State-dependent activity dynamics of hypothalamic stress effector neurons
Figures
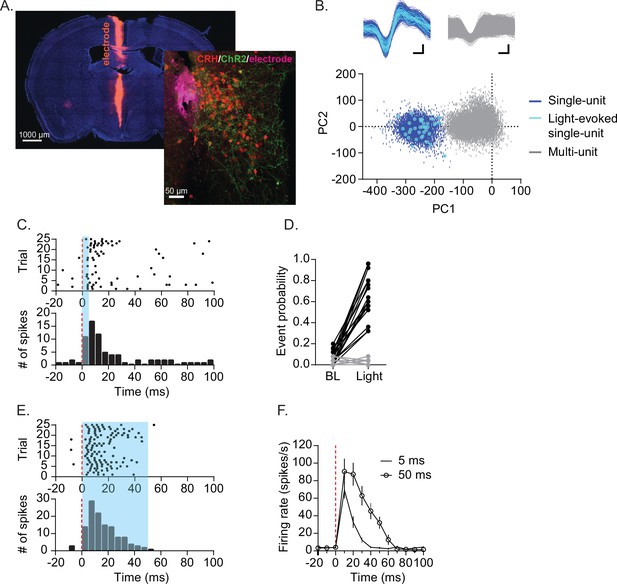
Optogenetic identification of CRHPVN neuron single-unit.
(A) Electrode tract (purple), TdTomato-expressing CRHPVN neurons (red) and ChR2-EYFP (green) expression. (B) An example for an isolated single-unit (blue) that also responded to light (cyan). (C) Raster plot (top) and peristimulus time histogram (PSTH, bottom) in response to 5 ms blue light from a representative unit. (D) A summary graph for the probability of firing before (−20–0 ms) and after blue light illumination (0–20 ms). Light-responsive units in black (n=18) and non-responsive units in gray (n=18). (E) Raster plot (top) and PSTH (bottom) for a representative single-unit responding to 50 ms blue light. (F) Summary of firing rate time course following 5 ms (tick) and 50 ms (open circle) blue light. n=18. SD is represented as error bars.
-
Figure 1—source data 1
Optogenetic identification of CRHPVN neuron single-unit.
- https://cdn.elifesciences.org/articles/76832/elife-76832-fig1-data1-v2.xlsx
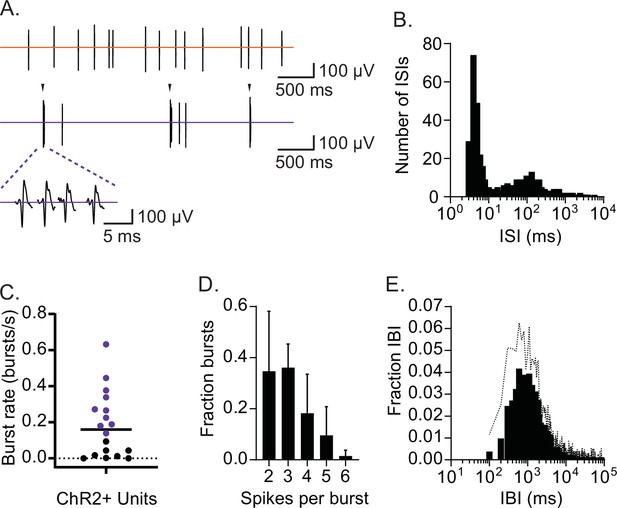
CRHPVN neurons fire in rhythmic bursts and single spikes.
(A) Two distinct spike patterns in a representative single-unit (top: single spikes; bottom: bursts indicated by arrow). (B) Interspike interval (ISI) distribution for the representative single-unit shown in A. (C) A summary plot for burst rates among CRHPVN neurons during non-stress baseline recordings. The criterion for burst firing neurons (purple) was set as 0.1 burst/s. A horizontal bar indicates the average. (D) A summary graph for burst length distribution (n=10). (E) Summary interburst interval (IBI) distribution (n=10). Standard deviation is represented in the graphs as the error bars (D) and dotted line (E).
-
Figure 2—source data 1
CRHPVN neurons fire in rhythmic bursts and single spikes.
