Hematopoietic plasticity mapped in Drosophila and other insects
Figures
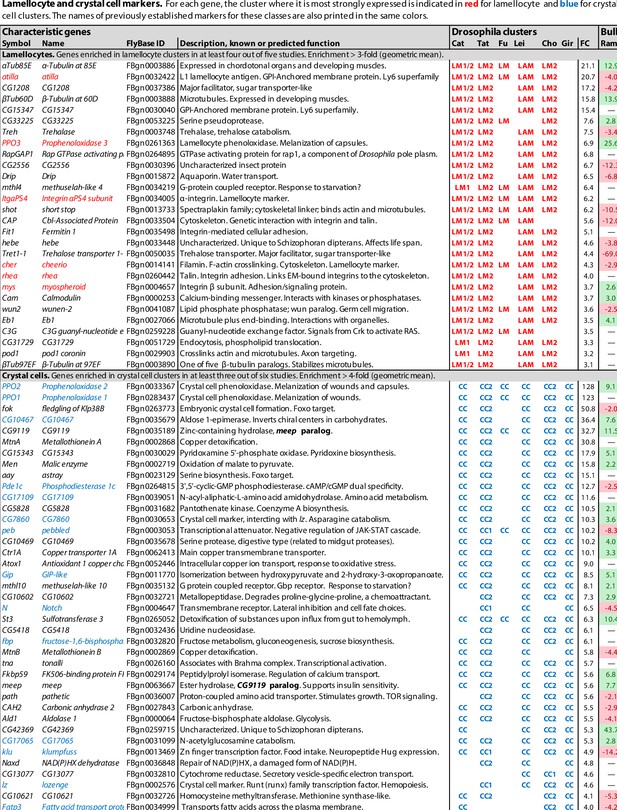
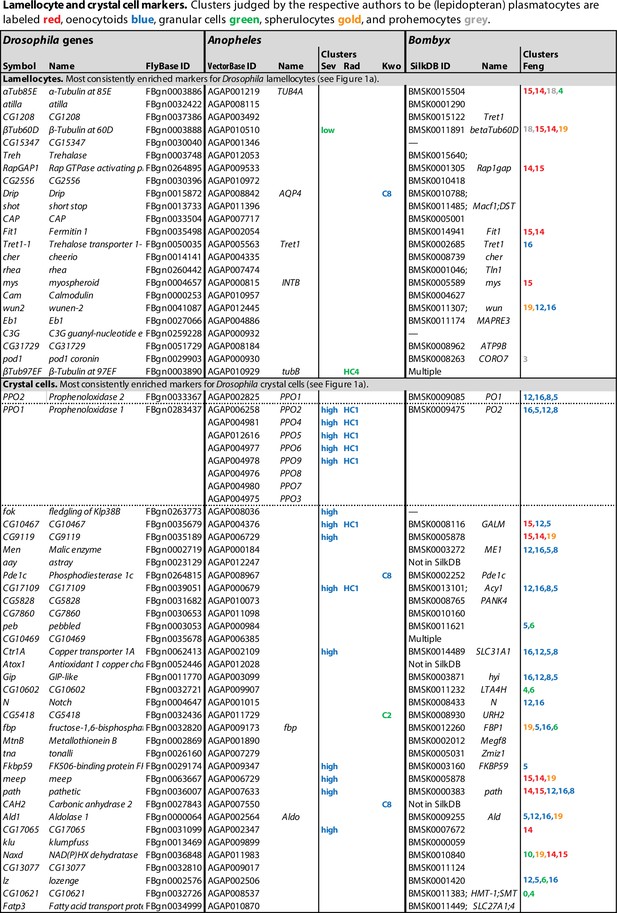
Lamellocyte and crystal cell marker genes.
Genes with enhanced expression in lamellocyte- and crystal cell-related clusters, as reported by Cattenoz et al., 2020 (Cat), Tattikota et al., 2020 (Tat), Fu et al., 2020 (Fu), Leitão et al., 2020 (Lei), Cho et al., 2020 (Cho), and Girard et al., 2021 (Gir). Relative expression (‘FC’) in bulk plasmatocytes compared to whole larvae, as reported by Ramond et al., 2020, is shown in a separate column (Ram). The figure summarizes the most consistently and strongly enhanced genes for each of these cell classes, and the average (geometric mean) fold enhancement (‘FC’). As we lack full data from Fu, we have only listed examples mentioned in the text and figures of that study. For a full list of all enhanced genes, see Figure 1—source data 1.
