Stimulation of hypothalamic oxytocin neurons suppresses colorectal cancer progression in mice
Figures
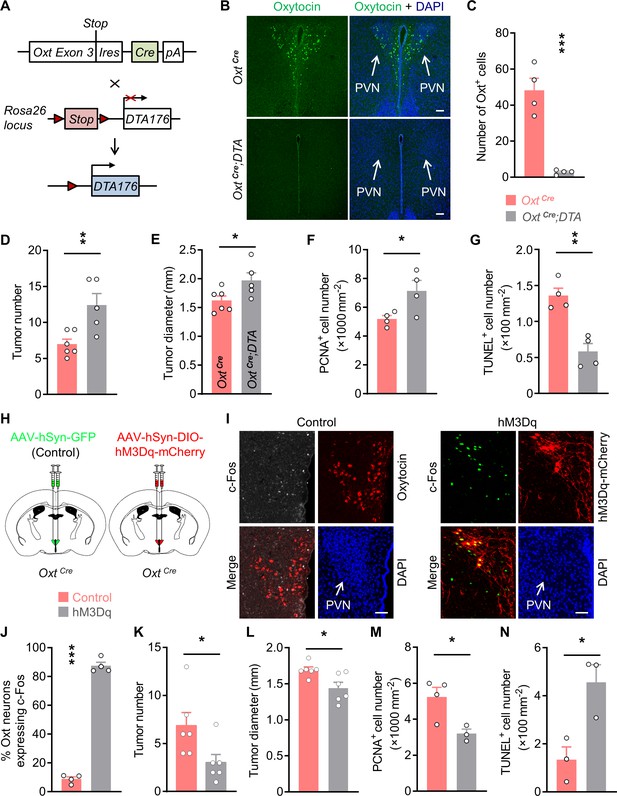
Oxytocin (Oxt) neurons modulate the progression of azoxymethane (AOM)/dextran sodium sulfate (DSS)-induced colitis-associated cancer (CAC) in mice.
(A) A schematic diagram showing the strategy of generating OxtCre;DTA mice. When Cre recombinase is present, loxP-flanked Stop cassette is excised, therefore allowing the expression of DTA176 in Oxt neurons. Triangles represent loxP sites. Ires, internal ribosome entry site. pA, simian virus 40 polyadenylation signal. (B) The CAC was induced in the 2-month-old OxtCre and OxtCre;DTA mice using AOM and DSS (see also Figure 1—figure supplement 1D). After completing the experiment, immunofluorescent staining for Oxt (green) indicated that Oxt neurons had been depleted in the paraventricular nucleus (PVN) of OxtCre;DTA mice. Cell nuclei were counterstained with DAPI (blue). Scale bars, 100 μm. (C) The number of Oxt-positive cells in the PVN. n = 4 mice per group. (D and E) The CAC was induced in the 2-month-old OxtCre and OxtCre;DTA mice using AOM and DSS. Tumor number (D) and diameter (E) in mice treated with AOM/DSS are shown. n = 6 (OxtCre) or 5 (OxtCre;DTA) mice per group. (F) The density of proliferating cell nuclear antigen (PCNA)-positive cells in the tumor tissues of AOM/DSS-treated OxtCre and OxtCre;DTA mice. n = 4 mice per group. (G) The density of terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL)-positive cells in tumor tissues. n = 4 mice per group. (H) Schematic diagrams showing that the indicated adeno-associated viruses (AAVs) were injected into mouse PVN. (I) Adult male OxtCre mice were injected with AAV-hSyn-GFP (control) or AAV-hSyn-DIO-hM3Dq-mCherry (hM3Dq) viruses into the PVN, and were then administered with AOM and DSS. The mice were i.p. injected with clozapine-N-oxide (CNO) every other day for 3 weeks (see also Figure 1—figure supplement 2D). Two hours after the final dose of CNO, mice were perfused with 4% paraformaldehyde (PFA). For control, we carried out double immunofluorescence staining for c-Fos (gray) and Oxt (red). For hM3Dq, immunostaining for c-Fos (green) was performed, and Oxt neurons were identified using hM3Dq-mCherry (red). DAPI staining is in blue. Scale bars, 50 μm. (J) The percentage of OxtPVN neurons expressing c-Fos. n = 4 mice per group. (K and L) Male OxtCre mice (2 months of age) were injected with the indicated AAV into PVN, and were then treated with AOM and DSS. Subsequently, mice were i.p. administered with CNO every other day for 3 weeks. The animals were then sacrificed and tumor number (K) as well as diameter (L) were assessed. n = 6 mice per group. (M) The density of PCNA-positive cells in tumor tissues. n = 4 (control) or 3 (hM3Dq) mice. (N) The density of TUNEL-positive cells in tumor tissues. n = 3 mice per group. Data are shown as means ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001, two-tailed Student’s t-test (C–G, J–N).
-
Figure 1—source data 1
Source data for Figure 1, panels C-G and J-N.
- https://cdn.elifesciences.org/articles/67535/elife-67535-fig1-data1-v2.xlsx
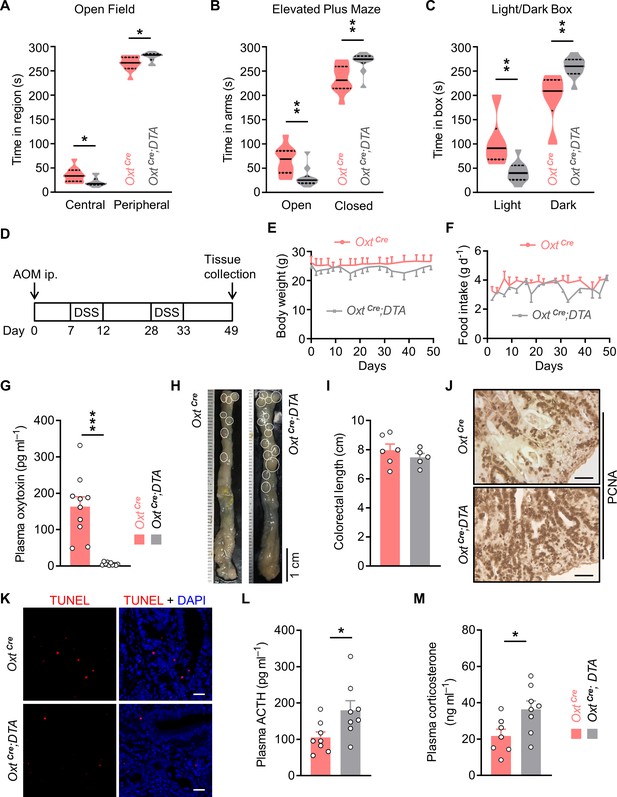
Depletion of oxytocin (Oxt) neurons increases anxiety level and promotes colitis-associated cancer (CAC) development in mice.