- https://cdn.elifesciences.org/articles/76832/elife-76832-fig2-data1-v2.xlsx
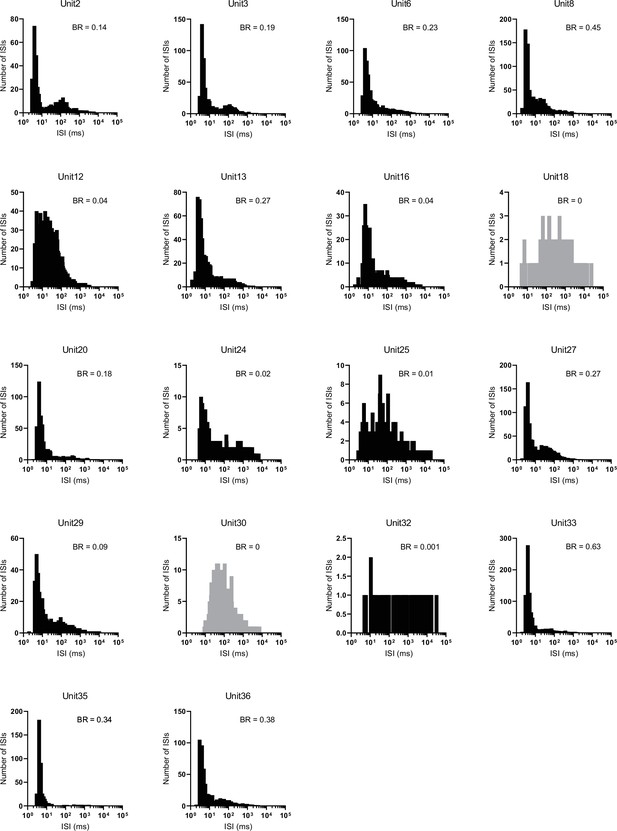
Interspike interval (ISI) histograms for all CRHPVN units.
Burst rate (BR) is indicated on the top-right corner of individual histograms. Unit 18 and 30 did not fire bursts and are shown in gray.
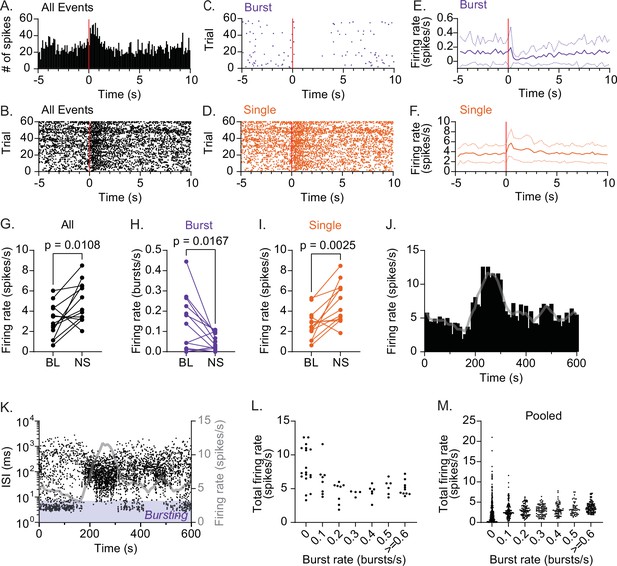
CRHPVN neurons are constrained to low activity during rhythmic bursting.
(A) Peristimulus time histogram (PSTH) for a representative single-unit responding to sciatic nerve stimulation (1.5 mA, 0.5 ms × 5 pulses at 20 Hz, red line). (B–D) Raster plots for the unit shown in A for all spikes (B), bursts (C) and single spikes (SS) (D). (E, F) Summary time course for bursts (E) and SS (F). n=13. (G–I) Summary changes in all spikes (G, paired t-test, n=13, p=0.0108), bursts (H, paired t-test, n=13, p=0.0167) and SS (I, paired t-test, n=13, p=0.0025) before (baseline, BL) and after sciatic nerve stimulation (NS). (J, K) Time course of firing rate (J) and interspike interval (ISI) (K) for a representative single-unit during baseline recording. Gray lines are the running average (30 s) of firing rate. (L) The relationship between burst rate and total firing rate for the representative single-unit shown in J and K. For each time bin (10 s), total spike rate was plotted against burst rate. (M) Pooled data for all units (n=18).
