A low-tech, cost-effective and efficient method for safeguarding genetic diversity by direct cryopreservation of poultry embryonic reproductive cells
Figures
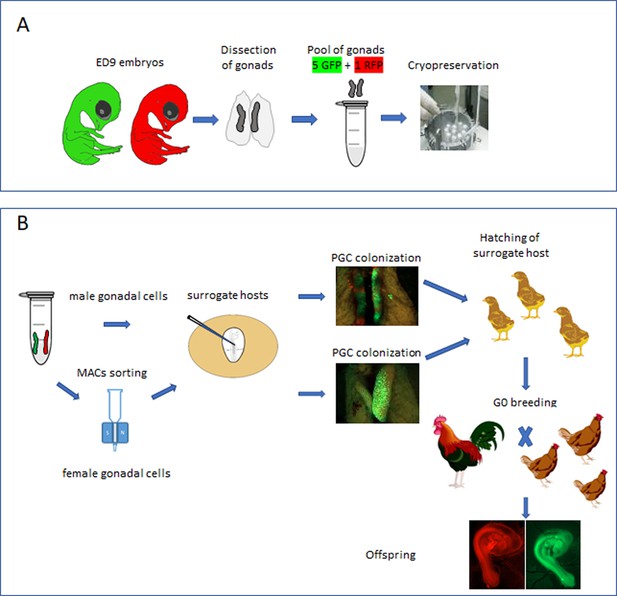
Isolation and cryopreservation of embryonic gonads followed by transmission through sterile surrogate hosts.
(A) Embryonic day (ED) 9 gonads are isolated from embryos, pooled by sex, and cryopreserved in liquid N2. (B) The frozen gonads are thawed, dissociated, and injected into sterile surrogate host embryos. The surrogate host embryos are incubated and hatched and bred to hatch donor gonadal offspring.
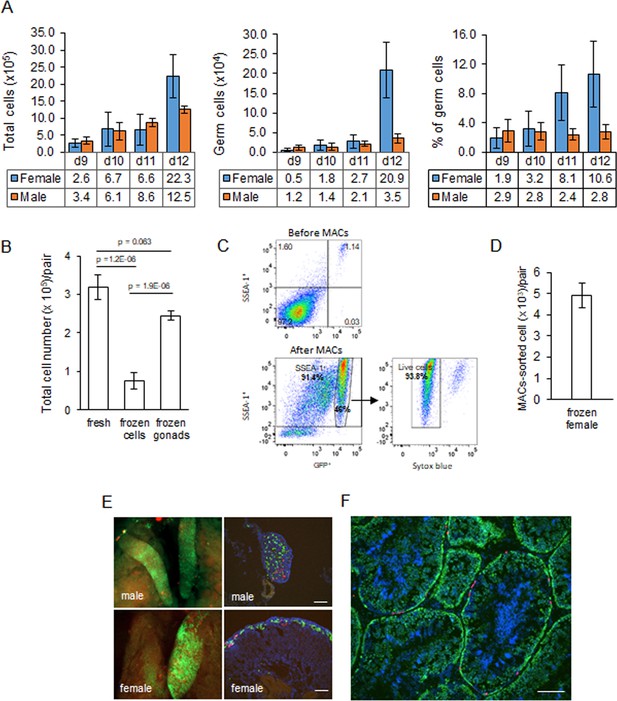
Characterisation and cryopreservation of gonadal germ cells.
