The mitotic spindle protein CKAP2 potently increases formation and stability of microtubules
Figures
Cytoskeleton-Associated Protein 2 (CKAP2) is an intrinsically disordered protein that increases microtubule formation.
(a) Schematic of mmCKAP2 protein domains and disorder prediction (Dosztányi et al., 2005; Obradovic et al., 2003). (b) Circular dichroism of 4.5 μM CKAP2. (c) Coomassie Blue-stained sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS–PAGE) gel of 1 μg of purified recombinant CKAP2 constructs and 5 μg of purified tubulin. (d) Light scattering assay (turbidity) schematic and data following microtubule formation as apparent absorbance (Abs) over time for 8 µM tubulin with increasing concentrations of CKAP2-mNeonGreen (CKAP2-mNG) (n = 1). (e) Turbidity data for tubulin alone (blue) and addition of CKAP2-mNG (n = 3, mean ± standard deviation [SD]).
-
Figure 1—source data 1
Protein purification gel.
- https://cdn.elifesciences.org/articles/72202/elife-72202-fig1-data1-v2.zip
Cytoskeleton-Associated Protein 2 (CKAP2) is an intrinsically disordered protein that increases microtubule assembly.
(a) Comparison of amino acid sequence conservation for CKAP2, TPX2, and chTOG between human (hs, Homo sapiens), mouse (ms, Mus musculus), and African clawed frog (xl, Xenopus laevis) and amino acid sequence alignment for CKAP2. Dark grey shading represents sequence identities, light grey shading sequence similarities. KEN box (green) and CKAP2_C domain (magenta) are underlined. (b) Turbidity assay with CKAP2-mNeonGreen (mNG) no-His construct (n = 1). (c) Turbidity assay with CKAP2 unlabelled (no-mNG) (n = 1). (d) Turbidity assay performed with CKAP2-mNG at 4°C (n = 2).
Cytoskeleton-Associated Protein 2 (CKAP2) lowers the critical concentration of microtubule growth and speeds up microtubule assembly rates.
(a) Dynamic microtubule growth assay schematic (upper panel). Microtubule dynamics for tubulin control and in the presence of CKAP2-mNeonGreen (mNG) are analyzed from space–time plots (kymographs, lower panel). (b) Plot of microtubule growth rates as a function of CKAP2-mNG concentration with 8 μM tubulin. Plotted as mean ± standard deviation (SD; n = 67, 24, 70, 67, 46 from 1 to 2 replicates) Tukey’s test and one-way analysis of variance (ANOVA) used to compare mean values of raw data. ****p ≤ 0.0001. (c) Plot of microtubule growth rates as a function of tubulin concentration for control (no CKAP2) and in the presence of CKAP2-mNG. Plotted as mean ± SD (blue; n = 272, 601, 347 from three replicates), 0.125 µM CKAP2-mNG (green; n = 146, 85, 91 from two replicates), 0.5 µM CKAP2-mNG (red; n = 48, 92, 91, 85, 55 from two replicates), and 1 µM CKAP2-mNG (purple; n = 68, 134, 143, 115 from two replicates). (d) Enlargement at low tubulin concentrations of microtubule growth rate plot from (c). (e) Plot of the apparent on-rate constant (ka) as a function of CKAP2-mNG concentration determined from linear regression fit of growth rates versus tubulin concentration. Error bars represent standard error (SE) of fit. (f) Plot of the apparent off-rate constant (kd) as a function of CKAP2-mNG concentration determined from linear regression fit of growth rates versus tubulin concentration. Error bars represent SE of fit. (g) Plot of the apparent critical concentration (Cc) as a function of CKAP2-mNG concentration determined from linear regression fit of growth rates versus tubulin concentration. Error bars represent SE of fit.
-
Figure 2—source data 1
The data and analysis for microtubule growth rates for different concentrations of Cytoskeleton-Associated Protein 2 (CKAP2).
