All-trans retinoic acid induces synaptopodin-dependent metaplasticity in mouse dentate granule cells
Figures
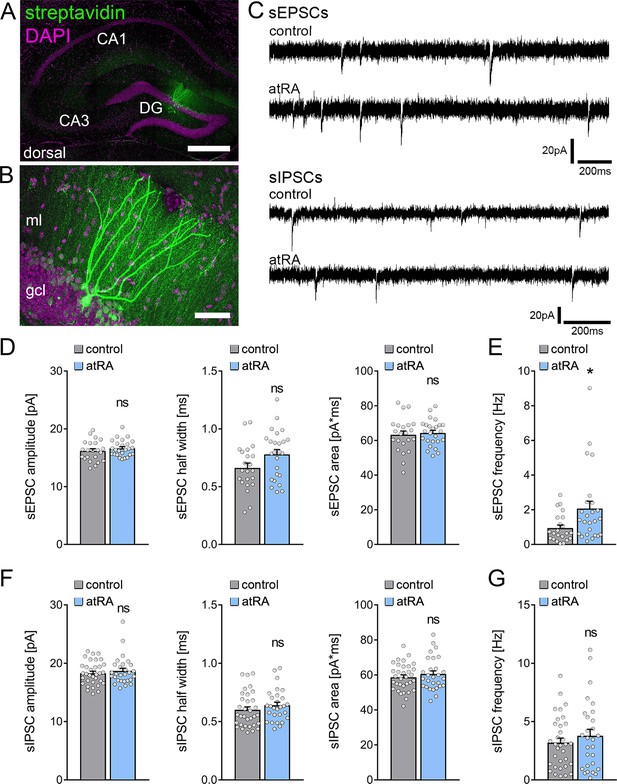
All-trans retinoic acid (atRA) induces no major changes in excitatory and inhibitory synaptic strength in dentate granule cells of the dorsal hippocampus.
(A, B) Example of patched and post hoc identified dentate granule cell in acute slices prepared from the dorsal hippocampus. Scale bar (upper panel)=500 µm; Scale bar (lower panel)=50 µm. (C) Sample traces of spontaneous excitatory postsynaptic currents (sEPSCs) and spontaneous inhibitory postsynaptic currents (sIPSCs) recorded from dentate granule cells of atRA (10 mg/kg; i.p.)-treated or vehicle-only (control) animals. (D, E) Group data of sEPSC recordings. A significant increase in the sEPSC frequency is observed (ncontrol=22 cells, natRA=25 cells in four animals; Mann-Whitney test, UsEPSC frequency=175). (F, G) Group data of sIPSC recordings (ncontrol=33 cells, natRA=28 cells in four animals; Mann-Whitney test). Individual data points are indicated by gray dots. Values represent mean ± SEM. (*, p<0.05; ns, non-significant difference). DG, dentate gyrus; gcl, granule cell layer; ml, molecular layer.
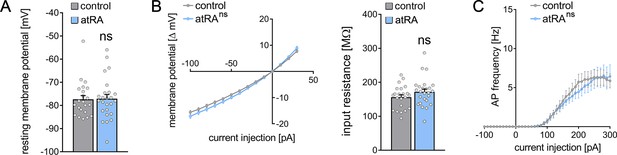
Passive or active membrane properties of dentate granule cells remain unchanged in the dorsal hippocampus following intraperitoneal administration of all-trans retinoic acid (atRA).
(A, B) Group data of resting membrane potentials, input-output curves, and input resistances. (C) A slight but not significant decrease in action potential (AP) frequency of dentate granule cells is observed in the atRA group (ncontrol = 22 cells, natRA = 25 cells in four animals each; Mann-Whitney test for column statistics, RM two-way ANOVA followed by Sidak’s multiple comparisons test for input-output curve and AP frequency analysis). Individual data points are indicated by gray dots. Values represent mean ± SEM. (ns, non-significant difference).