(A) Population of gonadal germ cells between embryonic day (ED) 9–12. The number of germ cells was determined by their expression of GFP protein in iCaspase9 transgenic embryos (n = 3–7 gonad pairs for each sex at each day). (B) Yield of viable dissociated cells directly from freshly isolated embryonic day 9 gonads (control), cryopreserved dissociated gonadal cells subsequently thawed (frozen cells), and cryopreserved whole gonads subsequently thawed then dissociated (frozen gonads). Cell viability was determined using a trypan blue exclusion assay. Data from 13 to 20 independent experiments using mixed male and female gonads. (C) Flow cytometric analysis of Magnetic Activated Cell (MAC)-sorted female gonadal cells. Frozen female ED 9 (HH35) gonads from iCaspase9 GFP+ embryos were MAC-sorted using an anti-SSEA-1 antibody. The purified cells were then immunostained by secondary antibody against SSEA-1 to detecting the percentage of SSEA-1 cells expressing GFP. The GFP+ population was analysed for viability using Syto blue; n = 5 independent experiments. (D) Yield of MAC-sorted cells from cryopreserved ED 9 (HH35) female gonads. The average number of GFP+ cells purified by MACS from a single iCaspase9 embryo using an anti-SSEA-1 antibody. Data from five independent experiments using 12–26 gonad pairs per experiment. (E) Colonisation of sterile iCaspase9 embryos by cryopreserved male and female gonadal cells. The host ED 14 gonads are shown on the left and transverse sections from those gonads are on the right. Day 2.5 iCaspase9 host embryos were injected with gonadal donor cells at a 5GFP+:1RFP+ ratio mixed with B/B compound; 45,000 male cells/embryo. Female cells were MAC-sorted before injecting 1400 female cells/embryo. Representative embryo shown (n > 5, for each sex). Scale bar = 100 μm. (F) Seminiferous tubule of an adult testis (>6 months) from a sterile surrogate host injected with donor male cells prepared as in (C). Representative testis section from n = 7 males. Scale bar = 100 μm.
-
Figure 2—source data 1
Source data for Figure 2A.
- https://cdn.elifesciences.org/articles/74036/elife-74036-fig2-data1-v1.xlsx
-
Figure 2—source data 2
Source data for Figure 2B.
- https://cdn.elifesciences.org/articles/74036/elife-74036-fig2-data2-v1.xlsx
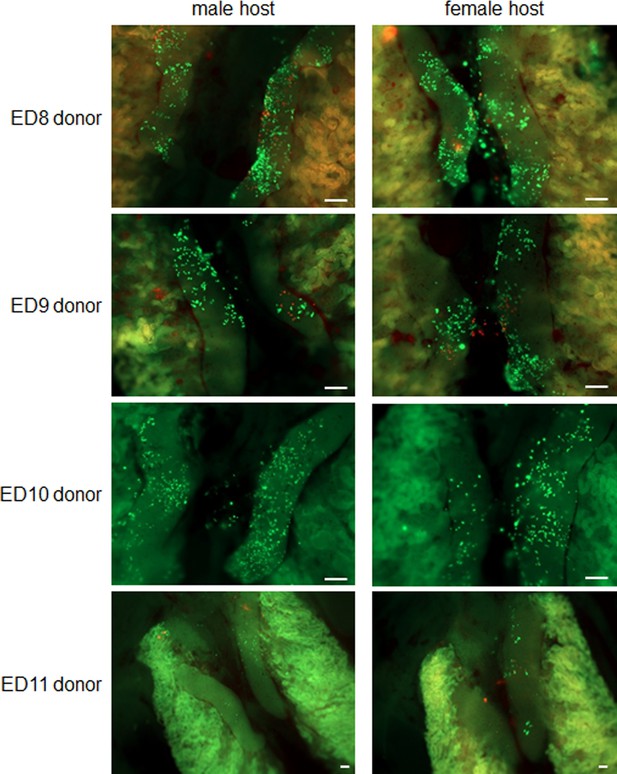
Recolonisation of surrogate host embryos by frozen gonadal germ cells.
Gonadal cell suspensions from cryopreserved gonads from embryonic day (ED) 8–11 GFP+ and RFP+ female embryos were injected into wildtype ED 2.5 (stage 16 HH) host embryos. Host embryos were examined at ED 8–9 of incubation for gonadal colonisation. 15,000 cells were injected per embryo (10,000 cells/embryos for ED 9 donor cells). The representative images are from at least three independent injection experiments. Scale bar = 200 μm.
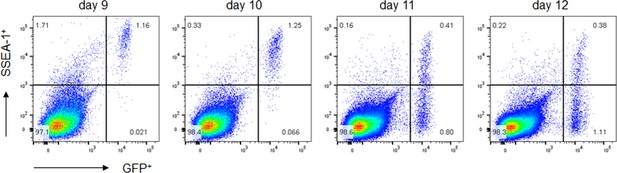
SSEA-1 expression of post-migratory gonadal germ cells.
iCaspase9 female embryos were used to identify putative germ cells using GFP expression. Gonadal cells were dissociated and examined by flow cytometry for GFP and SSEA-1 expression. SSEA-1 coexpression by GFP+ cells: 98.2% at day 9, 94.7 at day 10, 51.3% at day 11, and 34.2% at day 12. n = 4–8 embryos independently assayed for each time point.
