Lhx2 is a progenitor-intrinsic modulator of Sonic Hedgehog signaling during early retinal neurogenesis
Figures
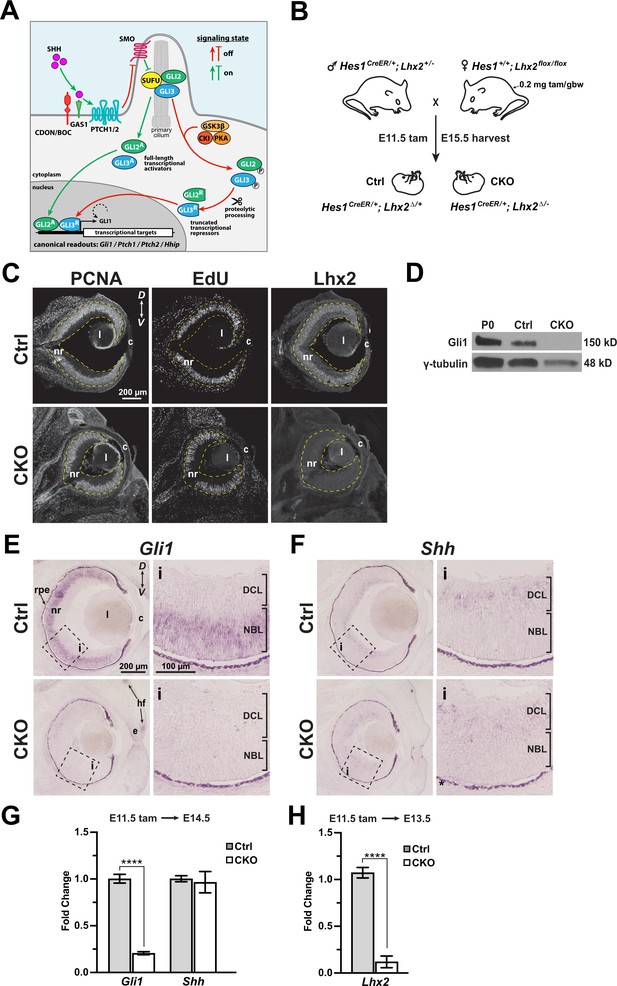
Lhx2 is required for Gli1 expression in RPCs.
(A) Overview of Hh signaling. See text for details and Briscoe and Thérond, 2013; Kong et al., 2019; Ramsbottom and Pownall, 2016 for more comprehensive pathway illustrations and descriptions. (B) Genetics and tamoxifen treatment paradigm. Female breeders are also homozygous for Rosaai14/ai14 and all embryos are Rosaai14/+, allowing rapid screening for recombined embryos with tdTomato expression. Recombined flox alleles are indicated by Δ. (C) Immunohistology for markers of RPC proliferation (PCNA, EdU) and Lhx2 expression in the E15.5 control and CKO eyes after tamoxifen treatment at E11.5. The retina is contained within the yellow dashed lines. (D) Western blot for Gli1 protein expression in P0 wild type, E15.5 Ctrl and CKO retinas following tamoxifen treatment at E11.5. γ-Tubulin served as an internal loading control. (E, F) in situ hybridizations for Gli1 expression in E15.5 Ctrl eyes following tamoxifen treatment at E11.5. (F) In situ hybridizations for Shh expression in E15.5 Ctrl and CKO eyes following tamoxifen treatment at E11.5. Dashed boxes reveal locations of close-up images (i). Dark appearance of the rpe is due to natural pigmentation and does not indicate gene expression. (G) qPCR for Gli1 and Shh expression in Ctrl and CKO retinas at E14.5 following tamoxifen at E11.5 (mean+/-S.E.M.; n=4 (Ctrl: Gli1); n=6 (CKO: Gli1); n=6 (Ctrl: Shh); n=8 (CKO: Shh); ****, padj=5x10-6; unpaired t-tests with multiple comparisons correction; see Supplementary file 3 for statistics). (H) qPCR for Lhx2 expression in Ctrl and CKO retinas at E13.5 following tamoxifen at E11.5 (mean+/-S.E.M.; n=4 (Ctrl); n=4 (CKO); ****, p<0.0001; unpaired t-test; see Supplementary file 3 for statistics). Abbreviations: D, dorsal; V, ventral; nr, neural retina; rpe, retinal pigment epithelium; l, lens; c, cornea; e, eyelid; hf, hair follicle; DCL, differentiated cell layer; NBL, neuroblast layer.
-
Figure 1—source data 1
Western blot probed with Gli1 antibody and reprobed with gamma-Tubulin antibody.
Filenames containing the word ‘unedited’ show multiple exposures of the same blot and antibody. Filenames containing the word ‘labeled’ show the exposure used in Figure 1 as indicated by the black boxes. The red boxes indicate the portions of the blots shown in Figure 1. The sizes of the protein standards are shown in kiloDaltons (kDa).
- https://cdn.elifesciences.org/articles/78342/elife-78342-fig1-data1-v1.zip
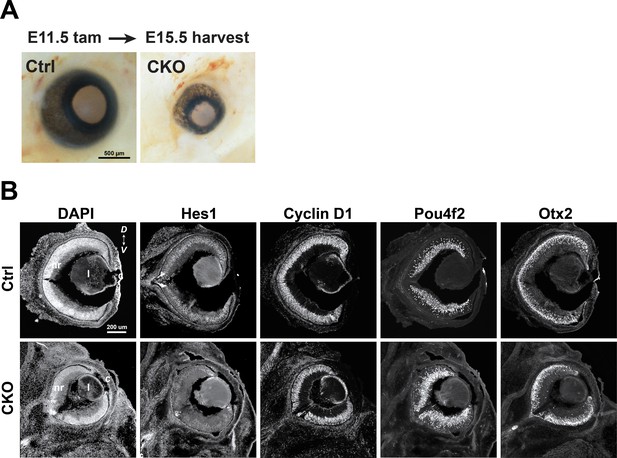
Immunohistology for Lhx2 CKO retina at E15.5 following tamoxifen treatment at E11.5.
(A) Relative eye sizes from control (left) and CKO (right) embryos. (B) Comparative immunohistology for control (top row) and CKO eyes (bottom row) showing tissue organization with DAPI, the RPC marker and progenitor maintenance factor Hes1, the RPC marker Cyclin D1, the RGC marker Pou4f2, and the photoreceptor precursor marker Otx2. Although the retina is noticeably smaller in the CKO compared to control, only Hes1 shows a qualitative change in expression at E15.5, 4 days after tamoxifen treatment.
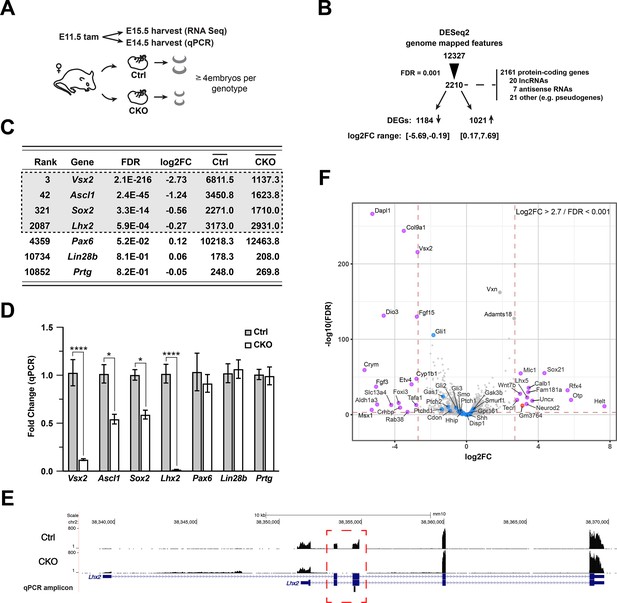
Gene expression changes due to Lhx2 inactivation.
