Tyrosine phosphorylation tunes chemical and thermal sensitivity of TRPV2 ion channel
Figures
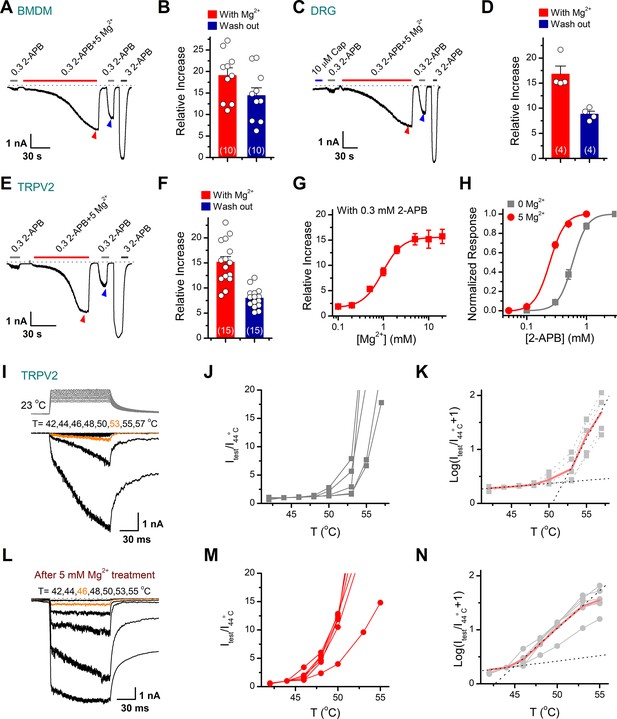
Transient receptor potential vanilloid 2 (TRPV2) activities are enhanced in the presence of Mg2+.
(A) Mg2+ potentiates 2-aminoethyl diphenylborinate (2-APB) responses in a representative rat bone marrow-derived macrophage (BMDM) cell. The cell was exposed to 0.3 mM 2-APB without or with 5 mM Mg2+ and 3 mM 2-APB as indicated by the bars. Membrane currents were recorded in whole-cell configuration, and the holding potential was –60 mV. Bars represent duration of drug application. The dotted line indicates zero current level. (B) Summary of relative currents evoked by 0.3 mM 2-APB in the presence of 5 mM Mg2+ (indicated by red triangle) and the subsequent application of 0.3 mM 2-APB (indicated by blue triangle). Numbers of cells are indicated in parentheses. (C) Whole-cell currents at –60 mV in a rat dorsal root ganglion (DRG) neuron treated with 10 μM Cap, 0.3 mM 2-APB, 0.3 mM 2-APB plus 5 mM Mg2+, and 3 mM 2-APB. (D) Summary of relative currents elicited with 5 mM Mg2+ (marked by red triangle) and the subsequent application of 0.3 mM 2-APB (marked by blue triangle). (E–F) Parallel whole-cell recordings in TRPV2-expressing HEK293T cells and the relative changes caused by Mg2+. (G) Dose dependence of Mg2+ effects on 2-APB response (0.3 mM). The solid line represents a fit by Hill’s equation with EC50 = 0.96 ± 0.03 mM and nH = 2.0 ± 0.1 (n ≥ 5). (H) Dose-response curves of 2-APB for activation of TRPV2 in the presence of 0 or 5 mM Mg2+. The solid lines corresponds to Hill’s equation with EC50 = 0.59 ± 0.01 mM and nH = 3.6 ± 0.1 for 0 Mg2+ (n = 11); and EC50 = 0.24 ± 0.01 mM and nH = 3.4 ± 0.1 for application of 5 mM Mg2+ (n = 21). (I) Representative responses to a family of temperature pulses for TRPV2-expressing HEK293T cells under control (n = 5). Temperature pulses stepped from room temperature generated by laser irradiation were 100 ms long and had a rise time of 2 ms. (J) Current vs. temperature relations at −60 mV obtained from experiments as in (I). Individual cells are shown with currents normalized by their amplitude at 44°C. (K) Plot of log(Itest/I44oC+1) obtained from the relations in (I). (L–N) Representative current traces, temperature-activation relations, and plot of log(Itest/I44oC+1) determinations for Mg2+ pretreated TRPV2-expressing cells (n = 6).
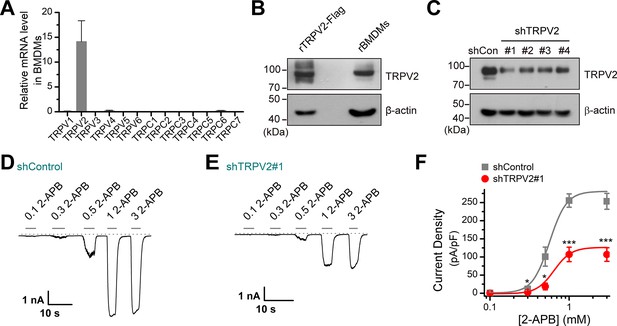
Expression of transient receptor potential vanilloid 2 (TRPV2) in bone marrow-derived macrophages (BMDMs).
(A) Relative mRNA expression levels of different Trpv and Trpc channels in BMDMs were assessed by qPCR. (B) Expression of TRPV2 protein in BMDMs. (C) Immunoblot analysis (with anti-TRPV2 or anti-β-actin) of BMDM cells transfected for 72 hr with TRPV2-targeting shRNA (shTRPV2#1, shTRPV2#2, shTRPV2#3, and shTRPV2#4) or shControl to test knockdown efficiency of shRNA. (D–E) Typical response of DMBMs transfected with shControl (D) or shTRPV2#1 (E) to 2-APB. (F) Summary plot of current density. The peak current densities evoked by 2-APB are normalized by membrane capacitance. The dotted line indicates zero current level. *p < 0.05, ***p < 0.001.
-
Figure 1—figure supplement 1—source data 1
Uncropped, unedited blots for Figure 1—figure supplement 1B.
- https://cdn.elifesciences.org/articles/78301/elife-78301-fig1-figsupp1-data1-v2.zip
-
Figure 1—figure supplement 1—source data 2
Uncropped, unedited blots for Figure 1—figure supplement 1C.
