tTARGIT AAVs mediate the sensitive and flexible manipulation of intersectional neuronal populations in mice
Figures
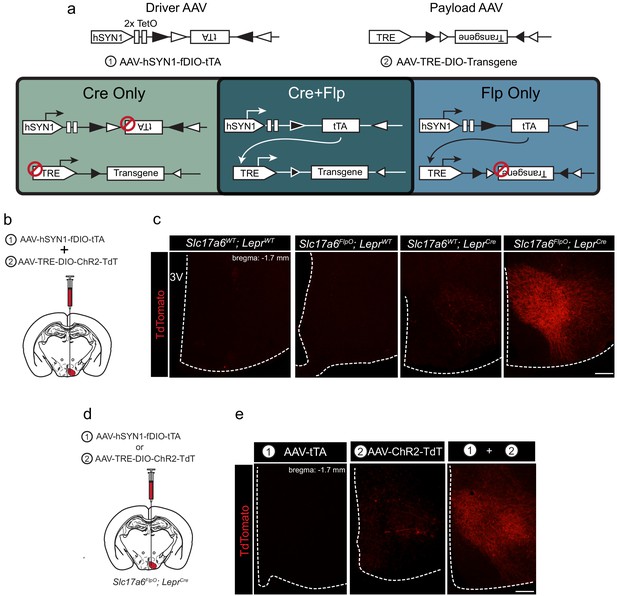
tTARGIT AAVs: a dual virus system to target intersectional populations.
(a) The tTARGIT system employs the combination of ‘Driver’ (AAV-hSYN-fDIO-tTA) and ‘Payload’ (AAV-TRE-DIO-Payload) AAVs. The Driver virus encodes a Flp-dependent tetracycline transactivator (tTA) under control of the human synapsin I (hSYN1) promoter and two tetracycline operators (TetO). The Payload virus encodes a Cre-dependent Payload transgene under control of the tetracycline response element (TRE). (b,c) Experimental scheme (b) and representative images (c) showing the detection of TdTomato (DSRed-IR, red) following the co-injection of AAV-hSYN-fDIO-tTA and AAV-TRE-DIO-ChR2-TdT into the VMH of (from left to right, as labeled) wild-type (WT), Slc17a6FlpO, LeprCre, or Slc17a6FlpO;LeprCre mice. (d,e) Experimental schematic (d) and representative images (e) showing the detection of TdTomato (DSRed-IR, red) following the injection of (from left to right, as labeled): (1) AAV-hSYN-fDIO-tTA, (2) AAV-TRE-DIO-ChR2-TdT, or the two viruses combined (1+2) into the VMH of Slc17a6FlpO;LeprCre mice. Scale bars = 100 μm.
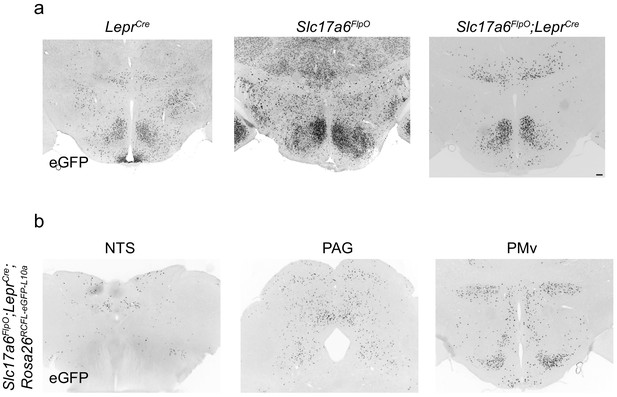
An intersectional approach for targeting LepRbVMH neurons.
Representative image of GFP-IR (black) in the (a) hypothalamus of adult LeprCre;Rosa26LSL-eGFP-L10a (LeprCre; left), Slc17a6FlpO;Rosa26FSF-eGFP-L10a (Slc17a6FlpO; middle), and Slc17a6FlpO;LeprCre;R26RCFL-eGFP-L10a (Slc17a6FlpO;LeprCre; right) mice. (b) GFP-IR (black) in the ventral premamillary nucleus (PMv), periaqueductal gray (PAG), and nucleus of the solitary tract (NTS) in Slc17a6FlpO;LeprCre;Rosa26RCFL-eGFP-L10a animals. Scale bar = 100 μm.