(A) Schematic of experimental design for RNA sequencing and qPCR. (B) Summary of DESeq2 analysis of RNA sequencing datasets. (C) DESeq2-derived statistics for progenitor genes with requirements during early retinal neurogenesis. Genes in gray box were within the 0.001 FDR cutoff for differential expression. Averaged counts per gene are shown in the last two columns. (D) Relative expression for progenitor genes at E14.5 by qPCR as a function of the fold change from the mean of control for each gene. Only significant comparisons are noted (mean+/-S.E.M.; *, padj<0.05; ****, padj <0.0001; n=4 (Ctrl: Vsx2, Ascl1, Lhx2); n=3 (Ctrl: Sox2, Pax6), n=6 (Ctrl: Lin28b, Prtg); n=6 (CKO: Vsx2, Ascl1, Lhx2); n=4 (CKO: Sox2, Pax6); n=8 (CKO: Lin28b, Prtg); unpaired t-tests with multiple comparisons correction; see Supplementary file 3 for statistics). (E) Coverage plot showing the mutant transcript is expressed and detected by RNA sequencing, indicating that nonsense mediated decay of mutant transcript is not occurring. The lack of reads in exons 2 and 3 (red box) of the CKO reveal the high degree of conditional deletion. Plots are the mean of counts for all samples. The qPCR amplicon located in the deleted exon 3, making the mutant transcript undetectable by qPCR with the Taqman probe used in the study. (F) Volcano plot showing selected Shh pathway genes (blue dots) relative to other DEGs. The most divergent DEGs gated on FDR (0.001) and absolute log2FC (2.7) cutoffs (red dashed lines) are highlighted (purple: mRNAs; red: lncRNA; gray: filtered out GMFs).
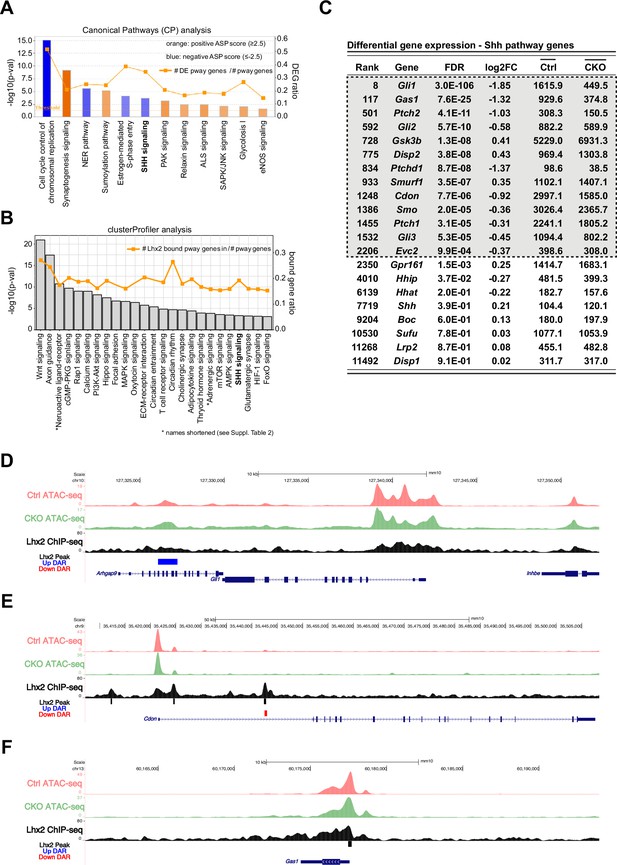
Lhx2 is required for the expression of multiple Hedgehog pathway genes.
(A) Canonical Pathways (CP) analysis for DEGs with an FDR of 0.001 or smaller. Pathways that surpassed the significance cutoff of 1.3 (x-axis) and have an absolute ASP score of 2.5 or higher are shown. Orange bars predict pathway activation and blue bars predict pathway inhibition. Color intensity is directly correlated to ASP score (scores are listed in Supplementary file 2). The line indicates the number of DEGs found in each pathway as a fraction of pathway genes (DEG ratio; right y-axis). (B) KEGG pathways associated with Lhx2 ChIP-seq peak distribution from E14.5 mouse RPCs using ClusterProfiler. The line indicates the number of genes associated with Lhx2 chromatin binding in each pathway as a fraction of pathway genes (bound gene ratio; right y-axis). (C) Differential gene expression values for canonical genes in the Shh pathway. Genes in the gray box passed the cutoff for DEG designation. (D–F) ATAC-seq and Lhx2 ChIP-seq genomic DNA tracks at Gli1 (D), Cdon (E), and Gas1 (F) loci from E14.5 RPCs. DARs identified in Lhx2 CKO RPCs are indicated by red and blue bars. Sites of Lhx2 chromatin binding are indicated by black bars from the ChIP-seq data.
ATAC-seq and Lhx2 ChIP-seq tracks for selected Hh pathway genes within the FDR cutoff for differential expression (0.001).
ATAC-seq and Lhx2 ChIP-seq tracks from E14.5 RPCs. Differentially accessible chromatin regions (DARs) in Lhx2 CKO RPCs compared to control are indicated by red (open) and blue bars (closed). Lhx2 ChIP-seq peak calls are indicated by grey bars.

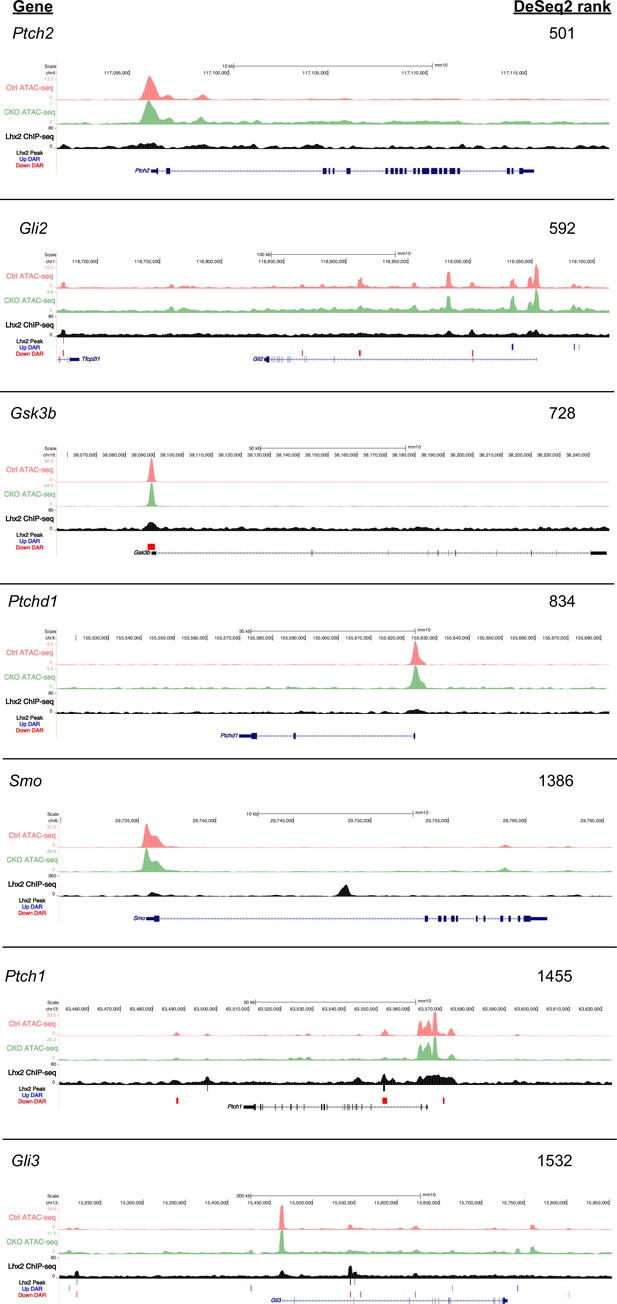
ATAC-seq and Lhx2 ChIP-seq tracks for selected Hh pathway genes outside the FDR cutoff for differential expression (0.001).
