Kidney organoids recapitulate human basement membrane assembly in health and disease
Figures
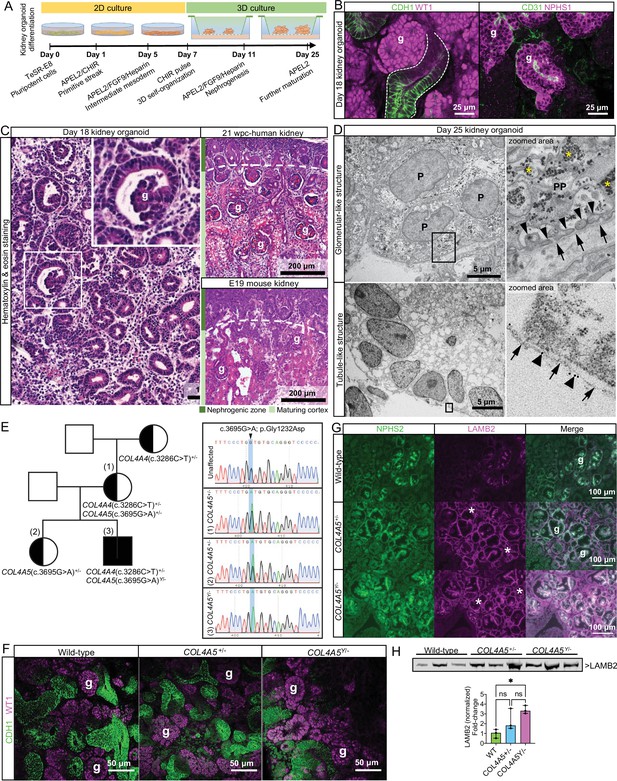
Kidney organoid basement membranes are altered in human disease.
(A) Schematic representing the differentiation of induced pluripotent stem cells (iPSCs) to 3D kidney organoids. (B) Whole-mount immunofluorescence for kidney cell types: left image shows glomerular structures (g) with WT1+ cells and CDH1+ tubule segments (dashed line); right image shows a glomerular-like structure (g) containing podocytes (NPHS1+) and endothelial cells (CD31+). (C) Representative photomicrographs of day 18 kidney organoids (left) and human and mouse fetal kidneys (right) to demonstrate the comparable histological structure; inset shows an organoid glomerular structure (g). (D) Transmission electron photomicrographs of glomerular- (upper panels) and tubule-like structures (lower panels) in a day 25 kidney organoid. In the top-right zoomed area, note the features of organoid podocytes (P): a primary process (PP) and distinct intercalating foot processes (thin arrowheads) lining a basement membrane (arrows). Asterisks indicate glycogen granules. In the lower panels, a tubule-like structure in the organoid, and a basement membrane (arrows) labeled with a 10 nm gold-conjugated anti pan-laminin antibody (see large arrowheads in the zoomed area). (E) Right: pedigree from a family with a likely pathogenic missense variant in COL4A5 (c.3695G>A; p.Gly1232Asp, posterior probability 0.988) and an uncertain significance variant in COL4A4 (c.3286C>T; p.Pro1096Ser [VUS], posterior probability 0.5). Left: Sanger sequencing data for the COL4A5 variant found in the mother and two siblings, which changes the amino acid from glycine to aspartic acid located in the triple-helical region of the collagen IV trimer. (F) Representative whole-mount immunofluorescence images of wild-type and Alport kidney organoids show glomerular structures (g) containing WT1+ cells and an intricate cluster of CDH1+ epithelial tubules. (G) Immunofluorescence for LAMB2 shows increased protein deposition in extraglomerular sites (*). NPHS2 was used as a podocyte marker to identify glomerular structures (g). (H) Immunoblotting for LAMB2 using total lysates from wild-type (n = 3) and Alport organoids (n = 3 per group): bar chart shows relative LAMB2 fold change (to wild-type). LAMB2 band optical density was normalized to Ponceau stain (total proteins) and compared by one-way ANOVA and Tukey’s multiple comparison tests (*p<0.05; ns, not significant). Pooled data are presented as median, and error bars indicate the 95% confidence interval for the median. See Figure 1—source data 1 (available at https://doi.org/10.6084/m9.figshare.c.5429628). See also Figure 1—figure supplements 1–3.
-
Figure 1—source data 1
Figure 1 - source data WB LAMB2.
- https://cdn.elifesciences.org/articles/73486/elife-73486-fig1-data1-v2.zip

Morphological characteristics of wild-type kidney organoids and human fetal kidney.
(A) Bright field images (left) and hematoxylin and eosin (H&E) staining (right) of human kidney organoids at days 11, 14, 18, and 25 of differentiation. (B) H&E staining of human fetal kidney at 8 weeks post conception (wpc), 12 wpc, 17 wpc, and 21 wpc highlights normal human kidney development. (C) Immunofluorescence for integrin beta-1 (ITGB1) in day 25 kidney organoid (wild-type). Antipan-laminin or pan-collagen IV antibodies were used to label basement membranes. In the glomerulus (g), note the distribution of ITGB1 adjacent to the basement membrane.
