Presynaptic NMDA receptors facilitate short-term plasticity and BDNF release at hippocampal mossy fiber synapses
Figures
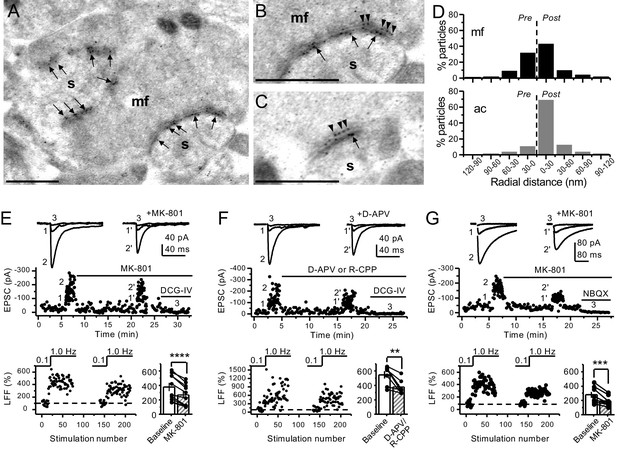
Anatomical and functional evidence for preNMDARs at mossy fiber synapses.
(A) Image of a mossy fiber (mf) giant bouton and postsynaptic spines (s). (B, C) Higher magnification of mf synapses. Arrows indicate postsynaptic GluN1, whereas arrowheads indicate presynaptic GluN1. Calibration bars: 500 nm. (D) Mossy fiber (mf) and associational–commissural (ac) synaptic GluN1 immuno-particle radial distribution (30 nm bins), mf: 34 synapses, 100 presynaptic particles; ac: 25 synapses, 24 presynaptic particles; three animals. (E) AMPAR-ESPCs were recorded at Vh = −70 mV in the presence of 0.5 µM LY303070 and 100 µM picrotoxin. Low-frequency facilitation (LFF), induced by stepping stimulation frequency from 0.1 to 1 Hz, was assessed before and after bath application of MK-801 (50 µM). MK-801 significantly reduced LFF (baseline 378 ± 57%, MK-801 270 ± 48%, n = 10 cells, nine animals; baseline vs MK-801, p=3.8×10−5, paired t-test). In all panels of this figure: representative traces (top), representative experiment (middle), and normalized LFF and summary plot (bottom). DCG-IV (1 µM) was applied at the end of all recordings to confirm mf-CA3 transmission. (F) D-APV (100 µM) or R-CPP (50 µM) application also reduced LFF (baseline 546 ± 50%, D-APV/R-CPP 380 ± 38%, n = 7 cells, five animals; baseline vs D-APV/R-CPP, p=0.00743, paired t-test). (G) KAR-EPSCs were recorded at Vh = −70 mV in the presence of 15 µM LY303070 and 100 µM picrotoxin. In addition, NMDAR-mediated transmission was blocked intracellularly by loading MK-801 (2 mM) in the patch-pipette. Bath application of MK-801 (50 µM) significantly reduced LFF (baseline 278 ± 40%, MK-801 195 ± 26% n = 8 cells, six animals; baseline vs MK-801, p=0.00259, paired t-test). Data are presented as mean ± s.e.m. **p<0.01; ***p<0.005; ****p<0.001.
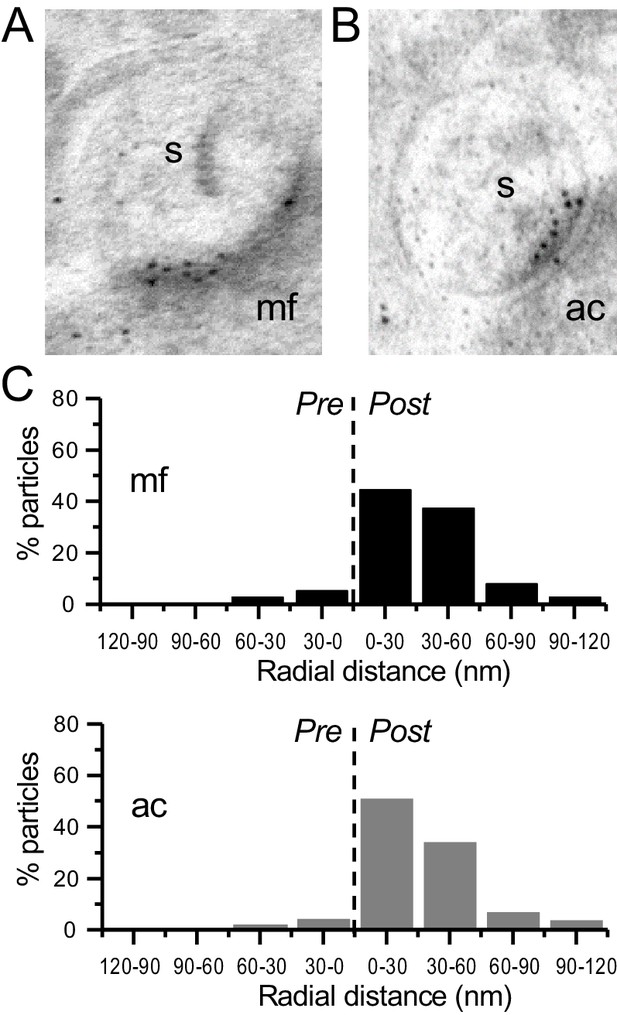
Immunogold-EM reveals negligible presynaptic AMPAR particle distribution.
(A, B) Images of mossy fiber (mf) and associational–commissural (ac) synapses, postsynaptic spines (s). (C) AMPAR immuno-particle distribution (30 nm bins), mf: 102 synapses, eight presynaptic particles; ac: 75 synapses, six presynaptic particles; three animals. Dashed line represents synaptic cleft.
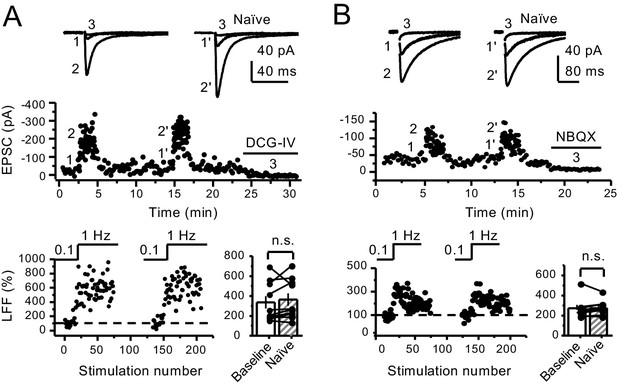
Stable low-frequency facilitation of mf-CA3 synaptic transmission in naïve slices.
(A) Stable low-frequency facilitation (LFF) of AMPAR-EPSCs. In naïve slices (interleaved experiments), LFF remained unchanged throughout the recording session (baseline 335 ± 62%, naïve 363 ± 63%, n = 10 cells, nine animals; p=0.185, Wilcoxon signed-rank test, baseline vs naïve). DCG-IV (1 µM) was applied at the end of all recordings to confirm mf-CA3 transmission. (B) LFF of KAR-EPSCs was also stable in interleaved, naïve slices (baseline 274 ± 33%, naïve 278 ± 25%, n = 9 cells, six animals; p=0.236, Wilcoxon signed-rank test, baseline vs naïve). NBQX (10 µM) was applied at the end of all recordings to confirm mf KAR transmission. Data are presented as mean ± s.e.m.