(A) Open field test. The time spent in the central and peripheral regions of OxtCre and OxtCre;DTA mice at 2 months of age. Solid and dotted lines indicate medians and quartiles. n = 11 mice per group. (B) Elevated plus maze test. The time spent in the open and closed arms of the indicated mice. n = 10 mice per group. (C) Light/dark box test. The time spent in the light and dark boxes. n = 10 mice per group. (D) Schematic diagram of the azoxymethane/dextran sodium sulfate (AOM/DSS) protocol. (E and F) Body weight (E) and food intake (F) in the mice under AOM/DSS treatment. n = 6 (OxtCre) or 5 (OxtCre;DTA) mice per group. (G) The plasma Oxt levels in mice at the end of the experiment. n = 10 mice per group. (H) Representative images of colon and rectum collected from the AOM/DSS-treated OxtCre and OxtCre;DTA mice. White eclipses indicate individual tumor. (I) Colorectal length. n = 6 (OxtCre) or 5 (OxtCre;DTA) mice per group. (J) Immunohistochemical staining for proliferating cell nuclear antigen (PCNA) of the tumor tissues collected from the AOM/DSS-treated mice. Scale bars, 50 μm. (K) Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) labeling (red) of tumor tissues. Cell nuclei were counterstained with DAPI (blue). Scale bars, 20 μm. (L and M) The plasma samples of OxtCre and OxtCre;DTA mice after the treatment were collected. Plasma adrenocorticotropin (ACTH) (L) and corticosterone (M) levels were then measured. n = 7 (OxtCre, corticosterone) or 8 (all other groups) mice per group. Data are presented as means ± SEM (E–G, I, L, M). *p < 0.05, **p < 0.01, ***p < 0.001, two-tailed Student’s t-test (A–C, G, I, L, M).
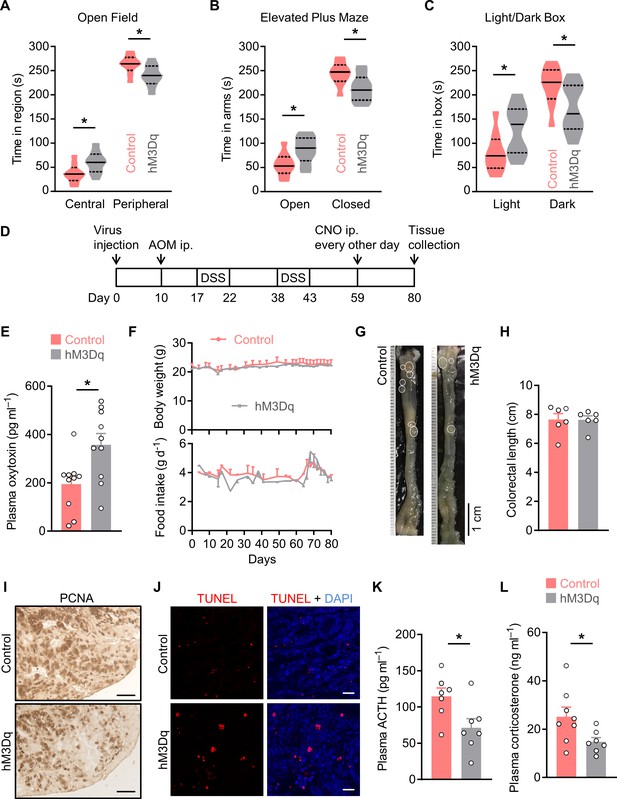
Excitation of OxtPVN neurons inhibits colitis-associated cancer (CAC) progression.
(A) Open field test. The control and hM3Dq adeno-associated viruses (AAVs) (hM3Dq) were injected into the paraventricular nucleus (PVN) of OxtCre mice. These mice were i.p. administered with clozapine-N-oxide (CNO) (3 mg kg–1) for 2 weeks, and then the open field test was performed. The time spent in the central and peripheral regions were recorded. Solid and dotted lines indicate medians and quartiles. n = 9 mice per group. (B) In elevated plus maze test, the time spent in the open and closed arms were assessed. n = 9 mice per group. (C) The time spent in the light and dark boxes in the light/dark box test. n = 9 mice per group. (D) Schematic diagram of the experimental design. The male OxtCre mice (2 months of age) were injected with the indicated AAV into the PVN, and were then treated with azoxymethane (AOM) and dextran sodium sulfate (DSS). Subsequently, CNO was i.p. administered every other day for 3 weeks. (E) The plasma oxytocin levels in mice at the end of the experiment. n = 10 mice per group. (F) Body weight (top) and food intake (bottom) in mice throughout the experiment. n = 6 mice per group. (G) Representative images of colorectal tissue. White eclipses indicate individual tumor. (H) Colorectal length. n = 6 mice per group. (I) Immunohistochemical staining for proliferating cell nuclear antigen (PCNA) of tumor tissues. Scale bars, 50 μm. (J) Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) labeling (red) of tumor tissues. Cell nuclei were counterstained with DAPI (blue). Scale bars, 20 μm. (K and L) Plasma adrenocorticotropin (ACTH) (K) and corticosterone (L) levels at the end of the experiment. n = 8 (control, corticosterone) or 7 (all other groups) mice per group. Data are presented as means ± SEM (E, F, H, K, L). *p < 0.05, two-tailed Student’s t-test (A–C, E, K, L).
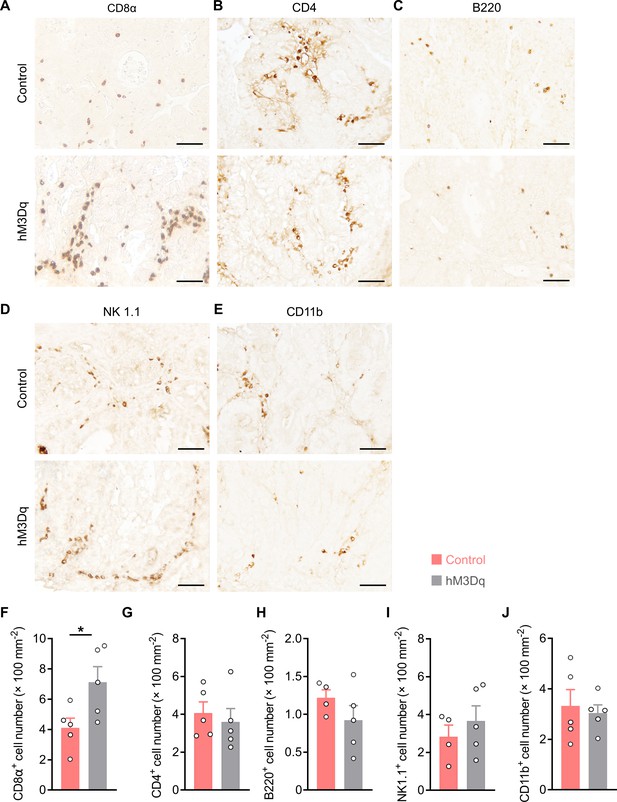
Density of immune cells in tumor tissues.
(A–E) The male OxtCre mice (2 months of age) were injected with the indicated adeno-associated viruses (AAVs) into the paraventricular nucleus (PVN), and were then treated with azoxymethane (AOM) and dextran sodium sulfate (DSS). Thereafter, clozapine-N-oxide (CNO) was i.p. injected every other day for 3 weeks. Immunohistochemical stainings for CD8α, CD4, B220, NK1.1, and CD11b of tumor tissue sections were carried out. Representative images are shown. Scale bars, 50 μm. (F–J) The density of immune cells in tumor tissue. n = 4 (control, B220 or control, NK1.1) or 5 (all other groups) mice per group. Data are presented as mean ± SEM. *p < 0.05, two-tailed Student’s t-test (F).