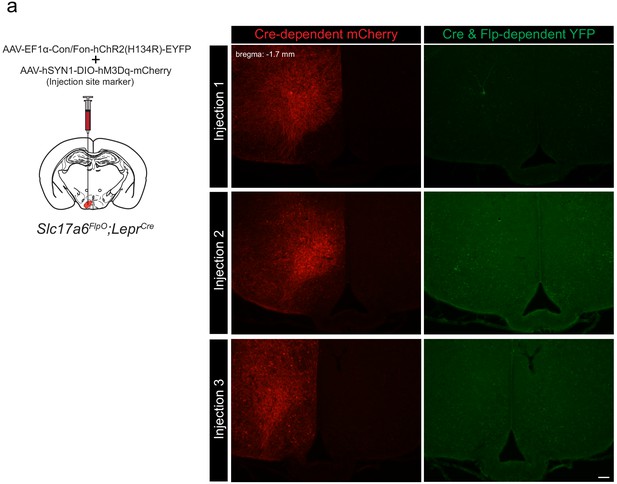
INTRSECT transgene expression in Slc17a6FlpO;LeprCre-defined LepRbVMH neurons.
(a) Schematic of unilateral injection strategy for INTERSECT virus and injection site marker into the VMH of Slc17a6FlpO;LeprCre mice. Representative images showing mCherry-IR (red, left) and YFP-IR (green, right) in the VMH of three separate Slc17a6FlpO;LeprCre mice co-injected with a Cre-dependent hM3Dq-mCherry AAV (an injection site marker) and INTERSECT Flp-ON/Cre-ON ChR2-eYFP AAV.
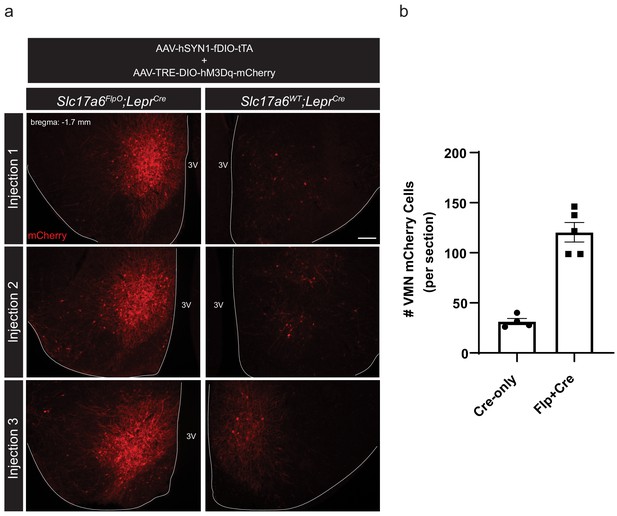
VMH hM3Dq-mCherry expression in Slc17a6FlpO;LeprCre and LeprCre-only mice.
Representative images (a) and quantification (b) of mCherry-IR in Slc17a6FlpO;LeprCre and LeprCre-only mice co-injected with AAV-hSYN1-fDIO-tTA and AAV-TRE-DIO-hM3Dq-mCherry. Data presented as mean ± SEM. Scale bar = 100 μm. 3V = third ventricle.
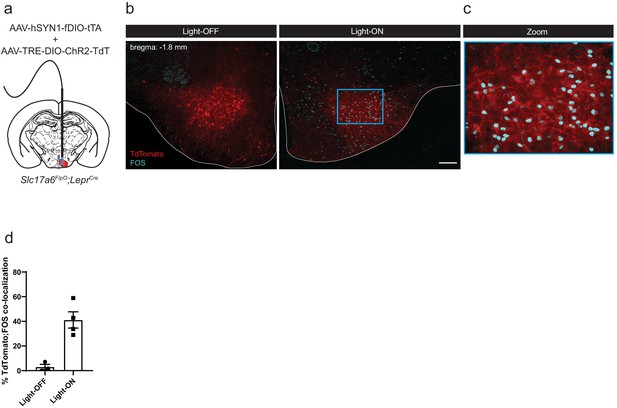
FOS response to photostimulation of tTARGIT AAV-driven ChR2 production in LepRbVMH cells.