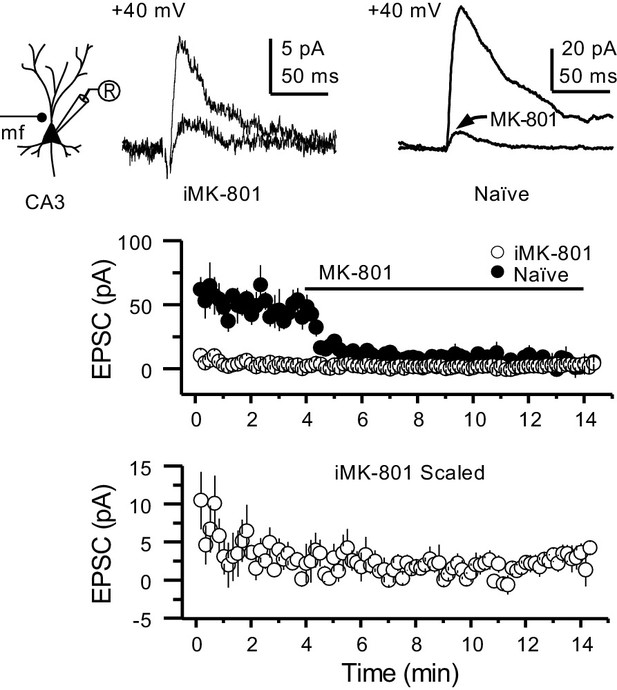
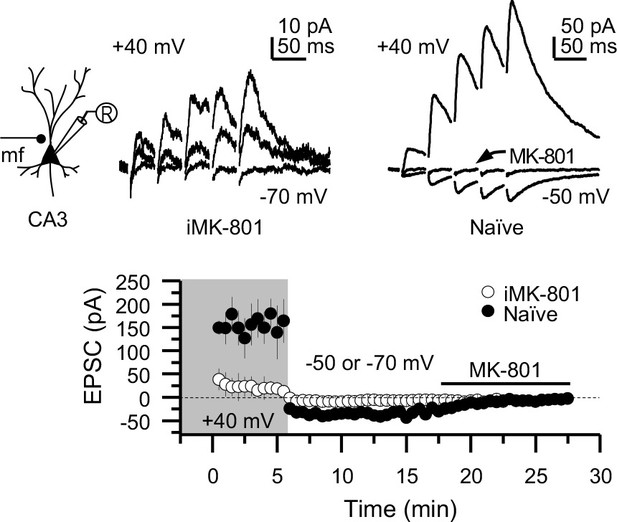
Intracellular MK-801 effectively blocked postsynaptic NMDARs.
Representative NMDAR-EPSCs (Vh = +40 mV) from CA3 pyramidal neurons patch-loaded with 2 mM MK-801 (left) or naïve internal solution (right). Mf inputs were stimulated with a bipolar electrode (theta-glass pipette) in stratum lucidum in the presence of picrotoxin (100 µM) and NBQX (10 µM). Bath application of MK-801 (50 µM) blocked NMDAR currents in naïve cells to a similar magnitude as cells patch-loaded with MK-801 (n = 5 cells, four animals in each condition; U = 0.676, Mann–Whitney test). Note that CA3 pyramidal neurons were loaded for at least 3–5 min before recording started at +40 mV.
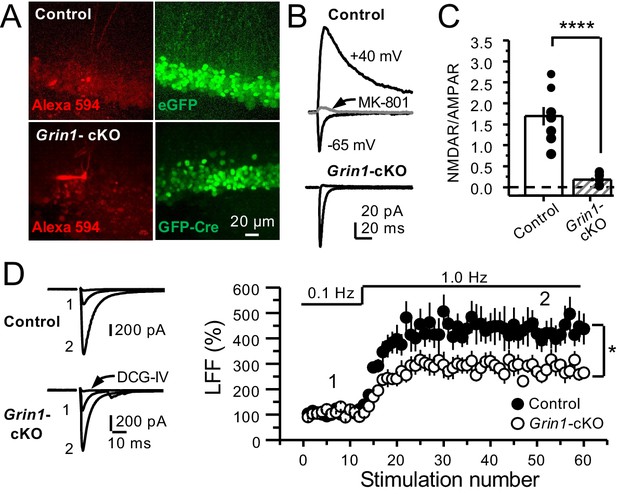
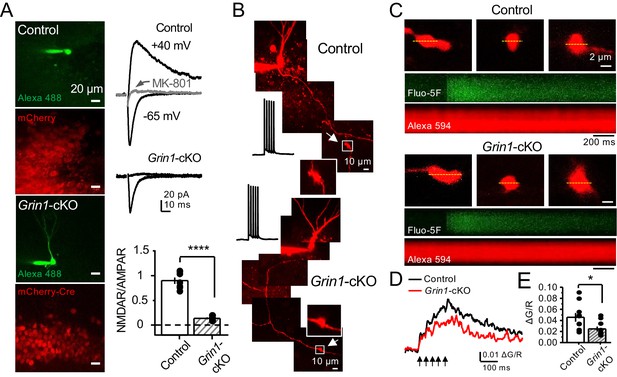
GluN1 deletion from GCs reduces mf-CA3 facilitation.
(A) Representative images showing GCs patch-loaded with Alexa 594 (35 µM) (left), and GFP expression in GCs (right). (B) Representative EPSCs recorded from control (GFP+) and Grin1-cKO (Cre-GFP+) GCs. Synaptic responses were elicited by activating medial perforant-path inputs. AMPAR-ESPCs were recorded at Vh = −65 mV in the presence of 100 µM picrotoxin, NMDAR-EPSCs were isolated with 10 µM NBQX and recorded at +40 mV. MK-801 (20 µM) was applied at the end of each recording. (C) Summary plot demonstrating that GluN1 deletion from GCs virtually abolished NMDAR-mediated transmission indicated by a strong reduction of NMDAR/AMPAR in Grin1-cKO GCs as compared to controls (control 1.61 ± 0.18, n = 9 cells, nine animals, Grin1-cKO 0.18 ± 0.04, n = 10 cells, 10 animals; control vs Grin1-cKO, p=9.2×10−6, unpaired t-test). (D) LFF was significantly reduced in GluN1-deficient animals (control, 430 ± 5%, n = 13 cells, 10 animals; Grin1-cKO, 291 ± 6%, n = 11 cells, 10 animals; p=0.0239, unpaired t-test). Representative traces (left) and summary plot (right). LFF was induced by stepping stimulation frequency from 0.1 to 1 Hz. DCG-IV (1 µM) was added at the end of each experiment. Data are presented as mean ± s.e.m. *p<0.05; ****p<0.001.