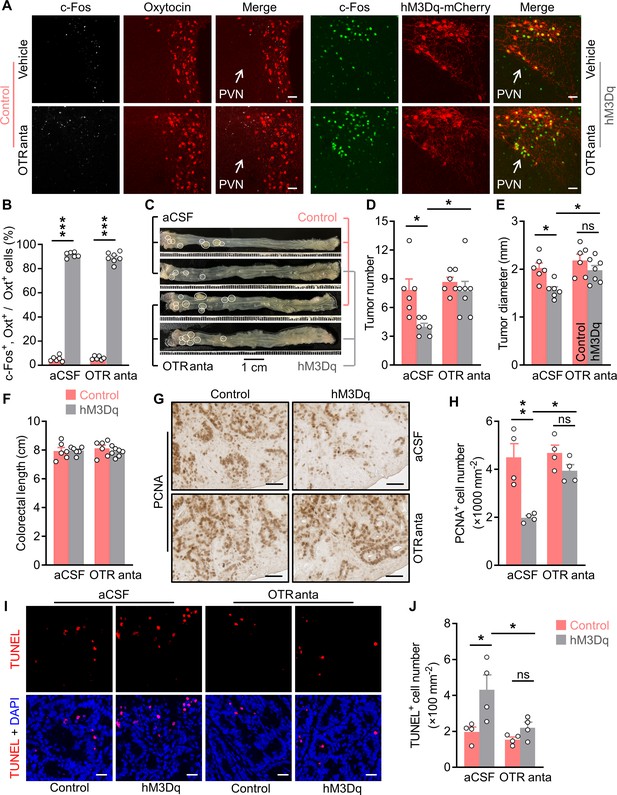
Brain oxytocin (Oxt) receptor is crucial for OxtPVN neuron activation to suppress colitis-associated cancer (CAC).
(A) Adult male OxtCre mice (2 months of age) were injected with AAV-hSyn-GFP (control) or AAV-hSyn-DIO-hM3Dq-mCherry (hM3Dq) viruses into the paraventricular nucleus (PVN), and then colitis-associated cancer (CAC) was induced using azoxymethane (AOM) and dextran sodium sulfate (DSS). Subsequently, these mice were administered with clozapine-N-oxide (CNO) (i.p.), and artificial cerebrospinal fluid (aCSF) or L-368,899 (i.c.v.), an Oxt receptor antagonist (OTR anta), every other day for 3 weeks. Mice were then perfused with 4% paraformaldehyde (PFA). For control, double immunofluorescence staining for c-Fos (gray) and Oxt (red) was performed. For hM3Dq, immunofluorescent staining for c-Fos (green) was performed, and Oxt neurons were identified using hM3Dq-mCherry (red). Cell nuclei were counterstained with DAPI (blue). Scale bars, 50 μm. (B) The percentage of Oxt neurons expressing c-Fos in the PVN. n = 7 (hM3Dq, OTR anta) or 6 (all other groups) mice per group. (C) The OxtCre mice (2 months of age) were injected with indicated adeno-associated viruses (AAVs) into the PVN, and then CAC was induced using AOM and DSS. Subsequently, these mice were administered with CNO (i.p.), as well as aCSF or L-368,899 (i.c.v.), the OTR antagonist (OTR anta), every other day for 3 weeks (see also Figure 2—figure supplement 1A). Representative images of colorectal tissue after the treatments are shown. White eclipse outlines the individual tumor. (D and E) Tumor number (D) and diameter (E). ns, not significant. n = 7 (hM3Dq, OTR anta) or 6 (all other groups) mice per group. (F) Colorectal length. n = 7 (hM3Dq, OTR anta) or 6 (all other groups) mice per group. (G and H) Immunohistochemical staining for proliferating cell nuclear antigen (PCNA) of tumor tissues. Representative images (G) and the density of PCNA-positive cells (H) are shown. Scale bars, 50 μm. ns, not significant. n = 4 mice per group. (I and J) Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay of tumor tissues. Representative images (I) and the density of TUNEL-positive cells (J) are shown. TUNEL labeling is in red. Cell nuclei were counterstained with DAPI (blue). Scale bars, 20 μm. ns, not significant. n = 4 mice per group. Data are presented as means ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001, one-way ANOVA with Bonferroni’s post hoc test.
-
Figure 2—source data 1
Source data for Figure 2, panels B, D-F, H and J.
- https://cdn.elifesciences.org/articles/67535/elife-67535-fig2-data1-v2.xlsx
Body weight and food intake in mice.
(A) A schematic diagram of the experimental design. The control and hM3Dq adeno-associated virus (AAV) were injected into the paraventricular nucleus (PVN) of OxtCre mice (2 months of age). These animals were then induced colitis-associated cancer (CAC) using azoxymethane (AOM) and dextran sodium sulfate (DSS). Subsequently, these mice were administered with clozapine-N-oxide (CNO) (i.p.) and artificial cerebrospinal fluid (aCSF) or L-368,899 (i.c.v.), an oxytocin (Oxt) receptor antagonist (OTR anta), every other day for 3 weeks. (B and C) Body weight (B) and food intake (C) in mice throughout the experiment. n = 8 (control+ CNO + OTR anta) or 7 (all other groups) mice per group. Data are presented as means ± SEM.
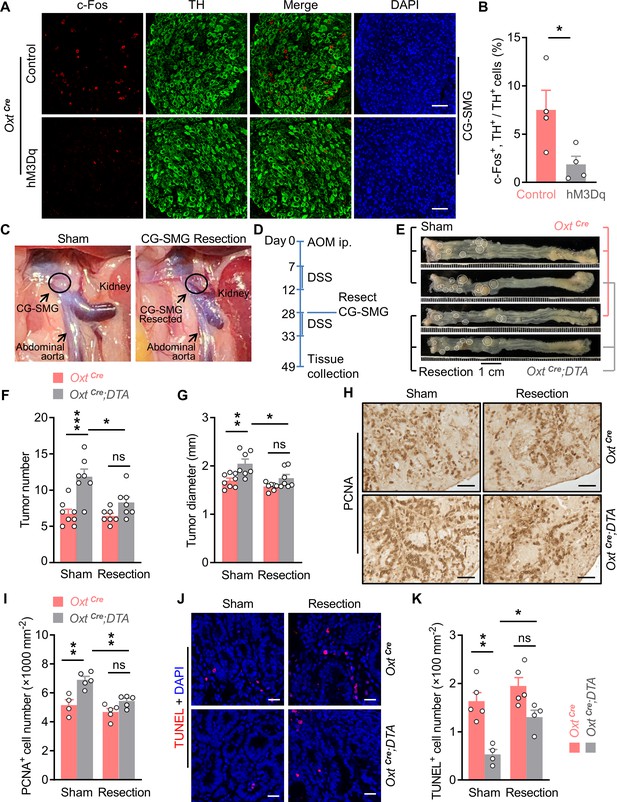
Surgical removal of celiac-superior mesenteric ganglion (CG-SMG) attenuates the tumor-promoting effect of oxytocin (Oxt) neuron depletion.