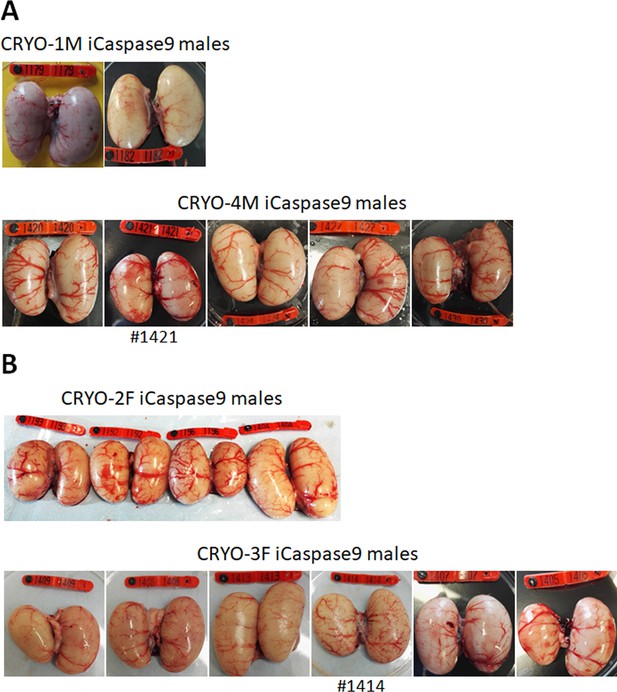
Adult gonads of iCaspase9 surrogate host cockerels.
(A) Male surrogate hosts injected with male gonadal germ cells examined at >25 weeks of age. (B) Male surrogate hosts injected with female gonadal germ cells examined at >25 weeks of age. Red wing tags = 5 cm.
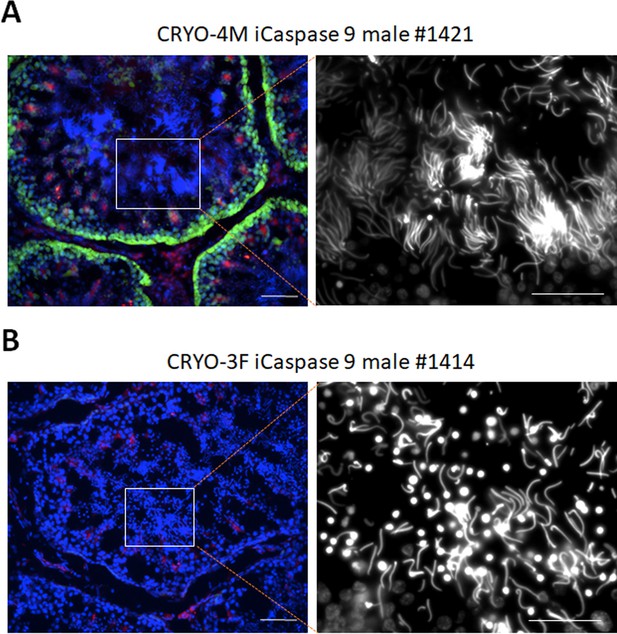
Cryosections of adult testes of iCaspase9 surrogate host cockerels.
(A) A male surrogate host injected with male gonadal germ cells examined at >25 weeks of age. Blue, Hoechst stain; right panel, Hoechst stain shown in white. * Lumen of tubule. (B) Male surrogate hosts injected with female gonadal germ cells examined at >25 weeks of age. Blue, Hoechst stain; right panel, Hoechst stain shown in white. Scale bars = 50 μm.