- https://cdn.elifesciences.org/articles/72202/elife-72202-fig2-data1-v2.xlsx
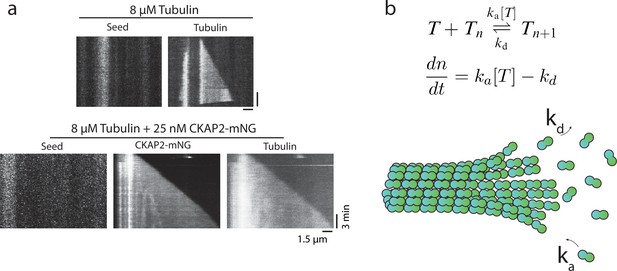
Cytoskeleton-Associated Protein 2 (CKAP2) lowers the critical concentration of microtubule growth and speeds up microtubule assembly rates.
(a) Representative kymograph of microtubule growth in the presence and absence of CKAP2-mNeonGreen (mNG). (b) Schematic of linear polymer growth equation used to derive apparent on-rate constant ka, the apparent off-rate constant kd, and critical concentration Cc (Oosawa et al., 1975). ka was calculated from the slope, kd, the y-intercept, and Cc the x-axis of the linear regression fit.
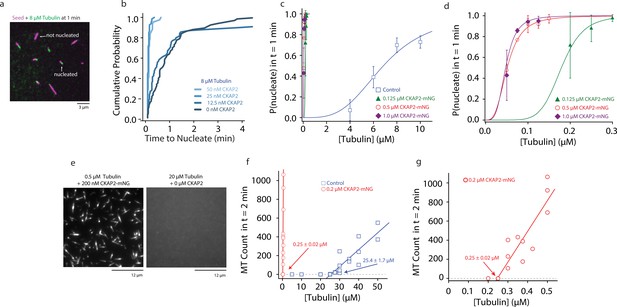
Cytoskeleton-Associated Protein 2 (CKAP2) increases templated and spontaneous microtubule nucleation.
(a) Representative field of view of guanosine-5′-[(α,β)-methyleno]triphosphate (GMPCPP)-stabilized microtubule seeds nucleating microtubules from 8 µM tubulin. (b) Plot of the cumulative probability distributions for time to nucleate as a function of CKAP2-mNeonGreen (mNG) concentration (n = 67, 70, 67, 46 from two replicates). (c) Plot of probability of GMPCPP seed to be nucleated within 1 min for control (blue; n = 102, 271, 115, 184 from ≥2 replicates), 0.125 µM CKAP2-mNG (green; n = 164, 213, 166 from two replicates), 0.50 µM CKAP2-mNG (red; n = 179, 216, 145, 126, 145 from two replicates), and 1 µM CKAP2-mNG (purple; n = 112, 186, 182, 148 from two replicates). Plotted as mean ± standard deviation (SD). Data were fit to a hill function forced to start at y = 0 and end at y = 1 (y = START + (END − START) × xn/(kn + xn)). Tubulin concentrations for half-maximal microtubule growth, C, and steepness of fit, s, for control (C = 6.85 ± 0.48 μM, s = 3.37 ± 0.87), 0.125 µM CKAP2-mNG (C = 0.18 ± 0.01 μM, s = 6.96 ± 1.39), 0.5 µM CKAP2-mNG (C = 0.05 ± 0.01 μM, s = 3.10 ± 0.20), and 1 µM CKAP2-mNG (C = 0.05 ± 0.01 μM, s = 3.79 ± 0.85). (d) Enlargement at low tubulin concentrations of probability to nucleate plot from (c). (e) Representative field of view of microtubules spontaneously nucleated in the absence of templates. (f) Quantification of spontaneous nucleation and linear fit showing a critical tubulin concentration for microtubule nucleation of 25.4 ± 1.7 µM for tubulin alone (blue; n = 20) and 0.25 ± 0.02 µM in the presence of 0.2 µM CKAP2 and 0.25 ± 0.02 µM (red; n = 14). (g) Enlargement at low tubulin concentrations of spontaneous nucleation in the presence of 0.2 µM CKAP2 from (e).
-
Figure 3—source data 1
The data and analysis for templated and spontaneous microtubule nucleation for different concentrations of Cytoskeleton-Associated Protein 2 (CKAP2).
- https://cdn.elifesciences.org/articles/72202/elife-72202-fig3-data1-v2.xlsx
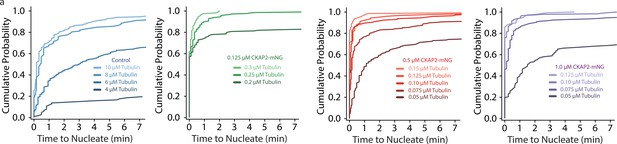
Cytoskeleton-Associated Protein 2 (CKAP2) increases templated and spontaneous microtubule nucleation.