(A) Adult male OxtCre mice were injected with control or hM3Dq adeno-associated viruses (AAVs) (hM3Dq) into the paraventricular nucleus (PVN). After surgical recovery, these mice were administered with clozapine-N-oxide (CNO). Two hours later, CG-SMG were dissected and fixed in 4% paraformaldehyde (PFA). Double immunofluorescence staining for c-Fos (red) and tyrosine hydroxylase (TH, in green) of the CG-SMG was performed. Cell nuclei were counterstained with DAPI (blue). Scale bars, 50 μm. (B) The percentage of TH-positive cells expressing c-Fos in the CG-SMG. n = 4 mice per group. (C) Representative images showing mouse abdominal cavity with (left panel), or without (right panel) CG-SMG (following the resection). (D) A schematic diagram of experimental design. The colitis-associated cancer (CAC) was induced in adult OxtCre and OxtCre;DTA mice using azoxymethane (AOM) and dextran sodium sulfate (DSS). After the first cycle of DSS treatment, sham operation and CG-SMG resection were performed in mice. (E) Representative images of colorectal tissue after the treatment. White eclipse was used to outline the individual tumor. (F and G) Tumor number (F) and diameter (G). ns, not significant. n = 8 (OxtCre, sham) or 7 (all other groups) mice per group. (H and I) Immunohistochemical staining for proliferating cell nuclear antigen (PCNA) of tumor tissue. Representative images (H) and the density of PCNA-positive cells (I) are shown. ns, not significant. Scale bars, 50 μm. n = 4 (OxtCre, sham) or 5 (all other groups) mice per group. (J and K) Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay of tumor tissue. Representative images (J) and the density of TUNEL-positive cells (K) are shown. TUNEL labeling is in red. Cell nuclei were counterstained with DAPI (blue). ns, not significant. Scale bars, 20 μm. n = 5 (OxtCre) or 4 (OxtCre;DTA). Data are presented as means ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001, two-tailed Student’s t-test (B) or one-way ANOVA with Bonferroni’s post hoc test (F, G, I, K).
-
Figure 3—source data 1
Source data for Figure 3, panels B, F, G, I and K.
- https://cdn.elifesciences.org/articles/67535/elife-67535-fig3-data1-v2.xlsx
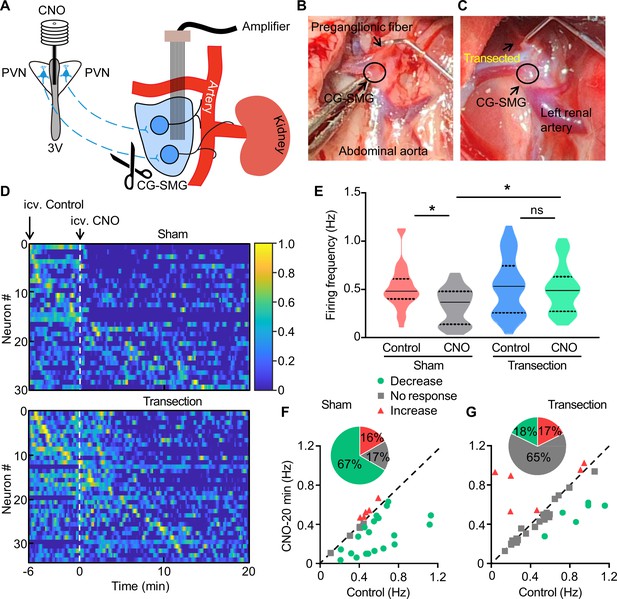
Transection of the preganglionic fiber of CG-SMG abolishes the inhibitory effect of Oxt neuron activation.
(A) Schematics of in vivo single-unit recordings in celiac-superior mesenteric ganglion (CG-SMG) with the transection of preganglionic fiber. Clozapine-N-oxide (CNO) was injected via an infusion cannula directed to third ventricle (3V). (B and C) Representative images displaying the CG-SMG with (B) or without (C) the preganglionic nerve fiber (following the transection). (D) Normalized firing rate of recorded CG-SMG neurons in response to CNO infusion in sham (top) and preganglionic fiber transection groups (bottom). Dashed line indicates the time point of CNO delivery. i.c.v., intracerebroventricular injection. n = 30 cells from 4 mice (sham) or 34 cells from 4 mice (transection). (E) Statistics of average firing frequency of CG-SMG neurons in response to CNO infusion in sham and preganglionic fiber transection groups. Solid and dotted lines indicate medians and quartiles, respectively. ns, not significant. n = 30 cells from 4 mice (sham) or 34 cells from 4 mice (transection). (F and G) Correlation of firing rate before and after CNO infusion in sham (F) and preganglionic fiber transection groups (G). Green filled circles represent individual units with significant lower firing frequency after CNO infusion. Red triangles represent the units with higher firing rate. Gray squares indicate neurons without significant change in firing rate. Inset: proportions of CG-SMG neurons with or without change in firing rate following CNO infusion. n = 30 cells from 4 mice (sham) or 34 cells from 4 mice (transection). Solid and dotted lines indicate medians and quartiles (E). *p < 0.05, one-way ANOVA with Bonferroni’s post hoc test (E).
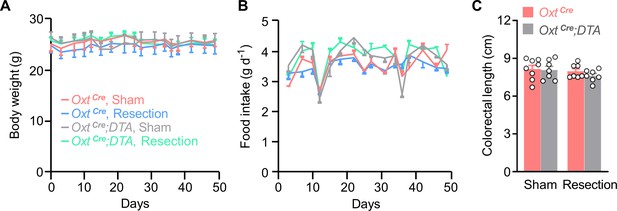
Body weight and food intake in mice.
(A and B) Body weight (A) and food intake (B) in mice throughout the experiment. n = 9 (OxtCre, sham), 10 (OxtCre, resection), 7 (OxtCre;DTA, sham), or 9 (OxtCre;DTA, resection) mice per group. (C) Colorectal length. n = 8 (OxtCre, sham) or 7 (all other groups) mice per group. Data are presented as means ± SEM.
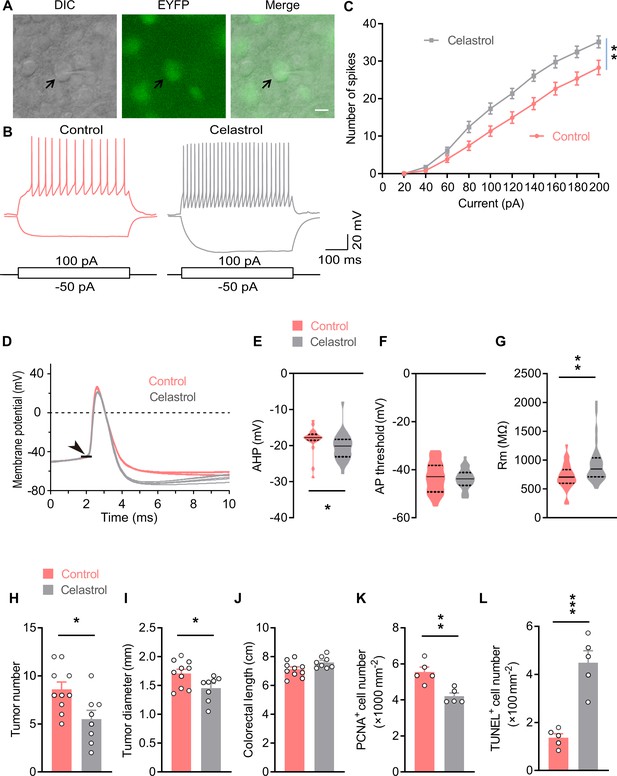
Celastrol enhances the excitability of OxtPVN neurons, and its administration in the brain inhibits colitis-associated cancer (CAC) progression.