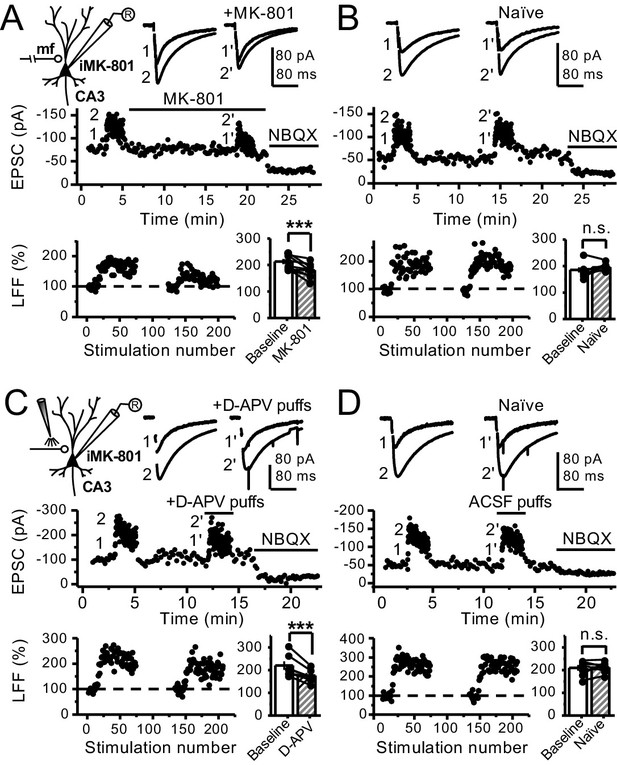
Reduced facilitation by NMDAR antagonism is independent of the GC somatodendritic compartment.
(A) KAR-EPSCs were recorded at Vh = −70 mV in the presence of 15 µM LY303070 and 100 µM picrotoxin. In addition, NMDAR-mediated transmission was blocked intracellularly by loading MK-801 (2 mM) in the patch-pipette. LFF of KAR-EPSCs was assessed as in Figure 1G but with transected mf axons (see Materials and methods). Bath application of MK-801 (50 µM) significantly reduced LFF (baseline 213 ± 9%, MK-801 181 ± 10%, n = 8 cells, seven animals; baseline vs MK-801, p=0.002, paired t-test). In all panels of this figure: recording arrangement (inset), representative traces (top), representative experiment (middle), normalized LFF and summary plot (bottom). (B) Stable LFF in transected, naïve slices (baseline 186 ± 10%, naïve 196 ± 5%, n = 8 cells, seven animals; baseline vs naïve, p=0.278, paired t-test). (C) LFF was induced before and during puff application of D-APV (2 mM) in stratum lucidum. This manipulation significantly reduced facilitation (baseline 220 ± 19%, D-APV puff 176 ± 11%, n = 7 cells, seven animals; baseline vs D-APV puff, p=0.003, paired t-test). (D) Stable LFF in acute slices during puff application of ACSF (baseline 210% ± 12, naïve 213% ± 9, n = 7 cells, seven animals; baseline vs naïve, p=0.778, paired t-test). NBQX (10 µM) was applied at the end of all recordings to confirm mf KAR transmission. Data are presented as mean ± s.e.m. ***p<0.005.
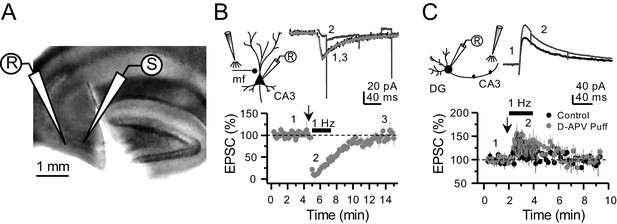
Targeting preNMDARs located in mf axons, but not granule cells.
(A) Field view of a representative hippocampal slice showing a surgical cut between DG and CA3. (B) Local D-APV puff application (vertical arrow, two puffs at 0.1 Hz) blocks NMDAR currents recorded at Vh = −50 mV and washes out in less than 10 min (n = 7 cells, five animals, p=5×10−8, paired t-test). Inset depicts the recording paradigm of the experiment (left), the representative NMDAR currents (top) and the summary time course (bottom) where arrows denote the onset of D-APV (2 mM) puff application. Mfs were stimulated with a bipolar electrode (theta-glass pipette) in stratum lucidum in the presence of 100 µM picrotoxin and 10 µM NBQX. (C) D-APV puff application in CA3 did not reduce NMDAR transmission in GCs (n = 6 cells, five animals, control vs D-APV puff, U = 0.594, Mann–Whitney test). Excitatory inputs were stimulated with a monopolar electrode placed in the medial molecular layer/inner molecular layer, in the presence of 100 µM picrotoxin and 10 µM NBQX, while GCs were clamped at Vh = +40 mV. Data are presented as mean ± s.e.m.
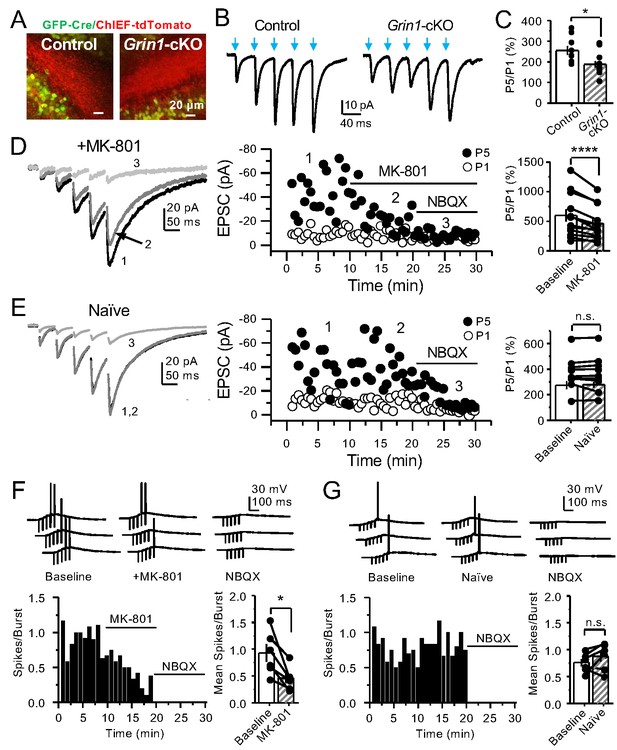
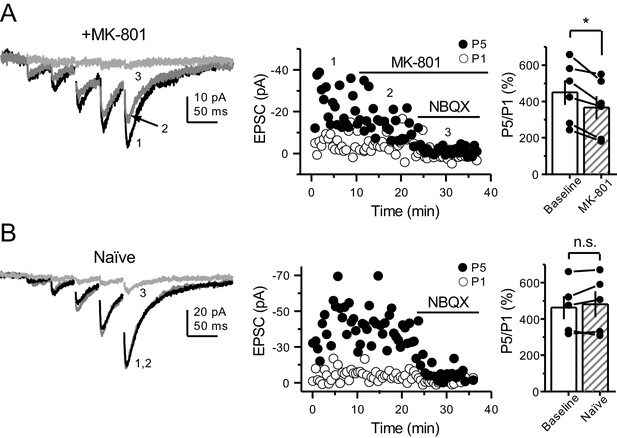
PreNMDARs contribute significantly to burst-induced facilitation and spike transfer.