ATAC-seq and Lhx2 ChIP-seq tracks from E14.5 RPCs. Differentially accessible chromatin regions (DARs) in Lhx2 CKO RPCs compared to control are indicated by red (open) and blue bars (closed). Lhx2 ChIP-seq peak calls are indicated by grey bars.
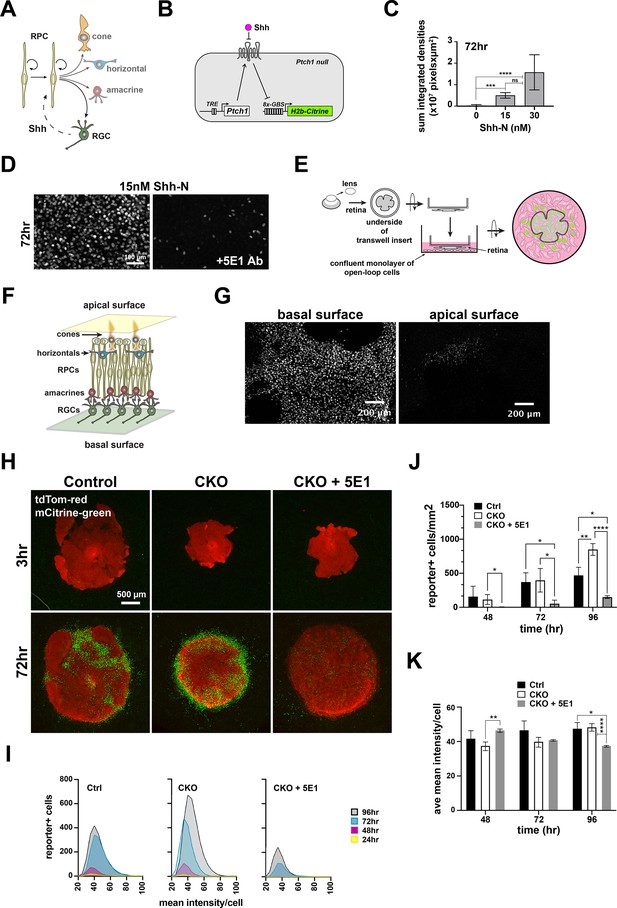
Evaluation of a live cell reporter system to test Shh bioavailability.
(A) Intra-lineage architecture of Hh signaling at the start of retinal neurogenesis. RPCs initiate neurogenesis and begin generating RGCs, the Shh producing cells. RPCs are the responder cells, placing Shh upstream and downstream of RPCs. (B) Configuration of the open-loop circuit in the responder NIH3T3 Ptch1-null cell line (open-loop cells). Ptch1 is produced from a doxycycline regulated transgene. Shh binds Ptch1, activating intrinsic signaling as well as Gli-dependent expression of mCitrine fused to Histone H2b through 8-multimerized Gli1 binding sites. (C) Dose response at 72 hr as a function of the total accumulation of signal intensities and area coverage of mCitrine + open loop cells (mean+/-S.D.; n=4 per condition; ns, not significant; ***, padj <0.001; ****, padj <0.0001; ANOVA followed by Tukey’s multiple comparisons; see Supplementary file 4 for statistics). (D) Addition of the Shh ligand-blocking monoclonal antibody, 5E1, diminishes mCitrine expression. (E) Design of coculture experiment. Freshly dissected embryonic retina is flat mounted to the underside of a transwell insert and placed into direct contact with a confluent monolayer of open-loop cells. mCitrine expression accumulates in open-loop cells that receive Shh from the retina. (F) Schematic cross-section of embryonic retina shows that the apical surface is comprised mainly of RPCs and developing photoreceptors (cones until ~E15.5, and a mix of rods and cones thereafter) and the basal surface is comprised of RGCs, the source of retinal SHH. Astrocytes and endothelial also reside on the basal surface (not shown). (G) E18.5 wild type retinas were cultured in opposite orientations such that the open-loop cells contacted the basal or apical surfaces of the retina. mCitrine expression was robustly induced in responder cells in close proximity to the basal surface but not the apical surface of the retina. (H) Cocultures for control (left), CKO (middle), and CKO incubated with 5E1 antibody (right) at 3 and 72 hr. Retinal tissues are tdTomato positive (red) and mCitrine positive nuclei are green. (I) Lowess-smoothed histograms showing the accumulation and fluorescence intensity distributions of mCitrine + responder cells at each timepoint during the co-culture period. The histograms are for the cocultures shown in A. (J) Quantification of mCitrine + cells at 48, 72, and 96 hr. Comparisons were done within timepoints only and the significant differences are shown (mean+/-S.D.; n=4 (Ctrl, all timepoints); n=6 (CKO, all timepoints); n=2 (CKO+5E1, all timepoints); *, padj <0.05; **, padj <0.01; ****, padj <0.0001; 2-way repeated measures ANOVA followed by Tukey’s multiple comparisons test; see Supplementary file 4 for statistics). (K) Quantification of the average mCitrine fluorescence intensities per cell at 48, 72, and 96 hr. Comparisons were done within timepoints only and the significant differences are shown (mean+/-S.D.; n=4 (Ctrl, all timepoints); n=6 (CKO, all timepoints); n=2 (CKO, all timepoints); *, padj <0.05; **, padj <0.01; ****, padj <0.0001; 2-way repeated measures ANOVA followed by Tukey’s multiple comparisons test; see Supplementary file 4 for statistics).
Responder cells express mCitrine near the basal surface of the retina.
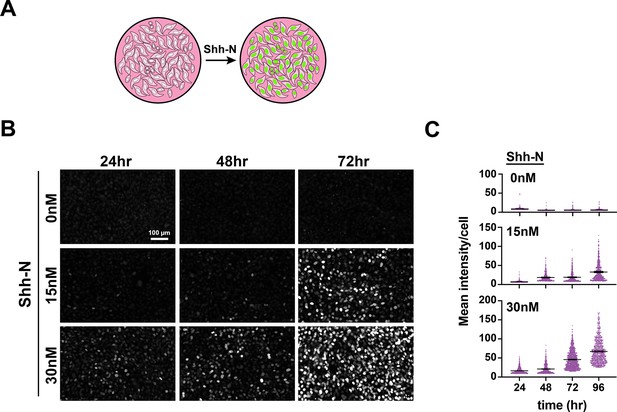
(A) Schematic showing that addition of recombinant Shh-N to a confluent monolayer of responder cells activates nuclear mCitrine expression. (B) Dose response and time course for accumulation of mCitrine expression in open-loop responder cells. Fields were randomly selected from the same cultures for each timepoint. (C) Scatter plots showing the accumulation of mCitrine + cells over time for each dose tested as a function of average mean fluorescence intensity per cell. Means are shown for each condition (lines).