- https://cdn.elifesciences.org/articles/78301/elife-78301-fig1-figsupp1-data2-v2.zip
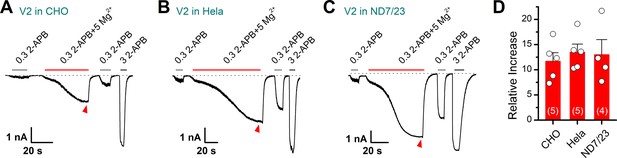
Mg2+ potentiates transient receptor potential vanilloid 2 (TRPV2) currents expressed in various cell lines.
(A–C) Representative whole-cell recordings in CHO, Hela, or ND7/23 that expressed TRPV2 showing the response to 0.3 mM 2-aminoethyl diphenylborinate (2-APB), 0.3 mM 2-APB plus 5 mM Mg2+ and 3 mM 2-APB. The dotted line indicates zero current level. (D) Summary of relative currents induced by 0.3 mM 2-APB plus 5 mM Mg2+ (marked by red triangle).
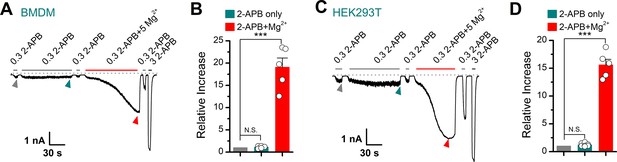
Effect of Mg2+ on 2-aminoethyl diphenylborinate (2-APB)-evoked transient receptor potential vanilloid 2 (TRPV2) currents.
(A) Representative whole-cell recordings in bone marrow-derived macrophages (BMDMs) showing the responses to 0.3 mM 2-APB, 0.3 mM 2-APB plus 5 mM Mg2+ and 3 mM 2-APB. The dotted line indicates zero current level. (B) Summary of relative currents evoked by 0.3 mM 2-APB (marked by dark cyan triangle) and 0.3 mM 2-APB plus 5 mM Mg2+ (marked by red triangle). (C) Whole-cell recordings from TRPV2-expressing HEK293T cells were consecutively challenged by 0.3 mM 2-APB, 0.3 mM 2-APB plus 5 mM Mg2+, and 3 mM 2-APB. (D) Summary of the relative changes.
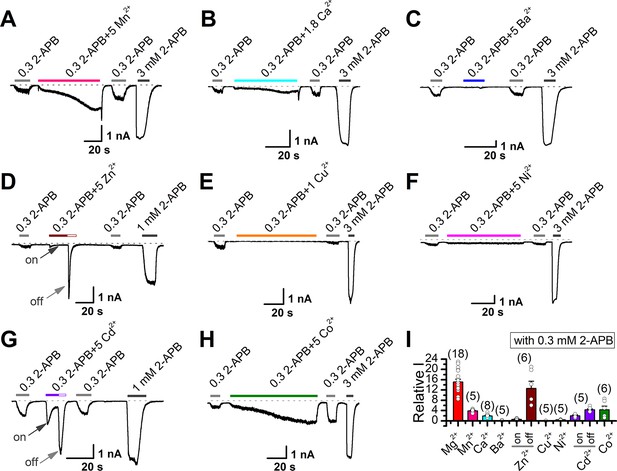
Effects of various divalent cations on 2-aminoethyl diphenylborinate (2-APB)-evoked transient receptor potential vanilloid 2 (TRPV2) currents.
(A–H) Representative whole-cell currents in TRPV2-expressing HEK293T cells induced by 0.3 mM 2-APB, the combination of 0.3 mM 2-APB and various divalent cations Mn2+ (A), Ca2+ (B), Ba2+ (C), Zn2+ (D), Cu2+ (E), Ni2+ (F), Cd2+ (G), and Co2+ (H), respectively. The dotted line indicates zero current level. (I) Summary of relative currents evoked by the combination of 0.3 mM 2-APB and different divalent cations vs. 0.3 mM 2-APB only.
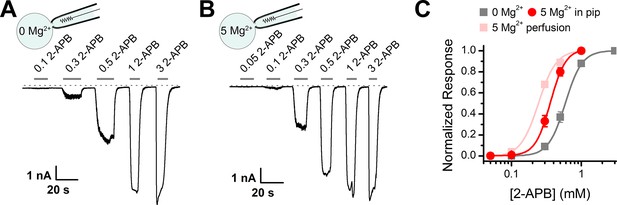
Effect of intracellular Mg2+ on dose responses to 2-aminoethyl diphenylborinate (2-APB) for activation of transient receptor potential vanilloid 2 (TRPV2).
(A–B) Representative whole-cell currents at –60 mV evoked by increasing concentrations of 2-APB in TRPV2-expressing HEK293T cells. Note the pipette solution contained 0 mM Mg2+ (A) or 5 mM Mg2+ (B). The dotted line indicates zero current level. (C) Concentration-response curves of 2-APB for activation of TRPV2. The solid lines correspond to Hill’s equation with EC50 = 0.56 ± 0.01 mM and nH = 3.9 ± 0.2 for 0 Mg2+ in pipette (n = 7); and EC50 = 0.36 ± 0.01 mM and nH = 3.7 ± 0.3 for 5 mM Mg2+ in the pipette solutions (n = 9).
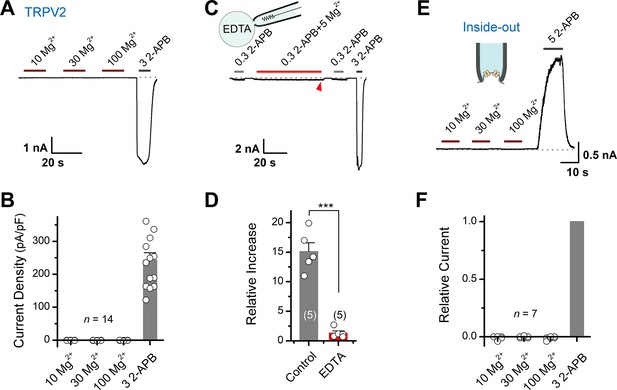
Mg2+ has an indirect effect on transient receptor potential vanilloid 2 (TRPV2) channels.