(A) Representative images showing expression of GFP-Cre (left) and ChIEF-tdTomato (right) in the DG of control and Grin1-cKO animals. (B) Representative AMPAR-EPSCs from control (left) and Grin1-cKO (right) CA3 pyramidal neurons recorded at Vh = −65 mV and evoked by optical burst-stimulation (5 pulses at 25 Hz) of stratum lucidum. Blue arrows indicate light stimulation. (C) Summary plot of burst-induced facilitation measured as P5/P1 ratio of optical responses; facilitation was significantly reduced in Grin1-cKO animals as compared to control (Grin1-cKO 187 ± 16%, n = 12 cells, nine animals; control 255 ± 22%, n = 9 cells, eight animals; Grin1-cKO vs control, p=0.0167, unpaired t-test). (D) Burst stimulation induced KAR-EPSCs were isolated and recorded as described in Figure 3, bath application of MK-801 (50 µM) significantly reduced facilitation (baseline 601 ± 107%, MK-801 464 ± 84%, n = 13 cells, 10 animals; baseline vs MK-801, p=0.00042, paired t-test). In (D) and (E) of this figure: representative traces (left), representative experiment (middle), and summary plot (right). (E) Burst-induced facilitation was stable in interleaved, naïve slices (baseline 369 ± 45%, naïve 367 ± 48%, n = 9 cells, nine animals; p=0.863, paired t-test). (F) Bath application of MK-801 (50 µM) reduced KAR-mediated action potentials induced by burst-stimulation (baseline 0.93 ± 0.17, MK-801 0.46 ± 0.09, n = 6 cells, five animals; p=0.036, Wilcoxon signed-rank test). In (F) and (G) of this figure: representative traces (top), representative experiment and summary plot (bottom). (G) Stable KAR-mediated action potentials in interleaved naïve slices (baseline 0.76 ± 0.07, naïve 0.88 ± 0.1, n = 6 cells, five animals; p=0.2084, Wilcoxon signed-rank test). NBQX (10 µM) was applied at the end of all experiments in (D–G). Data are presented as mean ± s.e.m. *p<0.05; ****p<0.001.
PreNMDARs contribute to burst-induced facilitation in more physiological conditions: 1.2 mM Mg+2, 1.2 mM Ca+2 and 35°C.
KAR-EPSCs were recorded from CA3 pyramidal cells loaded with 2 mM MK-801 in the presence of 15 µM LY303070 and 100 µM picrotoxin. (A) Bath application of MK-801 (50 µM) significantly reduced burst-induced facilitation elicited by 5 pulses, 25 Hz (baseline 450 ± 67%, MK-801 366 ± 63%, n = 6 cells, four animals; baseline vs MK-801, p=0.036, Wilcoxon signed-rank test). In (A) and (B) of this figure: representative traces (left), representative experiment (middle), and summary plot (right). (B) Burst-induced facilitation was stable in interleaved, naïve slices (baseline 462 ± 63%, naïve 481 ± 71%, n = 5 cells, four animals; p=0.281, Wilcoxon signed-rank test). Data are presented as mean ± s.e.m. *p<0.05.
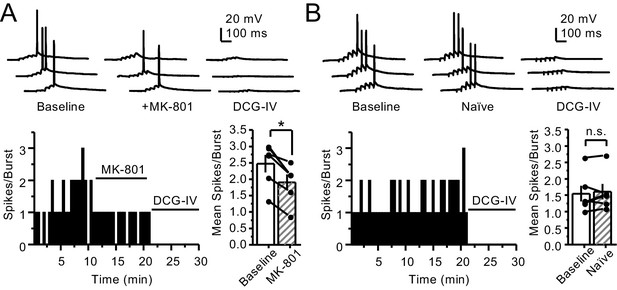
PreNMDARs contribute to action potential firing elicited by AMPAR-mediated transmission.
(A) Bath application of MK-801 (50 µM) reduced action potentials induced by 5 pulses at 25 Hz burst stimulation (baseline 2.47 ± 0.27, MK-801 1.9 ± 0.24, n = 6 cells, five animals; p=0.036, Wilcoxon signed-rank test). In (A) and (B) of this figure: representative traces (top), representative experiment, and summary plot (bottom). (B) Stable action potential firing in interleaved naïve slices (baseline 1.55 ± 0.24, naïve 1.61 ± 0.23, n = 6 cells, five animals; p=0.402, Wilcoxon signed-rank test). DCG-IV (1 µM) was applied at the end of all experiments in (A, B). Data are presented as mean ± s.e.m. *p<0.05.
Intracellular MK-801 effectively blocked postsynaptic NMDARs elicited by burst stimulation (5 pulses at 25 Hz).
Representative NMDAR-EPSCs (Vh = +40 mV) from CA3 pyramidal neurons patch-loaded with 2 mM MK-801 (left) or naïve internal solution (right). Mf inputs were stimulated with a bipolar electrode (theta-glass pipette) in stratum lucidum in the presence of picrotoxin (100 µM) and NBQX (10 µM). NMDAR currents were recorded at Vh = +40 mV (gray shaded area) followed by a voltage jump to −70 mV in iMK-801 conditions and −50 mV in naïve recordings. Bath application of MK-801 (50 µM) blocked NMDAR currents of the fifth pulse to a similar magnitude as iMK-801 (n = 5 cells, four animals per condition; U = 0.21, Mann–Whitney test). Data are presented as mean ± s.e.m.
preNMDARs contribute to presynaptic Ca2+ rise.
(A) Representative images showing GCs patch-loaded with Alexa 488 (35 µM) to confirm expression of mCherry (bottom). Representative AMPAR-EPSCs recorded from control (top) or Grin1-cKO (middle) GCs. Synaptic responses were elicited by activating medial perforant-path inputs. AMPAR-ESPCs were recorded at Vh = −65 mV in the presence of 100 µM picrotoxin, NMDAR-EPSCs were isolated with 10 µM NBQX and recorded at +40 mV. MK-801 (20 µM) was applied at the end of each experiment. Summary plot (bottom) demonstrating that GluN1 deletion from GCs virtually abolished NMDAR-mediated transmission indicated by a strong reduction of NMDAR/AMPAR in Grin1-cKO granule cells as compared to controls (control 0.90 ± 0.17, n = 7 cells, six animals; Grin1-cKO 0.13 ± 0.05, n = 6 cells, six animals; control vs Grin1-cKO, p=3.81×10−7, unpaired t-test). (B) Representative control and Grin1-cKO GCs patch-loaded with Fluo-5F (200 µM) and Alexa 594 (35 µM). Arrows indicate the identification of a mf giant bouton, magnified images in white box. (C) Three representative mf boutons (top) and line scan image of calcium transients (CaTs) elicited by five action potentials at 25 Hz (middle, Fluo-5F) and morphological dye (bottom, Alexa 594), in Control and Grin1-cKO animals. Dotted line (yellow) indicates line scan location. Red Channel, Alexa 594; Green Channel, Fluo-5F. (D, E) Peak analysis of the fifth pulse ΔG/R revealed a significant reduction in Ca2+ rise of Grin1-cKO animals as compared to Control (control 0.046 ± 0.01, n = 10 boutons, three line scans per bouton, eight animals; Grin1-cKO 0.025 ± 0.004, n = 10 boutons, eight animals; control vs Grin1-cKO, U = 0.017, Mann–Whitney test). Arrows indicate mf activation. Data are presented as mean ± s.e.m. *U < 0.05; ****p<0.001.
