AR-V7 exhibits non-canonical mechanisms of nuclear import and chromatin engagement in castrate-resistant prostate cancer
Figures
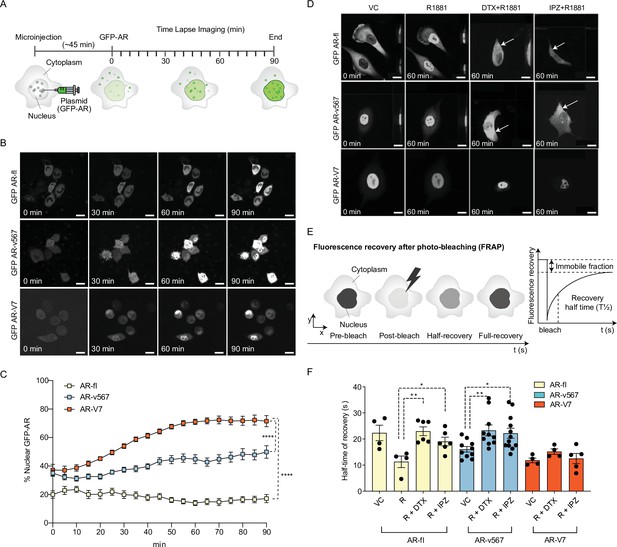
AR-V7 exhibits fast nuclear import kinetics independently of microtubules or the importin-α/β pathway, unlike AR-fl or AR-v567.
(A) Experimental design. Plasmids encoding GFP-tagged AR-fl, AR-v567, or AR-V7 were microinjected into the nuclei of the AR-null PC3 cells. As soon as GFP-tagged proteins were detected in the cytoplasm (~45 min post micro-injection), nuclear translocation kinetics was monitored by live-cell time-lapse confocal microscopy at 5 min intervals for a total of 90 min. (B) Representative time-lapse images showing subcellular localization of each GFP-tagged AR protein. Scale bar, 10 µm. (C) Quantitation of % nuclear GFP-AR protein in single cells (n=3–10 cells/condition/time point). (D) Effect of MT and importin-β inhibitors on AR-fl, AR-v567, and AR-V7 nuclear localization. M12 prostate cancer cells stably expressing GFP-tagged AR-fl or AR-v567 or AR-V7 were treated as indicated and subjected to live-cell time-lapse imaging. R1881: synthetic androgen; DTX: docetaxel, MT-stabilizing drug; IPZ: importazole, importin-β inhibitor. Representative images are shown. Arrows point to cells with cytoplasmic GFP-AR-fl or GFP-AR-v567. Scale bar, 10 µm. (E) Schematic overview of Fluorescence Recovery After Photobleaching (FRAP) assay and its quantitative output. (F) Effect of MT and importin-β inhibitors on AR-fl, AR-v567, and AR-V7 nuclear translocation kinetics following FRAP. T1/2 times in s are shown for each respective protein (n=4–12 cells/condition). Data represent mean ± SEM, p value (*p<0.05, **p<0.01, ****P<0.0001) was obtained using unpaired two-tailed t-test. Experiments were repeated at least twice.
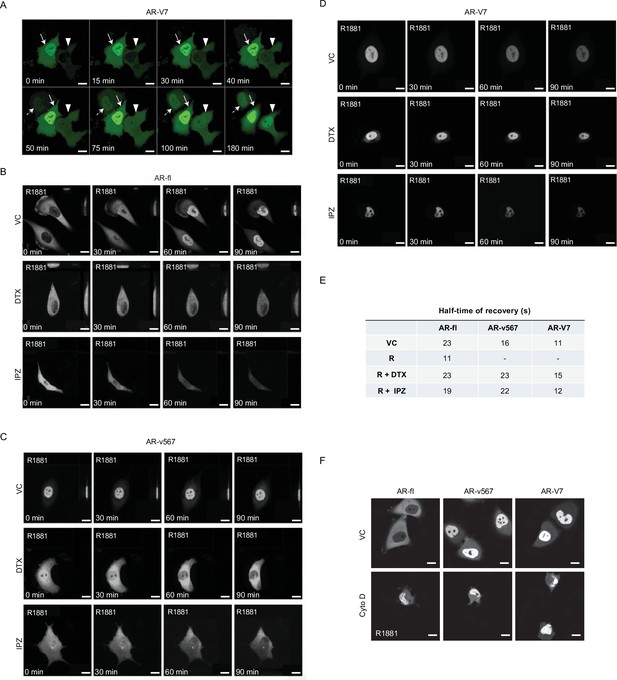
AR-V7 exhibits fast nuclear import kinetics independently of microtubules, actin, or the importin-α/β pathway.