(A) High concentrations of Mg2+ have no direct effect on TRPV2 channels from the extracellular side. Representative whole-cell currents at –60 mV in a TRPV2-expressing HEK293T cells consecutively treated with 10, 30, 100 mM Mg2+ and 3 mM 2-aminoethyl diphenylborinate (2-APB). (B) Comparison of current density evoked by different concentrations of Mg2+ and 3 mM 2-APB. (C) Whole-cell recordings from TRPV2-expressing HEK293T cells showing the response to 0.3 mM 2-APB, 0.3 mM 2-APB plus 5 mM Mg2+, and 3 mM 2-APB. Note the pipette solution contained 20 mM EDTA. (D) Average plot of the relative changes. p = 0.0006 by unpaired Student’s t-test. (E) Current traces recorded in inside-out configuration evoked by different concentrations of Mg2+ and 5 mM 2-APB. (F) Summary plot of relative currents elicited with 10, 30, 100 mM Mg2+ and 3 mM 2-APB. The dotted line indicates zero current level.
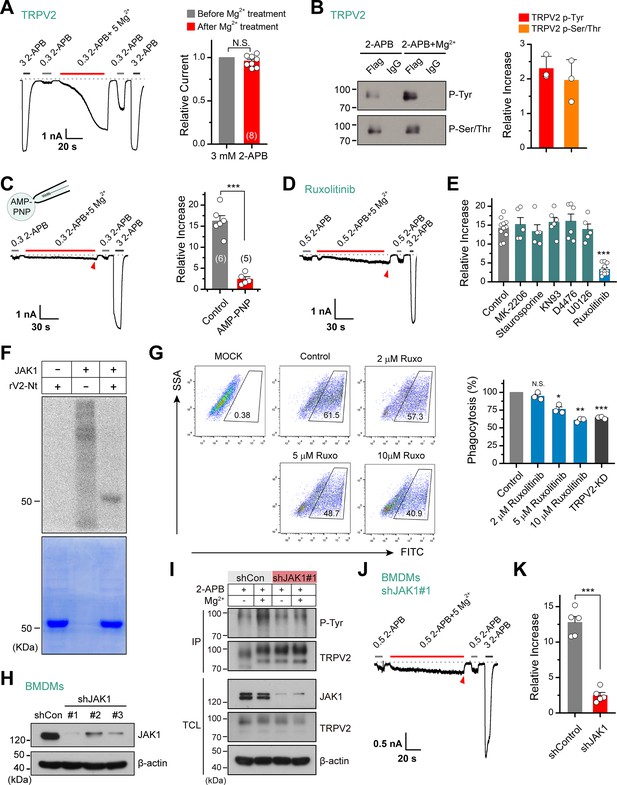
Tyrosine phosphokinase JAK1 upregulates channel activity via phosphorylation of transient receptor potential vanilloid 2 (TRPV2).
(A) Representative whole-cell recordings from TRPV2-expressing HEK293T cells showing the responses to 3 mM 2-aminoethyl diphenylborinate (2-APB) before and after the treatment by 0.3 mM 2-APB plus 5 mM Mg2+ (left). Average peak responses to 3 mM 2-APB before and after Mg2+ application (right). The dotted line indicates zero current level. The holding potential was –60 mV. p = 0.12 by one-sample t-test. (B) Tyrosine phosphorylation and serine/threonine phosphorylation of immunoprecipitated TRPV2-Flag transiently transfected in HEK293T cells in the absence and presence of 5 mM Mg2+ were determined by immunoblotting with anti-phosphotyrosine antibody (pTyr) and anti-Phospho-(Ser/Thr) Phe antibody (pSer/Thr). Inset, Protein amounts of tyrosine-phosphorylated or serine/threonine-phosphorylated immunoprecipitated TRPV2 proteins were quantified, and phospho-Tyr TRPV2/total TRPV2 and phospho-Ser/Thr TRPV2/total TRPV2 were calculated from at least three independent experiments. Error bars indicate SD. (C) Left, representative whole-cell currents at –60 mV in a TRPV2–expressing HEK293T cell treated with 0.3 mM 2-APB, 0.3 mM 2-APB plus 5 mM Mg2+ and 3 mM 2-APB. The pipette solution contained adenosine triphosphate ( ATP) nonhydrolyzable analog adenylyl imidodiphosphate (AMP-PNP). Right, summary of relative changes under different conditions. p = 9.29E-6 by unpaired Student’s t-test. (D) Whole-cell currents in response to 2-APB under inhibition of JAK1 by Ruxolitinib. (E) Summary plot of Mg2+ effects on TRPV2 currents under the various conditions. ***p < 0.001. (F) In vitro kinase assay with [32P]-γ-ATP, tyrosine kinase JAK1, and recombinant His-tagged rat TRPV2 N-terminus. Phosphorylation signals were detected by autoradiography. Loading amount of different TRPV2 proteins was accessed by coomassie blue staining. (G) Flow cytometry analysis for phagocytosis. Flow cytometry analysis was employed to determine the phagocytosed level of green fluorescent protein (GFP)-expressing Escherichia coli (GFP E. coli) by bone marrow-derived macrophages (BMDMs) treated with varying concentrations of Ruxolitinib or transfected with shTRPV2#1. Bar graph displaying the effects on phagocytosis under different conditions. *p < 0.05, **p < 0.01, ***p < 0.001. (H) Immunoblot analysis (with anti-JAK1 or anti-β-actin) of BMDM cells transfected for 72 hr with JAK-1-targeting shRNA (shJAK1#1, shJAK1#2, and shJAK1#3) or shControl to test knockdown efficiency of shRNA. (I) Immunoblot analysis of the tyrosine phosphorylation levels of TRPV2 in BMDM cells transfected with shJAK1#3 or shControl for 72 hr in the absence and presence of Mg2+, respectively. (J) Whole-cell recordings in BMDM cells transfected with shJAK1#3 showing the responses to 0.3 mM 2-APB, 0.3 mM 2-APB plus 5 mM Mg2+ and 3 mM 2-APB. (K) Comparison of relative increase under different conditions. p = 4.49E-6 by unpaired Student’s t-test. Error bars indicate standard error of the mean (SEM).
-
Figure 3—source data 1
Uncropped, unedited blots for Figure 3B.