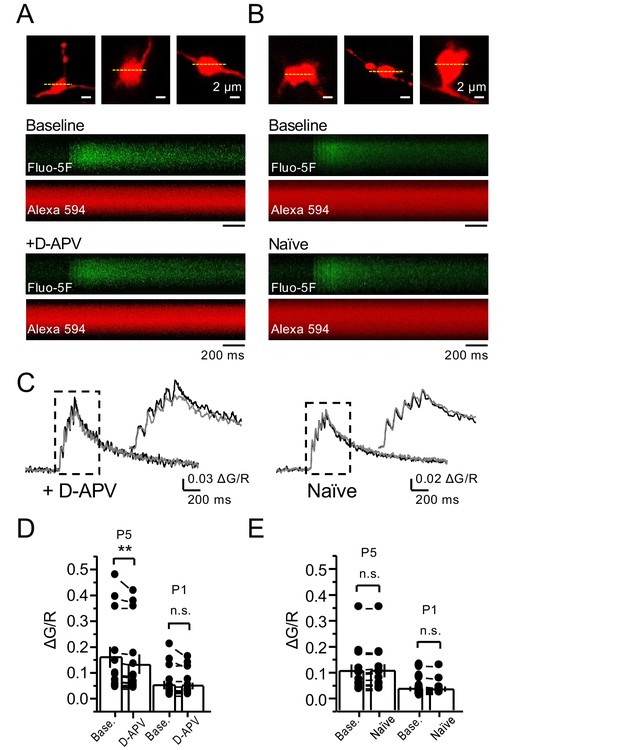
NMDAR antagonism reveals a reduction in presynaptic Ca+2 rise in the presence of 1.3 mM Mg+2 and 2.5 mM Ca+2.
(A, B) Granule cells were patch-loaded with Fluo-5F (200 µM) and Alexa 594 (35 µM). Line scan analysis of mf giant bouton calcium transients (CaTs) in response to action potential (AP) stimulation (5 APs, 25 Hz). (C) Line scan signals following D-APV application or naïve conditions. (D) D-APV (100 µM) significantly reduced the fifth peak (P5) of CaTs (baseline 0.155 ± 0.04, D-APV 0.138 ± 0.03, n = 13 boutons, three line scans per bouton, 10 animals; baseline vs D-APV, p=0.00642, Wilcoxon signed-rank test). (E) In naïve conditions P5 of CaTs is stable (baseline 0.104 ± 0.026, naïve 0.105 ± 0.026, n = 12 boutons, three line scans per bouton, 10 animals; baseline vs naïve, p=0.255, Wilcoxon signed-rank test). The first peak (P1) of CaTs is not affected by D-APV (baseline 0.05 ± 0.017; D-APV 0.047 ± 0.014; baseline vs D-APV, p=0.485, Wilcoxon signed-rank test) and is stable in naïve conditions (baseline 0.033 ± 0.009; naïve 0.032 ± 0.009, baseline vs naïve, p=0.196, Wilcoxon signed-rank test). Data are presented as mean ± s.e.m. **p<0.01.
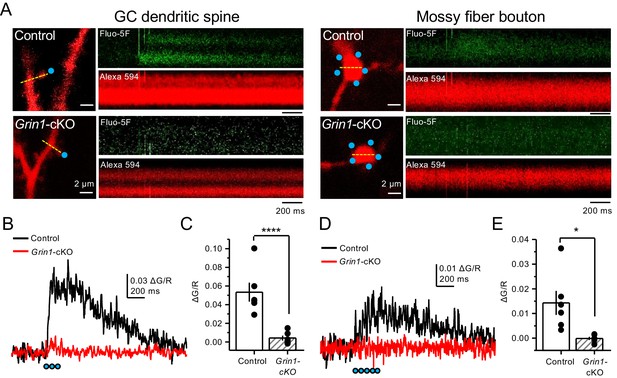
Uncaging glutamate induces Ca2+ rise in mossy fiber boutons.
(A) Representative images showing dendritic spines in GCs (left) and mf boutons (right), and the associated line scan image of calcium transients (CaTs) elicited by uncaging of MNI-glutamate (see Materials and methods), in control and Grin1-cKO animals. Blue dots indicate uncaging spots. Red channel, Alexa 594; Green channel, Fluo-5F. (B) Line scan analysis of CaTs measuring ΔG/R in dendritic spines when MNI-glutamate is uncaged in control or Grin1-cKO animals. Blue dots indicate location of two-photon uncaging (2PU) pulses. (C) Summary plot demonstrating a significant reduction in dendritic spine CaTs in Grin1-cKO as compared to Control (control 0.053 ± 0.01 ΔG/R, n = 6 dendritic spines, three line scans per spine, six animals; Grin1-cKO 0.004 ± 0.003 ΔG/R, n = 6 spines, three line scans per spine, six animals; ΔG/R control vs Grin1-cKO, p=0.00088, unpaired t-test). (D) Line scan analysis of CaTs measuring ΔG/R in mf boutons when MNI-glutamate is uncaged in control or Grin1-cKO animals. (E) Summary plot demonstrating significant CaTs in boutons of control as compared to Grin1-cKO (control 0.014 ± 0.005, n = 6 boutons, three line scans per bouton, six animals; Grin1-cKO −0.00012 ± −0.0006, n = 6 boutons, three line scans per bouton, six animals; control vs Grin1-cKO, p=0.015, unpaired t-test). Data are presented as mean ± s.e.m. *p<0.05; ****p<0.001.
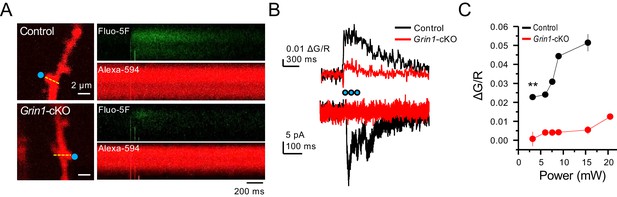
Grin1-cKO exhibit reduced CaTs at varying uncaging laser power intensities.
(A) Representative images of CaTs from control (top) and Grin1-cKO animals (bottom) after MNI-glutamate uncaging (2 mM, 3 pulses at 25 Hz) on GC dendritic spines. Dotted line (yellow) indicates line scan, and blue dots indicate 2PU spots. (B) Quantified ΔG/R signals (top) and uncaging induced NMDAR-EPSCs (bottom) from control and Grin1-cKO animals. Blue dots indicate when 2PU pulses were delivered. (C) Control animals display robust ΔG/R signals as compared to Grin1-cKO animals at varying laser power intensities (six spines, three line scans per spine, six animals per group, U = 0.00507 per power intensity, Mann–Whitney test). Data are presented as mean ± s.e.m. ** U < 0.01.
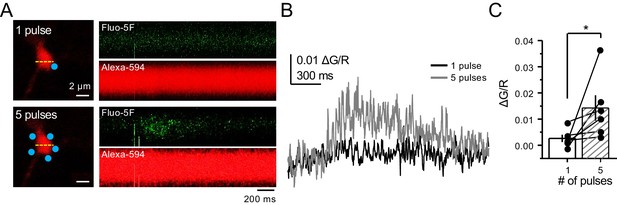
Bouton CaTs can be detected after repetitive uncaging of MNI-glutamate.
(A) Representative images of CaTs from single-trial: 1 pulse (top) and 5 pulses, 25 Hz (bottom) of MNI-glutamate uncaging (2 mM). Dotted line (yellow) indicates line scan, and blue dots indicate 2PU spots. (B) Quantified ΔG/R signals from 1 pulse (black) and 5 pulses (dark gray) from all trials. (C) Repetitive pulses result in larger ΔG/R signals as compared to single pulses (n = 6 boutons, three line scans per bouton, six animals, p=0.03603, Wilcoxon signed-rank test). Data are presented as mean ± s.e.m. *p<0.05.