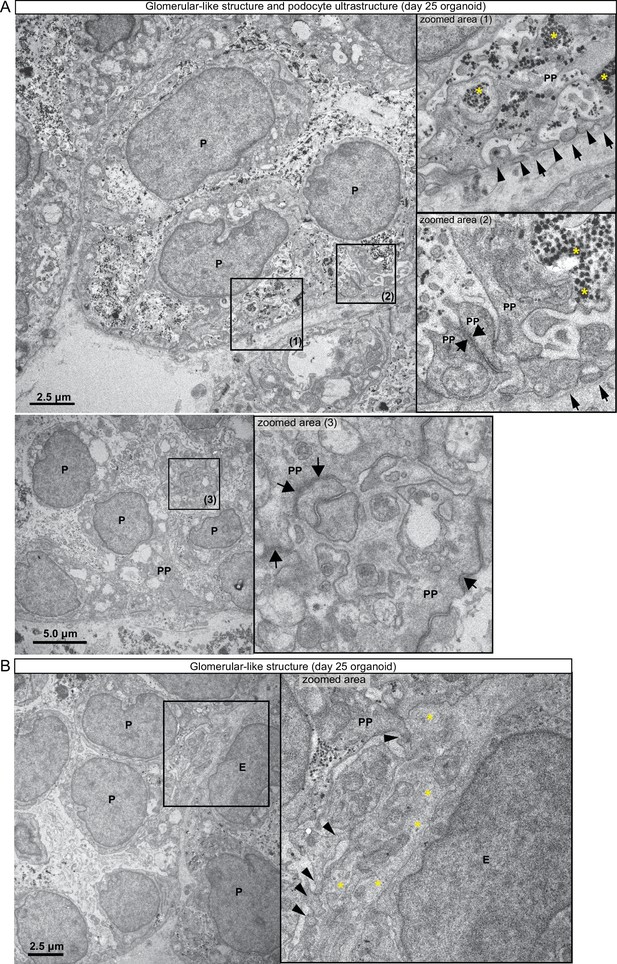
Ultrastructure of glomerular-like structures in day 25 human kidney organoids.
(A) Transmission electron microscopy of day 25 kidney organoids shows advanced differentiation of glomerular structures. In the zoomed areas (1 and 2) note podocytes (P) displaying long branching primary processes (PP) and a layer of intercalated foot processes (thin arrowheads) lining a distinct basement membrane (thin arrows). Asterisks indicate accumulation of glycogen granules, and large arrows indicate tight cell junctions between maturing podocyte processes (PP). In the lower panels, note in the zoomed area (3) deposition of basement membrane-like matrix between podocyte processes (PP) within a glomerular structure. (B) Note the presence of likely endothelial cells (E) and podocytes (P) in a glomerular structure. In the zoomed area, note a layer of podocyte foot processes (thin arrowheads) branching from a primary process (PP) and an endothelial cell (E); observe also deposition of matrix- and basement membrane-like electron-dense material (asterisks) between cells.
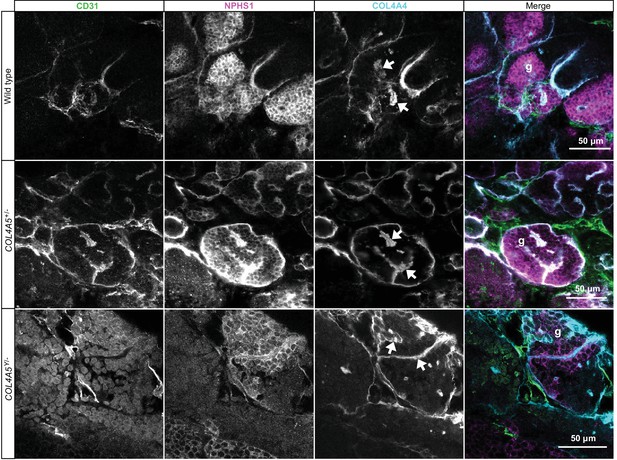
Differentiation of wild-type and Alport kidney organoids.
Whole-mount immunofluorescence for wild-type and Alport kidney organoids shows comparable deposition of COL4A4 in basement membrane-like structures (arrows) within NPHS1+/CD31+ glomerular structures (g).
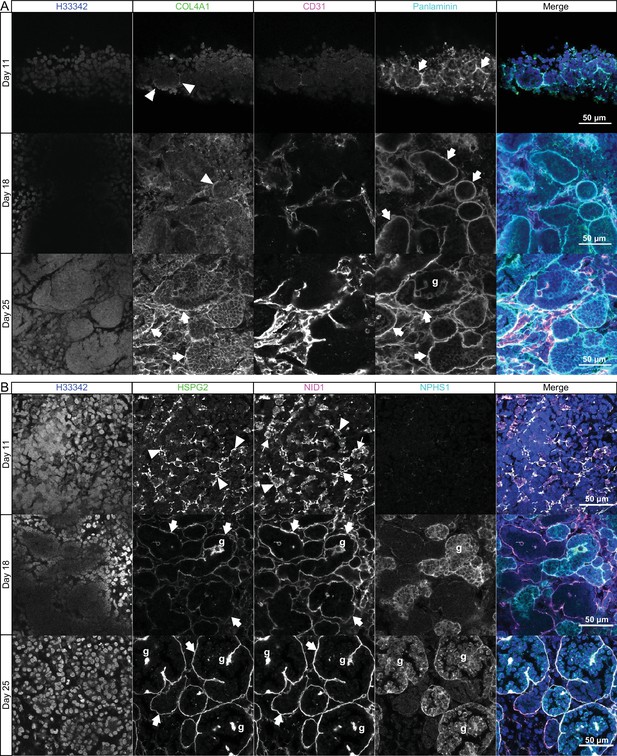
Sequential assembly of basement membrane components.