- https://cdn.elifesciences.org/articles/78301/elife-78301-fig3-data1-v2.zip
-
Figure 3—source data 2
Uncropped, unedited blots and gels for Figure 3F.
- https://cdn.elifesciences.org/articles/78301/elife-78301-fig3-data2-v2.zip
-
Figure 3—source data 3
Uncropped, unedited blots for Figure 3H.
- https://cdn.elifesciences.org/articles/78301/elife-78301-fig3-data3-v2.zip
-
Figure 3—source data 4
Uncropped, unedited blots for Figure 3I.
- https://cdn.elifesciences.org/articles/78301/elife-78301-fig3-data4-v2.zip
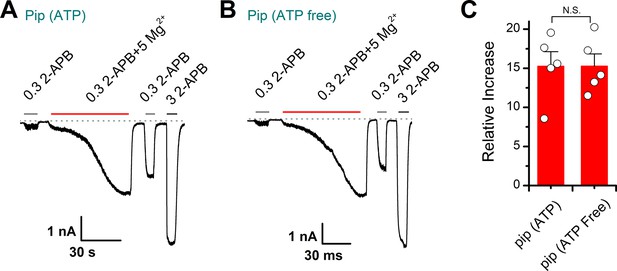
Effects of Mg2+ on transient receptor potential vanilloid 2 (TRPV2) responses with or without addition of adenosine triphosphate (ATP) in the pipette solutions.
(A–B) Representative whole-cell currents in a TRPV2-expressing HEK293T cell elicited with 0.3 mM 2-aminoethyl diphenylborinate (2-APB), 0.3 mM 2-APB plus 5 mM Mg2+ and 3 mM 2-APB. Please note the pipette solutions containing with 1 mM adenosine disodium triphosphate (Na2ATP) (A) or without ATP (B). The dotted line indicates zero current level. (C) Summary plot of the relative changes under different conditions. p = 0.998 by unpaired Student’s t-test.
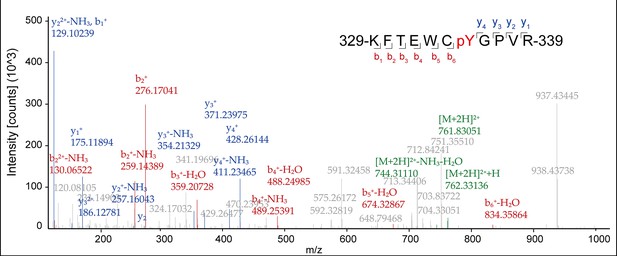
Liquid chromatography-tandem mass spectrometry (LC-MS/MS) analysis of the phosphorylation of transient receptor potential vanilloid 2 (TRPV2).
LC-MS/MS analysis showing the phosphorylation of TRPV2 in HEK293T cells after treatment by 0.3 mM 2-aminoethyl diphenylborinate (2-APB) plus 5 mM Mg2+, followed by immunoprecipitation (with anti-FLAG agarose). Peaks corresponding to phosphorylated TRPV2 peptides were obtained from MS. MS/MS ion spectrum with the matched b and y ions of the pY335-containing tryptic peptide KFTEWCpYGPVR was shown.
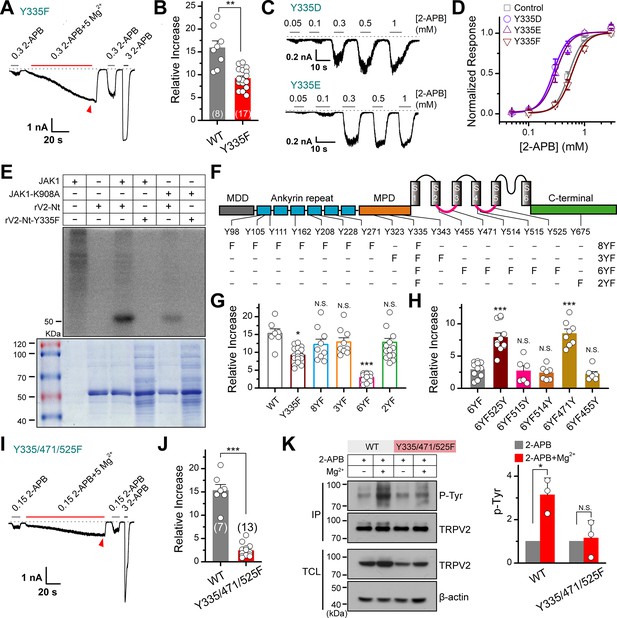
JAK1 has three phosphorylation sites on the transient receptor potential vanilloid 2 (TRPV2) channel.
(A) Representative whole-cell currents at –60 mV elicited with 0.3 mM 2-APB, 0.3 mM 2-APB plus 5 mM Mg2+ and 3 mM 2-APB in HEK293T cells that expressed TRPV2(Y335F). Bars represent duration of stimuli. The dotted line indicates zero current level. (B) Comparison of relative changes between wild-type TRPV2 and TRPV2(Y335F) following the treatment by Mg2+. p = 0.003 by unpaired Student’s t-test. (C) Representative whole-cell currents at –60 mV evoked by varying concentrations of 2-APB in HEK293T cells that expressed TRPV2(Y335D) or TRPV2(Y335E). (D) Concentration-response curves of 2-APB for TRPV2 mutants. Solid lines represent fits by a Hill’s equation with EC50 = 0.53 ± 0.01 mM and nH = 3.5 ± 0.1 for TRPV2-WT (n = 9); EC50 = 0.28 ± 0.01 mM and nH = 3.4 ± 0.2 for Y335D (n = 8); EC50 = 0.31 ± 0.01 mM and nH = 3.3 ± 0.1 for Y335E (n = 7) and EC50 = 0.60 ± 0.01 mM and nH = 3.4 ± 0.2 for Y335F (n = 8). (E) In vitro kinase assay with [32P]-γ-ATP, immunoprecipitated tyrosine kinase JAK1 and recombinant His-tagged wild-type or mutant TRPV2 N-terminus. Phosphorylation signals were examined by autoradiography. (F) Linear diagram of the TRPV2 channel topology, with all intracellular tyrosine residues labeled, and a summary of substitutions of tyrosine by phenylalanine used in this study. (G) Summary plot of the Mg2+-dependent enhancement in various mutants. All the TRPV2 mutants retained their normal responses to 2-APB. *p < 0.05, ***p < 0.001. (H) Statistic results for the Mg2+-dependent enhancement for mutants which were respectively reverse mutated from TRPV2-6YF. ***p < 0.001. (I) Representative whole-cell currents at –60 mV elicited with 0.15 mM 2-APB, 0.15 mM 2-APB plus 5 mM Mg2+ and 3 mM 2-APB in HEK293T cells that expressed TRPV2-Y335/471/525F. (J) Average plot of the relative changes of wild-type and Y335/471/525F currents following treatment by Mg2+. p = 2.30E-9 0.001 by unpaired Student’s t-test. (K) Immunoblotting analysis with anti-phosphotyrosine antibody (pTyr) showing the tyrosine phosphorylation levels in HEK293T cells transfected with TRPV2 or TRPV2-Y335/471/525F in the absence and presence of Mg2+. Right, quantitative analysis of the fold increase of tyrosine-phosphorylated TRPV2 proteins and TRPV2(Y335/471/525F) proteins following different treatments (n = 3; means ± SD [standard deviation]). Error bars indicate standard error of the mean (SEM).