-
Figure 1—source data 1
Genes with enhanced expression in specific cell classes.
Complete lists of differentially expressed genes.
- https://cdn.elifesciences.org/articles/78906/elife-78906-fig1-data1-v1.xlsx
-
Figure 1—source data 2
Gene Ontology (GO) terms.
Significantly enriched GO terms in differentially expressed genes in lamellocyte, crystal cell, and plasmatocyte clusters in at least two of the published studies.
- https://cdn.elifesciences.org/articles/78906/elife-78906-fig1-data2-v1.xlsx
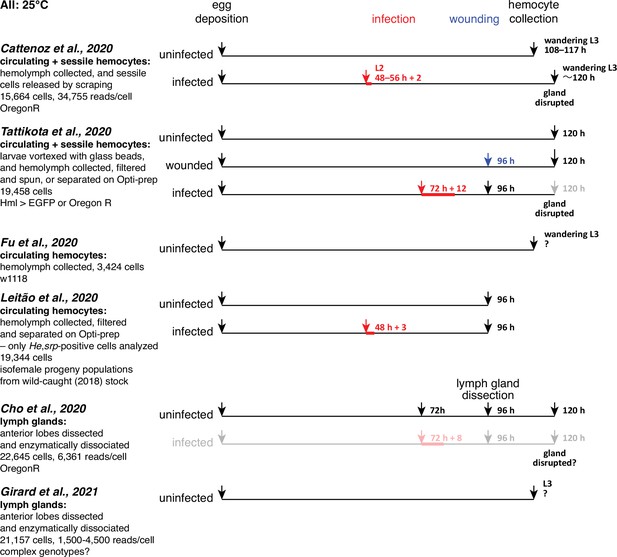
Timelines for six single-cell transcriptomic analyses of Drosophila hemocytes.
Data from the analyses shown in gray were not included in our comparisons.
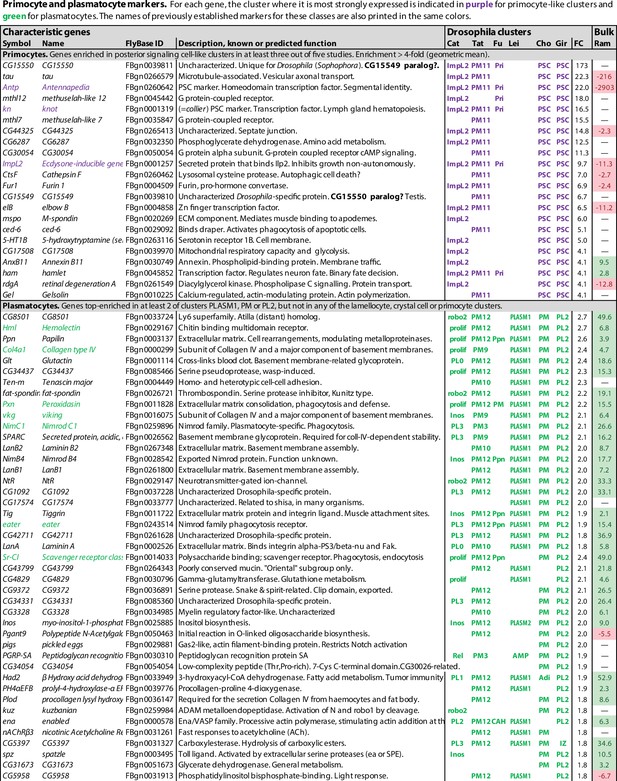
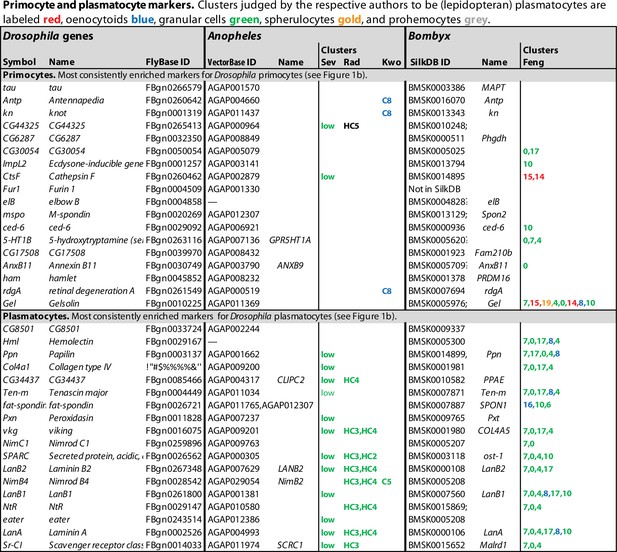
Primocyte and plasmatocyte marker genes.