(A) Plasmid encoding GFP-tagged AR-V7 was microinjected into nuclei of PC3 cells and as soon as GFP was detected in the cytoplasm (~45 min post micro-injection) the kinetics of GFP-AR-V7 nuclear import were monitored by live-cell time-lapse confocal microscopy at 5 min intervals for a total of 180 min. Representative time-lapse images are shown at the indicated time points. Solid arrow: cell with both cytoplasmic and nuclear AR-V7 at time 0; Arrowhead: cells with cytoplasmic only AR-V7 at time 0; Dashed Arrow: cell with primarily nuclear AR-V7 first detected at +50 min after the start of imaging. Enhanced AR-V7 nuclear translocation is observed over time for all cells. Notice that there are cells with already extensive nuclear accumulation of AR-V7 at 0 min, suggesting very fast nuclear import kinetics from the time of microinjection (–45 min). Scale bar, 10 µm. (B–D) Corresponds to Figure 1D with additional time points. Briefly, M12 prostate cancer cells stably expressing GFP-tagged (B) AR-fl, (C) AR-v567, or (D) AR-V7 were treated as indicated and subjected to live-cell time-lapse imaging. R1881: synthetic androgen used to stimulate AR-fl nuclear translocation; DTX: docetaxel, MT-stabilizing drug; IPZ: importazole, importin-β inhibitor. Representative images are shown. Scale bar, 10 µm. (E) Table with T1/2 (half-time recovery) values for each variant (related to Figure 1F). (F) PC3 cells were treated with 1 μg/ml cytochalasin D (Cyto D) or vehicle control (VC) for 1 hr at 37°C following plasmid micro-injection into the nuclei of PC3 cells. Cells were then treated with 10 nM R1881 for 4 hr and subjected to point-scanning confocal microscopy. Representative images showing are shown. Scale bar, 10 µm. Experiments were repeated at least twice.
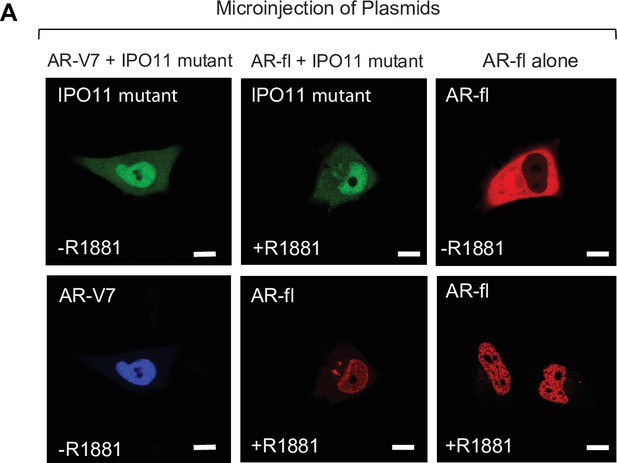
Dominant negative IPO11 does not abrogate the nuclear import of AR-fl or AR-V7.
(A) Plasmid encoding mCherry tagged AR-fl and/or a dominant negative mutant IPO11 was microinjected into PC3 cells.; 45 mins post microinjection, representative images were captured by live -cell confocal microscopy.
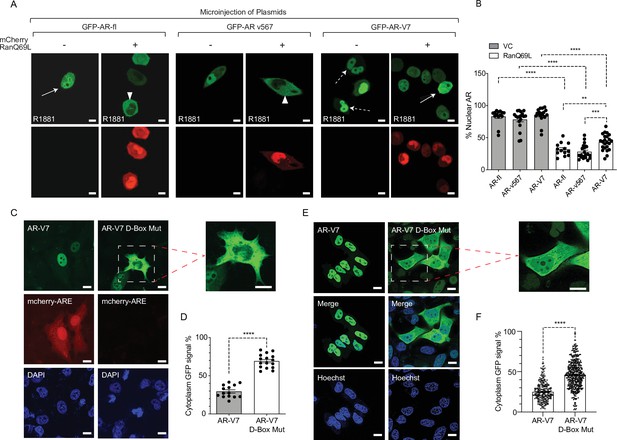
Inactivation of Ran-GTP-mediated nuclear transport affects differentially the subcellular localization of AR-variants.