(A) Electrophysiology of paraventricular nucleus (PVN) slice of 4-month-old OxtCre;EYFP mice. Left, a differential interference contrast (DIC) image of the recorded neuron (arrow). Middle, expression of enhanced yellow fluorescent protein (EYFP) (green) in the same cell suggests that it is an oxytocin (Oxt) neuron. Right, merged image. Scale bar, 10 μm. (B) Voltage response of Oxt neuron in response to 100 and –50 pA current injection in control and celastrol (5 μM in artificial cerebrospinal fluid [aCSF]) conditions. (C) Bath application of celastrol increased the number of action potentials (AP) fired across increasing current injections. n = 20 cells from five mice (control or celastrol). (D) Representative AP traces from control and celastrol conditions. Arrowhead indicates the AP threshold. (E–G) The size of afterhyperpolarization (AHP) (E), AP threshold, (F) and input resistance (Rm) (G) in control and celastrol conditions. Solid and dotted lines indicate medians and quartiles, respectively. n = 23 cells (E, F) or 27 cells (G) from five mice (control) or 28 cells from five mice (celastrol). (H and I) The CAC was induced in male C57 BL/6 mice (2 months of age) using azoxymethane (AOM) and dextran sodium sulfate (DSS). These animals were then i.c.v. administered with control versus celastrol every other day for 3 weeks. After the treatment, tumor number (H) and diameter (I) were determined. n = 10 (control) or 8 (celastrol) mice per group. (J) Colorectal length. n = 10 (control) or 8 (celastrol) mice per group. (K) The density of proliferating cell nuclear antigen (PCNA)-positive cells in tumor tissue. n = 5 mice per group. (L) The density of terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL)-positive cells in tumor tissue. n = 5 mice per group. Data are presented as means ± SEM (C, H–L). *p < 0.05, **p < 0.01, ***p < 0.001, two-way ANOVA with Bonferroni’s post hoc test (C), or two-tailed Student’s t-test (E, G, H, I, K, L).
-
Figure 4—source data 1
Source data for Figure 4, panels C and E-L.
- https://cdn.elifesciences.org/articles/67535/elife-67535-fig4-data1-v2.xlsx
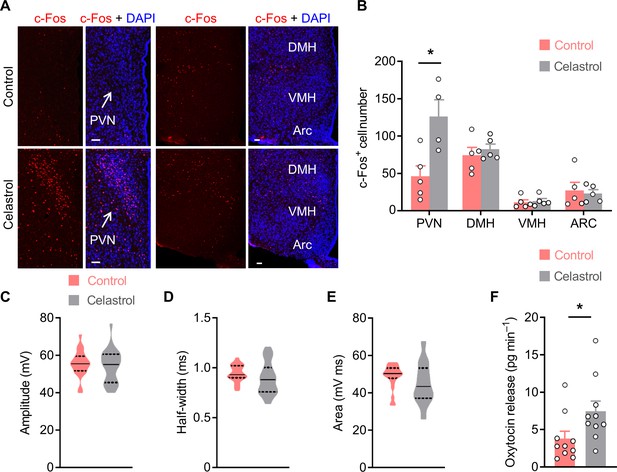
Celastrol excites neurons in the PVN and promotes Oxt release from PVN.
(A) Male C57 BL/6 mice (2 months of age) were i.c.v. administered with vehicle or celastrol (0.5 µg). Two hours later, the mice were anesthetized, and were then perfused with 4% paraformaldehyde (PFA). Immunofluorescent staining for c-Fos (red) of brain tissue sections was carried out. Representative images display the expression of c-Fos in the paraventricular (PVN), dorsomedial (DMH), ventromedial (VMH), and arcuate (Arc) nuclei. Cell nuclei were counterstained with DAPI (blue). Scale bars, 50 μm. (B) The number of c-Fos-positive cells in the neuronal nuclei. n = 4 (celastrol, PVN) or 5 (all other groups) mice per group. (C–E) Action potential amplitude (C), half-width (D), and area (E) in control and celastrol condition (5 μM in artificial cerebrospinal fluid [aCSF]). Solid and dotted lines indicate medians and quartiles, respectively. n = 23 cells from five mice (control) or 28 cells from five mice (celastrol). (F) The PVN tissue slices of male C57 BL/6 mice (2 months of age) were dissected from the brain. Basal and celastrol-elicited oxytocin (Oxt) release rates were then determined. n = 10 mice per group. Data are presented as means ± SEM (B, F). *p < 0.05, two-tailed Student’s t-test (B, F).
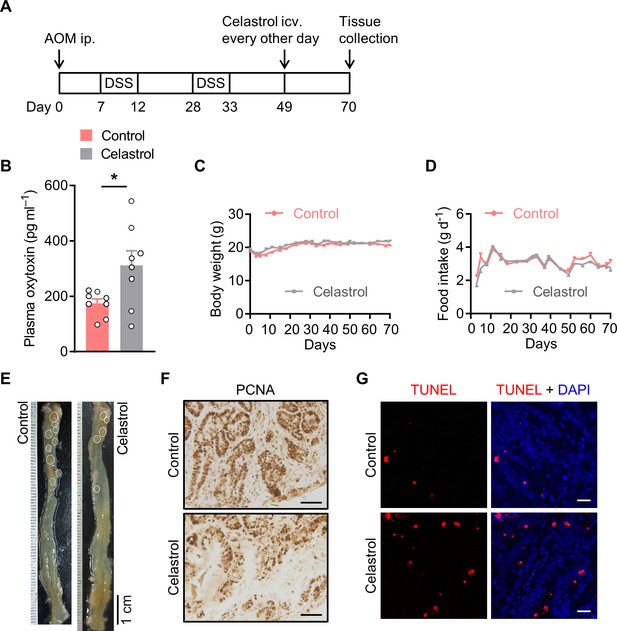
Brain treatment with celastrol suppresses colitis-associated cancer (CAC) progression in mice.
(A) Schematic diagram of the experimental design. The CAC was induced in the male C57 BL/6 mice (2 months of age) using azoxymethane (AOM) and dextran sodium sulfate (DSS). These mice were then i.c.v. administered with vehicle versus celastrol every other day for 3 weeks. (B) The plasma oxytocin (Oxt) levels at the end of the experiment. n = 8 mice per group. (C and D) The body weight (C) and food intake (D) in mice throughout the experiment. n = 10 (control) or 8 (celastrol) mice per group. (E) Representative images of the colorectal tissue. White eclipses outline individual tumor. (F) Immunohistochemical staining for proliferating cell nuclear antigen (PCNA) of tumor tissues. Scale bars, 50 μm. (G) Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay of tumor tissues. TUNEL labeling is shown in red. Cell nuclei were counterstained with DAPI (blue). Scale bars, 20 μm. Data are presented as means ± SEM. *p < 0.05, two-tailed Student’s t-test (B).
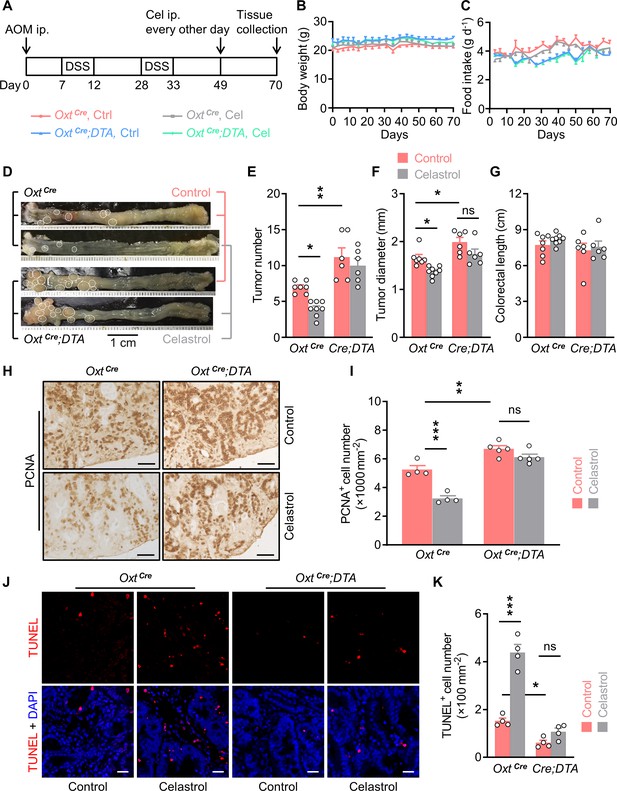
Depletion of oxytocin (Oxt) neurons attenuates the anti-tumor effect of celastrol.
