Insulator-based dielectrophoresis-assisted separation of insulin secretory vesicles
Figures
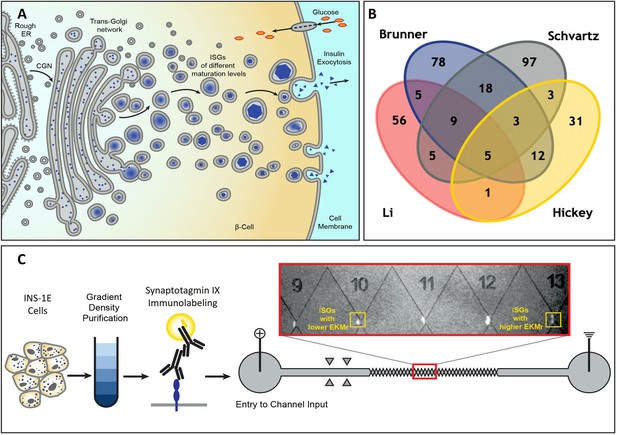
Schematic diagram for the formation of heterogenous insulin vesicles in INS-1E cells, graphical summation of disparate vesicle protein identifications, and processing of insulin vesicles including direct current insulator-based dielectrophoresis (DC-iDEP) device.
(A) Insulin vesicle formation and maturation in a pancreatic β-cell. Newly synthesized insulin is packed inside secretory vesicles which mature to store crystalline insulin in vesicles until secretion is stimulated through different signaling pathways. (B) Four published insulin vesicle proteomics studies (Schvartz et al., 2012; Li et al., 2018; Brunner et al., 2007; Hickey et al., 2009) aimed to identify the proteome of the heterogenous populations of secretory vesicles in INS-1E cells with only five proteins identified consistently. (C) Separation of insulin vesicles using a DC-iDEP device. Differential and density gradient centrifugation were used to enrich each sample for insulin vesicle populations. Samples were then immunolabeled and introduced into DC-iDEP device for high-resolution separation. Fluorescently labeled particles trapped near various gates in the channel are biophysically different subpopulations with varied EKMr values. The gates were constricted by increasing sizes of paired triangles, forming channel widths of 73 μm to 25 μm from inlet to the outlet. The different gates created and distributions for EKMr values.
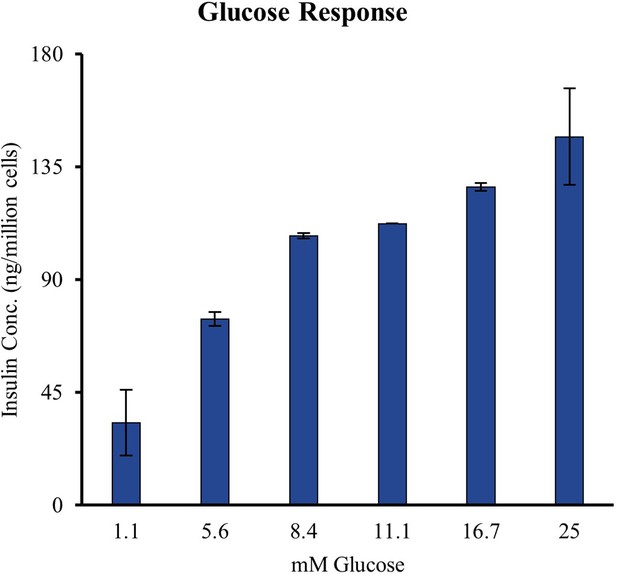
Glucose sensitivity of INS-1E insulinoma cells was tested by stimulation at increasing concentrations of glucose and measurement of insulin secretion by enzyme linked immunosorbent assay (ELISA).