(a) Plots of the cumulative probability distributions for time to nucleate as a function of tubulin concentration, same n as Figure 3C.
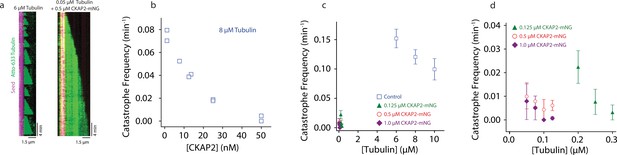
Cytoskeleton-Associated Protein 2 (CKAP2) suppresses catastrophe.
(a) Kymographs of representative microtubule growth in the absence and presence of CKAP2-mNeonGreen (mNG). (b) Plot showing the microtubule catastrophe frequencies as a function of CKAP2-mNG concentration. Each point represents the catastrophe frequency for an individual channel (n (MT growth events) = 30, 37, 24, 31, 49, 30, 35, 19, 25, number of catastrophes = 26, 34, 16, 16, 26, 8, 5, 0, 1 for channels CKAP2 concentrations 0, 0, 6.5, 12.5, 12.5, 25, 25, 50, 50 nM). (c) Plot showing the microtubule catastrophe frequencies as a function of tubulin concentration for control (blue; n (MT growth events) = 217, 676, 502, number of catastrophes = 217, 676, 502 from ≥2 replicates), 0.125 µM CKAP2-mNG (green; n (MT growth events) = 133, 222, 166, number of catastrophes = 21, 10, 4 from two replicates), 0.50 µM CKAP2-mNG (red; n = 161, 232, 95, 135, number of catastrophes = 9, 17, 2, 6 from two replicates), and 1 µM CKAP2-mNG purple (n (MT growth events) = 85, 171, 182, 148, number of catastrophes = 5, 7, 0, 1 from two replicates). Plotted as mean ± standard deviation (SD). (d) Enlargement at low tubulin concentrations of catastrophe frequency plot (b).
-
Figure 4—source data 1
The data and analysis for catastrophe frequency measurements.
- https://cdn.elifesciences.org/articles/72202/elife-72202-fig4-data1-v2.xlsx
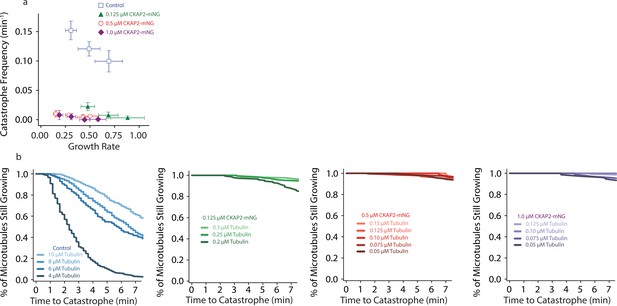
Cytoskeleton-Associated Protein 2 (CKAP2) suppresses catastrophe.
(a) Plot of tubulin growth rate versus catastrophe frequency. (b) Plot of the % of microtubules still growing (cumulative microtubule survival) as a function of time, same n as Figure 4B.
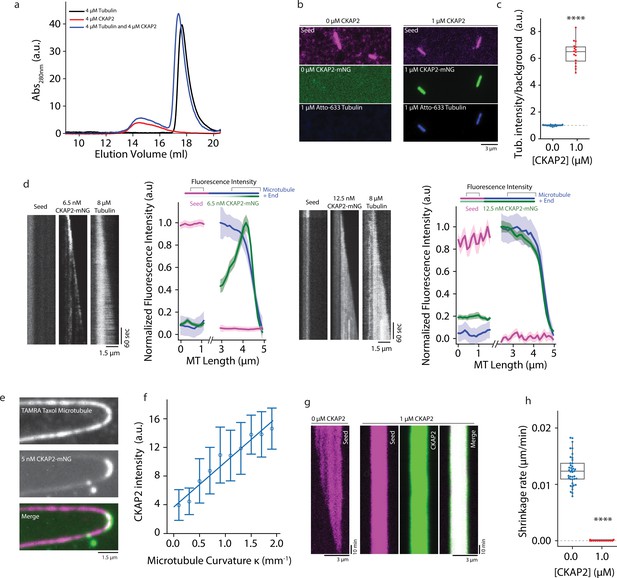
Cytoskeleton-Associated Protein 2 (CKAP2) recruits tubulin, recognizes lattice curvature, but does not catalyze microtubule depolymerization.