(A, B) Plasmids encoding GFP-tagged AR-fl, AR-v567, or AR-V7 were co-microinjected with plasmid encoding the catalytic mutant mCherry-tagged RanQ69L, into the nuclei of the AR-null PC3 cells. Cells expressing both tagged proteins were subjected to live-cell time-lapse imaging. Cells were treated with R1881 (10 nM) to induce AR-fl nuclear import and was kept present with the variants so that all conditions were the same. Expression of mCherry-tagged RanQ69L affected differentially the subcellular localization of each GFP-tagged AR proteins. Solid arrow: cell with both cytoplasmic and nuclear AR proteins; arrowheads: cytoplasmic AR proteins; dashed arrow: nuclear AR proteins. % Nuclear AR across conditions is graphically displayed in B (n>10 per condition). (C–F) AR-V7 nuclear import is impaired upon mutation of the dimerization box domain (D-box). PC3 cells stably expressing ARE-mCherry reporter were transfected with GFP-AR-V7 or GFP-AR-V7 D-box mutant (A596T and S597T). Representative images are shown (inset displays higher magnification of the indicated cell) and quantitative results are graphically displayed in (D) (n>10 cells per condition). (E, F) C4-2 cells stably expressing inducible GFP-AR-V7 or GFP-AR-V7 D-Box mutant were used to quantify subcellular AR-V7 localization following doxycycline induction. Representative images are shown (inset displays higher magnification of the indicated cell) and quantitative results are graphically displayed (F). ( n> 200 cells per condition). Data represent mean ± SEM, p value (**p<0.01, ***p<0.001, ****p<0.0001) was obtained using unpaired two-tailed t-test. Scale bar, 10 μm. Experiments were repeated at least twice.
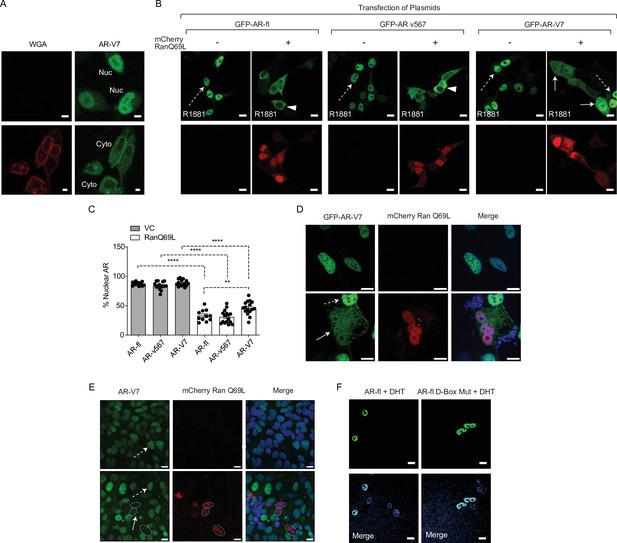
AR-V7 nuclear import requires active transport via the nuclear pore complex is dependent on Ran-GTP activity and is impaired upon mutation of the dimerization box domain (D-box).
(A) Wheat germ agglutinin (WGA) blocks AR-V7 nuclear import: we incubated cells with WGA, an inhibitor of nucleoporin-mediated nuclear transport and monitored GFP-AR-V7 localization by live-cell imaging. WGA kept AR-V7 in the cytoplasm in the presence of doxycycline suggesting that AR-V7 nuclear import requires active transport via the NPC. Representative confocal microscopy images were shown. Red: WGA labeling of membranes; Green: GFP-AR-V7. Nuc: nuclear AR-V; Cyto: cytoplasmic AR-V7. Scale bars represent 10 µm. (B) (related to Figure 2B) HEK293T cells were transfected with plasmids encoding GFP-tagged AR-fl, AR-v567 and AR-V7 in the presence of the catalytic mutant Ran-GTP (mCherry-tagged RanQ69L). Nuclear accumulation of each AR variant was calculated. Representative confocal microscopy images (63× magnification) for each condition are shown. Solid arrow: cell with both cytoplasmic and nuclear proteins; arrowheads: cells with cytoplasmic protein; dashed arrow: cell with nuclear protein. Scale bars represent 10 µm. (C) Graphic display of % Nuclear AR across 30 individual cells per condition. Data represent mean ± SEM with n>10, p-value (**p<0.01, ****p<0.0001) was obtained using unpaired two-tailed t-test. (D) C4-2 cells with endogenous AR-fl stably expressing inducible GFP-AR-V7 were transfected with the catalytic mutant mCherry-tagged RanQ69L. Representative confocal microscopy images are shown. Cytoplasmic enrichment for AR-V7 is shown when the mutant Ran is co-expressed in the same cell. (E). 22RV1 cells with endogenous AR-fl and AR-V7 were transfected with the catalytic mutant mCherry-tagged RanQ69L. AR-V7 was detected by immunostaining with the RevMab antibody. Downregulation of endogenous AR-V7 protein was observed in cells co-expressing RanQ69L. Solid arrow: cell with cytoplasmic and nuclear AR-V7; dashed arrow: non-transfected cells with endogenous nuclear AR-V7 Dotted ellipses: cells transfected with mutant Ran where AR-V7 appears absent or downregulated. Scale bar, 10 µm. (F). PC3 cells were transfected with GFP-AR-fl or GFP-AR-fl-D-box mutant (A596T and S597T) and they were treated for 4 hr with 10 nM DHT. There was no effect of the D-box mutations on AR-fl nuclear import. Experiments were repeated at least twice.