(A) Confocal immunofluorescence microscopy of wild-type kidney organoids showing the temporal emergence and co-distribution of COL4A1 and pan-laminin, and (B) perlecan and nidogen at days 11, 18, and 25 of differentiation. NPHS1 and CD31 were used as markers for podocyte and endothelial cells, respectively, in glomerular-like structures (g). Arrowheads indicate interrupted BM segments, large arrows indicate diffuse BM networks, and thin arrows indicate intracellular droplets of proteins.
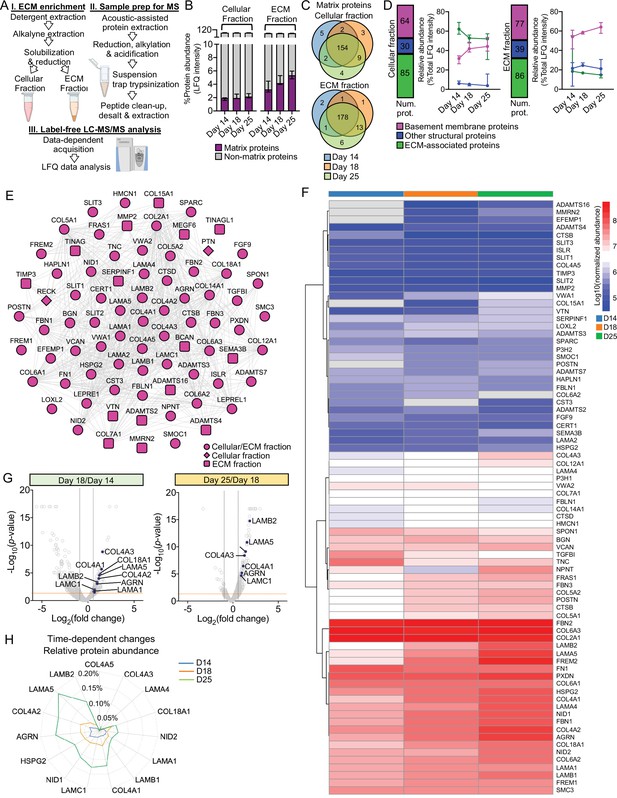
Time course proteomics reveals complex dynamics of basement membrane assembly.
(A) Schematic of sample enrichment for matrix (ECM) proteins for tandem mass spectrometry (MS) analysis (created with BioRender.com). (B) Bar charts show the relative abundance of matrix and non-matrix proteins identified by MS analysis in the cellular and ECM fractions of kidney organoids at days 14, 18, and 25 (n = 3 pools per time point). Pooled data are presented as median, and error bars indicate the 95% confidence interval for the median. (C) Venn diagrams showi the identification overlap for matrix proteins detected in kidney organoids at days 14, 18, and 25. (D) Matrix proteins are classified as basement membrane, other structural and ECM-associated proteins. Bar charts show the number of matrix proteins per matrix category in both cellular and ECM fractions, and line charts show the changes in their relative abundance (percentage of total matrix abundance) over the time course differentiation. Pooled data are presented as median, and error bars indicate the 95% confidence interval for the median. (E) Protein interaction network showing all basement membrane proteins identified over the kidney organoid time course MS study (nodes represent proteins and connecting lines indicate reported protein-protein interactions). (F) Heat map showing the log10-transformed abundance levels of basement membrane proteins identified in the ECM fraction along kidney organoid differentiation time course (proteins detected only at one time point are not shown). (G) Volcano plots show the log2-fold change (x-axis) versus -log10-p-value (y-axis) for proteins differentially expressed in the ECM fraction of kidney organoids from day 14 to day 18, and from day 18 to day 25 (n = 3 per time point). Key basement membrane proteins significantly upregulated (FC > 1.5, p-value<0.05, two-way ANOVA test, n = 3) are indicated. (H) Time-dependent changes in the relative abundance (percentage of total protein intensity) of key basement membrane proteins in the ECM fraction of kidney organoids during differentiation. Pooled data are presented as median. See Figure 3—figure supplement 1.
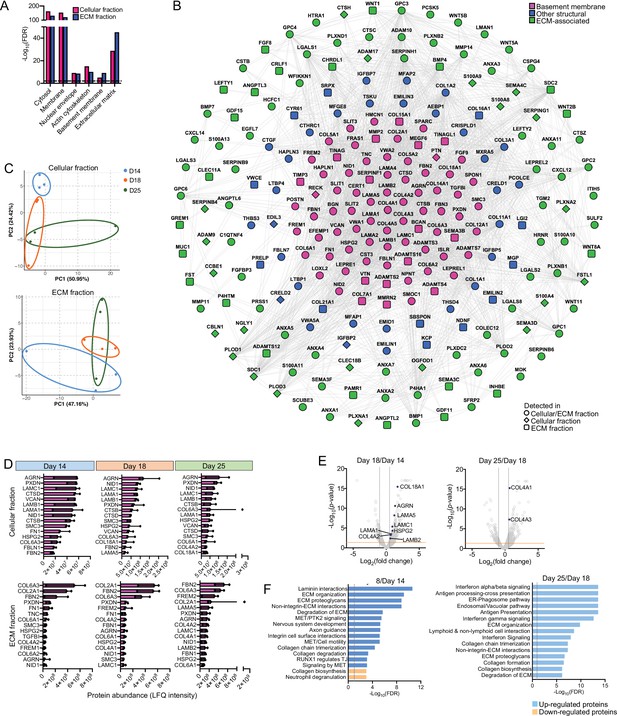
Time course proteomic analysis of kidney organoid differentiation.