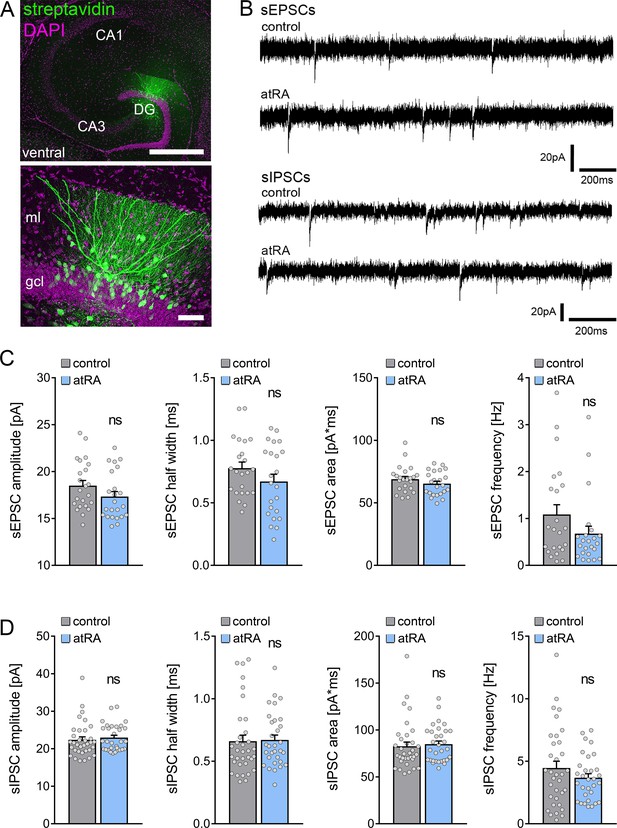
All-trans retinoic acid (atRA) does not induce changes in excitatory and inhibitory neurotransmission in the ventral hippocampus of adult mice.
(A) Example of patched and post hoc identified dentate granule cell in acute slices prepared from the ventral hippocampus. Scale bar (upper panel)=500 µm; Scale bar (lower panel)=50 µm. (B) Sample traces of spontaneous excitatory postsynaptic currents (sEPSCs) and spontaneous inhibitory postsynaptic currents (sIPSCs) recorded from dentate granule cells of atRA-treated or vehicle-only (control) animals. (C) Group data of sEPSC recordings (ncontrol=23 cells, natRA=23 cells in four animals each; Mann-Whitney test). (D) Group data of sIPSC recordings (ncontrol=34 cells, natRA=31 cells in four animals each; Mann-Whitney test). Individual data points are indicated by gray dots. Values represent mean ± SEM. (ns, non-significant difference). DG, dentate gyrus; gcl, granule cell layer; ml, molecular layer.
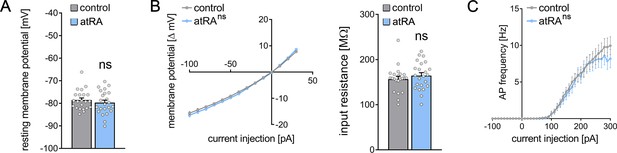
Passive or active membrane properties of dentate granule cells remain unchanged in the ventral hippocampus following intraperitoneal administration of all-trans retinoic acid (atRA).
(A–C) Group data of resting membrane potentials (A), input-output curves, and input resistances (B), and action potential (AP) frequency of dentate granule cells in the ventral hippocampus (ncontrol=22 cells, natRA=23 cells in four animals each; Mann-Whitney test for column statistics, RM two-way ANOVA followed by Sidak’s multiple comparisons test for input-output curve and AP frequency analysis). Individual data points are indicated by gray dots. Values represent mean ± SEM. (ns, non-significant difference).