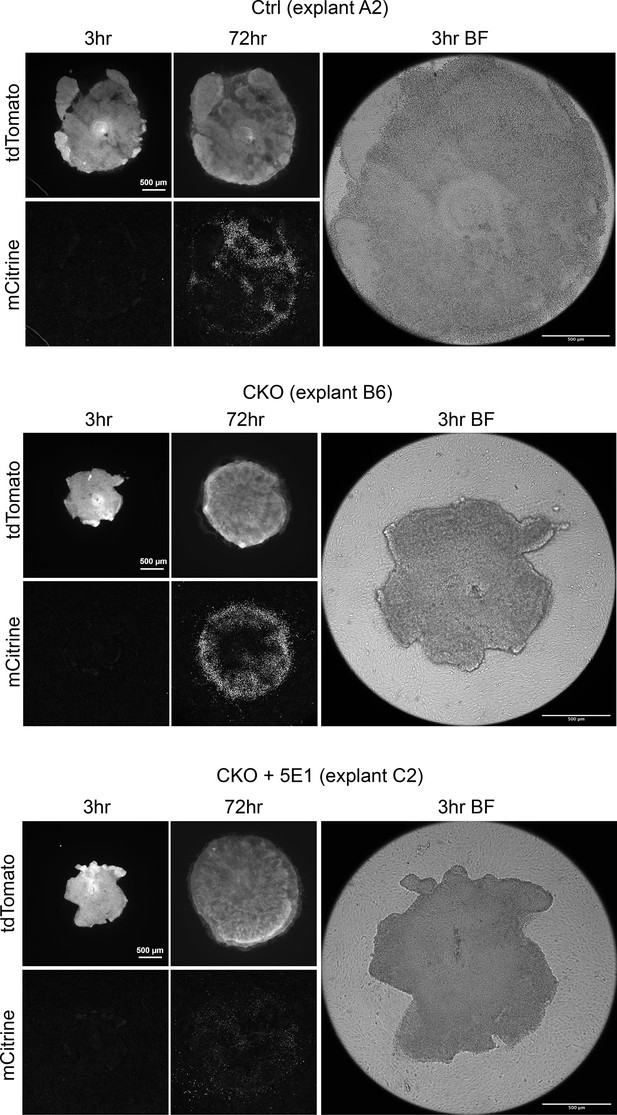
Single channel images Single channel fluorescence and bright field images for the cocultures shown in Figure 5B.
.
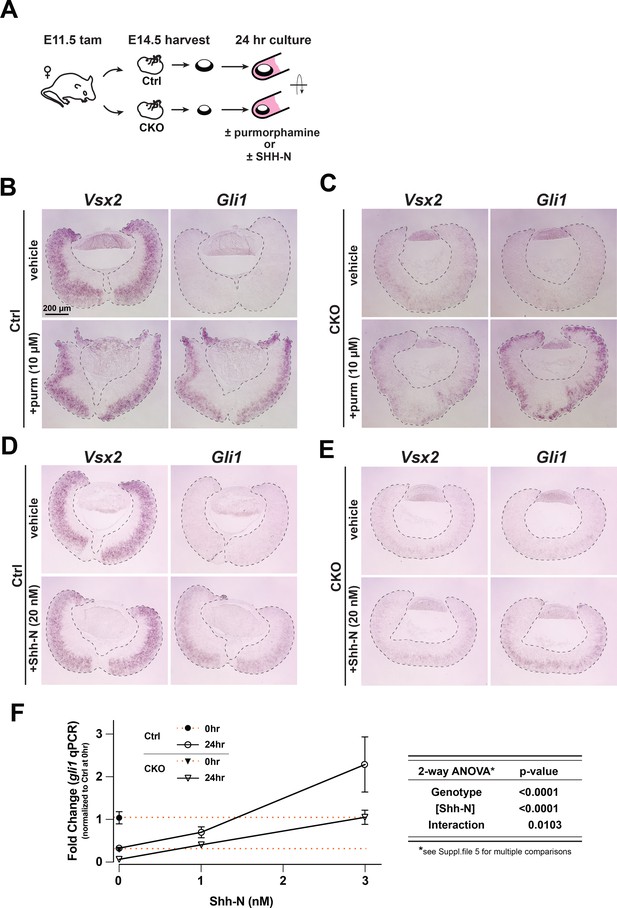
Purmorphamine and recombinant Shh-N stimulate signaling in Lhx2 CKO retinal explants.
(A) Experimental design of ex vivo retina-lens explant cultures. (B–E) in situ hybridizations of Vsx2 and Gli1 expression after 24 hr in culture to test Smo agonist purmorphamine or recombinant Shh-N. (B) Gli1 expression declines in the absence of purmorphamine (vehicle) whereas Vsx2 is maintained. Gli1 expression is restored with purmorphamine. (C) Vsx2 declines due to Lhx2 inactivation. Gli1 is expressed in response to purmorphamine. (D) Gli1 expression declines in the absence of Shh-N (vehicle) whereas Vsx2 is maintained. Gli1 is restored with Shh-N. (E) Vsx2 declines due to Lhx2 inactivation. Similar to purmorphamine, Gli1 expression is upregulated with Shh-N. (F) qPCR-based Gli1 expression in control and CKO retinal explants at the start of the culture (t=0) and after 24 hr at different concentrations of Shh-N, as determined from a pilot dose response with wild type retinal explants (Figure 5—figure supplement 1C, Supplementary file 5). Expression values are relative to the mean control value at t=0 (closed circle). Orange lines extend from t=0 values for the control (upper line) and the CKO (closed triangle, lower line). Note that the value for the 24 hr control in 0 nM Shh-N overlaps with the CKO at t=0. To the right of the graph is the summary table for two-way ANOVA showing that the main effects (genotype and Shh-N concentration) are significant and interact. mean+/-S.E.M.; n=4 all conditions except CKO, 1nM Shh-N (n=3); Two-way ANOVA followed by Tukey’s multiple comparisons test; See Supplementary file 5 for statistics including padj values for multiple comparisons.
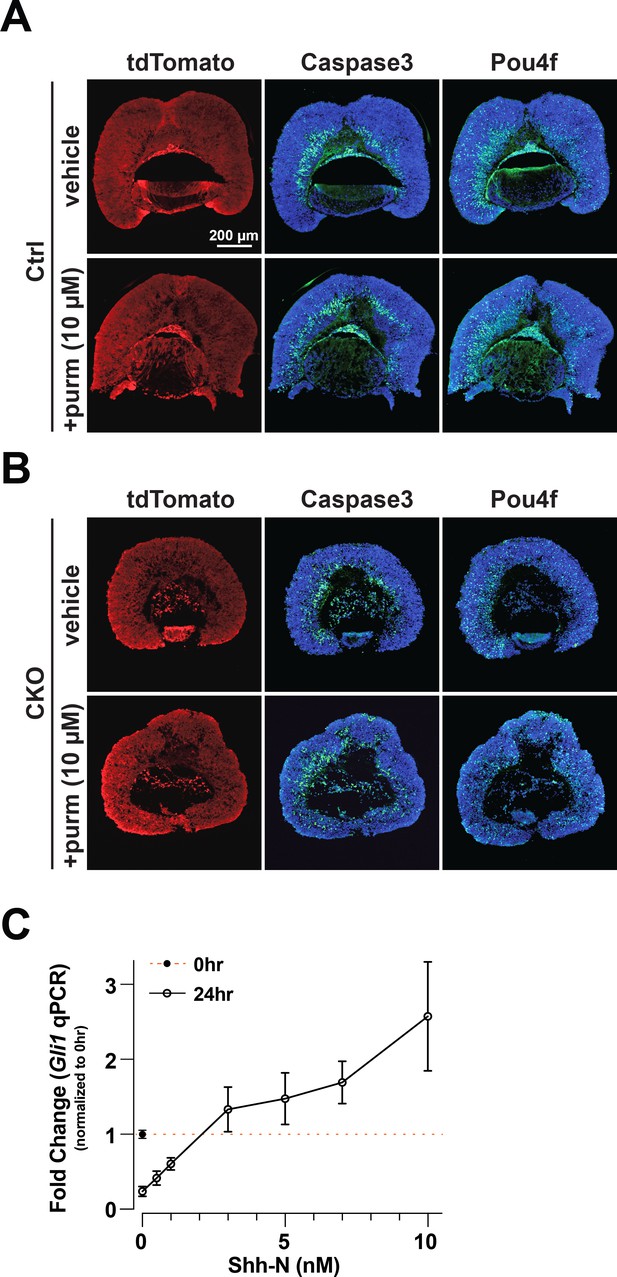
Markers of recombination, apoptosis, and RGCs after 24 hr, and dose response to estimate physiological range for recombinant Shh-N(C24II) protein.