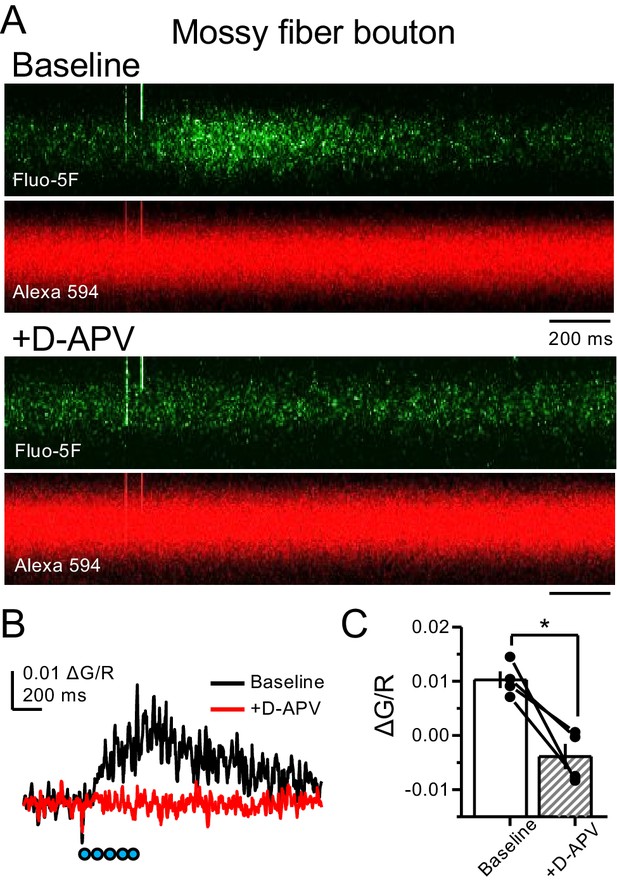
NMDAR antagonism with D-APV blocks CaTs elicited by glutamate 2PU.
(A) Representative image of baseline glutamate uncaging driven CaTs in mf boutons (top). D-APV application (100 µM) blocks CaTs (bottom). (B) Quantified ΔG/R signals before and after D-APV application. (C) Summary data of D-APV block on glutamate uncaging elicited CaTs (baseline 0.0103 ± 0.0016, D-APV −0.004 ± 0.0024, n = 4 boutons, three line scans per bouton, three animals; U = 0.0304, Mann–Whitney test, baseline vs D-APV). Data are presented as mean ± s.e.m. *U < 0.05.
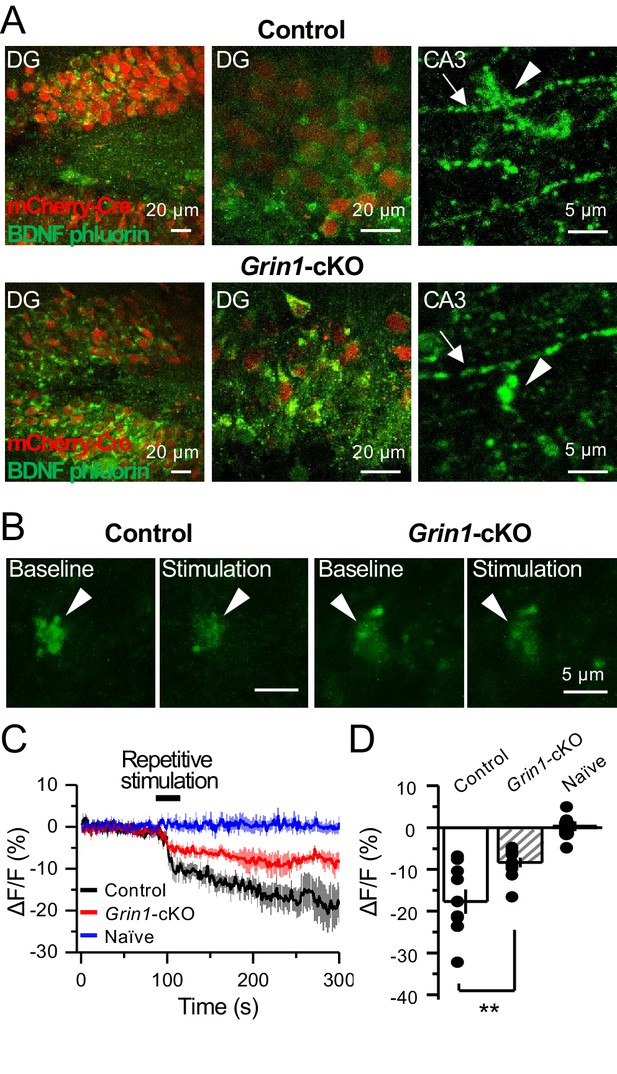
preNMDARs contribute significantly to BDNF release following repetitive activity.
(A) Representative images showing expression of BDNF-pHluorin in the DG and CA3 area (arrows indicate mf axon, arrowheads indicate mf boutons). Control images (top), Grin1-cKO images (bottom). (B) Representative images of BDNF-pHluorin signal intensity at baseline and after repetitive stimulation of mfs (125 pulses, 25 Hz, ×2). Control images (left), Grin1-cKO images (right), arrowhead indicates region of interest. (C) Time course of BDNF-pHluorin signal intensity measured as ΔF/F (%): control (black), Grin1-cKO (red), Naïve (blue). (D) Quantification of BDNF-pHluorin signal in (C) during the last 100 s reveals larger BDNF release in control animals as compared to Grin1-cKO (control −18% ± 3%, n = 9 slices, five animals; Grin1-cKO −8 ± 1%, n = 10 slices, five animals; Grin1-cKO vs control, p=0.00648, unpaired t-test). Data are presented as mean ± s.e.m. **p<0.01.
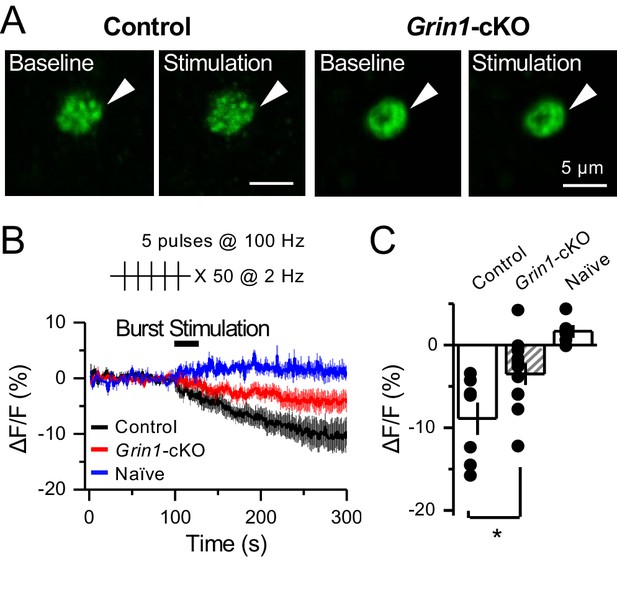
preNMDARs contribute significantly to BDNF release following a more physiological pattern of burst stimulation.