(a) Experimental schematic showing unilateral co-injection of the Flp-dependent tTARGIT Driver AAV and ChR2-TdTomato-expressing tTARGIT Payload AAV targeting the VMH of Slc17a6FlpO;LeprCre mice, with optic fiber implantation. (b,c) Representative images and quantification (d) of ChR2-Tdtomato (red) and FOS-IR (cyan) with either no photostimulation (left) or 1 hr photostimulation (right); (c) a digital zoom on the boxed region in (b). Data presented as mean ± SEM. Scale bar = 100 μm.
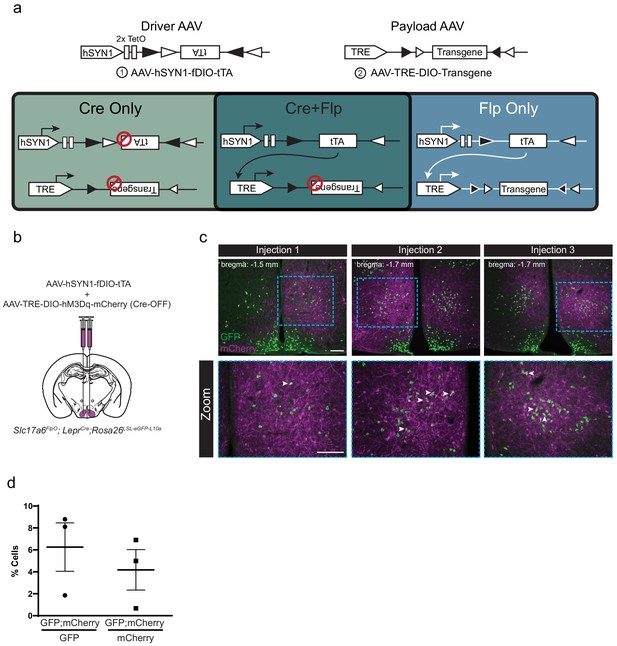
Design and validation of the tTARGIT AAV approach to Flp-ON/Cre-OFF genetics.
(a) The tTARGIT Flp-ON/Cre-OFF system is composed of a Flp-dependent tTA Driver virus (1) with a Payload virus (2) in which the transgene lies in the forward orientation unless Cre mediates its inversion. (b–d) Schematic of Flp-ON/Cre-OFF viral injection strategy, representative images (c) and quantification (d) of mCherry-IR (magenta) and GFP-IR (green) from three independent injections of the Flp-ON/Cre-OFF tTARGIT system using an hM3Dq-mCherry transgene into the VMH of Slc17a6FlpO;LeprCre; Rosa26LSL-eGFP-L10a mice (which express GFP in all LeprCre cells). Bottom images show zooms of the boxed regions in the top panels. White arrowheads indicate localization between GFP and mCherry. Data presented as mean± SEM. Scale bars = 100 μm.