(A) Control and (B) CKO explants treated with vehicle or 10 nM purmorphamine for 24 hr. Left column: tdTomato expression from heterozygous ai14 allele reveals CreER activity throughout the explants. Middle column: Caspase 3 immunofluorescence (green) reveals extent of apoptotic cells. Nuclei are stained with DAPI (blue). Right column: Pou4f2 immunofluorescence (green) marks RGCs. (C) Relative Gli1 expression in wild type E15.5 retinas treated with increasing concentrations of recombinant Shh-N. Data points are relative to the mean expression value from retinas harvested at the start of the experiment (t=0; orange line). Means and standard deviation are shown, and results are from a pilot experiment (n=3 for all conditions; One way ANOVA followed by Dunnett’s multiple comparisons test;see Supplementary file 5 for statistics including padj values for multiple comparisons).
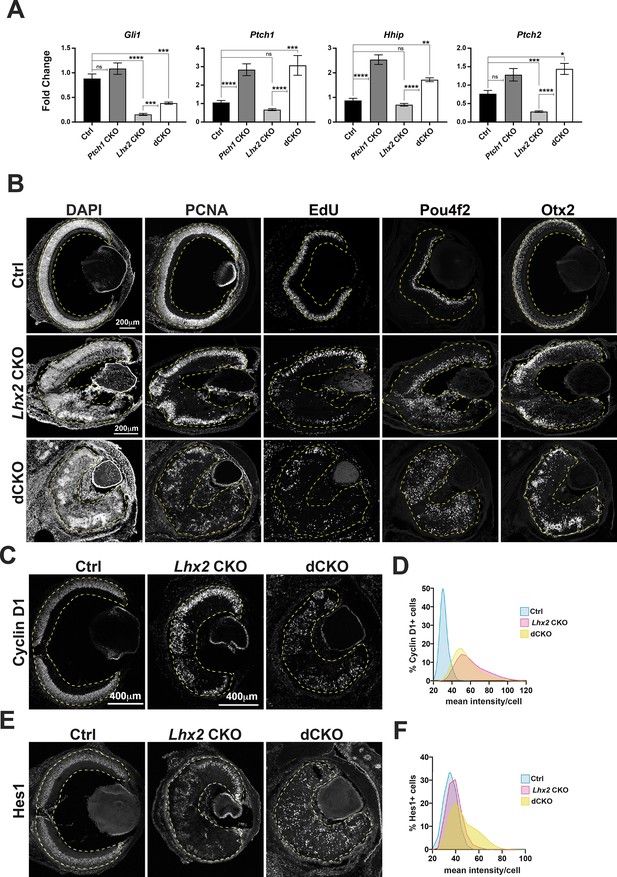
Hh signaling is enhanced in Lhx2-deficient RPCs by Ptch1 inactivation in vivo.
(A) Relative expression of Gli1, Ptch1, Hhip, and Ptch2 at E15.5 following tamoxifen treatment at E11.5 in retinas with the following genotypes: control, Ptch1 CKO, Lhx2 CKO and Lhx2; Ptch1 double CKO (dCKO). See Figure 6—figure supplement 1A for breeding scheme and genotypes assigned to control. For each gene, fold change values are relative to a specific control sample set as a reference. Shown are the comparisons for the three mutant genotypes compared to control and for the dCKO compared to the Lhx2 CKO (means+/-S.E.M.; n=6 (Ctrl, all genes); n=9 (Ptch1 CKO, all genes); n=7 (Lhx2 CKO, all genes); n=4 (dCKO, all genes); ns, not significant; * padj <0.05; ** padj <0.01; *** padj <0.001; **** padj <0.0001; ANOVA followed by Tukey’s multiple comparisons test; see Supplementary file 6 for statistics and complete multiple comparisons list). (B) DAPI staining and expression patterns for PCNA and EdU incorporation to identify RPCs and Pou4f2 and Otx2 to identify nascent RGCs and photoreceptors at E18.5 following tamoxifen treatment at E11.5 in retinas from control (top row), Lhx2 CKO (middle row) and dCKO (bottom row). (C) Cyclin D1 expression at E18.5 following tamoxifen treatment at E11.5 in control, Lhx2 CKO and dCKO retinas. (D) Lowess-smoothed histograms showing the distribution of Cyclin D1 + cells as a function of the mean fluorescence intensity per cell. Each histogram is normalized to the number of Cyclin D1 + cells within the respective genotype. (E) Hes1 expression at E18.5 following tamoxifen treatment at E11.5 in control, Lhx2 CKO and dCKO retinas. (F) Lowess-smoothed histograms showing the distribution of Hes1 + cells as a function of the mean fluorescence intensity per cell. Each histogram is normalized to the number of Hes1 + cells within the respective genotype.
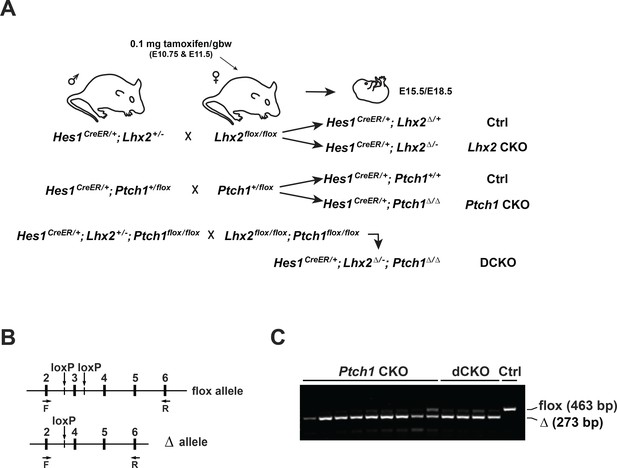
Genetics of Ptch1 and Lhx2 inactivation and validation of Ptch1 recombination.
(A) Overview of genetics and tamoxifen treatment. (B) Schematic of Ptch1 locus encompassing the regions containing exons 2–6. Exon 2 forward (ex2F) and Exon 6 reverse (ex6R) were used for RT-PCR to detect unrecombined and recombined transcripts from the flox and CKO alleles, respectively. (C) Agarose gel shows most samples were recombined (CKO) although some unrecombined (flox) was detected. All samples were used for qPCR.
-
Figure 6—figure supplement 1—source data 1
RT-PCR for non-deleted (flox) and deleted (Δ) Ptch1 mRNAs.
File with the word ‘unedited’ shows the original gel capture. File with the word ‘labeled’ shows the fragment sizes of the DNA standards in base pairs (bp). The red box indicates the portion of the gel shown in Figure 6—figure supplement 1. The sizes of the non-recombined and deleted Ptch1 mRNAs are shown in base pairs (bp).
