antibodies served as additional negative controls. The means + standard error of the mean (SEM) of results from at least three biological replicates are shown in (A)–(F). (G) shows means + SEM of technical triplicates from a representative experiment of three biological replicates. Values for M-C7.1 or other anti-MprF antibodies that were significantly different from those for the isotype control mAB L-1 in (A)–(F), calculated by Student’s paired t-test are indicated (*p < 0.05; **p < 0.01; ***p < 0.0001).
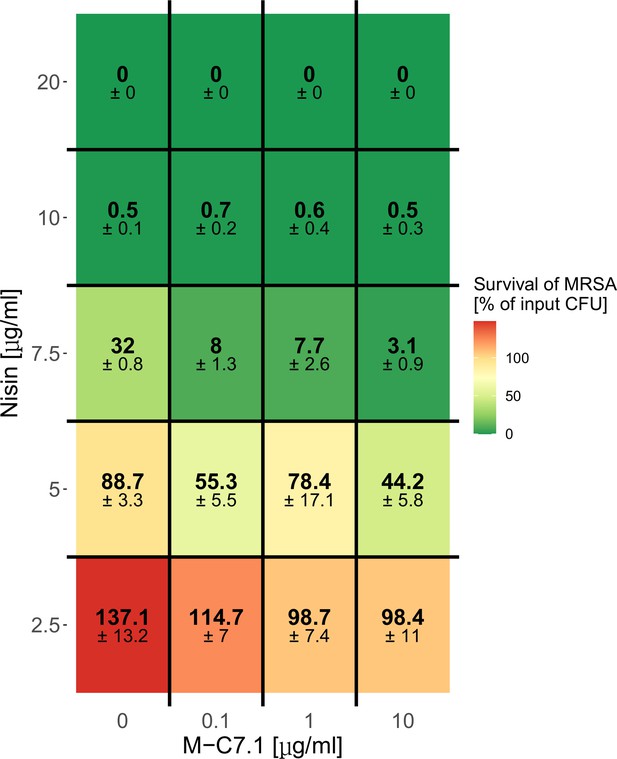
Dose-dependent support of M-C7.1 of S. aureus killing by nisin.
Survival of S. aureus USA300 in the presence of increasing concentrations of nisin and increasing concentrations of M-C7.1. Surviving colony-forming units (CFUs) of samples were analyzed after 2 hr incubation (‘killing’), the control CFU (0 µg/ml nisin) (‘input’) was set to 100%. Percentages of surviving CFUs are indicated and underlaid with a color scale. The means + standard error of the mean (SEM) of results from three independent experiments are shown.
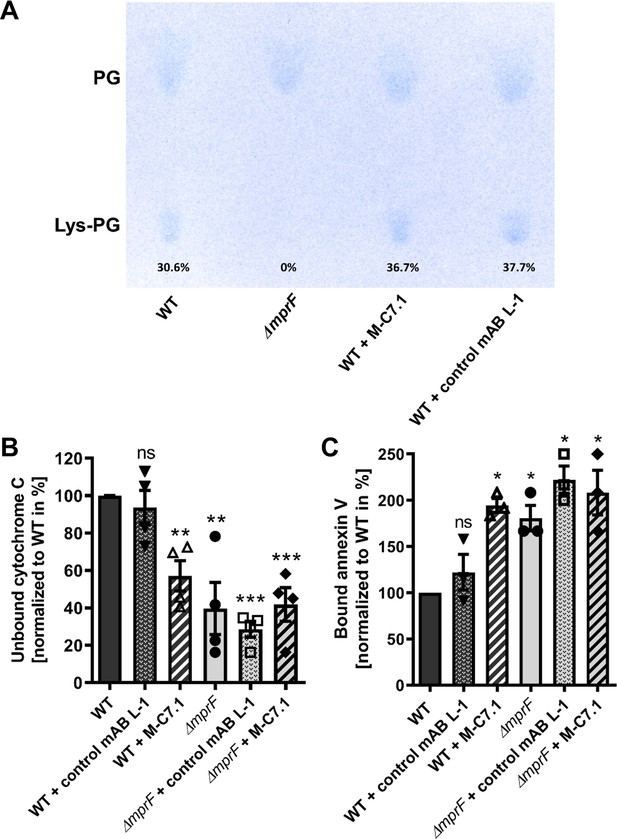
M-C7.1 inhibits the multiple peptide resistance factor (MprF) lysyl-phosphatidylglycerol (LysPG) flippase but not the LysPG synthase.