(A) Gene Ontology (GO) term enrichment analysis of cellular component annotations associated with proteins detected in the cellular and extracellular matrix (ECM) fractions of kidney organoids by mass spectrometry (MS). GO terms were considered enriched when false discovery rate (FDR) < 0.10. (B) Protein interaction network depicts matrisome proteins identified by MS over kidney organoid differentiation time course (nodes represent proteins and connecting lines indicate reported protein-protein interactions). (C) Principal component analysis (PCA) for matrix proteins identified by MS in the cellular (left plot) and ECM (right plot) fractions of kidney organoids. (D) Top 15 most abundant basement membrane proteins found in kidney organoids. Proteins were ranked according to their normalized abundance levels (LFQ-intensities). Pooled data are shown as median, and error bars indicate the 95% confidence interval for the median. (E) Volcano plots show the log2-fold change (x-axis) versus -log10-p-value (y-axis) for proteins differentially expressed in the cellular fraction of kidney organoids from day 14 to day 18, and from day 18 to day 25. Key basement membrane proteins significantly upregulated (FC > 1.5, p-value<0.05, two-way ANOVA test, n = 3) are indicated. (F) Pathway enrichment analysis of proteins differentially expressed during kidney organoid differentiation: bar charts depict log-transformed FDR for the top-most enriched pathways (FDR < 0.10). Pathway terms shown were simplified.
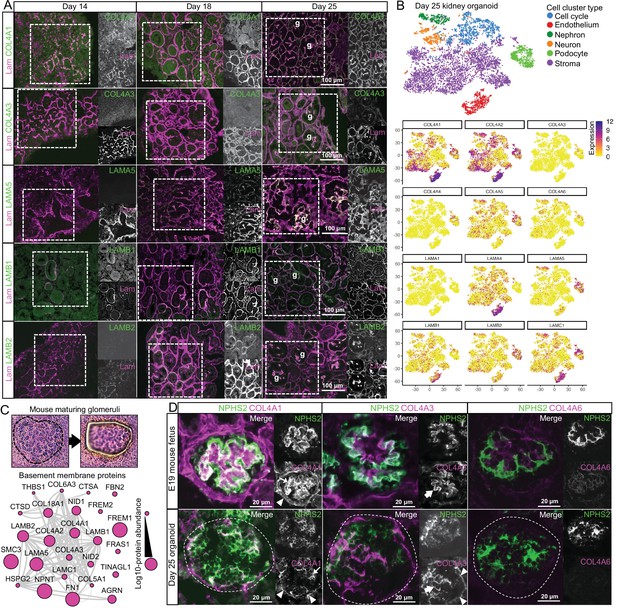
Key collagen IV and laminin isoform transitions occur during kidney organoid differentiation.
(A) Immunofluorescence for key collagen IV and laminin isoforms shows their emergence and distribution in kidney organoid basement membrane. Pan-laminin antibody was used to co-label organoid basement membranes; glomerular structures are indicated (g). (B) Reanalysis of a kidney organoid scRNA-seq dataset GSE114802 (Combes et al., 2019b) confirms cellular specificity for collagen IV and laminin isoform gene expression. tSNE plots represent the cell-type clusters identified, and color intensity indicates cell-specific level of expression for selected basement membrane genes. (C) Proteomic analysis of laser-captured maturing glomeruli from E19 mouse kidneys (n = 4). Histological images show a laser-microdissected glomerulus, and the protein interaction network shows the 25 basement membrane proteins identified (nodes represent proteins and connecting lines indicate reported protein-protein interactions). (D) Immunofluorescence for specific collagen IV isoforms in maturing glomeruli in E19 mouse kidney and in glomerular structures (indicated by dashed lines) in day 25 organoids. NPHS2 was used to label podocytes. Arrowheads indicate the Bowman’s capsule in the mouse or the glomerular surface in the organoid; large arrows indicate the GBM in the mouse or GBM-like matrix in the organoid; thin arrows indicate mesangial matrix in the mouse or internal glomerular matrix deposition in the organoid. See Figure 4—figure supplement 1.

Single-cell RNA-sequencing data analysis of human kidney organoids.
(A) Dot plot depicting the cell-specific level of expression of 160 basement membrane gene collection in 25-day kidney organoids. (B) tSNE plot indicates gene expression of LIMX1b and EPB41L5 in day 25 kidney organoids. Arrows indicate podocyte clusters. Data shown in (A) and (B) were obtained from a reanalysis of the publicly available scRNA-seq dataset GSE114802 (Combes et al., 2019b). (C) Bar charts show the time-dependent changes in protein abundance of EPB41L5 in kidney organoids (n = 3 per time point). Pooled data are shown as median, and error bars indicate the 95% confidence interval for the median. Two-way ANOVA test: *p-value< 0.05.