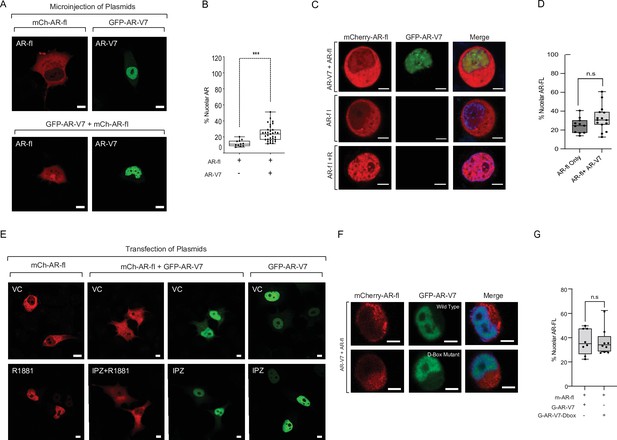
AR-V7 drives nuclear translocation of AR-fl in the absence of ligand.
(A, B) Plasmids encoding mCherry-AR-fl or GFP-AR-V7 were micro-injected in PC3 cells. Representative microscopic images (scale bar, 10 μm) and % nuclear AR is shown. Data represent a box plot with n>10cells per condition, p value (***p<0.001) was obtained using unpaired two-tailed t-test. (C, D) LNCaP cells were transfected with mCherry-ARfl and/or GFP-AR-V7, and cells were treated 10 nM R1881 as indicated (scale bar, 5 μm). Data quantification is shown as a box plot and was obtained using unpaired t-test with n≥10 cells per condition. (E) PC3 cells were transfected with mCherry-AR-fl, or GFP-AR-V7, and cells were treated 10 nM R1881 or 50 μM Importazole, as indicated. Representative confocal microscopy images are shown with arrows pointing to nuclear AR-fl across conditions. Scale bar, 10 μm. Experiments were repeated at least twice. (F, G) C4-2 cells stably expressing inducible GFP-AR-V7 or GFP-AR-V7 D-Box mutant were induced by doxycycline and transfected with mCherry-AR-fl (scale bar, 5 μm). Representative images are shown, data quantification is shown as a box plot, and obtained using a Mann-Whitney test with n≥ 8 per condition.
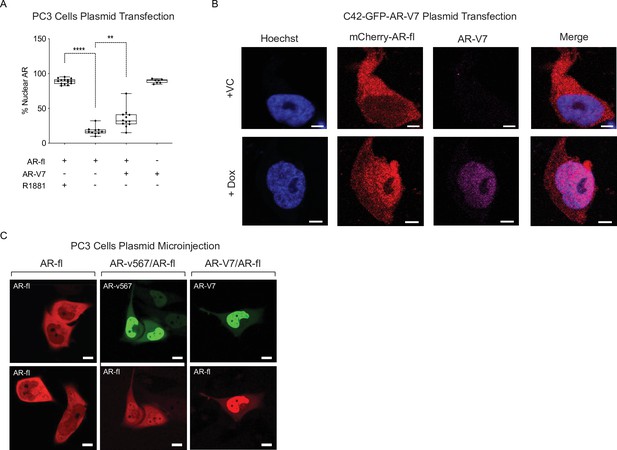
AR variants enhance nuclear localization of AR-fl in the absence of ligand.
(A) In a separate experiment, PC3 cells were transfected with plasmids encoding mCherry-AR-fl and/or GFP-AR-V7. % Nuclear AR-fl is graphically represented (n~7–14 cells/condition). Data represent mean ± SEM with n>10 cells per condition, p value (**p<0.01,****p<0.0001) was obtained using unpaired two-tailed t-test. (B) C4-2 cells stably expressing inducible GFP-AR-V7 were transfected with mCherry-AR-fl. Cells were fixed and immmunostained with rabbit monoclonal anti-AR-V7 (RevMab) and mouse monoclonal anti-mCherry. Nuclei are counterstained with Hoechst. Representative images of each condition are shown. Experiments were repeated at least twice. (C) Plasmids encoding mCherry-AR-fl, GFP-AR-V7, or GFP-AR-v567 were microinjected in PC3 cells. Representative images are shown. Scale bar, 10 µm.