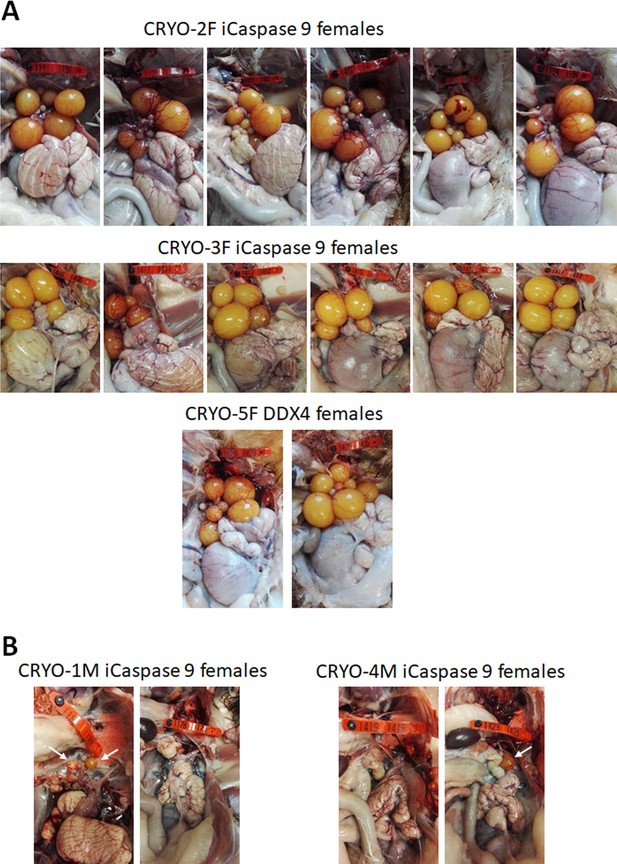
Adult gonads of iCaspase9 surrogate host hens.
(A) Female surrogate hosts injected with female gonadal germ cells examined at >25 weeks of age. (B) Female surrogate hosts injected with male gonadal germ cells examined at >25 weeks of age. White arrows, yellow follicles. Red wing tags = 5 cm.
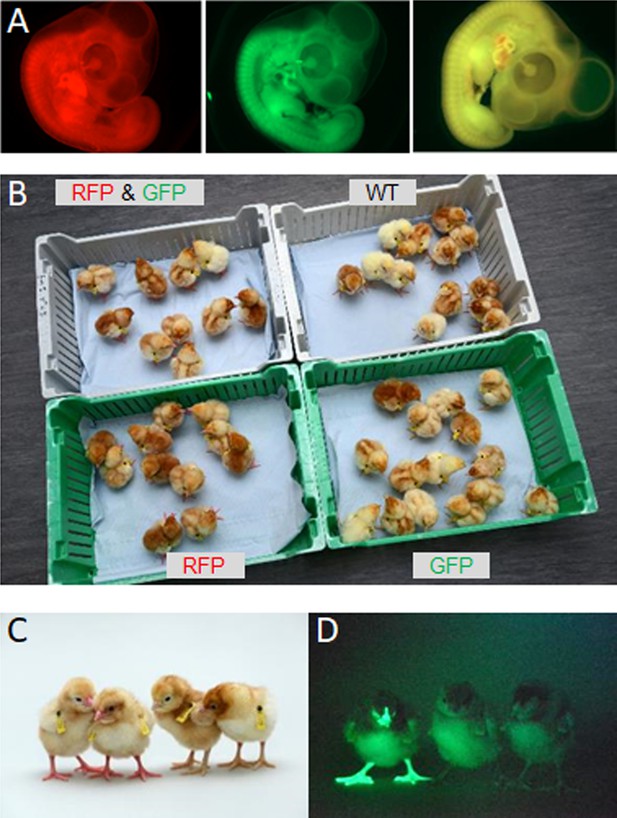
Hatchings from ♂ surrogate hosts injected with ♂ gonadal cells mated to ♀ surrogate hosts injected with ♀ gonadal germ cells.
(A) Embryonic day (ED) 5 embryos from Sire Dam Surrogate (SDS) mating displaying representative red, green, and ‘yellow’ fluorescence. (B) Hatched chicks from SDS mating grouped according to fluorescence. (C) RFP fluorescent chicks were apparent (chicks on left) under visible light. (D) GFP fluorescent chick (left) visualised under GFP illumination.