-
Figure 3—source data 1
CRHPVN neurons are constrained to low activity during rhythmic bursting.
- https://cdn.elifesciences.org/articles/76832/elife-76832-fig3-data1-v2.xlsx
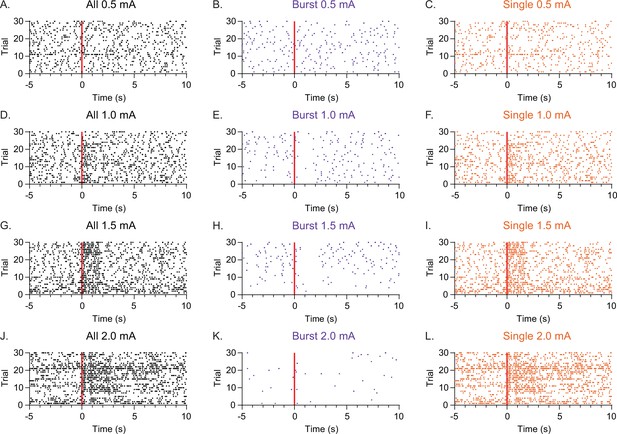
Firing pattern changes persist longer with higher intensity nerve stimulations.
Raster plots of a representative single unit responding to different intensities of sciatic nerve stimulations. (A–C) 0.5 mA, (D–F) 1.0 mA, (G–I) 1.5 mA, and (J–L) 2.0 mA. Raster plots for all spikes (A, D, G, J), burst firing (B, E, H, K) and single spikes (C, F, I, L). Note that the highest intensity stimulation (2.0 mA) caused persistent decrease of burst firing and increase of single spiking across trial durations compared to lower intensity (0.5–1.5 mA) stimulations.
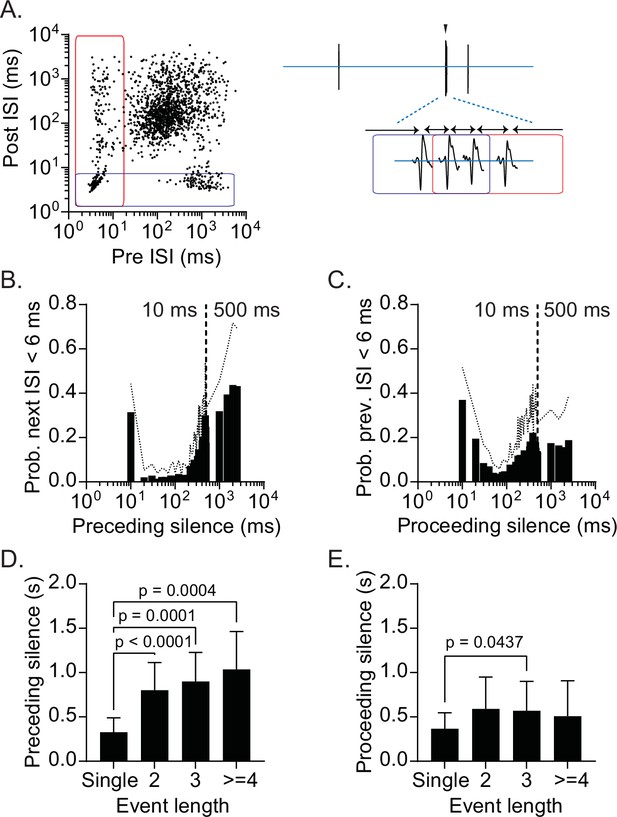
Prolonged silent periods precede burst firing.