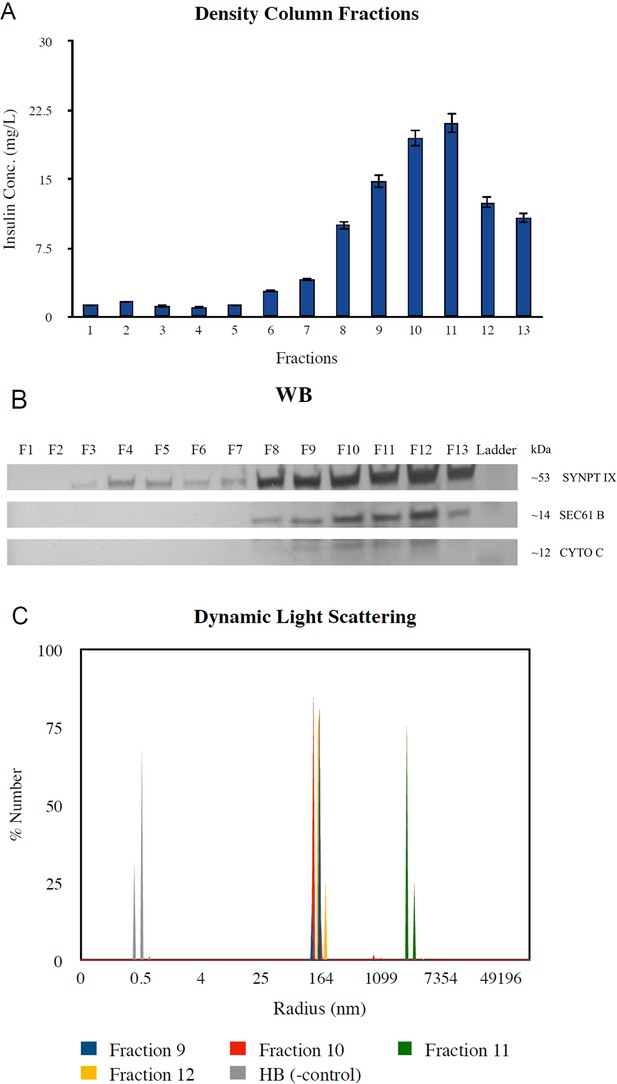
Selection of fractions for use in separation experiments.
(A) Fractions of the density column were screened for insulin content in the enzyme linked immunosorbent assay (ELISA) assay. (B) Western blotting of the density column fractions revealed high concentrations of insulin vesicle marker synaptotagmin IX in fractions with high insulin content as well as the presence of endoplasmic reticulum (ER) and mitochondria contaminants as indicated by organelle markers SEC61 and cytochrome c, respectively, in the same fractions. Raw blot in Figure 1—figure supplement 2—source data 1 and 2. (C) Dynamic light scattering (DLS) was performed on fractions of interest to validate the presence of particles of 150–200 nm in radius, corresponding to radii of insulin vesicles.
-
Figure 1—figure supplement 2—source data 1
Full image of western blotting (WB) with labels indicating synaptotagmin IX, SEC16 B, and cytochrome c.
- https://cdn.elifesciences.org/articles/74989/elife-74989-fig1-figsupp2-data1-v1.pdf
-
Figure 1—figure supplement 2—source data 2
Raw image of western blotting (WB).
- https://cdn.elifesciences.org/articles/74989/elife-74989-fig1-figsupp2-data2-v1.pdf
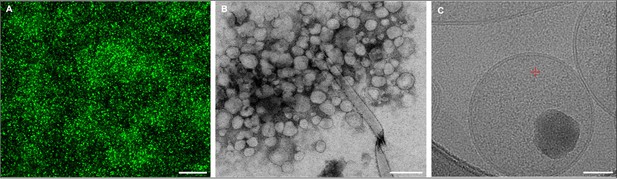
The identity of insulin vesicles was confirmed via several microscopy methods: confocal microscopy, transmission electron microscopy, and cryo-electron microscopy.
(A) Fluorescence confocal microscopy maximum projection image of enriched insulin vesicles. (B) Transmission electron microscopy (TEM) image of enriched insulin vesicles negatively stained with Nano-W. The diameters of these particles are characteristic of insulin vesicles. (C) Cryo-electron microscopy (cryo-EM) image of enriched insulin vesicles. Scale bars, 10 µm (A), 500 nm (B), 50 nm (C).
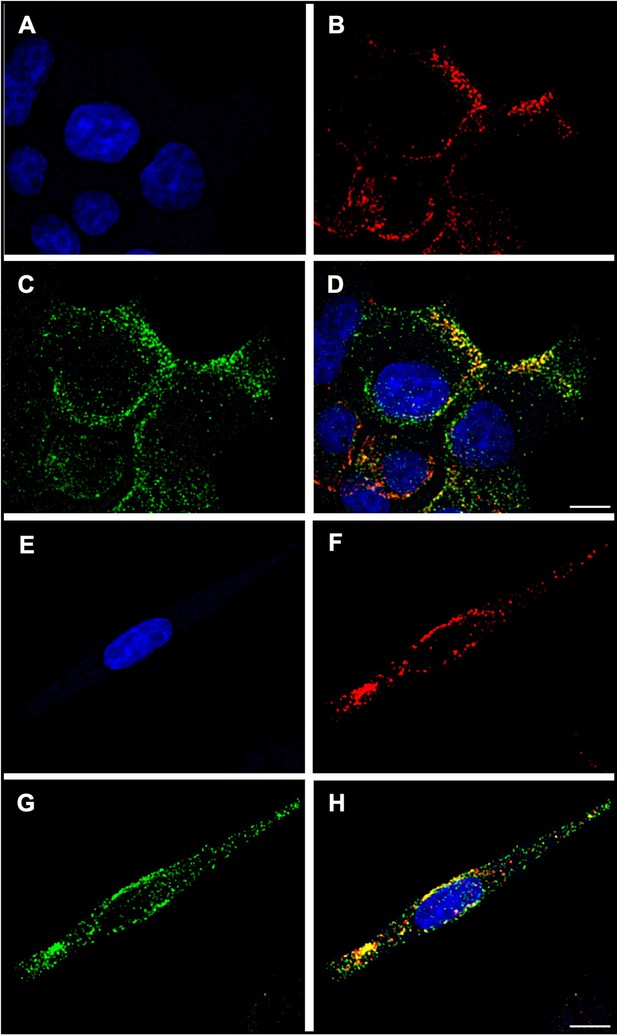
Colocalization of insulin vesicle marker used in this study (synaptotagmin IX) with insulin.