Genes with enhanced expression in primocyte- and plasmatocyte-related clusters. Details as in Figure 1.
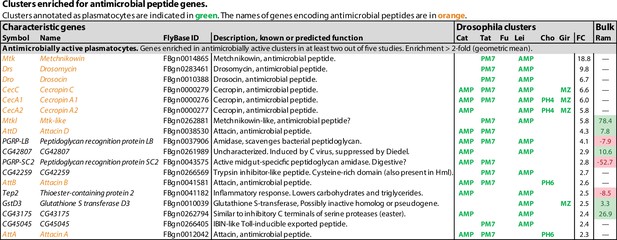
Clusters enriched for antimicrobial peptide genes.
Genes with enhanced expression in the AMP clusters, as reported by Cattenoz et al., 2020 (Cat), and Leitão et al., 2020 (Lei), and the PM7 cluster of Tattikota et al., 2020 (Tat). Clusters where the same genes are enriched as reported by Cho et al., 2020 (Cho) and Girard et al., 2021 (Gir) are also indicated. None of these genes were mentioned by Fu et al., 2020 (Fu). Relative expression (‘FC’) in bulk plasmatocytes compared to whole larvae, as reported by Ramond et al., 2020, is shown in a separate column (Ram). The figure summarizes the most consistently and strongly enhanced genes for each of these cell classes, and the average (geometric mean) fold enhancement (‘FC’). For a full list of all enhanced genes, see Figure 1—source data 1.
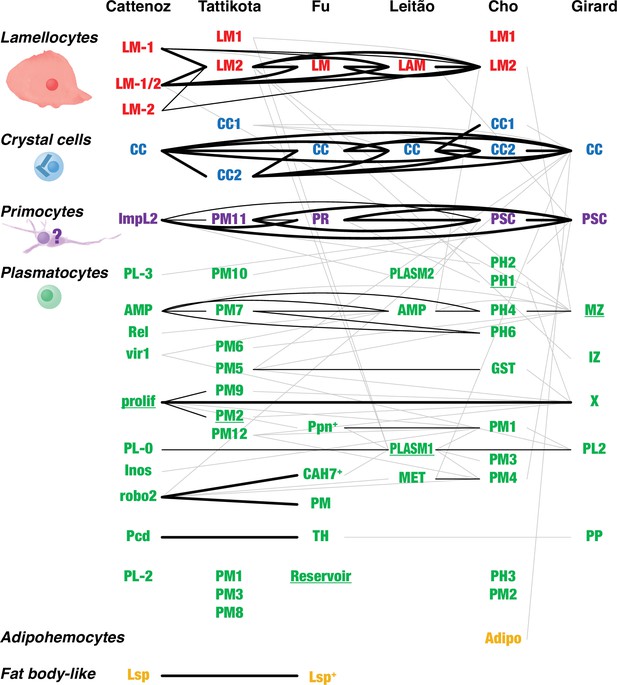
Overlap between Drosophila hemocyte clusters.