(A) Preceding and proceeding interspike interval (ISI) plotted for individual spikes recorded from a representative single-unit. Blue rectangle indicates spikes with proceeding ISI <6 ms. Red rectangle indicates spikes with preceding ISI <20 ms. Left. An example of a burst episode. (B) Summary of probability of burst firing relative to preceding silence (ISI). (C) Summary of probability of burst firing relative to proceeding silence (ISI). (D) Mean preceding silence as a function of event length. One-way ANOVA, p<0.0001; Tukey’s multiple comparisons test, single vs 2 (p<0.0001), 3 (p=0.0001), ≥ 4 (p=0.0004) (E) Mean proceeding silence as a function of event length. One-way ANOVA, p=0.0131; Tukey’s multiple comparisons test, single vs 2 (p=0.0573), 3 (p=0.0437), ≥ 4 (p=0.2885) SD is represented in the graphs as dotted lines (B, C) and the error bars (D, E).
-
Figure 4—source data 1
Prolonged silent periods precede burst firin.
- https://cdn.elifesciences.org/articles/76832/elife-76832-fig4-data1-v2.xlsx
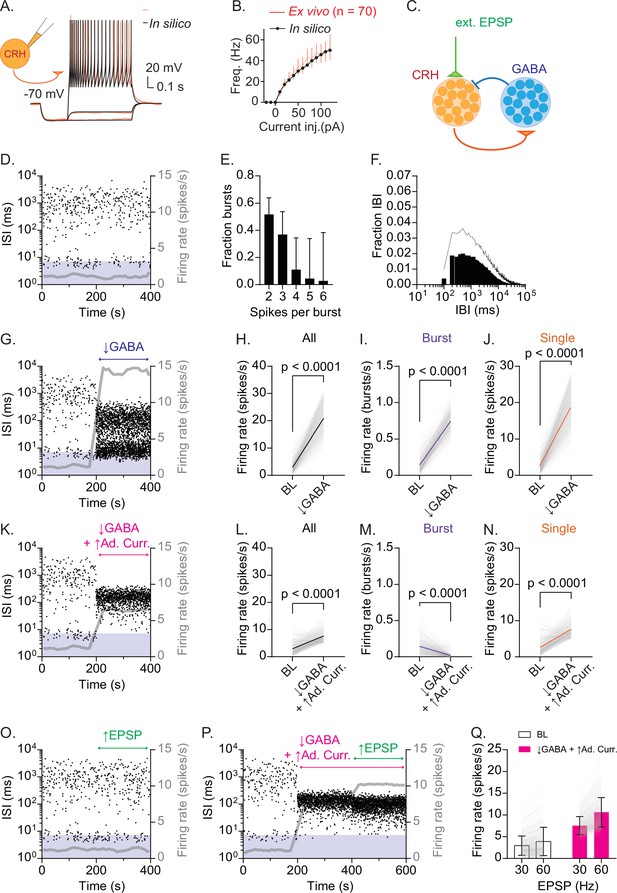
Recurrent inhibitory circuits generate burst firing and gate firing response to excitatory inputs in CRHPVN neurons.
(A) CRH model neuron in silico (black) fitted to current-clamp recordings of CRHPVN neurons ex vivo (red). (B) F-I curves for in silico (black) and ex vivo (red) neurons. (C) Network model diagram. (D) Time course of interspike interval (ISI) for a representative model CRH neuron. Gray line is the running average firing rate. (E) Summary burst length distribution for model simulations (n=283 for burst rate >0.1 Hz). (F) Summary IBI distribution for model simulations (n=283 for burst rate >0.1 Hz). (G) An example ISI time course before and after a drop of GABA release (Pr 0.8 → 0.1) from 200 s. Gray line is the running average firing rate. (H–J) Summary changes in all spikes (H, paired t-test, p<0.0001, n=500), bursts (I, paired t-test, p<0.0001, n=500) and single spikes (SS) (J, paired t-test, p<0.0001, n=500) before (BL) and after the GABA release drop. (K) An example ISI time course before and after a drop of GABA release (Pr 0.8 → 0.1) combined with an increase in spike-triggered adaptation current from 200 s. Gray line is the running average firing rate. (L–N) Summary changes in all spikes (L, paired t-test, p<0.0001, n=500), bursts (M, paired t-test, p<0.0001, n=500) and SS (N, paired t-test, p<0.0001, n=500) before (BL) and after combined removal of GABA release and increased spike-triggered adaptation current. (O) An example ISI time course before and after EPSP frequency increase (30 Hz → 60 Hz) from 200 s. Gray line is the running average firing rate. (P) An example ISI time course before and after the combined change in GABA release and spike-triggered adaptation current, followed by an increase in EPSP frequency (30 Hz → 60 Hz) from 400 s. (Q) Summary graph for changes in firing rate before (30 Hz) and after EPSP frequency increase (60 Hz) with (white) and without recurrent inhibition (pink). Two-way ANOVA (EPSP × Recurrent inhibition interaction, p<0.0001; Tukey’s multiple comparisons test, BL-30 Hz 2.942 ± 2.220 Hz vs BL-60 Hz 3.895 ± 3.252 Hz, p<0.0001; ↓GABA+↑Ad. Curr.-30 Hz 7.548 ± 2.068 Hz vs ↓GABA+↑Ad. Curr.-60 Hz 10.630 ± 3.387 Hz, p<0.0001). SD is represented in the graphs as the error bars.