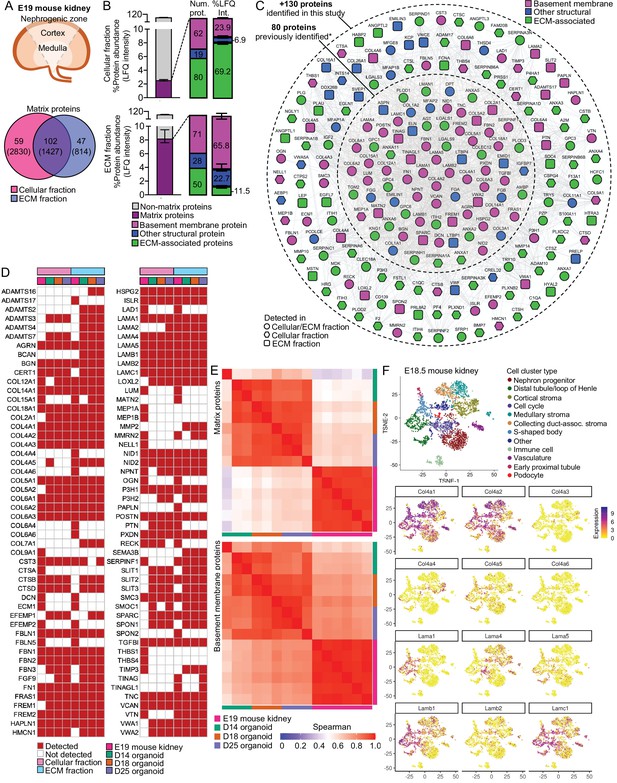
Basement membranes in mouse fetal kidneys are comparable to kidney organoids.
(A) Schematic representation of the E19 mouse kidney sampled for mass spectrometry (MS)-based proteomics, and a Venn diagram showing the overlap for matrix proteins identified in the cellular and extracellular matrix (ECM) fractions. (B) Bar charts show enrichment levels for matrix proteins in both cellular and ECM fractions (n = 6), as indicated by the number and relative abundance of proteins in each matrix category. Pooled data are presented as median, and error bars indicate the 95% confidence interval for the median. (C) Expanded mouse fetal kidney matrisome represented as a protein interaction network (nodes represent proteins identified in this and in a previous study [Lipp et al., 2021], and connecting lines indicate reported protein-protein interactions). (D) Comparison of basement membrane proteins identified in the E19 mouse kidney* (MFK) and human kidney organoids (HKOs) during differentiation (asterisks indicate that corresponding human ortholog for mouse proteins are shown). (E) Spearman’s rank correlation analysis of matrix and basement membrane protein abundance (in the ECM fraction) comparisons between E19 MFK and human kidney organoids (HKO) at days 14 (D14), 18 (D18) and 25 (25D). (F) Reanalysis of an E18.5 mouse kidney scRNA-seq dataset GSE108291 (Combes et al., 2019a) confirms cellular specificity for collagen IV and laminin isoform gene expression. tSNE plots represent the cell-type clusters identified, and color intensity indicates cell-specific level of expression for selected basement membrane genes. See Figure 5—figure supplements 1 and 2.
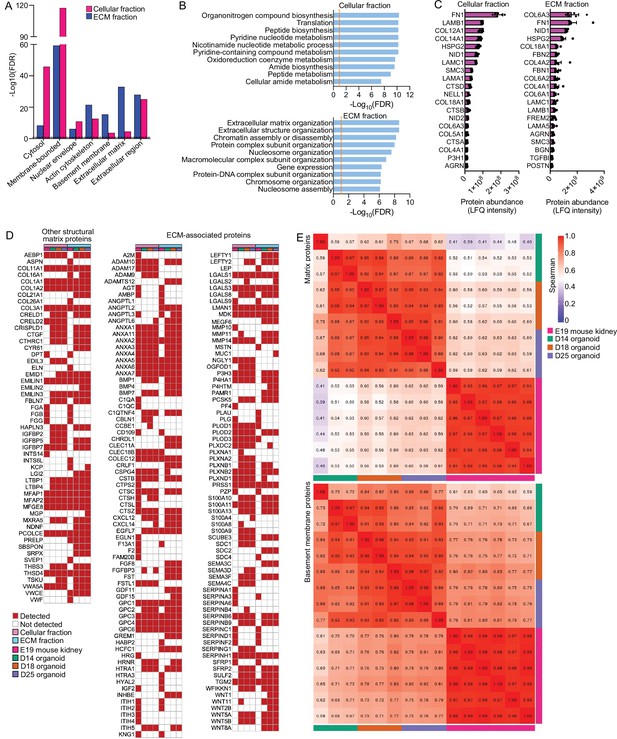
Proteomic analysis of embryonic day 19 (E19) mouse kidney and correlational comparison with kidney organoid proteomics.
(A) Gene Ontology (GO) term enrichment analysis of cellular component annotations associated with proteins detected by mass spectrometry (MS) in the cellular and extracellular matrix (ECM) fractions. (B) GO biological process annotations enriched for the top 100 most abundant proteins detected by MS in the cellular and ECM fractions. (C) Top 20 most abundant basement membrane proteins found in the E19 mouse kidney by MS. Proteins were ranked according to their normalized abundance levels (LFQ-intensities). Pooled data are presented as median, and error bars indicate the 95% confidence interval for the median. (D) Comparison of other structural matrix and ECM-associated proteins identified in the E19 mouse kidney and kidney organoids over differentiation time course. (E) Spearman’s rank correlation plots depicting the r coefficient values for matrix and BM protein abundance (ECM fraction) comparisons between the E19 mouse kidney and kidney organoids.