Lines connect clusters that share genes with >1.4-fold enriched transcripts. A fat line indicates that at least 50% of the genes enriched in one of the clusters are also enriched in the other one (but not necessarily reciprocally). A thin line indicates that both clusters share at least 10% of the enriched genes. A thin gray line indicates that one cluster shares 10% of the enriched genes with the other one, but not reciprocally. The different clusters were named after their presumed identity: lamellocytes (LM, LAM), crystal cells (CC), primocytes (PR), plasmatocytes (PL, PM, PLASM), prohemocytes (PH), proplasmatocytes (PP), medullary zone cells (MZ), intermediate zone cells (IZ), posterior signaling center (PSC), or after their presumed functions: metabolic (MET), reservoir. The clusters prolif and X represent mitotic cells. Some plasmatocyte clusters were named after characteristic genes or gene groups that are enriched in the clusters (ImpL2, AMP, Rel, vir1, Inos, robo2, Pcd, Lsp, Ppn, CAH7, GST). Two additional suggested classes, thanacytes (TH) and adipohemocytes (Adipo), were not reproducibly observed and are not further discussed here. Finally, no genes were preferentially expressed above our cutoff in clusters PL-1 (Cattenoz et al., 2020), PM4, and PM11 (Tattikota et al., 2020). The shape and size of circulating larval primocytes are unknown. Instead, the illustration is based on published images of primocyte-like cells in adults (Boulet et al., 2021) and primocytes in the posterior signaling center (Krzemień et al., 2007; Mandal et al., 2007).
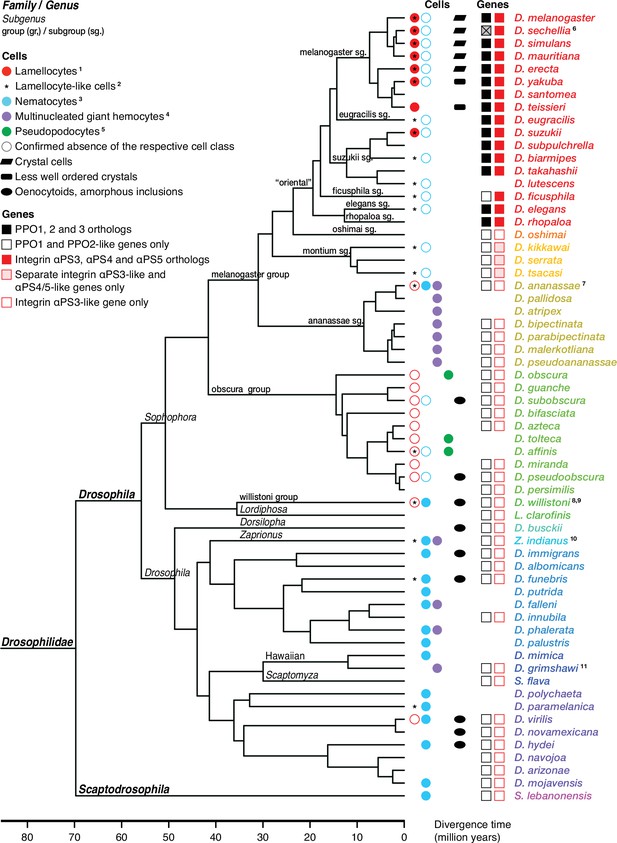
Occurrence of specialized effector cells (lamellocytes, nematocytes, multinucleated giant hemocytes, pseudopodocytes, and crystal cells) in parasitized drosophilid larvae, and correlation with presence or absence of PPO3 and ItgaPS4 genes.
Consensus phylogenetic tree from Russo et al., 2013, Thomas and Hahn, 2017, Miller et al., 2018, Kim et al., 2021, and Finet et al., 2021. Basic topology from Finet et al., 2021, time calibration from Russo et al., 2013, and taxonomy from Kim et al., 2021. 1Presence or absence of lamellocytes (Eslin and Doury, 2006; Eslin et al., 2009; Havard et al., 2009; Salazar-Jaramillo et al., 2014; Wan et al., 2019; Cinege et al., 2020). 2Kacsoh, 2012 documents lamellocyte-like cells from several species, sometimes at odds with reports elsewhere. Possible interpretations are discussed in the text. 3Presence or absence of nematocytes (Rizki, 1953; Srdic and Gloor, 1893; Kacsoh et al., 2014; Bozler et al., 2017). 4Presence or absence of multinucleated giant hemocytes (Márkus et al., 2015; Bozler et al., 2017; Cinege et al., 2020). 5Presence or absence of pseudopodocytes (Havard et al., 2009; Havard et al., 2012). 6PPO3 is pseudogenized in D. sechellia; the open reading frame is interrupted by a stop codon. Kacsoh et al., 2014 found no nematocytes in D. ananassae, but Márkus et al., 2015 observed ‘small filariform cells.’ 8Unusual hemocytes reminiscent of lamellocytes were observed in infected D. willistoni larvae (Salazar-Jaramillo et al., 2014). 9Nematocytes were found in D. willistoni by Rizki, 1953, but not by Kacsoh et al., 2014. 10Kacsoh et al., 2014 mention lamellocyte homologs but no such cells were observed by Cinege et al., 2020. 11Bozler et al., 2017 note ‘large multicellular, and multinuclear structures.’.