-
Figure 5—source data 1
Recurrent inhibitory circuits generate burst firing and gate firing response to excitatory inputs in CRHPVN neurons.
- https://cdn.elifesciences.org/articles/76832/elife-76832-fig5-data1-v2.xlsx
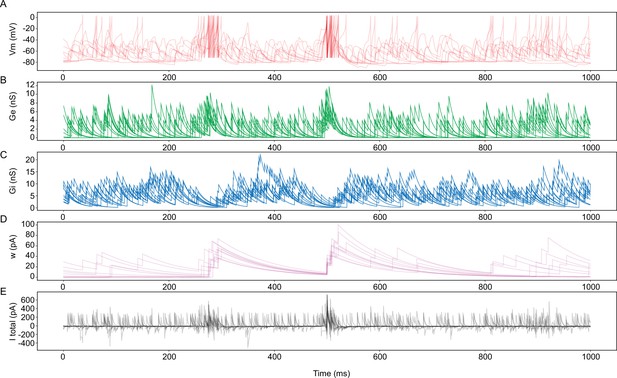
The temporal relationship between network and intrinsic properties with burst firing.
(A) Overlay of simulated membrane voltage (Vm; red) of representative traces of burst firing from model neurons (nneurons = 5; nbursts = 10). Note that spikes are truncated to 0 mV in this example. (B) The incoming excitatory conductance (Ge; green). (C) The incoming inhibitory conductance (Gi; blue). (D) The spike-triggered adaptation (w; pink). (E) The representative current (I total; black) that each neuron receives. The representative current is the proportional sum of the above network and intrinsic inputs (Ge, Gi, and w). Representative traces were selected based on an initial interspike interval lower than 6 ms and a preceding interburst interval (IBI) of 200–220 ms. Traces are aligned to the second burst.
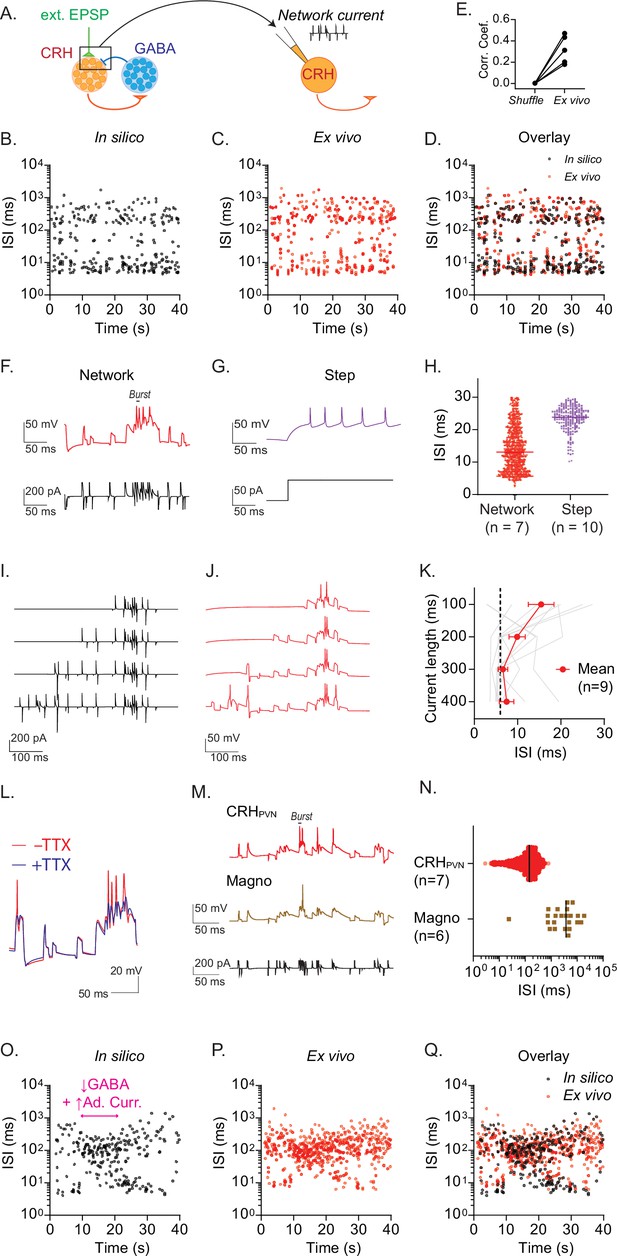
Recurrent inhibitory circuits underlie burst firing.
(A) Diagram of ‘network clamp’ experiment. (B–D) Time course of interspike interval (ISI) for a representative model CRH neurons (B), a biological CRHPVN neuron injected with network current (C) and overlay (D). (E) Correlation coefficient between the time series of model neuron spike train in silico, a biological CRHPVN neurons’ spike train ex vivo, and a shuffled spike train consisting of the model spike train with ISI’s shuffled (n=5). (F, G) Spike firing patterns in response to network current (F) and depolarizing current step (G) in a biological CRHPVN neuron. (H) ISI distribution of spikes