-
Figure 4—source data 1
Uncropped, unedited blots and gels for Figure 4E.
- https://cdn.elifesciences.org/articles/78301/elife-78301-fig4-data1-v2.zip
-
Figure 4—source data 2
Uncropped, unedited blots for Figure 4K.
- https://cdn.elifesciences.org/articles/78301/elife-78301-fig4-data2-v2.zip

Partial amino acid sequence alignment of transient receptor potential vanilloid 2 (TRPV2) channels.
Multiple alignments of TRPV2 amino acid sequences surrounding Y335, Y471, and Y525 from rat, mouse, human, rabbit, macaque, cattle, dog, horse, and whale. The residues of Y335, Y471, and Y525 are boxed in the sequence alignment.
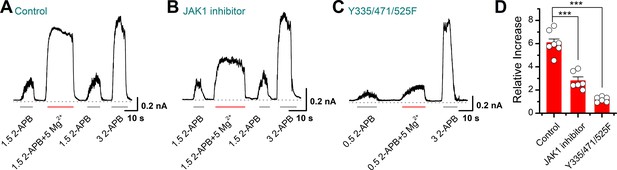
Inside-out recordings showing the effects of Mg2+ on 2-aminoethyl diphenylborinate (2-APB)-evoked currents.
(A–C) Representative currents at +60 mV recorded in an inside-out patch excised from transient receptor potential vanilloid 2 (TRPV2)(wild-type)-expressing cells (A), the TRPV2(wild-type)-expressing cells pretreated with 10 μM Ruxolitinib (B), and TRPV2(Y335/471/525F)-expressing cells (C). The dotted line indicates zero current level. (D) Average plot of the relative changes. p = 7.05E-7 for control vs. Ruxolitinib treatment and p = 2.78E-9 for control vs. Y335/471/525F treatment using one-way analysis of variance (ANOVA) t-test. Error bars indicate standard error of the mean (SEM).
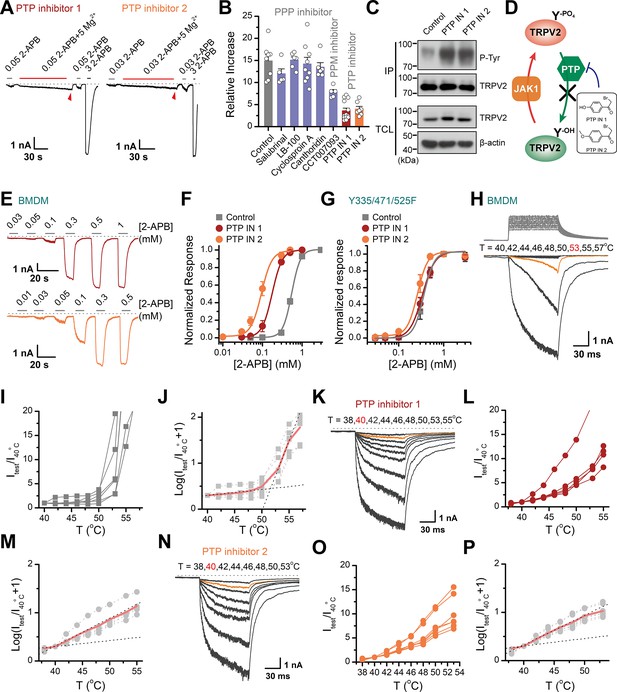
Increasing the phosphorylation level of transient receptor potential vanilloid 2 (TRPV2) by inhibition of dephosphorylase activity enhances the channel sensitivity to its stimuli.
(A) Whole-cell recordings from TRPV2-expressing HEK293T cell were consecutively challenged with 0.3 mM 2-aminoethyl diphenylborinate (2-APB), 0.3 mM 2-APB plus 5 mM Mg2+ and 3 mM 2-APB. The cells were pretreated with protein tyrosine phosphatase (PTP) inhibitor 1 and PTP inhibitor 2 for 5 min, respectively. The dotted line indicates zero current level. (B) Summary plot of effects of various phosphatase inhibitors on TRPV2 currents. (C) Immunoblotting analysis with anti-phosphotyrosine antibody exhibiting tyrosine phosphorylation of immunoprecipitated TRPV2-Flag in HEK293T cells under control conditions and after treatment with PTP inhibitor 1 or PTP inhibitor 2. (D) Schematic diagram showing increased TRPV2 tyrosine-phosphorylation levels caused by phosphokinase JAK1 or inhibition of PTP activity. (E) Representative whole-cell currents evoked by increasing concentrations of 2-APB for rat bone marrow-derived macrophages (rBMDMs). The cells were pretreated with PTP inhibitor 1 (top) and PTP inhibitor 2 (bottom). (F) Dose-response curves of 2-APB. Fitting by Hill’s equation resulted in the following: EC50 = 0.55 ± 0.01 mM and nH = 3.9 ± 0.2 for control (n = 6); EC50 = 0.18 ± 0.01 mM and nH = 3.4 ± 0.1 for treatment by PTP inhibitor 1 (n = 6) and EC50 = 0.09 ± 0.01 mM and nH = 3.3 ± 0.3 for treatment by PTP inhibitor 2 (n = 7). (G) Concentration-response curves of 2-APB in TRPV2-Y335/471/525F-expressing HEK293T cells under treatment by DMSO, PTP inhibitor 1 or PTP inhibitor 2. Fitting by Hill’s equation resulted in the following: EC50 = 0.36 ± 0.01 mM and nH = 3.8 ± 0.1 for control (n = 5); EC50 = 0.34 ± 0.01 mM and nH = 3.1 ± 0.1 for treatment by PTP inhibitor 1 (n = 6) and EC50 = 0.26 ± 0.01 mM and nH = 3.8 ± 0.7 for treatment by PTP inhibitor 2 (n = 6). (H–J) Representative current traces, temperature-activation relations, and plot of log(Itest/I40oC+1) determinations for DMSO pretreated rBMDMs. (K–M) Representative current traces, temperature-activation relations, and plot of log(Itest/I40oC+1) determinations for PTP inhibitor 1 pretreated rBMDMs. (N–P) Representative current traces, temperature-activation relations, and plot of log(Itest/I40oC+1) determinations for PTP inhibitor 2 pretreated rBMDMs.