(A, E) Nuclei of INS-1E insulinoma cells stained with NucBlue. (B, F) Synaptotagmin IX labeled with rabbit anti-synaptotagmin IX and goat anti-rabbit IgG (H+L), Alexa 647. (C, G) Insulin hormone labeled with mouse anti-insulin and goat anti-mouse IgG (H+L), Alexa 488. (D, H) Localization of synaptotagmin IX to insulin vesicles as apparent from the merged intensities of panels (B) and (C) or (F) and (G). A strong colocalization was observed between insulin and synaptotagmin IX. Pearson’s r value, 0.66 (D) and 0.64 (H). Microscopy was performed with a Leica Mica using a 63×/1.2NA water immersion objective on cells mounted in ProLong Glass Antifade Mountant. Scale bars, 5 µm.
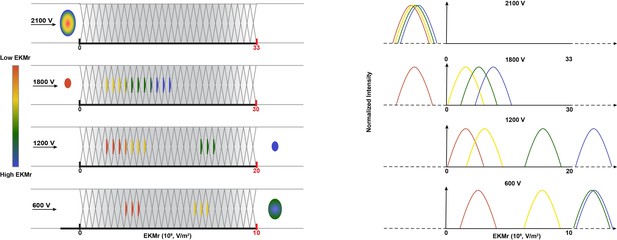
Schematic diagram of direct current insulator-based dielectrophoresis (DC-iDEP) system operated in a discovery or scanning mode.
As a function of the design of the sawtooth channel with different gate sizes along the channel, the applied voltage defines dielectrophoretic (DEP) and electrokinetic (EK) forces at each gate, and capture occurs when the EK force of the particle is equal to or smaller than the DEP force. At high voltages, only particles with high EKMr values can enter the channel; the highest applied voltage of 2100 V prevents all particles of the sample from entering the inlet of the device due to the induced dielectrophoretic forces (no fluorescent signal detected anywhere along the channel). Sequentially lower voltages allow the various subpopulations to enter and be separated throughout the channel. When a subpopulation’s EKMr value surpasses the channel’s DEP force limit, it travels freely and leaves the channel at the outlet. The right panel indicates fluorescent intensities of the captured particles are recorded along the channel at each voltage. Tracking these intensities allows the discovery and quantification of unknown subpopulations according to their biophysical properties.
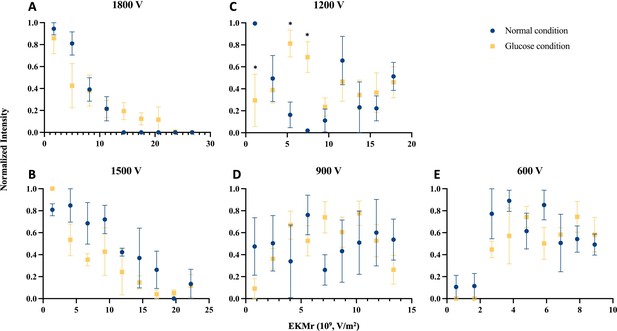
Comparison of distributions for the biophysical properties as reflected in EKMr values of n-insulin vesicles (blue circles) and g-insulin vesicles (yellow squares) with varied voltages applied.
Fluorescent intensities of captured n- and g-insulin vesicles with different EKMr values were recorded at each gate. Each data point reflects fluorescent intensities recorded at three subsequent gates with the same EKMr values, averaged out over biologically replicated experiments and normalized over all signals recorded at a given voltage. (A) Full profile of the sample’s biophysical distribution was recorded at 1800 V. (B–E) Insulin vesicle subpopulations were separated at subsequent applied voltages of 1500, 1200, 900, and 600 V. Values are mean ± SEM (n=3 for n-vesicles or 4 for g-vesicles for biologically independent experiments) (*p<0.05 using ANOVA with Bonferroni post hoc multiple comparison correction). Raw data in Figure 4—source data 1.
-
Figure 4—source data 1
The raw data depicted in Figure 4.
- https://cdn.elifesciences.org/articles/74989/elife-74989-fig4-data1-v1.xlsx