Tables
Injection and hatching of surrogate hosts.
| Injection set | Duration of storage (days) | Sex of donor cells | Enriched by MACS | No. of gonad pairs | Total cell yield | No. of donor cells injected per host embryo | No. of potential injections | Surrogate host genotype | ED 14 survival rate (% injected embryos) | No. and sex of hatchlings(% hatch rate) |
|---|---|---|---|---|---|---|---|---|---|---|
| CRYO-1M | 108 | M | No | 10 GFP + 2 RFP | 2,500,000 | 50,000 | 50 | iCaspase9 | 17/26 (65%) | 4F + 3M (41%) |
| CRYO-2F | 80 | F | Yes | 10 GFP + 4 RFP | 42,000 | 1000 | 42 | iCaspase9 | 12/18 (67%) | 6F + 4M (83%) |
| CRYO-3F | 103 | F | Yes | 17 GFP + 6 RFP | 100,000 | 1500 | 66 | iCaspase9 | 17/27 (63%) | 6F + 8M (82%) |
| CRYO-4M | 121 | M | No | 15 GFP + 3 RFP | 4,400,000 | 60,000 | 73 | iCaspase9 | 15/22 (68%) | 4F + 7M (73%) |
| CRYO-5F | 128 | F | Yes | 15 GFP + 3 RFP | 120,000 | 1500 | 80 | DDX4 | 19/23 (82%) | 4F* + 7M (58%) |
-
*Two females were wildtype (ZW) and two were DDX4 ZW- knockouts (ZW-).
-
MACS: magnetic-activated cell sorting; ED: embryonic day; M: male; F: female.
Germline transmission from ♂ surrogate hosts injected with ♂ gonadal primordial germ cells.
| Host mating groups | No. of eggs laid per week * | Eggs set | Fertility† (% eggs set) (%) | No. of RFP embryos (% fertile) | No. of GFP embryos (% fertile) | Transmission of surrogate iCaspase9 transgene(% fertile) |
|---|---|---|---|---|---|---|
| CRYO-1M_1181♂× 6 WT♀ | 6.7 | 147 | 96 | 4 (3%) | 101 (72%) | 0 |
| CRYO-1M_1182♂× 6 WT♀ | 6.6 | 148 | 99 | 9 (6%) | 89 (61%) | 9 (6%) |
| CRYO-1M_1179♂ × 6 WT♀ | 6.6 | 187 | 98 | 51 (28%) | 55 (30%) | 0 |
| Total | 482 | 98 | 64 (14%) | 245 (52%) | 9 (2%) | |
| CRYO-4M_1420♂ × 5 WT♀ | 4.5 | 89 | 91 | 4 (5%) | 64 (79%) | 0 |
| CRYO-4M_1424♂ × 5 WT♀ | 4.1 | 60 | 93 | 3 (5%) | 41 (73%) | 0 |
| CRYO-4M_1427♂ × 6 WT♀ | 6.9 | 74 | 85 | 13 (21%) | 29 (46%) | 0 |
| CRYO-4M_1430 ♂ × 6 WT♀ | 6.7 | 143 | 83 | 14 (12%) | 80 (68%) | 0 |
| Total | 366 | 87 | 34 (11%) | 214 (67%) | 0 |
-
*
Lay rate; eggs were counted over a 60-day period when hens were between 7 and 12 months of age and divided by the number of fertile hens present in pen. The maximum possible lay rate is 7.0 eggs per hen per week.
-
†
Fertility was assessed between embryonic day 4–7.
Germline transmission from ♀ surrogate hosts injected with ♀ gonadal primordial germ cells.
| Host mating groups | No. of eggs laid per week * | Eggs set | Fertility† (% eggs set) (%) | No. of RFP embryos (% fertile) | No. of GFP embryos (% fertile) | Transmission of surrogate iCaspase9 transgene (%) |
|---|---|---|---|---|---|---|
| WT ♂ ×6 CRYO-2F♀ | 6.3 | 371 | 98 | 51 (14%) | 56 (36%) | 0 |
| WT ♂ ×6 CRYO-3F♀ | 5.0 | 340 | 80 | 66 (24%) | 150 (55%) | 0 |
| WT ♂ ×2 DDX4♀ | 6.4 | 148 | 93 | 28 (20%) | 49 (36%) | NA |
-
*
Lay rate; eggs were counted over a 60-day period when hens were between 7 and 12 months of age and divided by the number of fertile hens present in pen. The maximum possible lay rate is 7.0 eggs per hen per week.
-
†
Fertility was assessed between embryonic day 4–7.