generated by network current (red) and steps of current (purple). Note that spikes with ISI smaller than 30 ms were used for comparison. (I, J) Network currents of variable length (I) and corresponding membrane voltage changes in CRHPVN neurons (J). (K) Summary graph for the shortest ISI triggered by network currents of variable lengths. SD is represented in the graph as the error bars. (L) CRHPVN neuron’s firing response to network current with (blue) and without tetrodotoxin (TTX, 1 µM, red). (M) Firing of CRHPVN neuron (orange) and magnocellular neuron (brown) in response to an identical network current (black). (N) Summary of ISI triggered by network current in CRHPVN and magnocellular neurons. (O–Q) A representative ISI time course for the transition between rhythmic burst and single spiking in a model CRH neuron (O), a biological CRHPVN neuron injected with the network current of the model neuron shown in O, and overlay (Q).
-
Figure 6—source data 1
Recurrent inhibitory circuits underlie burst firing.
- https://cdn.elifesciences.org/articles/76832/elife-76832-fig6-data1-v2.xlsx
Tables
Parameters for spiking network model.
| Neuron parameters | Mean | S | Synapse parameters | Value |
|---|---|---|---|---|
| Number of neurons | 1000 | Ee (mV) | 0 | |
| Number of CRH neurons | 500 | Ei (mV) | –80 | |
| Number of GABA neurons | 500 | τe (ms) | 12.5 | |
| Capacitance (pF) | 22.0 | 2.6 | τCRH (ms) | 232.6 |
| τm (ms) | 26.0 | 2.5 | τi (ms) | 20.4 |
| gL (nS) | 0.9 | 0.19 | we (nS) | 3.9 |
| ∆T (mV) | 12.1 | 2.0 | wCRH (nS) | 0.005 |
| EL (mV) | –67.9 | 2.9 | wi (nS) | 3.3 |
| VT (mV) | –47.2 | 6.5 | τbr (s) | 40 |
| VR (mV) | –58.8 | 2.5 | τp (s) | 80 |
| τw (ms) | 98.2 | 54.3 | ||
| a (nS) | 0.082 | 0.13 | ||
| b (pA) | 17.9 | 9.8 |