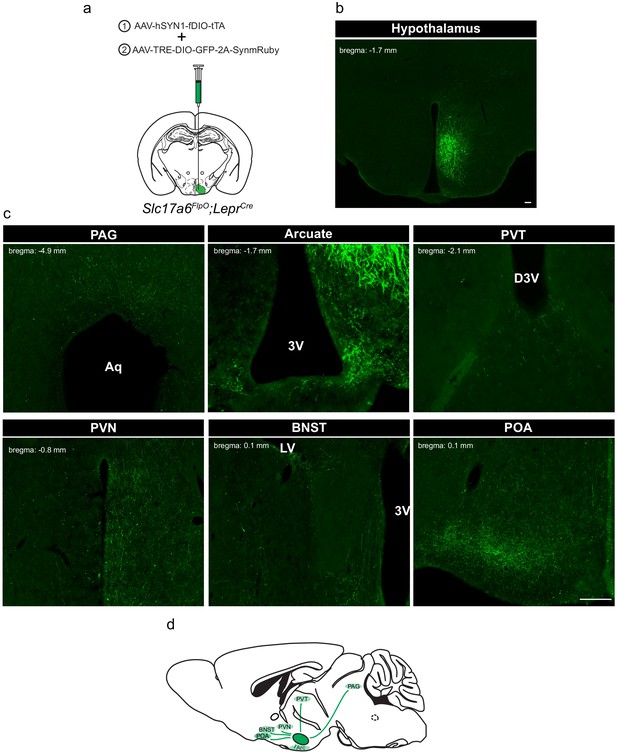
Defining the downstream projections of LepRbVMH neurons.
(a) Experimental schematic showing the injection of the Driver AAV with the Payload AAV encoding a GFP-2A-SynmRuby transgene into the VMH of Slc17a6FlpO;LeprCre animals. (b,c) Representative image of GFP-IR (green) showing viral transduction in the VMH (b) and projections (c) in the periaqueductal gray (PAG), arcuate nucleus (ARC), paraventricular hypothalamic nucleus (PVN), bed nucleus of the stria terminalis (BNST), and preoptic area (POA). (d) Cartoon showing the projection targets of LepRbVMH neurons.
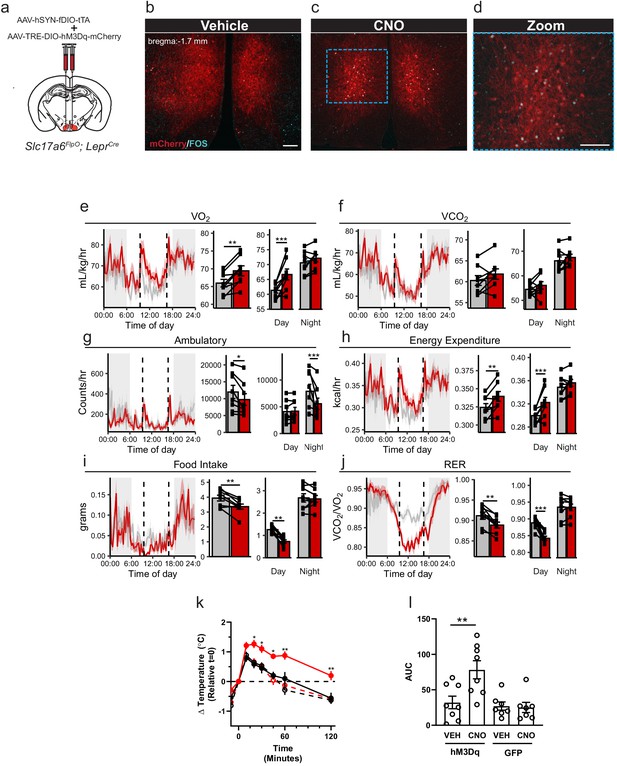
Activation of LepRbVMH neurons decreases food intake in addition to increasing brown adipose tissue (BAT) thermogenesis and energy expenditure.