-
Figure 5—source data 1
Uncropped, unedited blots for Figure 5C.
- https://cdn.elifesciences.org/articles/78301/elife-78301-fig5-data1-v2.zip
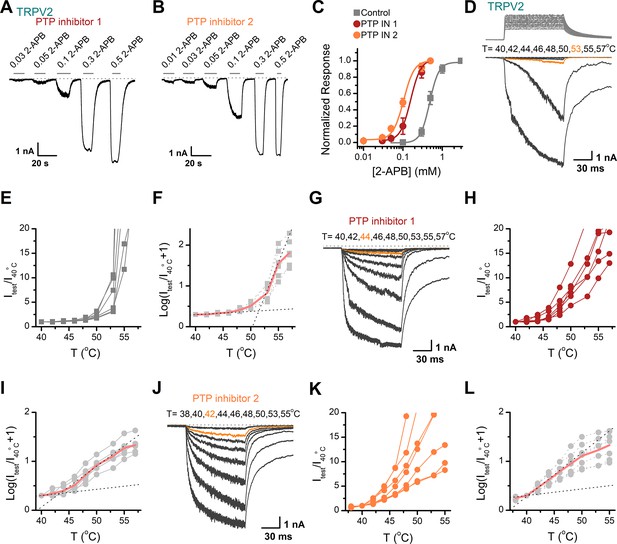
Inhibition of protein tyrosine phosphatase (PTP) activity by inhibitors enhanced the transient receptor potential vanilloid 2 (TRPV2) sensitivity to 2-aminoethyl diphenylborinate (2-APB) and heat in TRPV2-expressing HEK293T cells.
(A–B) Representative whole-cell recordings from TRPV2-expressing HEK293T cells pretreated with PTP inhibitor 1 (A) and PTP inhibitor 2 (B). The cells were exposed to increasing concentrations of 2-APB and the holding potential was –60 mV. The dotted line indicates zero current level. (C) Concentration-response curves of 2-APB. Solid lines indicate fits by a Hill’s equation, with EC50 = 0.48 ± 0.02 mM and nH = 3.7 ± 0.4 for control (n = 7); EC50 = 0.16 ± 0.01 mM and nH = 3.4 ± 0.2 for the treatment by PTP inhibitor 1 (n = 6), and EC50 = 0.10 ± 0.01 mM and nH = 3.2 ± 0.3 for the treatment by PTP inhibitor 2 (n = 7). (D–F) Representative current traces, temperature-activation relations, and plot of log(Itest/I40oC+1) determinations for DMSO pretreated TRPV2-expressing HEK293T cells. (G–I) Representative current traces, temperature-activation relations, and plot of log(Itest/I40oC+1) determinations for PTP inhibitor 1 pretreated TRPV2-expressing HEK293T cells. (J–L) Representative current traces, temperature-activation relations, and plot of log(Itest/I40oC+1) determinations for PTP inhibitor 2 pretreated TRPV2-expressing HEK293T cells. Error bars indicate standard error of the mean (SEM).
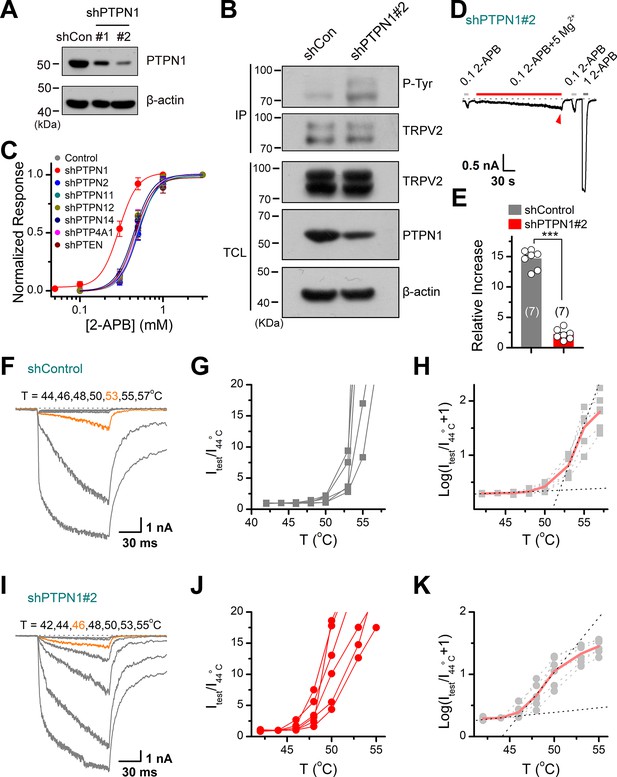
Protein tyrosine phosphatase non-receptor type 1 (PTPN1) is a phosphatase that mediates the dephosphorylation of transient receptor potential vanilloid 2 (TRPV2).