- https://cdn.elifesciences.org/articles/78342/elife-78342-fig6-figsupp1-data1-v1.zip
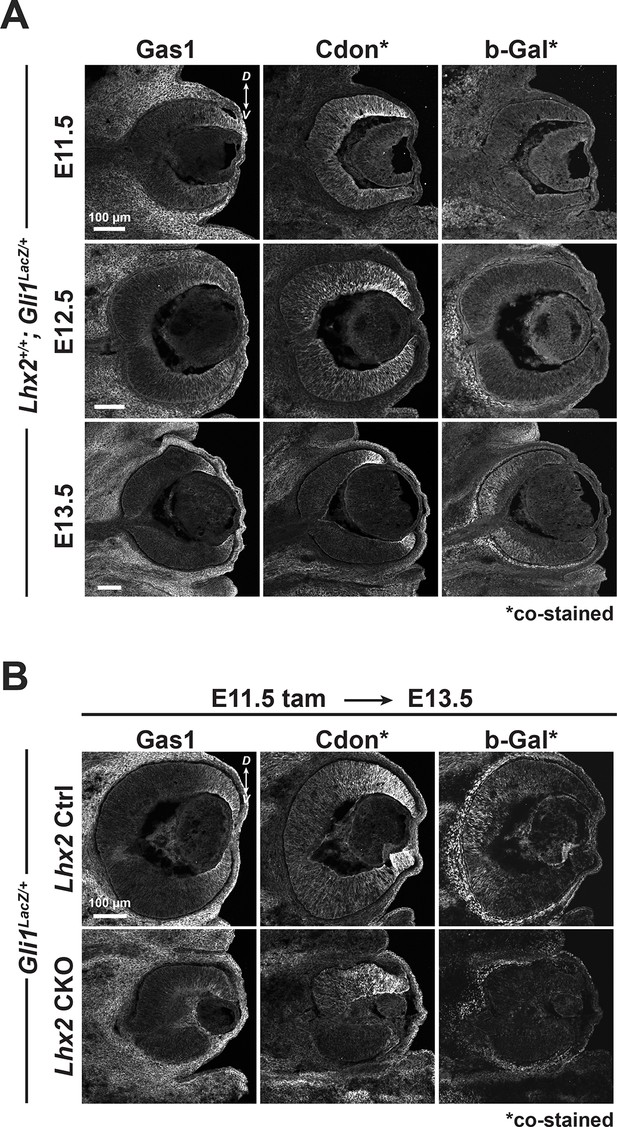
Cdon and Gas1 expression are dependent on Lhx2 prior to their downregulation at the start of Shh signaling.
(A) Temporal expression patterns of Gas1, Cdon, and β-Gal at E11.5, E12.5, and E13.5 in Gli1lacz/+ mice. Cdon and β-Gal were detected on the same tissue sections, Gas1 on adjacent sections (also in B). (B) Expression of Gas1, Cdon, and β-Gal in Ctrl and Lhx2 CKO; Gli1lacz/+ eyes at E13.5 following tamoxifen treatment at E11.5.
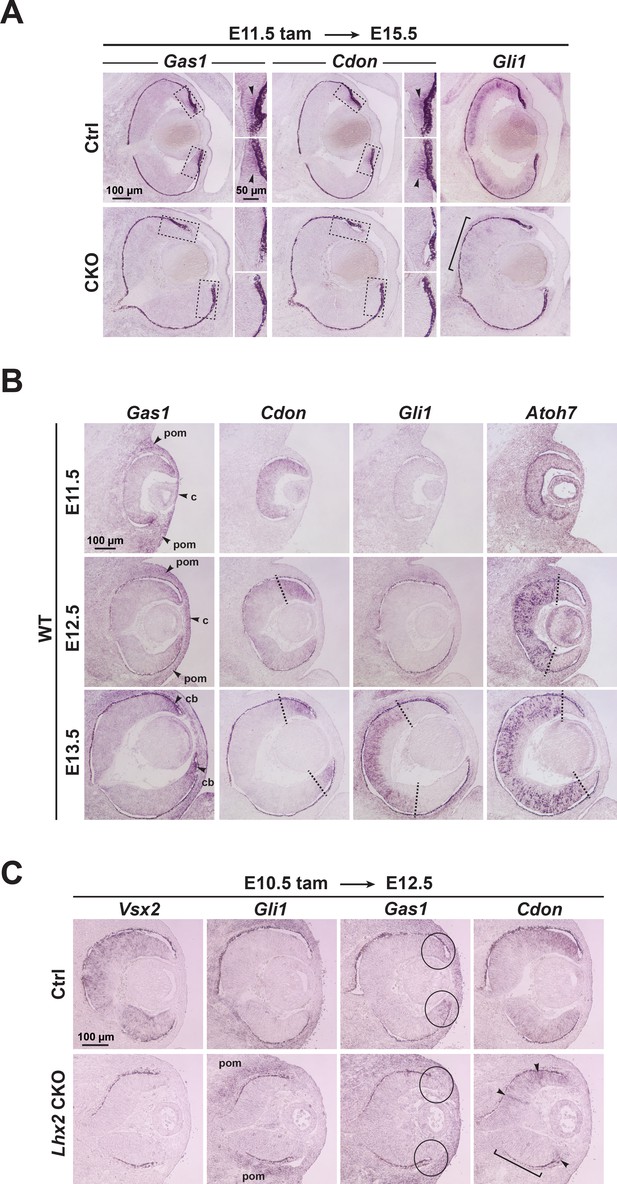
in situ hybridizations.
(A) Expression of Gas1, Cdon, and Gli1 at E15.5 after tamoxifen treatment at E11.5. Boxes in Gas1 and Cdon panels mark locations of insets, which correspond to the retinal periphery, where Gas1 and Cdon are expressed. Arrowheads in Ctrl panels show the restricted peripheral expression that is absent in the CKO. Bracket in CKO panel for Gli1 expression reveals low, but persistent, expression in the dorsal retina. (B) Temporal expression patterns of Gas1, Cdon, Gli1, and Atoh7 from E11.5 to E13.5. Dashed lines in E12.5 and E13.5 images show that temporal downregulation of Cdon mRNA has a tighter spatial complementarity with Atoh7 mRNA accumulation than Gli1. (C) Expression of Vsx2, Gli1, Gas1, and Cdon at E12.5 after tamoxifen treatment at E10.5. The presence of Gli1 in the periocular mesenchyme indicates that the staining protocol was sufficient to detect expression in other tissues. Circles in Gas1 images show regions of Gas1 expression in peripheral retina that is lost upon Lhx2 inactivation. Bracket in image of Cdon for the CKO image shows absence of Cdon in ventral domain, and arrowheads point to persistent Cdon expression. Abbreviations: c, cornea; cb, ciliary body; le, lens; onh, optic nerve head; nr, neural retina; pom, periocular mesenchyme.
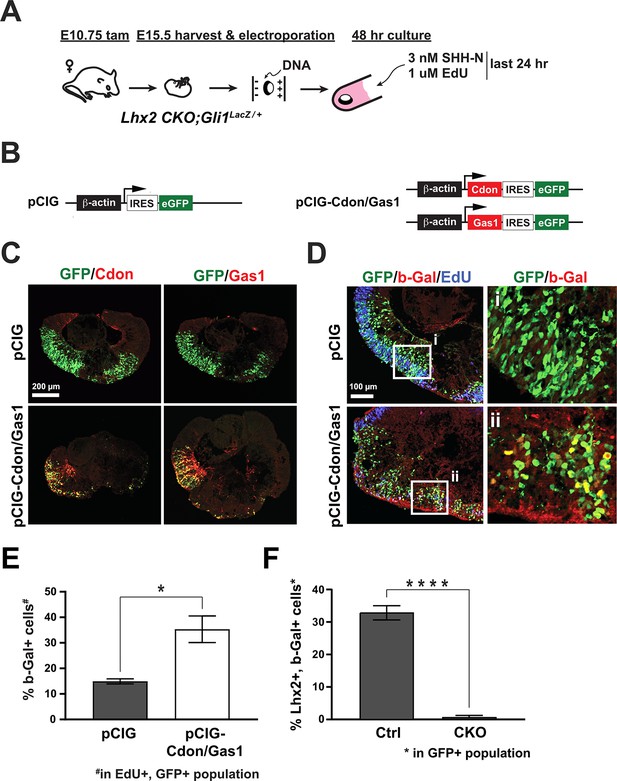
Cdon and Gas1 overexpression is sufficient to stimulate Shh signaling in the absence of Lhx2.