(A) Representative images of BDNF-pHluorin signal intensity at baseline and after burst stimulation of mfs (5 pulses, 100 Hz, ×50, every 0.5 s). Control images (left), Grin1-cKO images (right), arrowhead indicates region of interest. (B) Time course of BDNF-pHluorin signal intensity measured as ΔF/F (%): control (black), Grin1-cKO (red), Naïve (blue). (C) Quantification of BDNF-pHluorin signal in (B) during the last 100 s reveals larger BDNF release in control animals as compared to Grin1-cKO (control −8.9% ± 2%, n = 7 slices, five animals; Grin1-cKO −3.5 ± 1%, n = 11 slices, five animals; Grin1-cKO vs control, p=0.0305, unpaired t-test). Data are presented as mean ± s.e.m. *p<0.05.
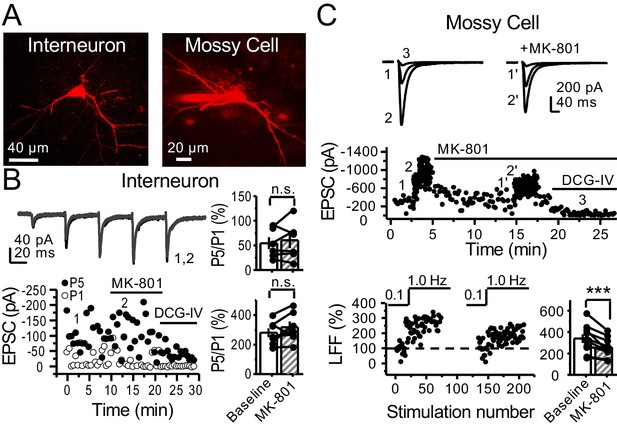
preNMDARs contribute to synaptic facilitation of mossy fiber inputs onto mossy cells, but not onto CA3 inhibitory interneurons.
(A) Representative images showing a CA3 IN and a hilar MC patch-loaded with Alexa 594 (35 µM) for morphological identification in acute rat hippocampal slices. (B) AMPAR-EPSCs were recorded from CA3 INs at Vh = −65 mV and burst stimulation was elicited by 5 pulses at 25 Hz, see traces (top). Representative experiment (bottom, left), and summary plots (right) showing bath application of MK-801 (50 µM) had no significant effect on depression (top, right) or facilitation (bottom, right) measured by P5/P1 ratio (baseline 54 ± 12%, MK-801 60 ± 16%, n = 6 cells; MK-801 vs baseline, p=0.675, Wilcoxon signed-rank test; baseline 281 ± 30%, MK-801 318 ± 37%, n = 7 cells; MK-801 vs baseline, p=0.178, paired t-test, five animals in each data set). (C) AMPAR-ESPCs were recorded at Vh = −70 mV from MCs, LFF was induced by stepping stimulation frequency from 0.1 to 1 Hz, see traces (top). Representative experiment (middle), normalized LFF and summary plot (bottom) indicating bath application of MK-801 (50 µM) reduced facilitation (baseline 339 ± 41%, MK-801 258 ± 29%, n = 10 cells, six animals; baseline vs MK-801, p=0.00152, paired t-test). DCG-IV (1 µM) was applied at the end of all experiments. Data are presented as mean ± s.e.m. ***p<0.005.
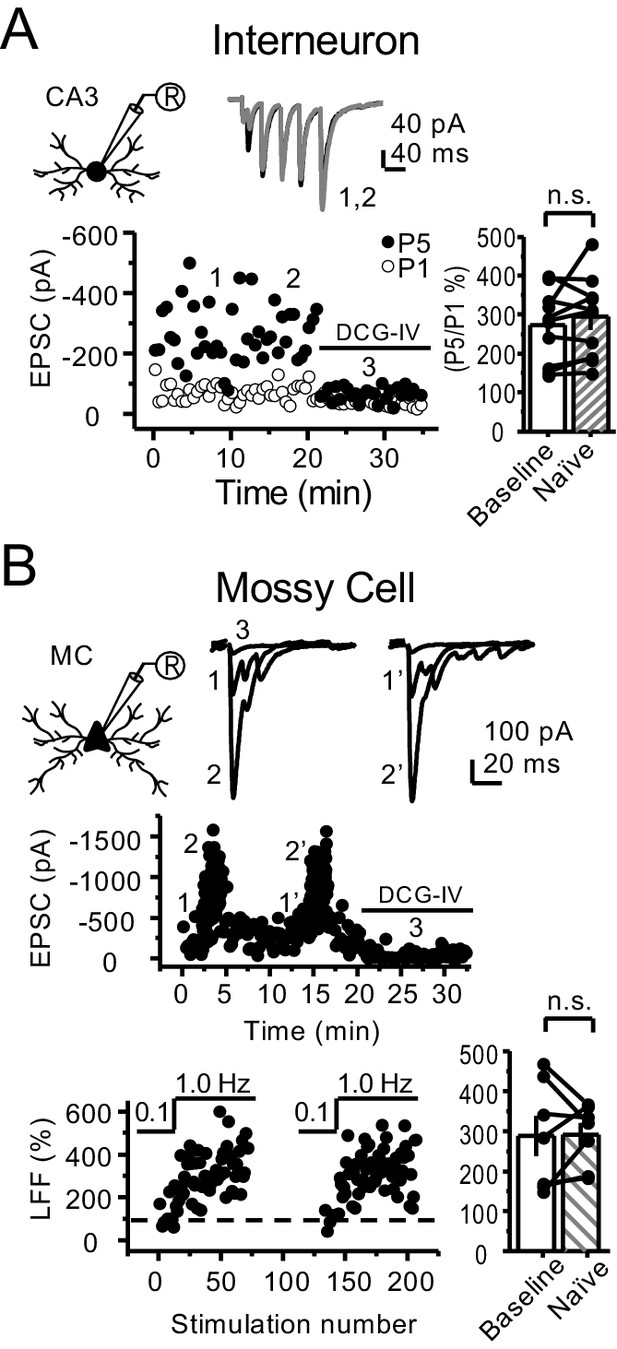
Stability experiments for mf-Interneuron and mf-mossy cell short-term plasticity.
