Multiple cullin-associated E3 ligases regulate cyclin D1 protein stability
Figures
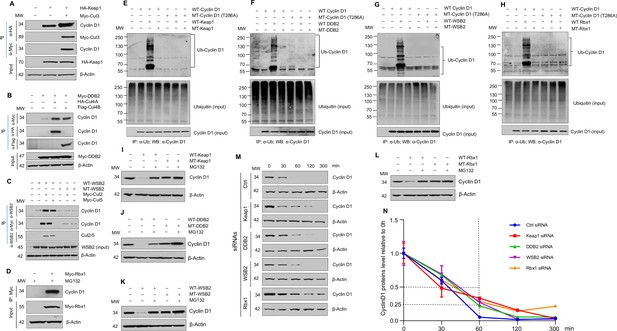
Cullin-associated E3 ligases mediate cyclin D1 ubiquitination and proteasome degradation.
(A) Co-immunoprecipitation (co-IP) of Keap1, CUL3 with endogenous cyclin D1. HA-Keap1 and Myc-CUL3 were co-transfected into HEK293 cells with the MG132 treatment (10 μM, 4 hr incubation). 24 hr after transfection, the cell lysates were collected. To detect interaction of Keap1 with cyclin D1 or CUL3, co-IP was performed using the anti-HA (α-HA) antibody followed by the western blot using the anti-cyclin D1 or anti-Myc (α-Myc) antibody. To detect the interaction between CUL3 with cyclin D1, co-IP assay was performed using the anti-Myc antibody followed by the western blot using the anti-cyclin D1 antibody. (B) Co-IP of DDB2 and CUL4A/4B with endogenous cyclin D1. Myc-DDB2, HA-CUL4A, and Flag-CUL4B were transfected into HEK293 cells with the MG132 treatment (10 μM, 4 hr incubation). Co-IP was performed using the anti-Flag (α-Flag), anti-HA, or anti-Myc antibody followed by the western blot using the anti-cyclin D1 antibody. (C) Co-IP of WSB2 and CUL2/5 with endogenous cyclin D1. Wild-type (WT) or mutant form of WSB2 (SOCS∆364–400) were co-transfected with Myc-CUL2/5 into HEK293 cells with MG132 treatment (10 μM, 4 hr incubation). Co-IP was performed using the anti-Myc or anti-WSB2 (α-WSB2) antibodies followed by the western blot using the anti-cyclin D1 antibody. (D) Co-IP of Rbx1 with endogenous cyclin D1. Myc-Rbx1 was transfected into HEK293 cells with MG132 treatment (10 μM, 4 hr incubation). Co-IP assay was performed using the anti-WSB2 or anti-Myc antibody followed by the western blot using the anti-cyclin D1 antibody. (E–H) Ubiquitination assay. WT or mutant cyclin D1 (T286A) were co-transfected with WT or mutant Keap1, DDB2, WSB2, or Rbx1 expression plasmids into HEK293 cells with the treatment of MG132 (10 μM, 4 hr incubation). Co-IP was performed using the anti-Ub (α-Ub) antibody followed by the western blot using the anti-cyclin D1 (α-cyclin D1) antibody. (I–L) WT or mutant Keap1, DDB2, WSB2, and Rbx1 expression plasmids were co-transfected with cyclin D1 expression plasmid into HEK293 cells with the MG132 treatment (10 μM, 4 hr incubation). Cyclin D1 protein levels were detected by the western blot analysis. (M, N) Protein decay assay. HEK293 cells were transfected with scramble siRNA (Ctrl) or Keap1, DDB2, WSB2, or Rbx1 siRNA. The cell lysates were collected 0, 30, 60, 120, or 300 min after cycloheximide treatment (80 μg/ml) and the cyclin D1 protein levels were detected by the western blot analysis and were quantified (n=3).
-
Figure 1—source data 1
Numerical data obtained during experiments represented in Figure 1.
- https://cdn.elifesciences.org/articles/80327/elife-80327-fig1-data1-v2.zip
-
Figure 1—source data 2
Original western blot files for Figure 1.
- https://cdn.elifesciences.org/articles/80327/elife-80327-fig1-data2-v2.zip
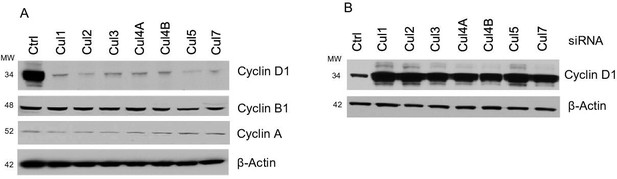
Cyclin D1 degradation is mediated by multiple cullins.
(A) Cullin1, 2, 3, 4A, 4B, 5 and 7 constructs were co-transfected with cyclin D1 into HEK293 cells. Cyclin D1, cyclin B1 and cyclin A protein levels were detected through western blotting. (B) Cyclin D1 expression plasmid was co-transfected with cullin siRNAs into HEK293 cells and changes in steady-state protein levels of cyclin D1 were analyzed by the western blot.