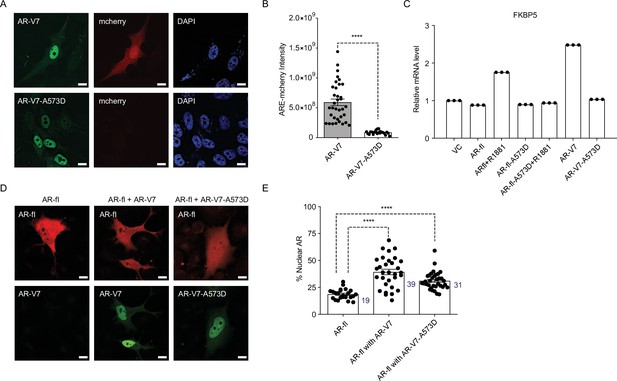
DBD mutation abrogates AR-V7 transcriptional activity (A, B) PC3 cells stably expressing ARE-mCherry reporter were transfected with the indicated plasmids for 48 hr and the expression of GFP protein with concomitant mCherry protein was analyzed by confocal imaging.
Representative images and quantitative results are shown (n>10 cells per condition). Scale bar, 10 µm. (C) RT-qPCR for the endogenous FKBP5 mRNA was quantified in PC3 cells after transfection of indicated plasmids. Data with AR-fl A573D are included as a control (n=3). (D) PC3 cells were transfected with mCherry-AR-fl or GFP-AR-V7 or GFP-AR-V7-A573D and imaged by confocal microscopy (scale bar, 10 µm) and (E). % Nuclear AR protein was quantified (n>23 cells per condition). Data represent mean ± SEM, p value (****p<0.0001) was obtained using unpaired two-tailed t-test. Experiments were repeated at least twice.
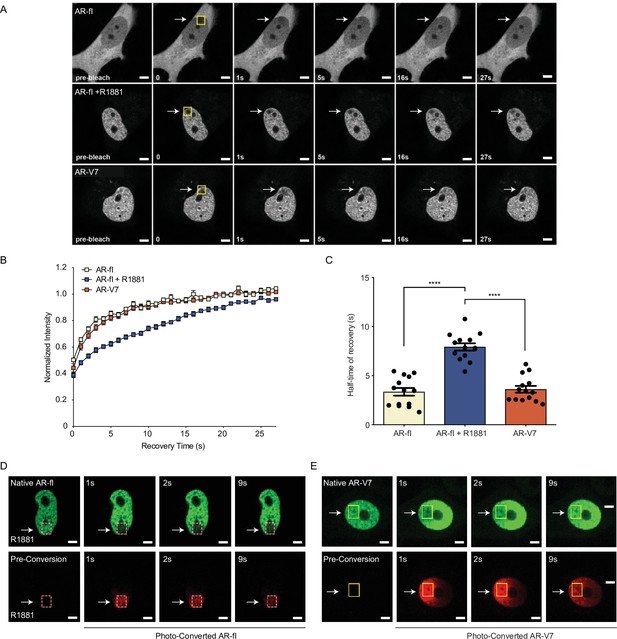
AR-V7 exhibits high intranuclear mobility compared with liganded AR-fl.
(A) FRAP was performed in PC3 cells transiently expressing GFP-AR-fl (in the absence or presence of 10 nM R1881) or GFP-AR-V7. FRAP was monitored at 1 s intervals. Representative images of cells at select time points are shown. (B) Kinetics of proteins recovery after photobleaching at 1 s intervals are graphically displayed, n=14. (C) Graphic display of half-time of recovery (T1/2) in seconds (s) for each condition, n=14. (D–E) FRAP was performed in PC3 cells transiently expressing photoconvertible mEos4b-AR-fl or mEos4b-AR-V7 protein. Cells were imaged at 1 s intervals to monitor fluorescence recovery of the non-converted proteins (Green) and nuclear distribution of photo-converted proteins (Red). Scale bar, 10 µm. Data represent mean ± SEM, p value (****p<0.0001) was obtained using unpaired two-tailed t-test. Experiments were repeated at least twice.