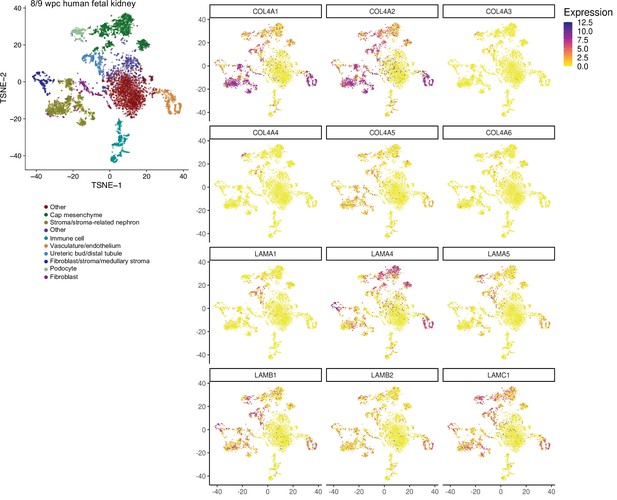
Single-cell RNA-sequencing analysis of human fetal kidney.
Reanalysis of 8/9 weeks post conception (wpc) human kidney scRNA-seq datasets (EGAS00001002325, EGAS00001002553; Young et al., 2018) confirms cellular specificity for collagen IV and laminin isoform gene expression. tSNE plots represent the cell-type clusters identified, and color intensity indicates cell-specific level of expression for the selected basement membrane genes.
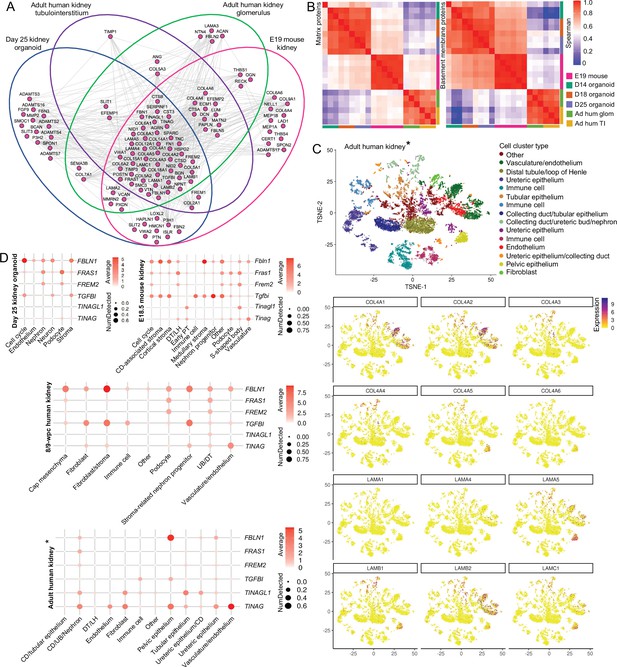
Basement membranes are dynamic through embryonic development to adulthood.
(A) Integrative interactome shows a common core of 44 basement membrane proteins across day 25 organoid, E19 mouse kidney, and adult human glomerular and tubulointerstitial compartments. Venn diagrams indicate in which dataset each BM protein was detected. Nodes represent proteins, and lines indicate reported protein-protein interactions. (B) Spearman’s rank correlation analysis of matrix and basement membrane protein abundance (in ECM fraction) comparisons between E19 mouse fetal kidney (MFK), human kidney organoids (HKO), and adult human glomerulus and kidney tubulointerstitium. (C) Reanalysis of an adult human kidney scRNA-seq dataset EGAS00001002553 (Young et al., 2018) confirms cellular specificity for collagen IV and laminin isoform gene expression. tSNE plots represent the cell-type clusters identified, and color intensity indicates cell-specific level of expression for the selected basement membrane genes (*proximal tubule cells were not included). (D) Cell expression of minor basement membrane components through kidney development to adulthood. Dot plots show the level of expression of target genes in all publicly available datasets reanalyzed in this study (*proximal tubule cells were not included). See Figure 6—figure supplement 1.