Silencing efficiency of the cullin siRNAs was determined by the luciferase assay.
Cyclin D1 expression plasmid and 3xE2F-luc reporter construct were co-transfected with specific siRNAs targeting cullin genes into HEK293 cells. Luciferase assay were performed 24h after the transfection (n=1).

Specificity of cullin 4A and 4B siRNAs.
Control siRNA (C) or siRNAs specific for each cullin (1, 2, 3, 4A, 4B, 5 and 7) were transiently tranfected into HEK293 cells. Western blotting was performed detecting the Cul4A and Cul4B protein expression using anti-cullin 4A or anti-cullin 4B antibody. The results showed that cullin 4A and 4B siRNAs specifically inhibited CUL4A and CUL4B protein expression.
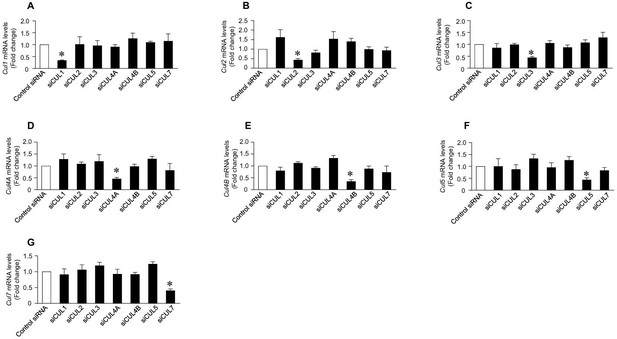
Specificity of cullin siRNAs.
Control siRNA or siRNAs specific for each cullin (1, 2, 3, 4A, 4B, 5 and 7) were transiently tranfected into HEK293 cells. Real-time PCR was performed to detect levels of each cullin mRNA. Data are presented as means±SE of three independent experiments. Figure (A-G) showed that transfection of cullin 1, 2, 3, 4A, 4B, 5 and 7 siRNAs specifically inhibited mRNA expression of cullin 1, 2, 3, 4A, 4B, 5 and 7. A-g showed that transfection of cullin 1, 2, 3, 4A, 4B, 5 and 7 siRNAs specifically inhibited mRNA expression of cullin 1, 2, 3, 4A, 4B, 5 and 7.
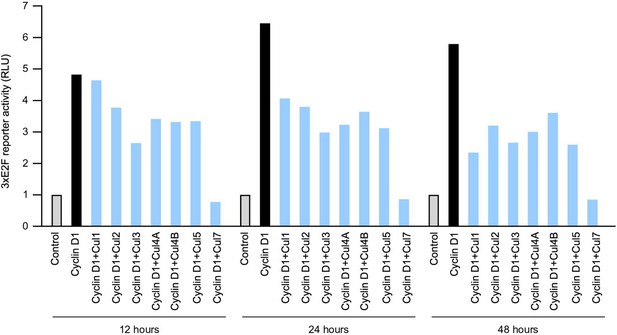
Cullins affect cyclin D1 activity.
Cyclin D1 expression plasmid and 3xE2F-luc reporter construct were co-transfected with each cullin iinto HEK293 cells. Luciferase assay were performed at three different time points: 12, 24 and 48 hours after the transfection.