Fertility and hatching rate from ♂ surrogate hosts injected with ♂ gonadal cells mated to ♀ surrogate hosts injected with ♀ gonadal germ cells.
| Host mating group | No. of eggs incubated | Fertility* (% eggs set) | No. of GFP embryos (%) | No. of RFP embryos (%) | No. of ‘yellow’† embryos(%) | No. of chicks hatched (% fertile eggs) | Transmission of host iCaspase9 transgene (%) |
|---|---|---|---|---|---|---|---|
| CRYO-1M_1179♂×6 CRYO-2F♀ | 15960 | 157 (99%)54 (90%) | 56 (36%)15 (31%) | 54 (34%)11 (22%) | 19 (12%)10 (20%) | NA49 (91%) | 00 |
-
Data is shown for two independent hatching cohorts.
-
*
Fertility was assessed between embryonic day 4–7.
-
†
‘Yellow’ embryos = both GFP and RFP positive.
Germline transmission from ♂ surrogate hosts injected with ♀ gonadal germ cells.
| Host mating groups | Eggs set | Fertility* (% eggs set) (%) | No. of GFP embryos (% fertile) | No. of RFP embryos (% fertile) | No. of WT embryos (% fertile) | Transmission of surrogate iCaspase9 transgene(% fertile) |
|---|---|---|---|---|---|---|
| CRYO-3F_1408♂CRYO-3F_1409♂× 6 WT♀ | 66 | 95 | 48 (76%) | 0 | 15 (24%) | 0 |
| CRYO-3F_1413♂CRYO-3F_1414♂× 5 WT♀ | 35 | 97 | 30 (88%) | 0 | 4 (12%) | 0 |
-
*
Fertility was assessed between embryonic day 4–9.
RFP transmission rates identify multiple transmission events.
| Bird ID | Total eggs set | Fertile eggs* (%) | No. of GFP(%) | No. of RFP(%) | No. of WT(%) | No. of iCaspase9(%) | No. of gonad pairs | Expected RFP (%) | p-Value |
|---|---|---|---|---|---|---|---|---|---|
| CRYO-1M: wb1181 | 147 | 14196% | 10172% | 43% | 3626% | 0 | 10 GFP+ 2 RFP | 11.1 | < 0.001 |
| CRYO-1M: wb1182 | 148 | 14799% | 8961% | 96% | 4833% | 96% | 10 GFP+ 2 RFP | 11.1 | 0.05 |
| CRYO-1M: wb1179 | 187 | 18498% | 5530% | 5128% | 7641% | 0 | 10 GFP+ 2 RFP | 11.1 | < 0.001 |
| Total | 482 | 47298% | 24552% | 6414% | 16034% | 92% | 10 GFP+ 2 RFP | 11.1 | 0.106 |
| CRYO-2F | 371 | 36298% | 13738% | 5114% | 17448% | 0 | 10 GFP+ 4 RFP | 19.0 | 0.016 |
| CRYO-3F | 340 | 27280% | 15055% | 6624% | 5520% | 0 | 17 GFP+ 6 RFP | 17.4 | 0.005 |
| CRYO-4M: wb1420 | 89 | 8191% | 6479% | 45% | 1316% | 0 | 15 GFP+ 3 RFP | 11.1 | 0.078 |
| CRYO-4M: wb1424 | 60 | 5693% | 4173% | 35% | 1120% | 0 | 15 GFP+ 3 RFP | 11.1 | 0.16 |
| CRYO-4M: wb1427 | 74 | 6385% | 2946% | 1321% | 2133% | 0 | 15 GFP+ 3 RFP | 11.1 | 0.043 |
| CRYO-4M: wb1430 | 143 | 11883% | 8068% | 1412% | 2420% | 0 | 15 GFP+ 3 RFP | 11.1 | 0.884 |
| Total | 366 | 31887% | 21467% | 3411% | 6922% | 0 | 15 GFP+ 3 RFP | 11.1 | 0.859 |
| CRYO-5F | 148 | 13893% | 4936% | 2820% | 5842% | 0 | 15 GFP+ 3 RFP | 11.1 | 0.003 |
-
A statistical analysis was performed to determine if the no. of observed RFP+ embryos differed significantly from the no. of expected RFP+ embryos. A p value of <0.05 was designated as the value at which the observed and expected numbers differed significantly. This number is shown in bold.
-
*
Fertility was measured for between embryonic day 4–6.