(A) Stable CA3 IN burst-induced facilitation of mf-CA3 transmission (baseline 273 ± 30%, naïve 294 ± 33%, n = 10 cells, six animals; p=0.298, paired t-test, baseline vs naïve). (B) Stable low-frequency facilitation (LFF) of AMPAR-EPSCs in hilar MCs (baseline 288 ± 51%, naïve 291 ± 29%, n = 7 cells, six animals; p=0.937, paired t-test, baseline vs naïve). DCG-IV (1 µM) was applied at the end of all recordings to confirm mf-CA3 transmission. Data are presented as mean ± s.e.m.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information | |
|---|---|---|---|---|---|
| Strain, strain background (Rattus norvegicus male and female) | Rat: Sprague-Dawley | Charles River | Strain code: 400 | ||
| Strain, strain background (Mus musculus, male and female) | Mouse: Grin1fl/fl(B6.129S4-Grin1tm2Stl/J) | Dr. Michael Higley/The Jackson Laboratory | RRID:IMSR_JAX005246 | ||
| Strain, strain background (Mus musculus male and female) | Mouse: C57Bl6/J | Charles River | Strain code: 027 | ||
| Antibody | (Include host species and clonality) Mouse, Monoclonal, anti-NMDAR1 | Millipore | Cat# MAB363 | 10 μg/mL | |
| Antibody | Rabbit, Polyclonal, anti-GluA1-4, (pan-AMPA) | Dr. Elek Molnar/Bristol University | Generated by Dr. Elek Molnar | 10 μg/mL | |
| Antibody | Goat anti-rabbit IgG conjugated gold particles | Nanoprobes Inc | #2003–0.5 ML | (1:100) | |
| Recombinant DNA reagent | AAV5-CaMKII-GFP-Cre | Penn Vector Core | AV-5-PV2521 | Available on Addgene | |
| Recombinant DNA reagent | AAV5-CaMKII-eGFP | Penn Vector Core | AV-5-PV1917 | Available on Addgene | |
| Recombinant DNA reagent | AAV5-CaMKII-mcherry-Cre | UNC Vector Core | See website | https://wwwmed.unc.edu/genetherapy/vectorcore/in-stock-aav-vectors/ | |
| Recombinant DNA reagent | AAV5-CaMKII-mcherry | UNC Vector Core-Dr. Karl Deisseroth Control Fluorophores | See website | https://wwwmed.unc.edu/genetherapy/vectorcore/in-stock-aav-vectors/ | |
| Recombinant DNA reagent | AAV-DJ-flex-OChIEF-tdTomato | Dr. Pascal Kaeser PMID:29398114 | Generated at UNC Vector Core | Custom Order | |
| Recombinant DNA reagent | AAV-DJ-DIO-BDNF-phluorin | Dr. Hyungju Park PMID:25467984 | Generated at UNC Vector Core | Custom Order | |
| Chemical compound, drug | Ketamine | Merial | Cat# 03661103001904 | ||
| Chemical compound, drug | Xylazine | Calier | Cat# 20100–003 | ||
| Chemical compound, drug | Paraformaldehyde | Scharlau | PA0095 | ||
| Chemical compound, drug | Glutaraldehyde | Electron Microscopy Sciences | Cat# 16210 | ||
| Chemical compound, drug | Picric Acid | Panreac | Cat# 141048.1609 | ||
| Chemical compound, drug | Phosphate Buffer | Scharlau | SO03321000 | ||
| Chemical compound, drug | Human serum albumin | SigmaMillipore | A-1653 | ||
| Chemical compound, drug | TBS | TRIZMA BASE | SigmaMillipore | T1503 | |
| Trizma HCl | T3253 | ||||
| Chemical compound, drug | Triton X-100 | SigmaMillipore | T8787 | ||
| Chemical compound, drug | Polyethylene glycol | SigmaMillipore | 25322-68-3 | ||
| Chemical compound, drug | Uranyl acetate | Electron Microscopy Sciences | Cat# 22400 | ||
| Chemical compound, drug | Reynold’s lead citrate | Electron Microscopy Sciences | #17800 | ||
| Chemical compound, drug | Picrotoxin | SigmaMillipore | Cat# P1675 | ||
| Chemical compound, drug | LY303070 | ABX Chemical Co. | N/A | Custom Order | |
| Chemical compound, drug | MK-801 | Tocris Bioscience | Cat# 0924 | ||
| Chemical compound, drug | DCG-IV | Tocris Bioscience | Cat# 0975 | ||
| Chemical compound, drug | D-APV | Tocris Bioscience | Cat# 0106 | ||
| chemical compound, drug | D-APV | NIMH Chemical Synthesis Program | N/A | ||
| Chemical compound, drug | R-CPP | Tocris Bioscience | Cat# 0247 | ||
| Chemical compound, drug | NBQX | Cayman Chemical Co. | Cat# 14914 | ||
| Chemical compound, drug | Fluo5-F pentapotassium salt cell impermeant | Invitrogen Molecular Probes | Cat# F14221 | ||
| Chemical compound, drug | Alexa Fluor 594 Hydrazide | Invitrogen Molecular Probes | Cat# A10438 | ||
| Chemical compound, drug | Alexa Fluor 488 Hydrazide | Invitrogen Molecular Probes | Cat# A10436 | ||
| Chemical compound, drug | D-Serine | Tocris Bioscience | Cat# 0226 | ||
| Chemical compound, drug | MNI-caged-L-glutamate | Tocris Bioscience | Cat# 1490 | ||
| Chemical compound, drug | Sucrose | SigmaMillipore | Cat# S9378 | ||
| Chemical compound, drug | KCl | SigmaMillipore | Cat# P3911 | ||
| Chemical compound, drug | NaH2PO4 | SigmaMillipore | Cat# S9638 | ||
| Chemical compound, drug | CaCl2 | SigmaMillipore | Cat# C8106 | ||
| Chemical compound, drug | MgCl2 | SigmaMillipore | Cat# M2670 | ||
| Chemical compound, drug | MgSO4 | SigmaMillipore | Cat# M1880 | ||
| Chemical compound, drug | Glucose | SigmaMillipore | Cat# G8270 | ||
| Chemical compound, drug | NaCl | SigmaMillipore | Cat# S7653 | ||
| Chemical compound, drug | NaHCO3 | SigmaMillipore | Cat# S6014 | ||
| Chemical compound, drug | Cesium hydroxide | SigmaMillipore | Cat# 23204 | ||
| Chemical compound, drug | D-gluconic acid | SigmaMillipore | Cat# G1951 | ||
| Chemical compound, drug | EGTA | SigmaMillipore | Cat# E4378 | ||
| Chemical compound, drug | HEPES | SigmaMillipore | Cat# H3375 | ||
| Chemical compound, drug | Potassium gluconate | SigmaMillipore | Cat# G4500 | ||
| Chemical compound, drug | MgATP | SigmaMillipore | Cat# A9187 | ||
| Chemical compound, drug | Na3GTP | SigmaMillipore | Cat# G0635 | ||
| Chemical compound, drug | NMDG | SigmaMillipore | Cat# M2004 | ||
| Chemical compound, drug | Sodium ascorbate | SigmaMillipore | Cat# A4034 | ||
| Chemical compound, drug | Thiourea | SigmaMillipore | Cat# T8656 | ||
| Chemical compound, drug | Sodium pyruvate | SigmaMillipore | Cat# P2256 | ||
| Chemical compound, drug | KMeSO4 | SigmaMillipore | Cat# 83000 | ||
| Chemical compound, drug | Na2ATP | SigmaMillipore | Cat# A2383 | ||
| Chemical compound, drug | NaGTP | SigmaMillipore | Cat# 51120 | ||
| Chemical compound, drug | Sodium phosphocreatine | SigmaMillipore | Cat# P7936 | ||
| Chemical compound, drug | NH4Cl | SigmaMillipore | Cat# A9434 | ||
| Chemical compound, drug | KOH | EMD Millipore | Cat# 109108 | ||
| Chemical compound, drug | HCl | Fisher Chemical | Cat# SA49 | ||
| Software, algorithm | IgorPro7 | Wavemetrics | https://www.wavemetrics.com/ | ||
| Software, algorithm | Origin Pro 9 | Origin Lab | https://www.originlab.com/ | ||
| Software, algorithm | ImageJ | ImageJ | http://imagej.net/Welcome | ||
| Software, algorithm | Multiclamp 700B | Molecular Devices | https://www.moleculardevices.com/ | ||
| Software, algorithm | Prairie View 5.4 | Bruker Corp. | https://www.pvupdate.blogspot.com/ | ||
Additional files
-
Source data 1
Source datasets for all figures.
- https://cdn.elifesciences.org/articles/66612/elife-66612-data1-v2.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/66612/elife-66612-transrepform-v2.pdf