Orthologs of Drosophila lamellocyte and crystal cell markers expressed in mosquito and silkworm hemocyte clusters.
Data from single-cell RNAseq studies by Severo et al., 2018 (Sev), Raddi et al., 2020 (Rad), Kwon et al., 2021a (Kwo), and Feng et al., 2021 (Feng). Drosophila markers for which no orthologs could be identified were excluded from the analysis. Clusters where the genes are significantly enriched are indicated, with highest enrichment first. Non-hemocyte clusters are omitted.
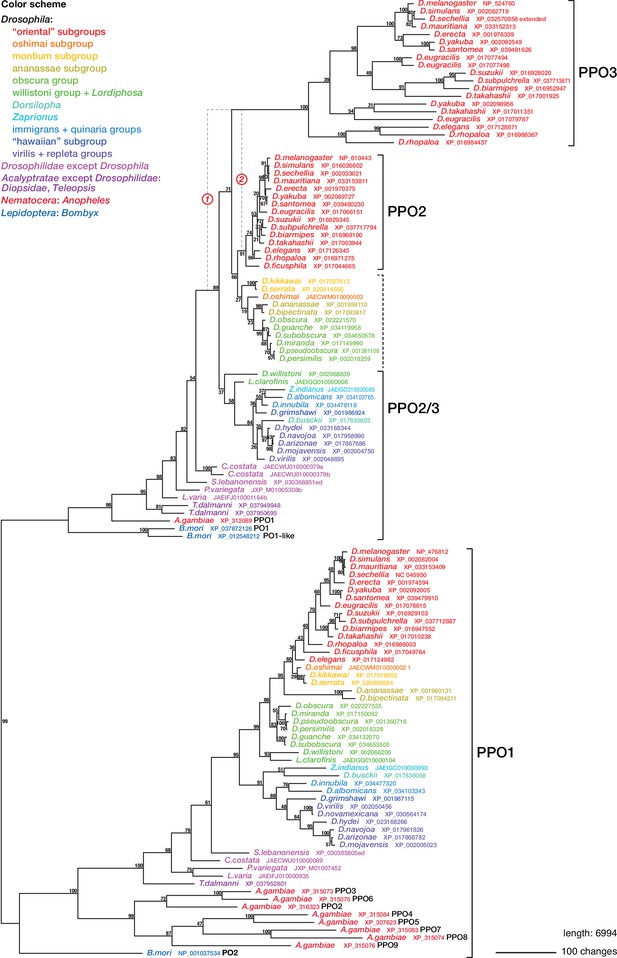
Phylogenetic relationships between insect phenoloxidases.
Maximum parsimony tree of protein sequences found by blastp search of all annotated sequences from the family Drosophilidae and from Anopheles gambiae and Bombyx mori, in the refseq_protein database. Additional selected protein sequences were modeled from genomic sequences retrieved in a tblastn search of the refseq_genomes and wgs databases. Bootstrap values are percent support after 1000 replicates, using the PPO1-like proteins as outgroup. Note that the PPO3 homolog is pseudogenized in D. sechellia, and there is no trace of a PPO3 homolog in D. ficusphila. Consequently, although D. sechellia has lamellocytes, it is unable to encapsulate the eggs of parasitoid wasps (Kacsoh, 2012; Salazar-Jaramillo et al., 2014). D. ficusphila can encapsulate and kill parasites, but the capsules are not melanized (Kacsoh, 2012).