(a) Approach for activating LepRbVMH neurons by co-injecting the AAV-hSYN-fDIO-tTA Driver and the AAV-TRE-DIO-hM3Dq-mCherry Payload viruses into the VMH of Slc17a6FlpO;LeprCre animals. (b–d) Representative images showing mCherry detection (DSRed-IR, red) and FOS-IR (cyan) in LepRbVMH-Dq mice administered Vehicle (b) or CNO (1 mg/kg) (c,d). The right-hand panel of (c) shows a digital zoom of the boxed region in the left panel. Scale bars = 100 μm. (e–j) Results from metabolic cage analysis of LepRbVMH-Dq mice (n = 6) treated with either vehicle (gray) or 1 mg/kg CNO (red) at 9:30 and 16:30 (dotted lines). Lines in left panels denotes mean value; shading denotes SEM. Each animal was treated with vehicle for 2 days followed by CNO for 2 days to allow pairing. Bar graphs to the right show the average for each mouse at each time point across 24 hr and separated by time in light cycle (day = light, night = dark). (k, l) Changes to intrascapular temperatures over 120 min in LepRbVMH-Dq mice (n = 8) or GFP-injected (n = 7) controls following vehicle (gray) or CNO (red; 1 mg/kg) administration at 30°C. (l) Shows area under the curve (AUC) for each treatment condition in (k). For metabolic cage studies, statistical significance was determined using either a paired t-test (full-day data) or a linear mixed model for effects by time of day. For interscapular temperature measurements, significance was determined by paired t-test. *p<0.05, **p<0.01, ***p<0.001.
-
Figure 3—source data 1
Raw Data from metabolic cages.
- https://cdn.elifesciences.org/articles/66835/elife-66835-fig3-data1-v2.xlsx
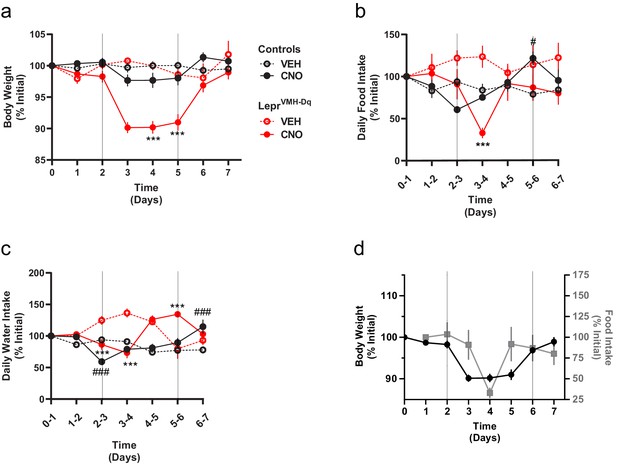
Chronically activating LepRbVMH neurons promotes negative energy balance.
(a–c) Body weight, food intake, and water intake of chow-fed LeprCre-only controls injected with AAV-hSYN1-fDIO-tTA and AAV-TRE-DIO-hM3Dq-mCherry (black lines; VEH n = 5; CNO n = 8) and LepRbVMH-Dq mice (red lines n = 8 both conditions) receiving 2 days normal drinking water (days 0–2), followed by 3 days exposure to CNO-laced drinking water (days 2–5), followed by an additional 2 days of normal drinking water. Onset and termination of CNO treatment are denoted by vertical lines in each panel. Vehicle controls (dashed red and black lines) did not receive CNO on days 2–5. (d) Comparisons between body weight (closed black circles) and food intake (gray squares) over time in LepRbVMH-Dq animals. Data is presented as mean ± SEM. Significance was determined by linear mixed model; ***p<0.001 between CNO and VEH LepRbVMH-Dq animals. #p<0.05, ###p<0.001 between CNO and VEH LeprCre-only Control animals.
-
Figure 4—source data 1
Raw data from chronic CNO dosing.
- https://cdn.elifesciences.org/articles/66835/elife-66835-fig4-data1-v2.xlsx
Tables
Available tTARGIT Vectors.