(A) Immunoblot analysis (with anti-PTPN1 or anti-β-action) of HEK293T cells transfected for 48 hr with PTPN1-targeting shRNA (shPTPN1#1 and shPTPN1#2) or shControl to test knockdown efficiency of shRNA. (B) Immunoblot analysis of the tyrosine phosphorylation level of TRPV2 in HEK293T cells transfected with shControl or shPTPN1#2 for 48 hr. (C) Concentration-response curves of 2-aminoethyl diphenylborinate (2-APB). Whole-cell recordings were performed in HEK293T transfected with various protein tyrosine phosphatase-targeting shRNA. (D) Whole-cell recordings in TRPV2-expressing HEK293T cells that were transfected for 48 hr with shPTPN1#2 showing the response to 0.1 mM 2-APB, 0.1 mM 2-APB plus 5 mM Mg2+ and 1 mM 2-APB. The dotted line indicates zero current level. (E) Comparison of relative changes under different conditions. p = 3.88E-10 by unpaired Student’s t-test. (F–H) Representative current traces, temperature-activation relations, and plot of log(Itest/I44oC+1) determinations for TRPV2-expressing HEK293T cells which were transfected for 48 hr with shControl. (I–K) Representative current traces, temperature-activation relations, and plot of log(Itest/I44oC + 1) determinations for TRPV2-expressing HEK293T cells which were transfected for 48 hr with shPTPN1#2.
-
Figure 6—source data 1
Uncropped, unedited blots for Figure 6A.
- https://cdn.elifesciences.org/articles/78301/elife-78301-fig6-data1-v2.zip
-
Figure 6—source data 2
Uncropped, unedited blots for Figure 6B.
- https://cdn.elifesciences.org/articles/78301/elife-78301-fig6-data2-v2.zip
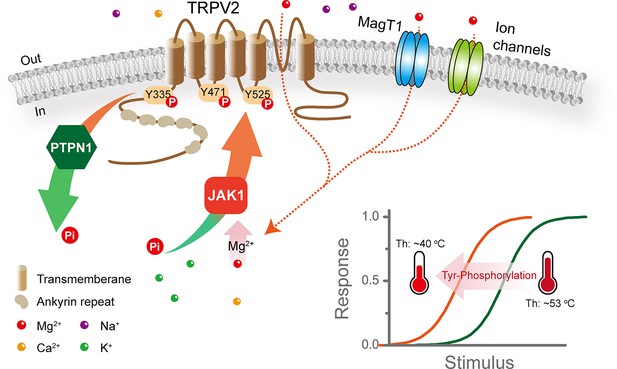
Tyrosine phosphorylation sets the agonist and heat sensitivity of transient receptor potential vanilloid 2 (TRPV2).
This study demonstrates that JAK1 phosphokinase mediates Mg2+-dependent phosphorylation of TRPV2 at Y335, Y471, and Y525 residues. And, increasing tyrosine phosphorylation of TRPV2 lowers its thermal activation threshold and enhances its sensitivity to agonistic stimuli. Furthermore, protein tyrosine phosphatase non-receptor type 1 (PTPN1) is the tyrosine phosphatase that mediates the dephosphorylation of the TRPV2 channel.
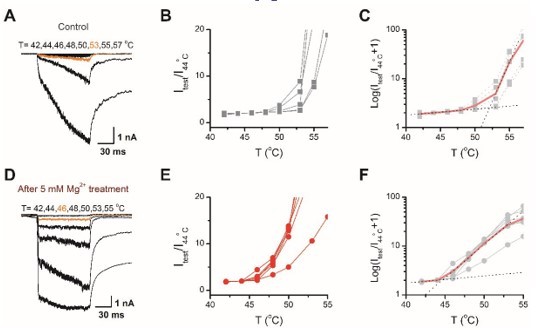
Determination of the temperature threshold for activation of TRPV2.
(A) Representative current traces for TPRV2-expressing HEK293T cells in response to a family of temperature pulses ranging from 42 oC to 57 oC. (B) Current vs. temperature relations at −60 mV obtained from experiments as in (A). Individual cells are shown with currents normalized by their amplitude at 44 oC. (C) Plot of log(Itest/I44oC+1) obtained from the relations in (A). (D-F) Representative current traces, temperature-activation relations, and Plot of log(Itest/I44oC+1) determinations for Mg2+ pretreated TRPV2expressing cells.
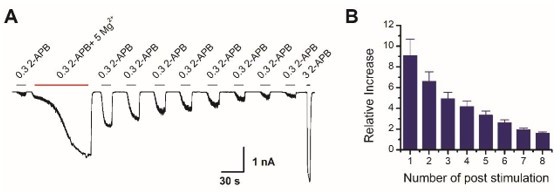
Mg2+-induced sensitization of TRPV2 is reversible.
(A) Representative whole-cell of TRPV2 currents evoked by 0.3 mM 2-APB, 0.3 mM 2-APB plus 5 mM Mg2+, subsequently repeated applications of 0.3 mM 2-APB and 3 mM 2-APB. (B) Time courses of peak currents elicited by repeated applications of 0.3 mM 2-APB after being sensitized by Mg2+. Currents were normalized to that evoked by 0.3 mM 2-APB before Mg2+ treatment.
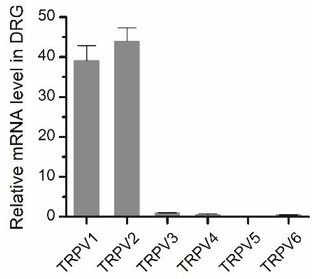
Expression of TRPV2 in DRG neurons.
Relative mRNA expression levels of different Trpv channels in BMDMs were assessed by qPCR.
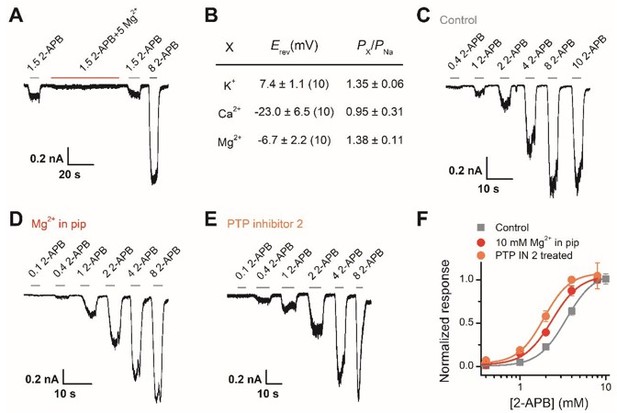
Effect of Mg2+-dependent phosphorylation on human TRPV2.