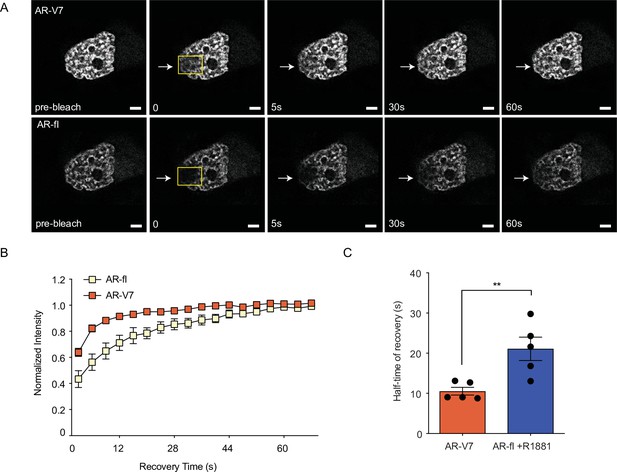
AR-V7 intranuclear mobility is not affected by co-expression of ligand-bound AR-fl.
(A). FRAP was performed in PC3 cells co-microinjected with mCherry-AR-fl and GFP-AR-V7 followed by treatment with 10 nM R1881 for 2 hr. Representative images from the same single cell co-expressing the two AR proteins are shown. (B). Kinetics of protein recovery after photobleaching for each protein, when co-expressed in the same single cell, are graphically displayed. The fluorescence intensity in the bleached area was measured and depicted as the normalized recovery. (C). Half-time of recovery (T1/2) required for the fluorescence intensity to reach 50% of its pre-bleach intensity was compared, n=5. Data represent mean ± SEM, p value (**p<0.01) was obtained using unpaired two-tailed t-test. Experiments were repeated at least twice.
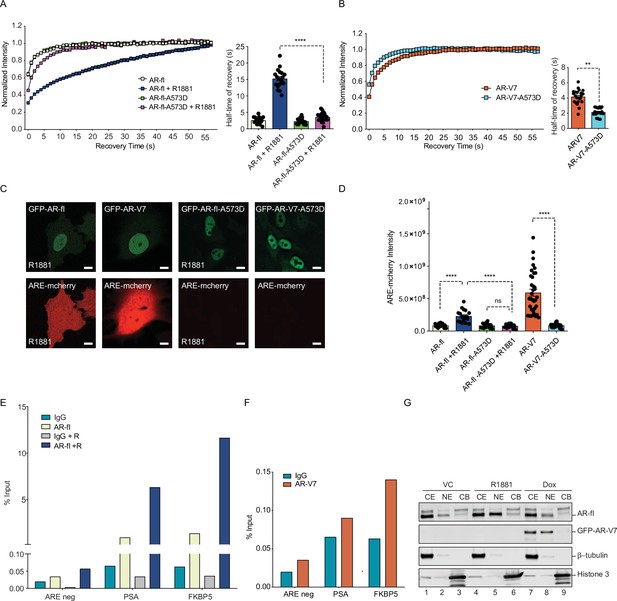
DNA-binding mutation increases the intranuclear mobility and abrogates the transcriptional activity of AR-fl and AR-V7.
(A, B) FRAP was performed in PC3 cells transiently expressing GFP-AR-fl or GFP-AR-V7 or their respective DBD mutants (A573D). Kinetics of protein recovery after photobleaching are graphically displayed and their half-time of recovery, is obtained for each condition, n>14. (C) PC3 cells stably expressing ARE-mCherry reporter were transfected with indicated plasmids, in the presence or absence of ligand (10 nM R1881). Representative images of each condition are shown. (D) quantitation of mCherry fluorescence intensity in single cells (n>17). (E, F) The binding of (E) AR-fl or (F) AR-V7 on the enhancer of PSA or FBKP5 in 22RV1 was analyzed by ChIP-QPCR assay. Cells in charcoal stripped media were treated with vehicle or 10 nM R1881 for 24 hr. (G) Immunoblot for AR-fl and AR-V7 following subcellular fractionation CE, cytosolic extract; NE, nuclear extract, CB, chromatin-bound nuclear extract. Histone H3 and β-tubulin were used as controls for the fractionation. Data represent mean ± SEM, p value (**p<0.01, ****p<0.0001) was obtained using unpaired two-tailed t-test. Experiments were repeated at least twice.
-
Figure 6—source data 1
DNA-binding mutation increases the intranuclear mobility and abrogates the transcriptional activity of AR-fl and AR-V7.