(A) A schematic diagram of experimental design. The colitis-associated cancer (CAC) was induced in the OxtCre and OxtCre;DTA mice (2 months of age), in which control solution and celastrol (Cel) were i.p. administered every other day for 3 weeks. (B and C) Body weight (B) and food intake (C) in mice throughout the experiment. n = 7 (OxtCre, Ctrl), 8 (OxtCre, Cel), or 6 (OxtCre;DTA) mice per group. (D) Representative images of colorectal tissue after the indicated treatments. White eclipses indicate individual tumor. (E and F) Tumor number (E) and diameter (F). Cre;DTA, OxtCre;DTA. ns, not significant. n = 7 (OxtCre, Ctrl), 8 (OxtCre, Cel), or 6 (OxtCre;DTA) mice per group. (G) Colorectal length. n = 7 (OxtCre, Ctrl), 8 (OxtCre, Cel), or 6 (OxtCre;DTA) mice per group. (H and I) Immunohistochemical staining for proliferating cell nuclear antigen (PCNA) of tumor tissue. Representative images (H) and the density of PCNA-positive cells (I) are shown. ns, not significant. Scale bars, 50 μm. n = 4 (OxtCre) or 5 (OxtCre;DTA) mice per group. (J and K) Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay of tumor tissue. Representative images (J) and the density of TUNEL-positive cells (K) are shown. TUNEL labeling is in red. Cell nuclei were counterstained with DAPI (blue). ns, not significant. Scale bars, 20 μm. n = 4 mice per group. Data are presented as means ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001, one-way ANOVA with Bonferroni’s post hoc test (E, F, I, K).
-
Figure 5—source data 1
Source data for Figure 5E–G,I,K.
- https://cdn.elifesciences.org/articles/67535/elife-67535-fig5-data1-v2.xlsx
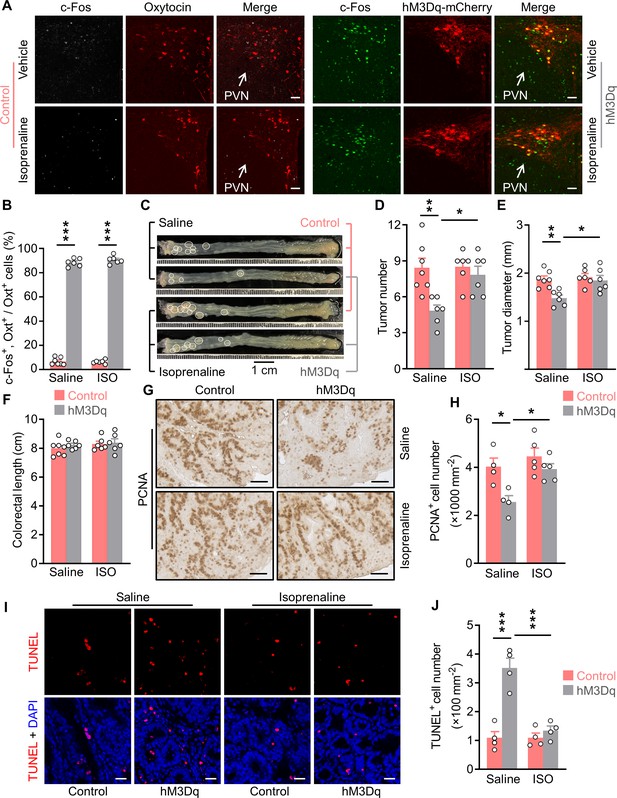
Treatment with an agonist for β2 adrenergic receptor attenuates the anti-tumor effect of OxtPVN neuron activation.
(A) Control and AAV-hSyn-DIO-hM3Dq-mCherry (hM3Dq) viruses were injected into the paraventricular nucleus (PVN) of male adult OxtCre mice. Colitis-associated cancer (CAC) was then induced using azoxymethane (AOM) and dextran sodium sulfate (DSS). These mice were i.p. administered with clozapine-N-oxide (CNO) every other day and i.p. injected with saline or isoprenaline, a β2 adrenergic receptor agonist, on a daily basis. After 3 weeks of treatment, mice were perfused with 4% paraformaldehyde (PFA). For control, double immunofluorescence staining for c-Fos (gray) and oxytocin (Oxt) (red) was performed. For hM3Dq, immunofluorescent staining for c-Fos (green) was performed and Oxt neurons were identified using hM3Dq-mCherry (red). Cell nuclei were counterstained with DAPI (blue). Scale bars, 50 μm. (B) The percentage of Oxt neurons expressing c-Fos in the PVN. ISO, isoprenaline. n = 7 (control, saline) or 6 (all other groups) mice per group. (C) Adult OxtCre mice were injected with adeno-associated viruses (AAVs) into the PVN. CAC was then induced using AOM and DSS. Subsequently, these mice were i.p. administered with CNO every other day and i.p. injected with saline or isoprenaline on a daily basis. These treatments were continued for 3 weeks (see also Figure 6—figure supplement 1C). Representative images of colorectal tissue after the treatments are shown. White eclipse outlines individual tumor. (D and E) Tumor number (D) and diameter (E). n = 7 (control, saline) or 6 (all other groups) mice per group. (F) Colorectal length. n = 7 (control, saline) or 6 (all other groups) mice per group. (G and H) Immunohistochemical staining for proliferating cell nuclear antigen (PCNA) of tumor tissue. Representative images (G) and the density of PCNA-positive cells (H) are shown. Scale bars, 50 μm. n = 4 (saline) or 5 (ISO) mice per group. (I and J) Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay of tumor tissue. Representative images (I) and the density of TUNEL-positive cells (J) are shown. TUNEL labeling is in red. Cell nuclei were counterstained with DAPI (blue). Scale bars, 20 μm. n = 4 mice per group. Data are presented as means ± SEM. *p < 0.05, **p < 0.01, ***p < 0.001, one-way ANOVA with Bonferroni’s post hoc test (B, D, E, H, J).
-
Figure 6—source data 1
Source data for Figure 6, panels B, D-F, H and J.
- https://cdn.elifesciences.org/articles/67535/elife-67535-fig6-data1-v2.xlsx
Body weight and food intake in mice.