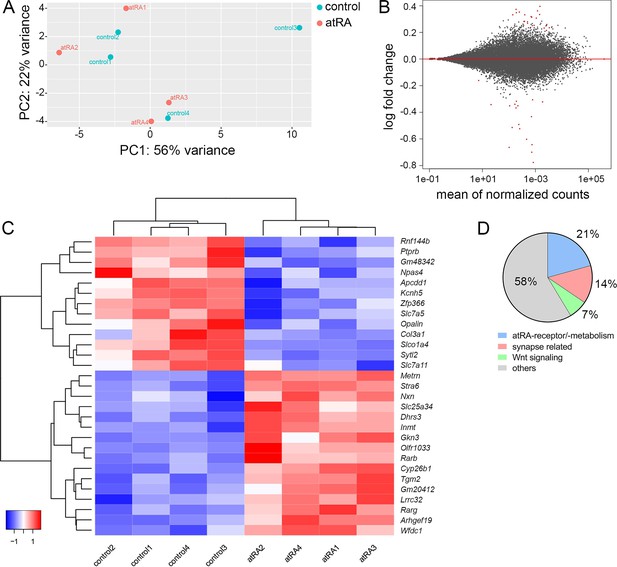
Hippocampal transcriptome analysis reveals no major differences in synapse-related genes following intraperitoneal injection of all-trans retinoic acid (atRA).
(A) Principal component analysis with ‘treatment’ as primary factor reveals no treatment-specific clustering of hippocampal mRNA samples (n=4 animals, one hippocampus each). (B) DESeq2-Analysis reveals differential expression of genes with a moderate │log2FC│<1 (visualization by MA plot). (C) Heatmap showing the z-scores of 29 differentially expressed genes. The differential expression of genes depends on the atRA treatment, as indicated by the z-score clustering. (D) Subsets of genes can be attributed to atRA-signaling or atRA-metabolism, synaptic transmission, and Wnt-signaling, respectively.
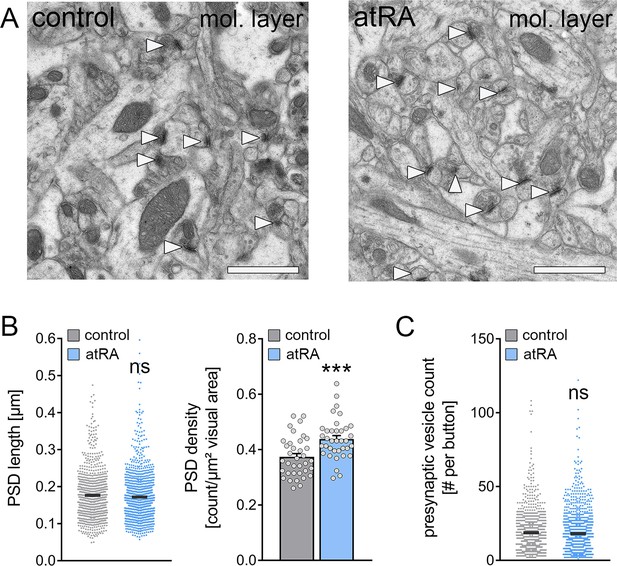
The numbers of cortico-hippocampal synapses in the dentate gyrus of the dorsal hippocampus are increased following intraperitoneal administration of all-trans retinoic acid (atRA).
(A) Representative electron microscopy images of asymmetric synapses (arrowheads) in the outer two-thirds of the molecular layer (mol. layer) of the dorsal hippocampus from atRA- or vehicle-only-injected mice. Scale bar=1 µm. (B) Group data of postsynaptic density (PSD) counts (ncontrol=36 individual images, natRA=36 individual images in three different animals with 12 images per sample; Mann-Whitney test, U=340) and analysis of PSD lengths of asymmetric synapses (ncontrol=908 PSDs, natRA=1189 PSDs in three different animals, one data point outside the axis limits in the control group; Mann-Whitney test). (C) Presynaptic vesicle counts are not significantly different between the groups (ncontrol=885 presynaptic buttons, natRA=1151 presynaptic buttons in three different animals, one data point outside the axis limits in the atRA group; Mann-Whitney test). Individual data points are indicated by gray dots. Values represent mean ± SEM. (***, p<0.001; ns, non-significant difference).