- https://cdn.elifesciences.org/articles/73396/elife-73396-fig6-data1-v2.pdf
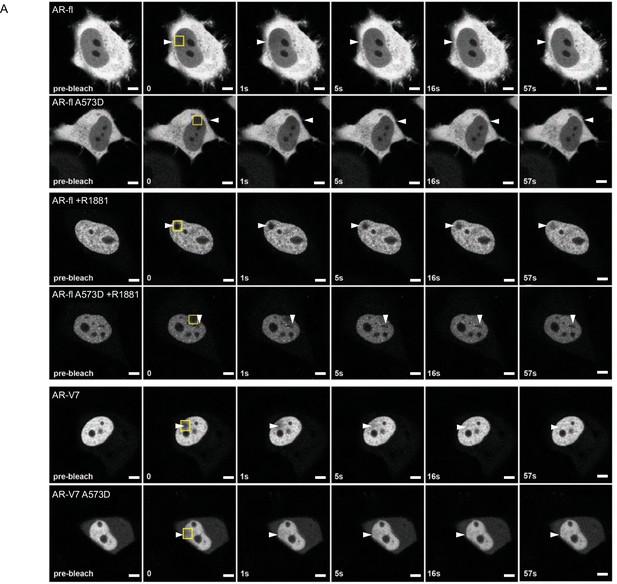
DBD mutation increases the intranuclear mobility of liganded-AR-fl and AR-V7.
(A) FRAP was performed in the nuclei of PC3 cells following transient expression of GFP-AR-fl or GFP-AR-fl A573D (in the presence of 10 nM R1881) or GFP-AR-V7 or GFP-AR-fl A573D. Representative images of cells at the indicated time points are shown. Arrow heads show the photo-bleaching area. Scale bar, 10 µm. Experiments were repeated at least twice.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Cell line (Homo sapiens) | PC3 | ATCC | RRID: CVCL_4885 | |
| Cell line (H. sapiens) | LNCaP | ATCC | RRID: CVCL_0395 | |
| Cell line (H. sapiens) | C4-2 | ATCC | RRID: CVCL_4782 | |
| Cell line (H. sapiens) | HEK-293T | ATCC | RRID: CVCL_0063 | |
| Cell line (H. sapiens) | 22rv1 | ATCC | RRID: CVCL_1045 | |
| Cell line (H. sapiens) | M12 | ATCC | RRID: CVCL_4860 | |
| Antibody | (Rabbit polyclonal) anti-AR-N-terminal (AR-N-21) | This paper | Custom antibody produced by the Giannakakou Lab (1:500) | |
| Antibody | (Rabbit monoclonal) anti-AR-V7 | RevMab | Cat #: 31-1109-00 RRID: AB_2716436 | (1:500) |
| Antibody | (Rabbit monoclonal) anti-AR-C-terminal | Abcam | Cat #: ab52615 RRID: AB_867653 | (1:1000) |
| Antibody | (Rabbit polyclonal) anti-actin | Sigma-Aldrich | Cat #: A2066 RRID: AB_476693 | (1:500) |
| Antibody | (Rabbit polyclonal) anti-beta tubulin | Abcam | Cat #: ab6046 RRID: AB_2210370 | (1:500) |
| Antibody | (Rabbit polyclonal) anti-Histone H3 | Abcam | Cat #: ab1791 RRID: AB_302613 | (1:1000) |
| Antibody | (Rabbit polyclonal) anti-GFP | Novus Biologicals | Cat #: NB600-303 RRID: AB_10001300 | (1:500) |
| Antibody | (Mouse monoclonal) anti-mCherry 1C51 | Novus Biologicals | Cat #: NBP1-96752 RRID: AB_11034849 | (1:500) |
| Antibody | Wheat Germ Agglutinin (WGA) - Alexa Fluor 594 Conjugate | Molecular Probes | Cat #: W11262 RRID: AB_2334867 | 5 µg/ml |
| Antibody | (Goat anti-Rabbit) IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor 488 | Thermo Fisher Scientific | Cat#: A-11008 | (1:10,000) |
| Antibody | (Goat anti-Mouse) IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor 568 | Thermo Fisher Scientific | Cat#: A-11004 | (1:10,000) |
| Chemical compound, drug | Docetaxel (Taxotere) | Sigma-Aldrich | Cat #: 01885 | 1 µM |
| Chemical compound, drug | Importazole | Sigma-Aldrich | Cat #: SML0341 | 50 µM |
| Chemical compound, drug | Cytochalasin D | Sigma-Aldrich | Cat #: C8273 | 1 µg/ml |
| Chemical compound, drug | Hygromycin | Santa Cruz Biotechnology | Cat #: sc-29067 | (1:1000) |
| Chemical compound, drug | Doxycycline | Enzo Life Sciences | Cat #: ALX380273G005 | 1 µg/ml |
| Chemical compound, drug | R1881 | Sigma-Aldrich | Cat #: R0908 | 10 nM |
| Chemical compound, drug | Puromycin Dihydrochloride | Thermo Fisher Scientific | Cat #: A1113803 | 1 µg/ml |
| Chemical compound, drug | Hoechst 33342, Trihydrochloride, Trihydrate | Thermo Fisher Scientific | Cat #: H3570 | 10 µg/ml |