(A) Male C57 BL/6 mice (2 months of age) were i.p. administered with saline or isoprenaline (10 mg kg–1). Two hours later, celiac-superior mesenteric ganglion (CG-SMG) were dissected and fixed in 4% paraformaldehyde (PFA). Double immunofluorescence staining of c-Fos (red) and tyrosine hydroxylase (TH, in green) of the CG-SMG was performed. Cell nuclei were counterstained with DAPI (blue). Scale bars, 50 μm. (B) The percentage of TH-positive cells expressing c-Fos in the CG-SMG. n = 5 (saline) or 4 (isoprenaline) mice per group. (C) A schematic diagram illustrating the experimental design. The adult OxtCre mice were injected with control or hM3Dq adeno-associated viruses (AAVs) into the paraventricular nucleus (PVN). The colitis-associated cancer was then induced using azoxymethane (AOM) and dextran sodium sulfate (DSS). Subsequently, these mice were i.p. administered with clozapine-N-oxide (CNO) (3 mg kg–1) every other day, as well as saline or isoprenaline (ISO), a β2 adrenergic receptor agonist, on a daily basis. These treatments were continued for 3 weeks. (D and E) Body weight (D) and food intake (E) in mice throughout the experiment. n = 7 (control+ CNO + saline), 6 (control+ CNO + ISO), or 5 (hM3Dq) mice per group. Data are presented as means ± SEM.
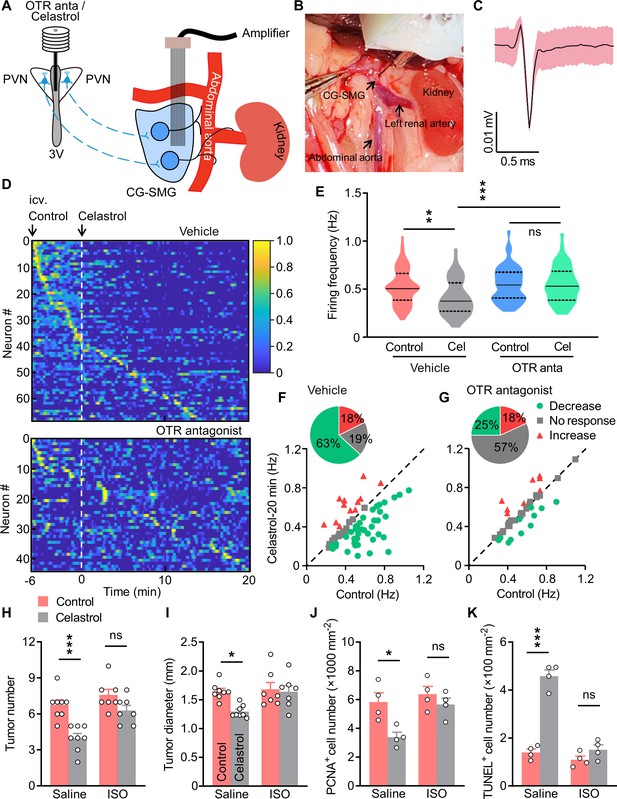
Treatment with isoprenaline abolished the anti-tumor effect of celastrol.
(A) Schematics of in vivo single-unit recordings in celiac-superior mesenteric ganglion (CG-SMG). L-368,899, the Oxt receptor (OTR) antagonist (OTR anta), and celastrol were applied through a guide cannula directed to third ventricle (3V). (B) A CG-SMG image was taken during the operation. (C) Example waveform of the single unit detected. (D) Normalized firing rate of recorded CG-SMG neurons in response to celastrol infusion in vehicle (top) and OTR antagonist (bottom) groups. Dashed line indicates the time point of celastrol delivery. i.c.v., intracerebroventricular injection. n = 68 cells (vehicle) from 7 mice or 44 cells (OTR antagonist) from 6 mice. (E) Statistics of average firing frequency of CG-SMG neurons in response to celastrol infusion in vehicle and OTR antagonist groups. Solid and dotted lines indicate medians and quartiles, respectively. n = 68 cells (vehicle) from 7 mice or 44 cells (OTR antagonist) from 6 mice. (F and G) Correlation of firing rate before and after celastrol infusion in vehicle (F) and OTR antagonist (G) groups. Green filled circles represent individual units with significantly lower firing frequency after celastrol infusion. Red triangles represent the units with higher firing rates. Gray squares indicate neurons without significant difference in firing rates. Inset: proportions of CG-SMG neurons with significantly decreased rates, increased rates, or no change in rates after celastrol infusion in vehicle (F) and OTR antagonist group (G). n = 68 cells (vehicle) from 7 mice or 44 cells (OTR antagonist) from 6 mice. (H and I) Colitis-associated cancer (CAC) was induced in male C57 BL/6 mice (2 months of age). These mice were then i.c.v. administered with vehicle (control) or celastrol every other day. In the meantime, the mice were i.p. injected with saline or isoprenaline (ISO) on a daily basis. These treatments were continued for 3 weeks (see also Figure 7—figure supplement 2A). Tumor number (H) and diameter (I) are shown. ns, not significant. n = 8 (saline) or 7 (ISO) mice per group. (J) The density of proliferating cell nuclear antigen (PCNA)-positive cells in tumor tissue. ns, not significant. n = 4 mice per group. (K) The density of terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL)-positive cells in tumor tissue. ns, not significant. n = 4 mice per group. Data are presented as means ± SD (C) or means ± SEM (H–K). *p < 0.05, **p < 0.01, ***p < 0.001, one-way ANOVA with Bonferroni’s post hoc test (E, H–K).
-
Figure 7—source data 1
Source data for Figure 7, panels E-K.
- https://cdn.elifesciences.org/articles/67535/elife-67535-fig7-data1-v2.xlsx
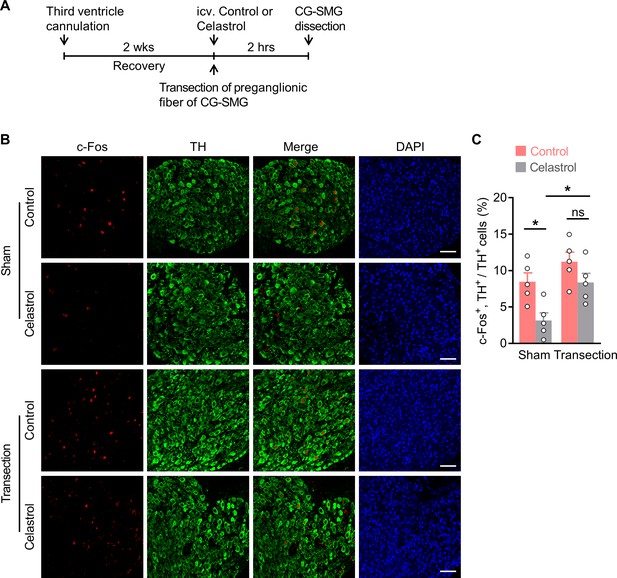
The preganglionic nerve fiber is crucial for brain administered celastrol to suppress neuronal activities in celiac-superior mesenteric ganglion (CG-SMG).
(A) A schematic diagram of the experimental design. Male C57 BL/6 mice (2 months of age) were implanted with a guide cannula directed to the third ventricle. After 2 weeks of recovery, the preganglionic nerve fiber of CG-SMG was transected. The other groups of mice were administered with sham operations. Subsequently, these mice were i.c.v. administered with vehicle (control) or celastrol. Two hours later, CG-SMG was dissected and fixed in 4% paraformaldehyde (PFA). (B) Double immunofluorescence staining for c-Fos (red) and tyrosine hydroxylase (TH, in green) of the CG-SMG. Cell nuclei were counterstained with DAPI (blue). Scale bars, 50 μm. (C) The percentage of TH-positive cells expressing c-Fos in the CG-SMG. ns, not significant. n = 5 mice per group. Data are presented as means ± SEM. *p < 0.05, one-way ANOVA with Bonferroni’s post hoc test (C).