(A) Experimental design for ex vivo electroporation and explant culture. Lhx2 CKO; Gli1LacZ/+ explants were electroporated at the beginning of the culture. 3 nM Shh-N and 1 µM EdU were added after 24 hr and cultured for an additional 24 hr. (B) DNA constructs used for electroporation. pCIG served as the control and pCIG-Cdon and pCIG-Gas1 were co-electroporated. (C) Upper panels: explants were electroporated with pCIG and co-stained for GFP and Cdon (left panel) or Gas1 (right panel). Lower panels: explants were co-electroporated with pCIG-Cdon and pCIG-Gas1 and co-stained for GFP and Cdon (left panel) or Gas1 (right panel). (D). Electroporated explants were co-stained for GFP, β-Gal, and EdU. Insets (i and ii) show GFP and β-Gal staining only. (E) Quantification of the percentage β-Gal+ cells in the EdU+, GFP+ cell populations from GFP (control) and Cdon/Gas1 electroporated Lhx2 CKO; Gli1LacZ/+ explants (mean+/-S.E.M.; n=3, both conditions; *, p<0.05; n=3, both conditions; unpaired t-test). (F) Quantification of the percentage of Lhx2+, β-Gal+ cells in the GFP+ cell populations from control (Lhx2Δ⁄+; Gli1LacZ/+) and Lhx2 CKO; Gli1LacZ/+ explants electroporated with pCIG (mean+/-S.D.; n=7 (Ctrl); n=4 (CKO); *, p<0.0001; unpaired t-test) See Supplementary file 6 for statistics. Single channel images for C and D and representative images for Ctrl (Lhx2Δ⁄+; Gli1LacZ/+) and Lhx2 CKO; Gli1LacZ/+ explants used for quantification in F are presented in Figure 8—figure supplement 1.
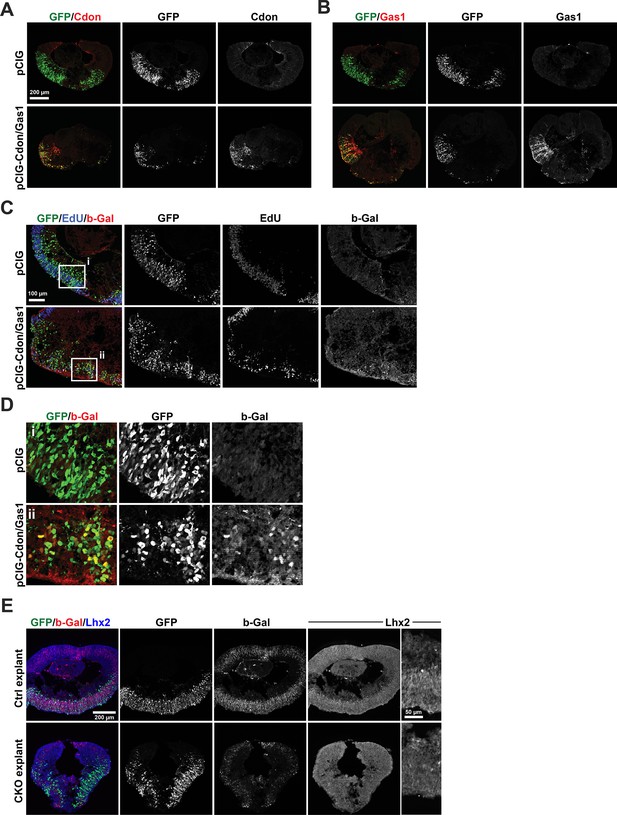
Single channel images for Figure 8C and D.
(A) Merged and single channel images for left panels in 8 C. (B) Merged and single channel images for right panels in 8 C. (C) Merged and single channel images for left panels in 8D. (D) Merged and single channel images for right panels in 8D. b-Gal expression in C and D detects LacZ expression from the Gli1LacZ allele. (E) Merged and single channel images for representative control and CKO explants electroporated with pCIG and stained for GFP, b-Gal, and Lhx2. Zoomed in views for Lhx2 are shown to emphasize the loss of Lhx2 protein in the CKO. The low signal to noise ratio for Lhx2 is due to the use of a secondary antibody conjugated to fluorophore in the 405 nm channel, which yields weak signal and elevated tissue autofluorescence and was necessary to perform the co-expression analysis (tdTomato occupied the 568 nm channel).
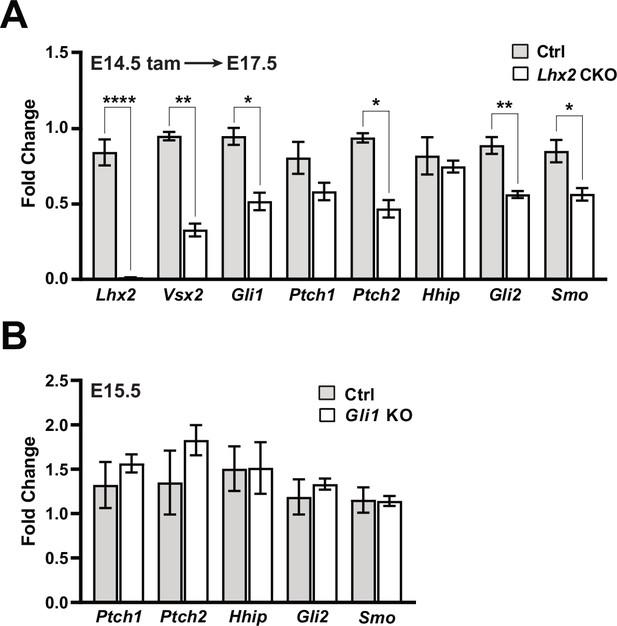
Lhx2 is required for sustained Shh signaling.
(A) qPCR-based expression for Lhx2, Vsx2, Gli1, Ptch1, Ptch2, Hhip, Gli2, and Smo from E17.5 control and Lhx2 CKO retinas following tamoxifen treatment at E14.5. For each gene, fold change values are relative to a specific control sample set as a reference. Only significant comparisons are noted (mean+/-S.E.M.; n=3 (Ctrl, all genes); n=4 (CKO, all genes); * padj <0.05; ** padj <0.01; **** padj <0.0001; unpaired t-tests with correction for multiple comparisons; see Supplementary file 3 for statistics) (B) qPCR-based expression for Ptch1, Ptch2, Hhip, Gli2, and Smo from E15.5 control (Gli1LacZ/+) and Gli1 KO retinas. For each gene, fold change values are relative to a specific control sample set as a reference. None of the comparisons were significant (mean+/-S.E.M; n=4 (both conditions, all genes); unpaired t-tests with correction for multiple comparisons; see Supplementary file 3 for statistics).