(a) Size exclusion chromatography traces for 4 µM CKAP2, 4 µM tubulin, and a combination of both. (b) Field of view showing 1 µM CKAP2-mNeonGreen (mNG) recruiting Atto-633 tubulin to guanosine-5′-[(α,β)-methyleno]triphosphate (GMPCPP) seeds. (c) Box plot of tubulin intensity on the GMPCPP lattice over background for control and 1 µM CKAP2-mNG (blue; n = 17 from one representative channel) and 1 µM CKAP2-mNG (red; n = 16 from one representative channel). Tukey’s test and one-way analysis of variance (ANOVA) used to compare mean values of raw data. ****p ≤ 0.0001. (d) Kymographs representative of 6.5 and 12.5 nM CKAP2-mNG on a growing microtubule seed and plus end. Plot of normalized average fluorescence intensity of CKAP2-mNG, microtubule seed, and dynamic microtubule lattice along microtubule seed and + end (6.5 nM) (n = 42 seed, 54 tip), 12.5 nM (n = 17 seed, 26 tip). Plotted as normalized background subtracted average ± standard error (SE). (e) Field of view of 5 nM CKAP2-mNG preferentially binding to curved regions of tetramethylrhodamine (TAMRA)-labelled paclitaxel-stabilized microtubules. (f) Plot of CKAP2-mNG intensity versus curvature (κ) measured using Kappa (Mary and Brouhard, 2019) (n = 73 from two replicates. Plotted as binned mean ± standard deviation [SD]). (g) GMPCPP seed depolymerization kymographs representative of n = 41 (control) and n = 16 (1 µM CKAP2-mNG). (h) Box plot displaying the shrinkage rate of GMPCPP microtubule seeds for control (blue; n = 41 from one representative channel) and in the presence of 1 µM CKAP2-mNG (red; n = 16 from one representative channel). Tukey’s test and one-way ANOVA used to compare mean values of raw data. ****p ≤ 0.0001.
-
Figure 5—source data 1
The data and analysis for tubulin recruitment by Cytoskeleton-Associated Protein 2 (CKAP2) and shrinkage rate for guanosine-5′-[(α,β)-methyleno]triphosphate (GMPCPP) microtubules in the presence and absence of CKAP2.
- https://cdn.elifesciences.org/articles/72202/elife-72202-fig5-data1-v2.xlsx

Cytoskeleton-Associated Protein 2 (CKAP2) recruits tubulin, recognizes lattice curvature, but does not catalyze microtubule depolymerization.
(a) Guanosine-5′-[(α,β)-methyleno]triphosphate (GMPCPP) ‘seed’ depolymerization kymograph for 1 µM CKAP2-mNeonGreen (mNG) over 10 hr.
Tables
| Published concentration for Xenopus (Table S2/S3, Wuehr et al. 2014) | |||||
|---|---|---|---|---|---|
| Α Tubulins | ng | Β Tubulins | ng | ||
| TUBA4A | Tubulin α-4A chain | 4741.14 | TUBB | Tubulin β chain | 5931.21 |
| TUBA3C | Tubulin α-3C/D chain | 1686.89 | TUBB2B | Tubulin β-2B chain | 1357.19 |
| TUBA1B | Tubulin α-1B chain | 1143.46 | TUBB4B | Tubulin β-4B chain | 1173.38 |
| TUBA1A | Tubulin α-1A chain | 790.93 | |||
| TUBA1C | Tubulin α-1C chain | 439.50 | |||
| 8801.91 | 8461.78 |
Additional files
-
Supplementary file 1
Available plasmids used for Cytoskeleton-Associated Protein 2 (CKAP2) protein expression.
- https://cdn.elifesciences.org/articles/72202/elife-72202-supp1-v2.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/72202/elife-72202-transrepform1-v2.docx