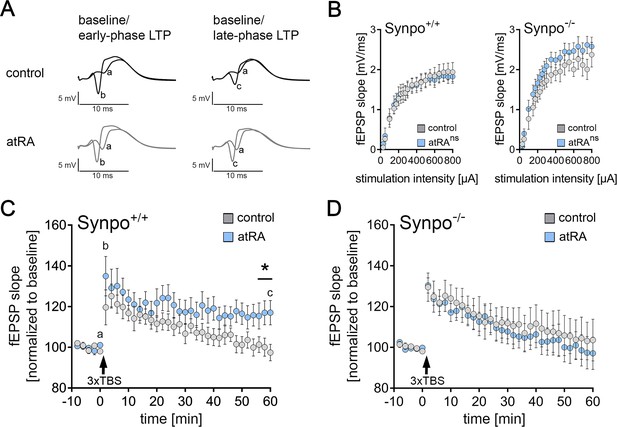
Intraperitoneal injection of all-trans retinoic acid (atRA) improves synaptic plasticity in the dentate gyrus of wild-type but not synaptopodin-deficient mice.
(A) In vivo long-term potentiation (LTP) experiments on perforant path synapses were carried out in anesthetized mice using a weak theta-burst stimulation (TBS) protocol. Representative traces of field excitatory postsynaptic potential (fEPSP) recordings in wild-type mice at indicated points in time (a–c) after induction of LTP in vehicle-only controls and atRA-injected mice (10 mg/kg, i.p.; 3–6 hr prior to recordings). (B) Input-output properties of wild-type and synaptopodin-deficient animals (Synpo+/+: ncontrol=9 animals, natRA=9 animals; Synpo−/−: ncontrol=7 animals, natRA=8 animals. RM two-way ANOVA with Sidak’s multiple comparisons). (C) Group data of fEPSP slopes in wild-type mice (Synpo+/+: ncontrol=9 animals, natRA=9 animals; Mann-Whitney test, U=13–17 for three terminal data points). (D) Group data of fEPSP slopes in synaptopodin-deficient mice (Synpo−/−: ncontrol=7 animals, natRA=8 animals; Mann-Whitney test). Values represent mean ± SEM. (*, p<0.05; ns, non-significant difference).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Chemical compound, drug | All-trans retinoic acid (atRA) | Sigma-Aldrich | Cat#: R2625 | Final concentration:10 mg/kgInjection vehicle: Corn oil+5% DMSO |
| Chemical compound, drug | Dimethyl Sulfoxide(DMSO) | Sigma-Aldrich | Cat#: D2650 | |
| Chemical compound, drug | Paraformaldehyde (PFA) | Carl Roth | Cat#: 0335.3 | Final concentration: 4% (w/v) in PB or PBS |
| Chemical compound, drug | Glutardialdehyd | Carl Roth | Cat#: 4157.2 | Final concentration: 2% (w/v) in PB |
| Chemical compound, drug | CNQX | Biotrend | Cat#: BN0153 | Final concentration: 10 µM |
| Chemical compound, drug | D-APV | Abcam | Cat#: ab120003 | Final concentration: 10 µM |
| Chemical compound, drug | DAPI (1 mg/ml in water) | Thermo Fisher Scientific | Cat#: 62248 | IF and post hoc labeling (1:5000) |
| Commercial assay or kit | Monarch Total RNA Miniprep Kit | New England Biolabs | #T2010S | |
| Genetic reagent (Mus musculus) | B6.129-Synpotm1Mndl/Dllr; Synpo−/− | Vlachos et al., 2013 PMID:23630268 | RRID:MGI:6423115 | Obtained from Deller Lab (Frankfurt); male |
| Peptide, recombinant protein | Streptavidin, Alexa Fluor 488-Conjugate | Invitrogen | Cat#: S32354 RRID:AB_2315383 | Post hoc labeling (1:1000) |
| Software, algorithm | Prism | GraphPad | RRID:SCR_002798 | |
| Software, algorithm | Clampfit (pClamp software package) | Molecular Devices | RRID:SCR_011323 | |
| Software, algorithm | ImageJ | RRID:SCR_003070 | ||
| Software, algorithm | Photoshop | Adobe | RRID:SCR_014199 | |
| Strain, strain background (M. musculus) | C57BL/6J; Synpo+/+ | Jackson Laboratory | RRID:IMSR_JAX:000664 |