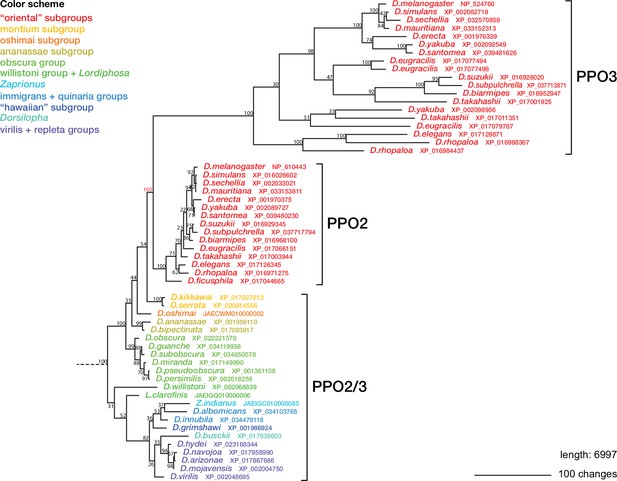
Maximum parsimony tree with forced monophyly of PPO2 + PPO3 sequences from the oriental subgroups.
Data are the same as in Figure 6, but for simplicity only the Drosophila PPO2/3 branch of the tree is shown. Bootstrap values are percent support after 1000 replicates, using the PPO1-like proteins as outgroup.
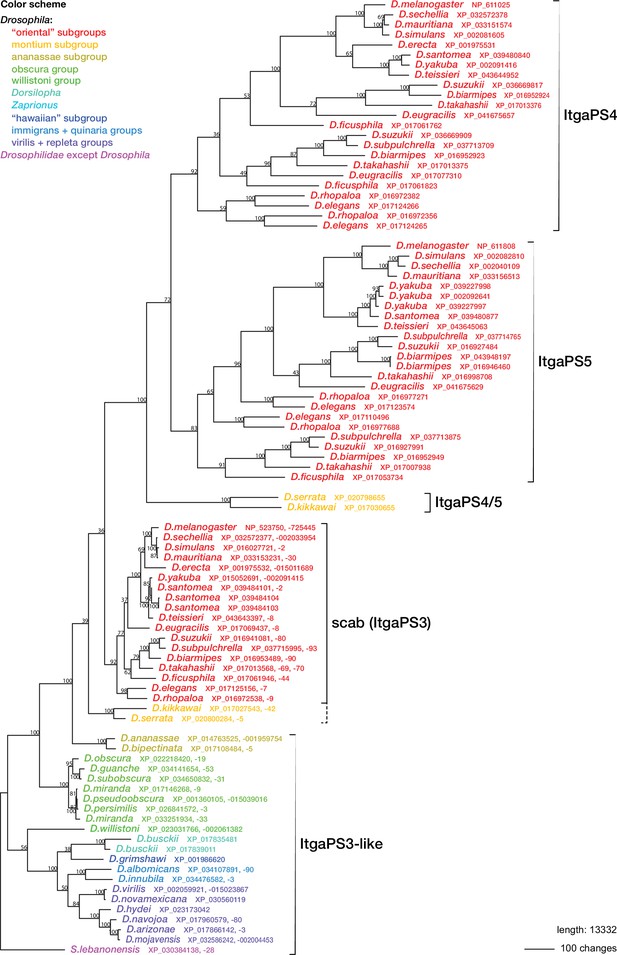
Phylogenetic relationships between Drosophilid integrin alphaPS3, 4, and 5 homologs.
Maximum parsimony tree of protein sequences found by blastp search of all Drosophilidae sequences annotated in the refseq_protein database. Most ItgaPS3 and ItgaPS3-like genes have two alternative splice forms, A and B, with different approximately 63 amino acid leader sequences. The A- and B-form leaders were concatenated before the sequences were aligned. The ItgaPS4 and ItgaPS5 sequences have only an A-form leader. A few partial or chimaeric forms were excluded from the analysis. Bootstrap values are percent support after 1000 replicates, using the Scapto Drosophila lebanonensis protein as outgroup.