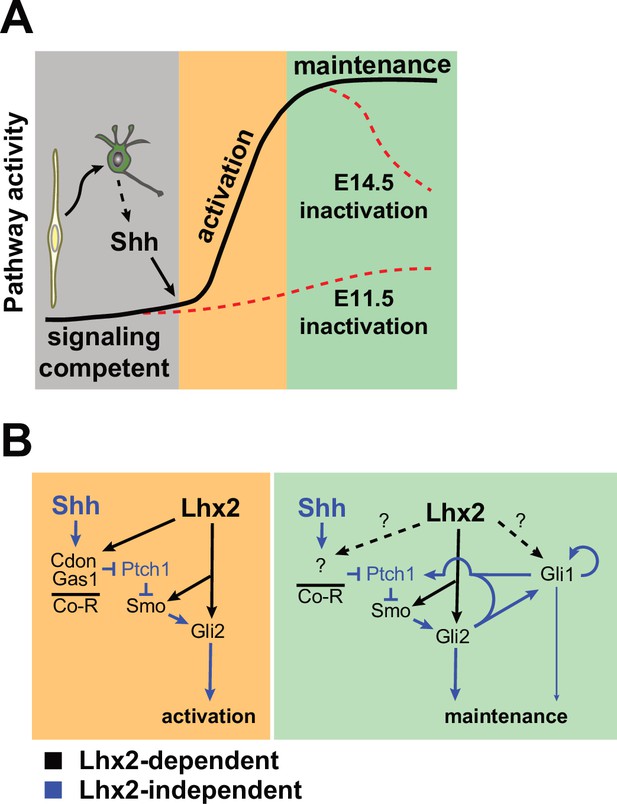
Models of interaction between Lhx2 and Shh signaling during embryonic retinal neurogenesis.
(A) At the cellular level, Lhx2 promotes signaling competence in RPCs and once neurogenesis begins, RGCs produce Shh leading to pathway activation. This is revealed by Lhx2 inactivation at E11.5. Lhx2 also promotes the correct level of signaling in RPCs as evidenced by the drop in pathway readout gene expression when Lhx2 is inactivated at E14.5. (B) Mechanistically, Lhx2 promotes signaling competence and efficient activation by promoting the expression of the coreceptors (Co-R) for ligand reception, Smo for signal transduction, and Gli2 for target gene activation. During the maintenance phase, Lhx2 promotes signaling again by promoting Smo and Gli2. Lhx2 may also regulate other coreceptors, Gli1, or other factors to promote efficient signaling.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (Mus musculus) | Lhx2 | GenBank | Gene ID:16870 | |
| Genetic reagent (Mus musculus) | Lhx2flox | in lab >5 yr; from Ed Monuki, University of California, Irvine | MGI:3772179 | |
| Genetic reagent (Mus musculus) | Lhx2 KO | in lab >5 yr; from Heiner Westphal, National Institutes of Health | MGI:1890208 | |
| Genetic reagent (Mus musculus) | Lhx2 CKO | experimentally generated; not maintained | This paper | |
| Genetic reagent (Mus musculus) | Hes1CreERT2 | in lab >5 yr; from Charles Murtaugh, University of Utah | MGI:4412375 | |
| Genetic reagent (Mus musculus) | Gli1lacz | Jax stock 8211 | MGI:2449767 | |
| Genetic reagent (Mus musculus) | Ptch1flox | from Michael Lewis, Baylor College of Medicine | MGI:2675356 | |
| Genetic reagent (Mus musculus) | Ptch1 CKO | experimentally generated; not maintained | This paper | |
| Genetic reagent (Mus musculus) | Rosa26ai14 | Jax stock 7914 | MGI:3809524 | |
| Genetic reagent (Mus musculus) | Rosa26mTmG | present in Ptch1flox strain | MGI:3716464 | |
| Cell line (Mus musculus) | NIH 3T3 open-loop responder cells | Pulin Li, Massachusetts Institute of Technology; Li et al., 2018 | ||
| Antibody | Anti-Caspase 3 (Rabbit polyclonal) | BD Biosciences | PPID:AB_397274 | IF (1:500) |
| Antibody | Anti-Pou4F (Goat polyclonal) | Santa Cruz | RRID:AB_673441 | IF (1:500) |
| Antibody | Anti-Otx2 (Goat polyclonal) | Gene Tex | RRID:AB_2157172 | IF (1:600) |
| Antibody | Anti-PCNA (Mouse monoclonal) | Santa Cruz | RRID:AB_628110 | IF (1:500) |
| Antibody | Anti-Cyclin D1 (Rabbit polyclonal) | Abcam | RRID:AB_443423 | IF (1:400) |
| Antibody | Anti-b-Gal (Rabbit polyclonal) | Cappel | RRID:AB_2313707 | IF (1:5,000) |
| Antibody | Anti-Cdon (Goat polyclonal) | R&D Systems | RRID:AB_2078891 | IF (1:600) |
| Antibody | Anti-Gas1 (Goat polyclonal) | R&D Systems | RRID:AB_2107951 | IF (1:300) |
| Antibody | Anti-GFP (Chicken polyclonal) | Aves Labs | RRID:AB_10000240 | IF (1:2,000) |
| Antibody | Anti-Shh (Rat monoclonal) | DHSB | 5E1, RRID:AB_528466 | Blocking antibody (15 nM) |
| Antibody | Anti-Gli1 (Mouse monoclonal) | Cell Signalling Technologies | RRID:AB_2294746 | Western blot (1:3000) |
| Recombinant DNA reagent | pCIG-GFP | Guoqiang Gu, Vanderbilt University; Megason and McMahon, 2002 | Electroporation (3 µg/µl) | |
| Recombinant DNA reagent | pCIG-Cdon | Ben Allen, University of Michigan; Allen et al., 2007 | Electroporation (3 µg/µl) | |
| Recombinant DNA reagent | pCIG-Gas1 | Ben Allen, University of Michigan; Allen et al., 2007 | Electroporation (3 µg/µl) | |
| Sequence-based reagent | Oligonucleotides | Supplementary file 7 | ||
| Peptide, recombinant protein | Shh-N | R&D Systems | Cat#:1845-SH-025 | |
| Commercial assay or kit | Click-iT EdU Alexa Fluor 647 imaging kit | Thermo Fisher Scientific | Cat#:C10340 | |
| Commercial assay or kit | Cytiva Protein G HP SpinTrap Columns | Thermo Fisher Scientific | Cat#:45001485 | Affinity purification for 5E1 antibody |
| Commercial assay or kit | Amicon Ultracel-30 filter | EMD/Millipore | Cat#:UFC503024 | Buffer exchange for 5E1 antibody |
| Chemical compound, drug | Purmorphamine | EMD Biosciences | Cat#:540220 | |
| Software, algorithm | GraphPad Prism (version 9.0) | GraphPad Software, Inc | Graphing and statistics analysis | |
| Software, algorithm | Ingenuity Pathway Analysis Suite | Qiagen, Inc | Pathway analysis for RNA seq data | |
| Other | DAPI | Sigma | Cat#:D9542 | IF (300 nM) |
Additional files
-
Supplementary file 1
Differential gene expression analysis of RNA-seq data with DESeq2.
- https://cdn.elifesciences.org/articles/78342/elife-78342-supp1-v1.xlsx
-
Supplementary file 2
KEGG pathways identified by Ingenuity Pathway Analysis and clusterProfiler.
- https://cdn.elifesciences.org/articles/78342/elife-78342-supp2-v1.xlsx
-
Supplementary file 3
Statistics and tests for multiple pairwise comparisons of qPCR-based measurements of gene expression.
- https://cdn.elifesciences.org/articles/78342/elife-78342-supp3-v1.xlsx
-
Supplementary file 4
Statistics and test results for experiments with open-loop responder cells.
- https://cdn.elifesciences.org/articles/78342/elife-78342-supp4-v1.xlsx
-
Supplementary file 5
Statistics and test results for Shh-N dose responses in retinal explants.
- https://cdn.elifesciences.org/articles/78342/elife-78342-supp5-v1.xlsx
-
Supplementary file 6
Statistics and test results for cell counts following electroporation in Lhx2 CKO, Gli1Lacz/+ retinal explants.
- https://cdn.elifesciences.org/articles/78342/elife-78342-supp6-v1.xlsx
-
Supplementary file 7
Oligonucleotides.
- https://cdn.elifesciences.org/articles/78342/elife-78342-supp7-v1.xlsx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/78342/elife-78342-mdarchecklist1-v1.docx