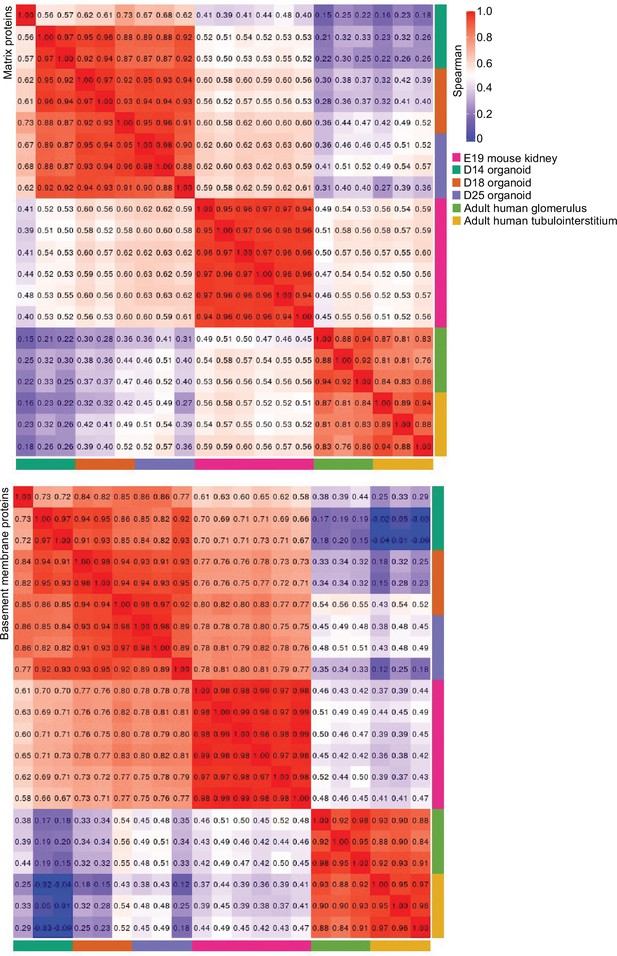
Integrated correlational analysis of organoid and in vivo kidney datasets.
Spearman’s rank correlation plots depicting r coefficient values or matrix and basement membrane protein abundance (in ECM fraction) comparisons between E19 mouse kidney, kidney organoids and adult human kidney proteomic datasets.
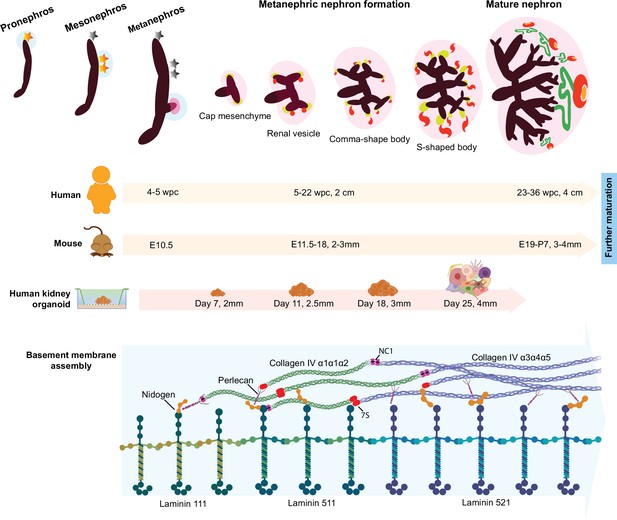
Overview of the developmental milestones in human and mouse kidney morphogenesis and a comparison to human kidney organoids.
Differentiation is dated in in weeks post conception (wpc) for human, embryonic days (E) for mouse, and in days of differentiation for kidney organoids; and measures (cm or mm) indicate specimen size in each model. Three sets of embryonic kidneys develop in mammals in a temporal sequence: from the pronephros to the mesonephros (both temporary), and then to the metanephros (permanent). Metanephric nephron formation (nephrogenesis) commences in humans at 5 wpc, in mice at E10.5, and in kidney organoid from day 11, when laminin starts to deposit as basement membrane networks. Following the same developmental stage in human and mouse kidneys, kidney organoids start to form discernible renal vesicles at day 14, and distinct comma- and S-shaped bodies at day 18; day 25 organoids, which most closely resemble late capillary loop stage nephrons in vivo, comprise more mature structures including glomeruli with capillary lumens, proximal tubules, and distal tubules. Meanwhile, a conserved sequence of basement membrane assembly is detected in kidney organoids, and laminin and collagen IV developmental isoform transitions are identified between day 14 and day 25.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mus musculus) | Swiss | University of São Paulo (Brazil) | - | 2 months old, male and female mice |
| Cell line (Homo sapiens) | iPSC | HipSci | CGAP-38728; CGAP-4852B; CGAP-581E8 | Derived from patients with Alport syndrome |
| Cell line (Homo sapiens) | iPSC | Wood et al., 2020 | - | Derived from peripheral blood mononuclear cells |
| Biological sample (Homo sapiens) | Embryonic and fetal kidneys | Joint MCR/Wellcome Trust HDBR | Kidney_ID: 13878; 11295; 13891; 13842; 1358 | FFPE samples |
| Antibody | Anti-CD31, clone 89C2 (mouse monoclonal) | Cell Signaling | Cat# 3582 | IF (1:100)WM (1:100) |
| Antibody | Anti-E-cadherin, clone M168 (mouse monoclonal) | Abcam | RRID:AB_1310159 | IF (1:300)WM (1:300) |
| Antibody | Anti-WT1, clone C-19 (rabbit polyclonal) | Santa Cruz Biotechnology | RRID:AB_632611 | IF (1:100)WM (1:100) |
| Antibody | Anti-human nephrin (sheep polyclonal) | R&D Systems | RRID:AB_2154851 | IF (1:200)WM (1:400) |
| Antibody | Anti-human collagen IV α1 chain NC1 domain, clone H11 (rat monoclonal) | Chondrex | Cat# 7070 | IF (1:100)WM (1:400) |
| Antibody | Anti-human collagen IV α3 chain NC1 domain, clone H31 (rat monoclonal) | Chondrex | Cat# 7076 | IF (1:100) |
| Antibody | Anti-human collagen IV α4 chain NC1 domain, clone H43 (rat monoclonal) | Chondrex | Cat# 7073 | IF (1:100) |
| Antibody | Anti-human collagen IV α6 chain NC1 domain, clone H63 (rat monoclonal) | Chondrex | Cat# 7074 | IF (1:50) |
| Antibody | Anti-laminin (rabbit polyclonal) | Abcam | RRID:AB_298179 | IF (1:250)WM (1:250) |
| Antibody | Anti-nidogen, clone 302,117 (mouse monoclonal) | Invitrogen | RRID:AB_2609420 | IF (8.3 μg/ml)WM (8.3 μg/ml) |
| Antibody | Anti-perlecan, clone A7L6 (rat monoclonal) | Millipore | RRID:AB_10615958 | IF (1:250)WM (1:250) |