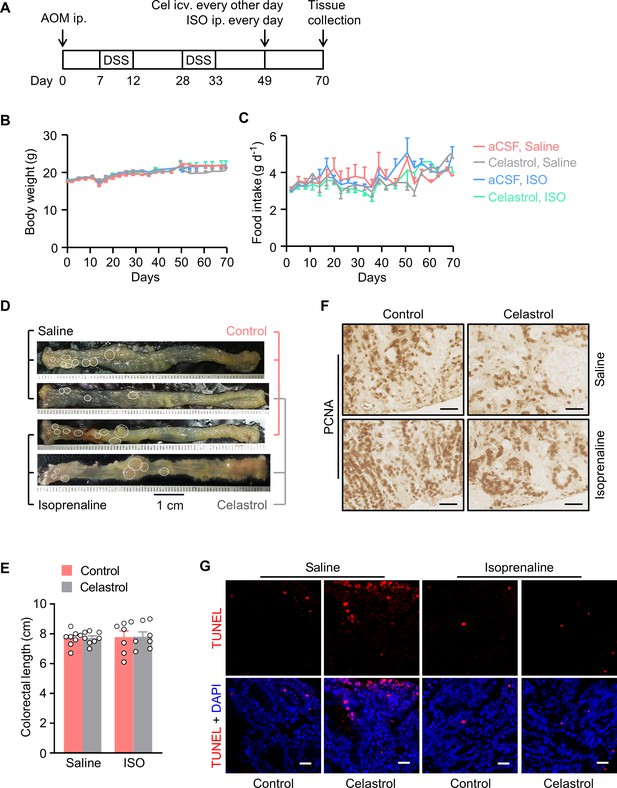
Activation of β2 adrenergic receptor abolishes the tumor suppression effect of centrally administered celastrol.
(A) A schematic diagram of the experimental design. The colitis-associated cancer was induced in the male C57 BL/6 mice (2 months of age) using azoxymethane (AOM) and dextran sodium sulfate (DSS). Subsequently, these mice were i.c.v. administered with vehicle (control) or celastrol (Cel) every other day. In the meanwhile, these mice were i.p. injected with saline or isoprenaline (ISO), a β2 adrenergic receptor agonist, on a daily basis. These treatments were continued for 3 weeks. (B and C) Body weight (B) and food intake (C) in mice throughout the experiment. n = 8 (saline) or 7 (ISO) mice per group. (D) Representative images of colorectal tissue. White eclipses outline the individual tumor. (E) Colorectal length. n = 8 (saline) or 7 (ISO) mice per group. (F) Immunohistochemical staining for proliferating cell nuclear antigen (PCNA) of tumor tissues. Scale bars, 50 μm. (G) Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay of tumor tissues. TUNEL labeling is shown in red. Cell nuclei were counterstained with DAPI (blue). Scale bars, 20 μm. Data are presented as means ± SEM.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent (Mus. musculus) | OxtCre | Jackson Laboratory | 024234 | PMID:23028821 |
| Genetic reagent (Mus. musculus) | Rosa26DTA176 | PMID:16407399 | ||
| Genetic reagent (Mus. musculus) | Rosa26-LSL-EYFP | PMID:11299042 | ||
| Strain, strain background (AAV) | AAV-hSyn-GFP | Obio Technology | AOV062 | |
| Strain, strain background (AAV) | pAAV-hSyn-DIO-hM3Dq-mCherry | Obio Technology | HYMBH2482 | |
| Antibody | (Rabbit polyclonal) anti-c-Fos | Santa Cruz Biotechnology | Cat# sc-7202; RRID:AB_2106765 | IF, (1:150) |
| Antibody | (Goat polyclonal) anti-c-Fos | Santa Cruz Biotechnology | Cat# sc-52-G; RRID:AB_2629503 | IF, (1:25) |
| Antibody | (Mouse monoclonal) anti-TH | Santa Cruz Biotechnology | Cat# sc-25269; RRID:AB_628422 | IF, (1:200) |
| Antibody | (Mouse monoclonal) anti-CD4 | Santa Cruz Biotechnology | Cat# sc-19641; RRID:AB_10554681 | IHC, (1:50) |
| Antibody | (Mouse monoclonal) anti-CD11b | Santa Cruz Biotechnology | Cat# sc-53086; RRID:AB_628894 | IHC, (1:100) |
| Antibody | (Rabbit polyclonal) anti-c-Fos | Abcam | Cat# ab190289; RRID:AB_2737414 | IF, (1:2000) |
| Antibody | (Rabbit polyclonal) anti-Oxt | Immunostar | Cat# 20068; RRID:AB_572258 | IF, (1:400) |
| Antibody | (Mouse monoclonal) anti-PCNA | Boster Biological | Cat# BM0104 | IHC, (1:200) |
| Antibody | (Rabbit polyclonal) anti-CD8α | Bioss | Cat# bs-0648R; RRID:AB_10857537 | IHC, (1:250) |
| Antibody | (Rat monoclonal) anti-B220 | BD Biosciences | Cat# 553087; RRID:AB_394617 | IHC, (1:300) |
| Antibody | (Mouse monoclonal) anti-NK1.1 | BD Biosciences | Cat# 550627; RRID:AB_398463 | IHC, (1:400) |
| Commercial assay or kit | Oxytocin EIA kit | Enzo Life Sciences | Cat# ADI-900–153 A; RRID:AB_2815012 | |
| Commercial assay or kit | Corticosterone ELISA kit | Enzo Life Sciences | Cat# ADI-900–097; RRID:AB_2307314 | |
| Commercial assay or kit | ACTH ELISA kit | Aviva Systems Biology | Cat# OKEH00628 | |
| Commercial assay or kit | In Situ Cell Death Detection Kit, TMR red | Sigma-Aldrich | Cat# 12156792910 | |
| Commercial assay or kit | SABC-POD kit | Boster Biological | Cat# SA1021 | |
| Chemical compound, drug | Azoxymethane | Sigma-Aldrich | Cat# A5486 | |
| Chemical compound, drug | Avertin | Sigma-Aldrich | Cat# T48402 | |
| Chemical compound, drug | Isoprenaline | Sigma-Aldrich | Cat# I5627 | |
| Chemical compound, drug | Proteinase K | Sigma-Aldrich | Cat# 3115879001 | |
| Chemical compound, drug | Dextran sulfate sodium | TdB Labs | Cat# 9011-18-1 | |
| Chemical compound, drug | CNO | MedChemExpress | Cat# HY-17366 | |
| Chemical compound, drug | Celastrol | Mengry Bio-Technology | Cat# MR80328 | |
| Chemical compound, drug | L-368,899 | Santa Cruz Biotechnology | Cat# sc-204037 | |
| Softwares, algorithm | Pclamp 10 acquisition | Molecular Devices | ||
| Softwares, algorithm | OmniPlex neural recording data acquisition system | Plexon | ||
| Softwares, algorithm | Offline Sorter V4.0 | Plexon | ||
| Softwares, algorithm | Neuroexplorer V5.0 | Plexon | ||
| Softwares, algorithm | Matlab R2019b | MathWorks | ||
| Softwares, algorithm | Photoshop | Adobe | ||
| Softwares, algorithm | Prism 8 | GraphPad Software | RRID:SCR_002798 | |
| Softwares, algorithm | ImageEP software | PMID:19229173 | ||
| Softwares, algorithm | ImageLD software | PMID:18704188 | ||
| Softwares, algorithm | ImageOF software | https://cbsn.neuroinf.jp/modules/xoonips/detail.php?id=ImageOF |