(A) Detection of phospholipids from S. aureus SA113ΔmprF and wild-type (WT) treated or not treated with 100 µg/ml M-C7.1 or the isotype control monoclonal antibody (mAB) L-1. Polar lipids were separated by thin layer chromatography (TLC) and stained with the phosphate group-specific dye molybdenum blue to detect the well-documented phosphatidylglycerol (PG) and LysPG pattern of S. aureus WT and mprF deletion mutant (Slavetinsky et al., 2012). Percentages of LysPG in relation to total phospholipid content are given below LysPG spots. (B) The repulsion of positively charged cytochrome C corresponds to MprF LysPG synthase plus flippase activity while the synthase activity alone does not affect repulsion. To assess MprF flippase efficiency, unbound cytochrome C in the supernatant was quantified photometrically after incubation with the S. aureus SA113 WT without pretreatment, or with pretreated with M-C7.1 or the isotype control mAB L-1 (WT set to 100%). SA113ΔmprF with or without mAB pretreatment served as positive control. The means + standard error of the mean (SEM) of results from three biological replicates are shown. (C) Annexin V binding to S. aureus SA113 WT compared to incubation with M-C7.1 or the isotype control mAB L-1 was quantified by measuring cell-bound annexin V by fluorescence-activated cell sorting (FACS) and untreated WT samples were set to 100%. SA113ΔmprF with and without mAB incubation served as positive control. Data are expressed as % of untreated WT cells. The means + SEM of results from three biological replicates are shown in (B) and (C). Significant differences compared to WT samples were calculated by Student’s paired t-test (*p < 0.05; **p < 0.01; ***p < 0.0001).
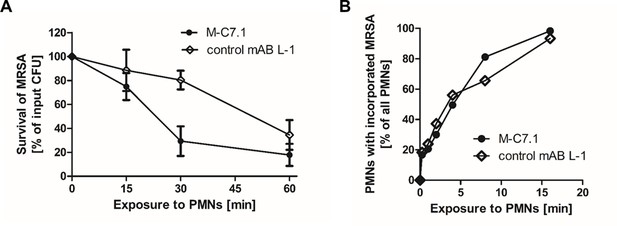
M-C7.1 supports S. aureus clearance by isolated human polymorphonuclear leukocytes (PMNs).
(A) Kinetics of killing of community-associated methicillin-resistant S. aureus (CA-MRSA) strain USA300 wild-type (WT) treated with M-C7.1 compared to isotype control monoclonal antibody (mAB) L-1 by freshly isolated human PMNs. Viable bacteria (colony-forming units, CFU) after incubation with PMNs are shown as percentage of initial CFU counts. The means + standard error of the mean (SEM) of results from three biological replicates are shown. (B) Kinetics of phagocytosis of USA300 WT treated with M-C7.1 compared to isotype control mAB L-1 by freshly isolated human PMNs. Percentages of PMNs bearing fluorescein-5-isothiocyanate (FITC)-labeled USA300 are given. Means of three counts from a representative experiment are shown.
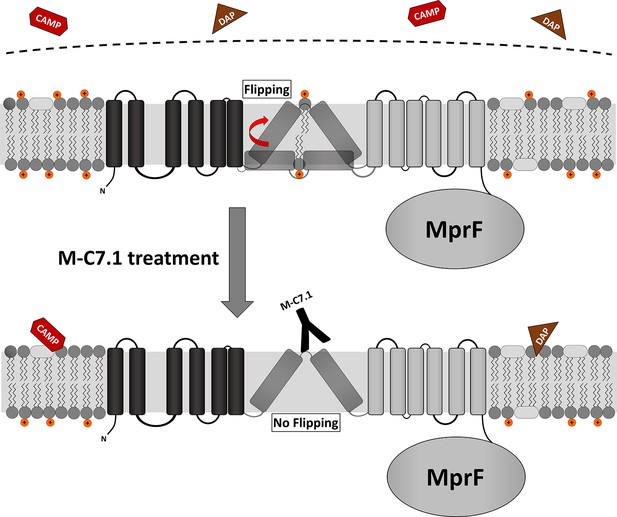
Proposed model for multiple peptide resistance factor (MprF) inhibition by M-C7.1.
Flipping of positively charged lysyl-phosphatidylglycerol (LysPG) probably by transmembrane segment (TMS) 7 and 8 of MprF results in a more positively charged staphylococcal membrane, which is better able to repulse cationic antimicrobial peptides (CAMPs) or daptomycin (DAP). Binding of M-C7.1 to MprF loop 7 blocks the flippase, which results in a more negatively charged staphylococcal membrane and subsequently in an increased S. aureus membrane disruption by CAMPs and daptomycin.
Additional files
-
Supplementary file 1
Multiple peptide resistance factor (MprF)-directed antibodies, bacterial strains, plasmids, and primers.
(a) MprF-directed antibodies and its target peptides. (b) Bacterial strains used in this study. (c) Plasmids used in this study. (d) Primers used in this study.
- https://cdn.elifesciences.org/articles/66376/elife-66376-supp1-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/66376/elife-66376-transrepform1-v2.docx