| Driver viruses | Notes |
|---|---|
| AAV-hSYN1-fDIO-tTA | Drives expression in the absence of DOX |
| AAV-hSYN1-fDIO-rtTA | Drives expression in the presence of DOX; not tested |
| Payload viruses | |
| Flp-ON/Cre-ON (Payload inverted) | |
| AAV-TRE-DIO-hM3Dq-mCherry | Tested |
| AAV-TRE-DIO-ChR2-TdTomato | Tested |
| AAV-TRE-DIO-Cas9 | Not tested |
| AAV-TRE-DIO-TVA-oG-mCherry | Not tested |
| AAV-TRE-DIO-eGFP-2A-TetanusToxin | Not tested |
| AAV-TRE-DIO-SwiChR-eYFP | Not tested |
| AAV-TRE-DIO-eGFP-L10a | Not tested |
| AAV-TRE-DIO-GCaMP6s | Not tested |
| AAV-TRE-DIO- proCaspase-3-TEVp | Not tested |
| AAV-TRE-DIO-GFP-2A-SynmRuby | Tested; GFP expression permits tracing; limited SynmRuby expression |
| Flp ON/Cre-OFF (Payload Sense orientation) | |
| AAV-TRE-DIO-hM3Dq-mCherry | Tested; may require strong Cre driver for complete inactivation in Cre neurons |
| AAV-TRE-DIO-ChR2-TdTomato | |
| AAV-TRE-DIO-eGFP-2A-TetanusToxin |
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Antibody | Anti-FOS (Rabbit monoclonal) | Cell Signaling | Cat# 2250; RRID:AB_2247211 | (1:1000) |
| Antibody | Anti-GFP (Chicken polyclonal) | Aves Laboratories | Cat# GFP1020; RRID:AB_10000240 | (1:1000) |
| Antibody | Anti-dsRed (Rabbit polyclonal) | Takara | Cat # 632496; RRID:AB_10013483 | (1:1000) |
| Cell line (E. coli) | Stbl3 E. coli | Thermofisher | Cat # C737303 | |
| Recombinant DNA reagent | pAAV-nEF Con/Fon hChR2(H134R)-EYFP | Fenno et al., 2014; Addgene | Addgene Plasmid ID:55644 | |
| Recombinant DNA reagent | AAV-hSYN1-fDIO-tTA | This manuscript | Addgene plasmid ID: 166597 | hSYN1-driven, Flp-dependent expression of tTA |
| Recombinant DNA reagent | AAV-TRE-DIO-hM3Dq-mCherry (Cre-ON) | This manuscript | Addgene plasmid ID: 166599 | TRE-driven, Cre-dependent expression of hM3Dq-mCherry |
| Recombinant DNA reagent | AAV-TRE-DIO-hM3Dq-mCherry (Cre-OFF) | This manuscript | Addgene plasmid ID: 166609 | TRE-driven, Cre-dependent inactivation of hM3Dq-mCherry |
| Recombinant DNA reagent | AAV-TRE-DIO-ChR2-Tdtomato | This manuscript | Addgene plasmid ID: 166600 | Cre-dependent expression of ChR2-TdTomato downstream of a TRE |
| Recombinant DNA reagent | AAV-TRE-DIO-GFP-2A-SynmRuby | This manuscript | Addgene plasmid ID:166608 | Cre-dependent expression of GFP-2A-Synaptophysin-mRuby downstream of a TRE |
| Chemical compound, drug | CNO | Tocris | Cat# 4936 | |
| Genetic reagent (M. musculus) | LeprIRES-Cre | Jackson Laboratories | Strain #: 032457 | Mouse: Lepr-IRES-Cre |
| Genetic reagent (M. musculus) | Slc17a6IRES-FlpO | This manuscript | N/A | Slc17a6 FlpO knockin mouse |
| Genetic reagent (M. musculus) | C57Bl6J | Jackson Laboratories | Strain #: 000664 | Wild-type animals |
| Genetic reagent (M. musculus) | Rosa26LSL-eGFP-L10a | Krashes et al., 2014 | N/A | Rosa26 targeted Cre-dependent eGFP_L10a allele |
| Genetic reagent (M. musculus) | Rosa26FSF-eGFP-L10a | This manuscript | N/A | Rosa26 targeted Flp and flp-dependent eGFP_L10a allele |
| Genetic reagent (M. musculus) | Rosa26RCFL-eGFP-L10a | This manuscript | N/A | Rosa26 targeted Flp-dependent eGFP_L10a allele |
| Software, algorithm | Prism V8 | Graphpad | https://www.graphpad.com/scientific-software/prism/ | |
| Software, algorithm | ImageJ | NIH | https://imagej.nih.gov/ij/download.html |