| Antibody | Anti-laminin α5 chain, clone 4C7 (mouse monoclonal) | Abcam | RRID:AB_443652 | IF (1:100) |
| Antibody | Anti-laminin β1 chain, clone 4E10 (mouse monoclonal) | Millipore | RRID:AB_571039 | IF (1:100) |
| Antibody | Anti-laminin S/laminin β2 chain, clone CL2979 (mouse monoclonal) | Novus Biologicals | Cat# NBP-42387 | IF (1:50)WB (1:1000) |
| Antibody | Anti-podocin (rabbit polyclonal) | Millipore | RRID:AB_261982 | IF (1:200) |
| Antibody | Anti-NPHS2 (rabbit polyclonal) | Abcam | RRID:AB_882097 | IF (1:200) |
| Antibody | Anti-integrin β1 chain, clone 9EG7 (rat monoclonal) | Lenter et al., 1993 | - | IF (1:100) |
| Antibody | Anti-rat IgG conjugated with Alexa Fluor 488 (donkey) | Invitrogen Antibodies | RRID:AB_141709 | IF (1:400) |
| Antibody | Anti-rat IgG conjugated with Alexa Fluor 594 (donkey) | Invitrogen Antibodies | RRID:AB_2535795 | IF (1:400) |
| Antibody | Anti-mouse IgG conjugated with Alexa Fluor 488 (donkey) | Invitrogen Antibodies | RRID:AB_141607 | IF (1:400) |
| Antibody | Anti-mouse IgG conjugated with Alexa Fluor 594 (donkey) | Invitrogen Antibodies | RRID:AB_141633 | IF (1:400) |
| Antibody | Anti-rabbit IgG conjugated with Alexa Fluor 488 (donkey) | Invitrogen Antibodies | RRID:AB_2535792 | IF (1:400) |
| Antibody | Anti-rabbit IgG conjugated with Alexa Fluor 647 (donkey) | Invitrogen Antibodies | RRID:AB_2536183 | IF (1:400) |
| Antibody | Anti-rabbit IgG conjugated with Alexa Fluor 594 (goat) | Invitrogen Antibodies | RRID:AB_141359 | IF (1:400) |
| Antibody | Anti-sheep IgG conjugated with Alexa Fluor 594 | Invitrogen Antibodies | RRID:AB_2534083 | IF (1:400) |
| Antibody | Anti-sheep IgG conjugated with Alexa Fluor 680 | Invitrogen Antibodies | RRID:AB_1500713 | IF (1:400) |
| Antibody | Anti-mouse IgG DyLight 800 4X PEG conjugated (goat) | Cell Signaling Technology | RRID:AB_10697505 | WB (1:1000) |
| Antibody | Anti-rabbit IgG labeled with 10 nm gold (goat) | Abcam | RRID:AB_954434 | Immunogold (1:400) |
| Chemical compound, drug | CHIR99021 | Tocris Bioscience | 4423/10 | - |
| Chemical compound, drug | FGF-9 | PeproTech | 100-23 | - |
| Chemical compound, drug | Heparin | Sigma-Aldrich | H3393 | - |
| Chemical compound, drug | TeSR-E8 medium | STEMCELL | 05991; 05992 | - |
| Chemical compound, drug | STEMdiff APEL 2 medium | STEMCELL | 05270 | - |
| Software, algorithm | Proteome Discoverer v.2.3.0.523 | Thermo Fisher Scientific | RRID:SCR_014477 | - |
Additional files
-
Supplementary file 1
Human fetal kidney and human induced pluripotent stem cell (hiPSC) general information.
- https://cdn.elifesciences.org/articles/73486/elife-73486-supp1-v2.xlsx
-
Supplementary file 2
Human kidney organoid proteome and matrix proteins.
- https://cdn.elifesciences.org/articles/73486/elife-73486-supp2-v2.xlsx
-
Supplementary file 3
Single-cell RNA-sequencing kidney datasets: cell clustering and expression data.
- https://cdn.elifesciences.org/articles/73486/elife-73486-supp3-v2.xlsx
-
Supplementary file 4
Embryonic day 19 mouse maturing glomerulus proteome and matrix proteins.
- https://cdn.elifesciences.org/articles/73486/elife-73486-supp4-v2.xlsx
-
Supplementary file 5
Embryonic day 19 mouse kidney proteome and matrix proteins.
- https://cdn.elifesciences.org/articles/73486/elife-73486-supp5-v2.xlsx
-
Supplementary file 6
Human adult kidney glomerular and tubulointerstitial proteome and matrix proteins.
- https://cdn.elifesciences.org/articles/73486/elife-73486-supp6-v2.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/73486/elife-73486-transrepform1-v2.docx