(A) Whole-cell currents at -60 mV in a hTRPV2-expressing HEK293T cell treated with 1.5 mM 2-APB, 1.5 mM 2-APB plus 5 mM Mg2+, and 3 mM 2-APB. (B) Relative cationic permeabilities (PX/PNa) of human TRPV2 channels assessed by reversal potentials. (C-E) Representative whole-cell currents evoked by increasing concentrations of 2-APB for human TRPV2-expressing HEK293T cells. The cells were under control contions (A), with addition of 10 mM Mg2+ in the pipette solution (B), and pre-treated with PTP inhibitor 2 (C). (F) Dose-response curves of 2-APB. Fitting by Hill’s equation resulted in the following: EC50 = 3.4 ± 0.1 mM and nH = 2.7 ± 0.2 for control (n = 7); EC50 = 2.4 ± 0.2 mM and nH = 2.7 ± 0.5 for addition of Mg2+ (n = 6) and EC50 = 1.9 ± 0.1 mM and nH = 2.9 ± 0.5 for treatment by PTP inhibitor 2 (n = 6).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Antibody | Anti-Phosphotyrosine antibody (Rabbit monoclonal) | Abcam | Cat#ab179530; RRID: AB_828379 | WB (1:1000) |
| Antibody | Anti-Phospho-(Ser/Thr)Phe antibody (Rabbit polyclonal) | Abcam | Cat#ab17464; RRID: AB_443891 | WB (1:1000) |
| Antibody | Anti-Flag antibody (Rabbit polyclonal) | Proteintech | Cat#20543–1-AP; RRID: AB_11232216 | WB (1:3000) |
| Antibody | Anti-TRPV2 antibody (Rabbit polyclonal) | Alomone Labs | Cat#ACC-032; RRID: AB_2040266 | WB (1:500), IP (1:200) |
| Antibody | Anti-mouse IgG (H+L) (Goat polyclonal) | Jackson Immunoresearch | Cat#115-035-003; RRID: AB_10015289 | (5 μg) |
| Antibody | Anti-rabbit IgG (H+L) (Goat polyclonal) | Jackson Immunoresearch | Cat#111-005-003; RRID: AB_2337913 | (5 μg) |
| Antibody | Anti-JAK1 antibody (Rabbit monoclonal) | Abcam | Cat#ab133666 | WB (1:1000) |
| Antibody | Anti-PTPN1 antibody (Rabbit monoclonal) | Abcam | Cat#ab244207; RRID: AB_2877148 | WB (1:1000) |
| Antibody | Anti-Flag Affinity Gel (Mouse monoclonal) | Bimake | Cat#B23102; RRID: AB_2728745 | (15 μl) |
| Other | ProteinIso Protein G Resin | TransGen | Cat#DP401 | (30 μl) |
| Chemical compound, drug | 2-APB | Sigma-Aldrich | Cat#D9754, CAS: 524-95-8 | TRPV2 agonist |
| Chemical compound, drug | MgCl2·6H2O | Sigma-Aldrich | Cat#M2393, CAS: 7791-18-6 | |
| Chemical compound, drug | Na2-ATP | Sigma-Aldrich | Cat#A2383; CAS: 34369-07-8 | |
| Chemical compound, drug | EDTA | Biosharp | Cat#BS107; CAS: 60-00-4 | |
| Chemical compound, drug | AMP-PNP | Sigma-Aldrich | Cat#A2647; CAS: 25612-73-1 | |
| Chemical compound, drug | MK-2206 | TargetMol | Cat#T1952; CAS: 1032350-13-2 | Akt inhibitor |
| Chemical compound, drug | Staurosporine | TargetMol | Cat#T6680; CAS: 62996-74-1 | PKC inhibitor |
| Chemical compound, drug | KN-93 Phosphate | TargetMol | Cat#T2606; CAS: 1188890-41-6 | CaMKII inhibitor |
| Chemical compound, drug | D4476 | TargetMol | Cat#T2449; CAS: 301836-43-1 | CK1 inhibitor |
| Chemical compound, drug | U0126-EtOH | TargetMol | Cat#T6223; CAS: 1173097-76-1 | MEK1/2 inhibitor |
| Chemical compound, drug | Ruxolitinib | TargetMol | Cat#T1829; CAS: 941678-49-5 | JAK1 inhibitor |
| Chemical compound, drug | Salubrinal | TargetMol | Cat#T3045; CAS: 405060-95-9 | PP1 inhibitor |
| Chemical compound, drug | LB-100 | MCE | Cat#HY-18597; CAS: 1632032-53-1 | PP2A inhibitor |
| Chemical compound, drug | Cyclosproin A | TargetMol | Cat#T0945; CAS: 59865-13-3 | PP2B inhibitor |
| Chemical compound, drug | Cantharidin | Aladdin | Cat#c111020; CAS: 56-25-7 | PP1 and PP2A inhibitors |
| Chemical compound, drug | CCT007093 | TargetMol | Cat#T1927; CAS:176957-55-4 | PPM1D inhibitor |
| Chemical compound, drug | PTP inhibitor 1 | TargetMol | Cat#T7084; CAS: 2491-38-5 | PTPs inhibitor |
| Chemical compound, drug | PTP inhibitor 2 | TargetMol | Cat#T7541; CAS: 2632-13-5 | PTPs inhibitor |
| Cell lines (species) | Human embryo kidney (HEK) 293T (human) | ATCC | Cat#CRL-3216; RRID: CVCL_0063 | |
| Software, algorithms | QStudio | Developed by Dr Feng Qin from University of New York at Buffalo | ||
| Software, algorithms | Micro-Manager 1.4 | Vale Lab, UCSF | ||
| Software, algorithms | Clampfit | Molecular Devices, Sunnyvale, CA | ||
| Software, algorithms | IGOR | Wavemetrics, Lake Oswego, OR | ||
| Software, algorithms | SigmaPlot | SPSS Science, Chicago, IL | ||
| Software, algorithms | OriginPro | OriginLab Corporation, Northampton, MA | ||
| Software, algorithms | ImageJ | Schneider et al., 2012 |