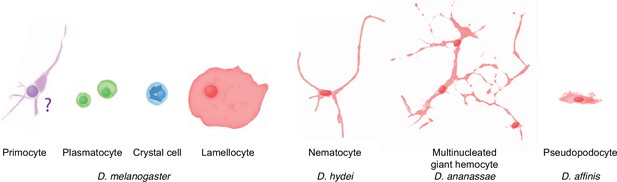
Schematic overview of drosophilid hemocyte morphologies.
Plasmatocyte, crystal cell, and lamellocyte cartoons are sketched from images of D. melanogaster hemocytes (Rizki, 1957), the D. hydei nematocyte from Kacsoh et al., 2014, the Zaprionus indianus multinucleated giant hemocyte from Cinege et al., 2020, and the D. affinis pseudopodocyte from Havard et al., 2012. The primocyte illustration is based on published images of primocyte-like cells in adults (Boulet et al., 2021) and primocytes in the posterior signaling center (Krzemień et al., 2007; Mandal et al., 2007). The morphology of circulating larval primocytes is unknown.
Orthologs of Drosophila primocyte and plasmatocyte markers expressed in mosquito and silkworm hemocyte clusters.
Details as in Figure 5.
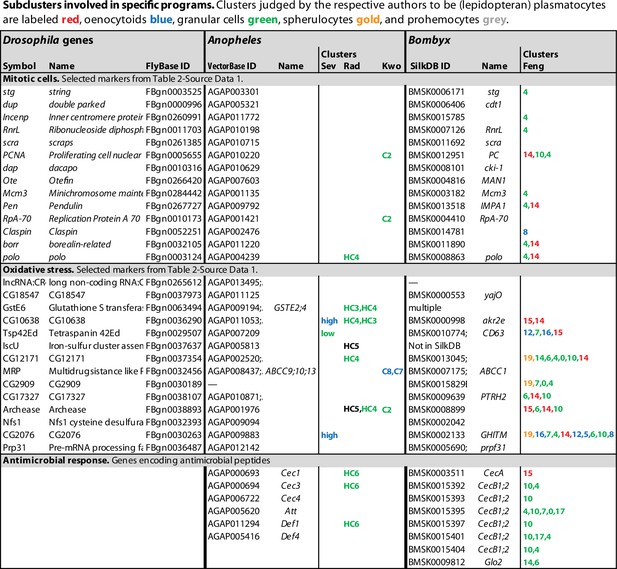
Antimicrobial peptide genes expressed in mosquito and silkworm hemocyte clusters.
All antimicrobial peptide genes expressed in mosquito and silkworm hemocytes are listed. Extensive paralogies prevent us from orthology assignments. Clusters where the genes are significantly enriched are indicated, with highest enrichment first. Non-hemocyte clusters are omitted. Data come from single-cell RNAseq studies by Severo et al., 2018 (Sev), Raddi et al., 2020 (Rad), Kwon et al., 2021b (Kwo), and Feng et al., 2021 (Feng).
Tables
Insect hemocyte terminology.
| Function | Drosophila melanogaster (and related ‘oriental’ subgroup species) | Other drosophilids | Mosquitoes | Lepidopterans(similar terminology in other insect orders) |
|---|---|---|---|---|
| Phagocytes | Plasmatocytes | Plasmatocytes | Granulocytes/ granular cells | Granulocytes/ granular cells |
| Melanization | Crystal cells | Oenocytoids (commonly called ‘crystal cells,’ but they lack crystals) | Oencytoids | Oencytoids |
| Encapsulation | Lamellocytes | Other encapsulating cell types: Nematocytes Multinucleated giant hemocytes Pseudopodocytes Activated plasmatocytes | — | Plasmatocytes (not homologous to Drosophila plasmatocytes – uncertain homology to Drosophila lamellocytes, as discussed in the text) |
| ? | Primocytes (novel class) | ? | ? | ? |
| Cuticle formation? | — | — | — | Spherule cells |
| Hemocyte precursors | Prohemocytes (In lymph gland; few, if any, in circulation) | ? | Prohemocytes | Prohemocytes (in hematopoietic organ only?) |