| Transfected construct | pmCherry-AR-fl (PC3) | This paper | Produced by Giannakakou Lab | |
| Transfected construct | pEGFP-C1-AR-fl (PC3, M12) | This paper, Thadani-Mulero et al., 2014 | Produced by Giannakakou Lab | |
| Transfected construct | pEGFP-C1-AR-v567 (PC3, M12) | This paper, Thadani-Mulero et al., 2014 | Produced by Giannakakou Lab | |
| Transfected construct | pEGFP-AR-V7 (PC3,M12) | This paper, Thadani-Mulero et al., 2014 | Produced by Giannakakou Lab | |
| Transfected construct | pEGFP-C1-AR-fl A573D/ (PC3) | This paper | Produced by Giannakakou Lab | |
| Transfected construct | pEGFP-C1-AR-V7 A573D/ (PC3) | This paper | Produced by Giannakakou Lab | |
| Transfected construct | pEGFP-C1-AR-fl (A596T/S597T) | This paper | Produced by Giannakakou Lab | |
| Transfected construct | pEGFP-C1-AR-V7 (A596T/S597T) | This paper | Produced by Giannakakou Lab | |
| Recombinant DNA reagent | mEos4b-C1 | Paez-Segala et al., 2015 | Obtained from Dr. Michael Davidson | |
| Transfected construct | mEos4b-AR-fl (PC3) | This paper | Produced by Giannakakou Lab | |
| Transfected construct | mEos4b-AR-v567 (PC3) | This paper | Produced by Giannakakou Lab | |
| Transfected construct | mEos4b-AR-V7 (PC3) | This paper | Produced by Giannakakou Lab | |
| Transfected construct | pCW57.1-GFP-AR-V7 (C42) | This paper | Produced by Giannakakou Lab | |
| Transfected construct | pCW57.1-GFP-AR-V7 DBox (C42) | This paper | Produced by Giannakakou Lab | |
| Transfected construct | pmCherry-C1-RanQ69L (22rv1, PC3) | Kazgan et al., 2010 | Obtained from Dr. Jay Brenman | |
| Transfected construct | CS-GS241B-mCHER-LV152 lentivirus (PC3) | Azeem et al., 2017 | Obtained from Dr. Karl-Henning Kalland | |
| Recombinant DNA reagent | pCW57.1 tet-on vector | Addgene | Cat #: 41393 RRID: Addgene_41393 | Obtained from Dr. David Root |
| Software, algorithm | Zeiss Zen Software | Zeiss | RRID:SCR_013672 | https://www.zeiss.com/ microscopy/ us/products/microscope-software/zen-lite.html |
| Software, algorithm | GraphPad Prism | GraphPad | RRID:SCR_005375 | http://www.graphpad.com/scientific-software/prism/ |
| Software, algorithm | Fiji | ImageJ | RRID:SCR_002285 | https://imagej.net/Fiji |
| Chemical compound | Fast SYBR Green Mastermix | Thermo Fischer Applied Biosystems | Cat #: 4385612 | |
| Commercial assay, kit | Macherey-Nagel Kit | Macherey-Nagel | Cat #: 740609.250 | |
| Commercial assay, kit | Nucelofector Kit R | Lonza Bioscience | Cat #: VVCA-1001 | Used LNCaP program T007 with Amaxa II |
| Commercial assay, kit | FuGENE HD Transfection Reagent | Promega Corporation | Cat #: E2691 | |
| Sequence-based reagent | Q-PCR Amplification Primer FKBP5e: Fwd: GGT TCC TGG GCA GGA GTA AG Rev: AAC GTG GAT CCC ACA CTC TC | IDT DNA | ||
| Sequence-based reagent | Q-PCR Amplification Primer PSAe: Fwd-TGG GAC AAC TTG CAA ACC TG Rev-GAT CCA GGC TTG CTT ACT GT | IDT DNA | ||
| Sequence-based reagent | Q-PCR Amplification Primer AREneg: Fwd: GCT GAT TCA ATT ACC TCC CAG AA Rev: AGT TTG GGA CAG ACG GGA AA | IDT DNA |