Silencing of cullins enhances cyclin D1 activity.
Cyclin D1 expression plasmid and 3xE2F-luc reporter construct were co-transfected with each siRNA specific for each cullin into HEK293 cells. Luciferase assay were performed 24 hours after the transfection. Data are presented as means ± SD of three independent experiments.
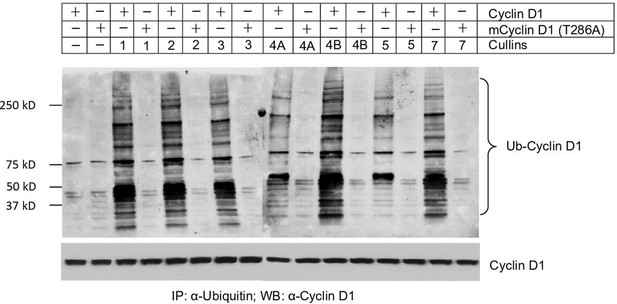
Cyclin D1 ubiquitination is mediated by multiple cullins.
WT or mutant cyclin D1 (T286A) expression plasmids were co-transfected with cullins expression plasmids into HEK293 cells with the treatment of proteasome inhibitor MG132 (10 μM, 4h incubation). 24 hours after transfection, the cell lysates were collected, and ubiquitylated proteins were pulled down using a specific ubiquitin-binding affinity matrix (UbiQapture-Q, Biomol) and polyubiquitylated cyclin D1 was detected using the anti-cyclin D1 antibody.

Proteasomal inhibitor reverses cullin-mediated cyclin D1 degradation.
Cyclin D1 and cullin expression plasmids were co-transfected into HEK293 cells with or without MG132 treatment (10 μM, 4h incubation). Changes in cyclin D1 protein levels were detected by the western blot analysis.
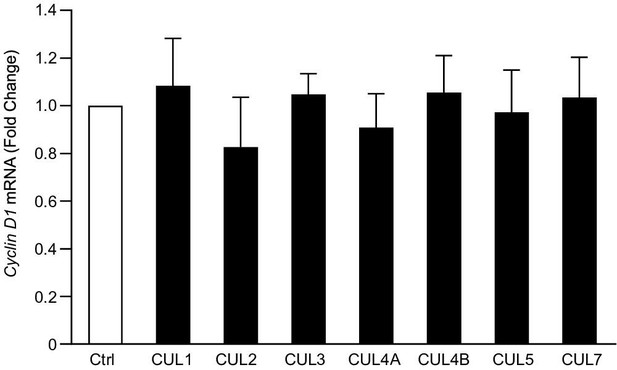
Effects of cullins on cyclin D1 mRNA expression.
HCT-116 cells were transiently transfected with CUL1, 2, 3, 4A, 4B, 5 or 7 respectively. Total RNAs were harvested 24 hours after transfection. Changes in cyclin D1 mRNA expression was detected by real-time PCR. Data were expressed as the fold changes compared to the control group and were presented as means ± SD of three independent experiments.

Cullin-associated ubiquitin ligases siRNA library screening.
(A and B) 468 siRNAs targeting 156 genes (triplicate) were coated in 96-well plates (silencer Custom siRNA library, Ambion, Austin, USA). Cyclin D1 expression plasmid and 3xE2F-luc reporter construct were co-transfected with siRNAs specific for each ligase subunit into NIH3T3 cells. Luciferase assay was performed 24 hours after transfection. Data are presented as means ± SD of three independent experiments.

Five different cullin-associated ubiquitin ligases mediate cyclin D1 degradation.
Cyclin D1 expression plasmid was co-transfected with Fbxw8 (associated with CUL1), Keap1 (associated with CUL3), DDB2 (associated with CUL4A and 4B), WSB2 (associated with CUL2 and 5) and Rbx1 (Ring-finger domain protein interacting with the C-terminal of cullins) expression plasmids into HEK293 cells. Changes in cyclin D1 protein levels were detected by the western blot analysis.

Silencing effects of Keap1, DDB2, WSB2, and Rbx1 siRNAs are specific to their target genes.
Control (Ctrl) siRNA or siRNAs specific for Keap1, DDB2, WSB2 and Rbx1 were transiently tranfected into HEK293 cells. Expression of Keap1 and KLHL12 (interact with CUL3), DDB2 and DDB1 (interact with CUL4A and CUL4B), WSB2 and WSB1 (interact with CUL2 and CUL5) and Rbx1 and ROC2 (interact with CUL1-7) was detected through real-time PCR assay to detect the silencing efficiency and specificity of Keap1, DDB2, WSB2 and Rbx1 siRNAs. Data are presented as means ± SD of three independent experiments.
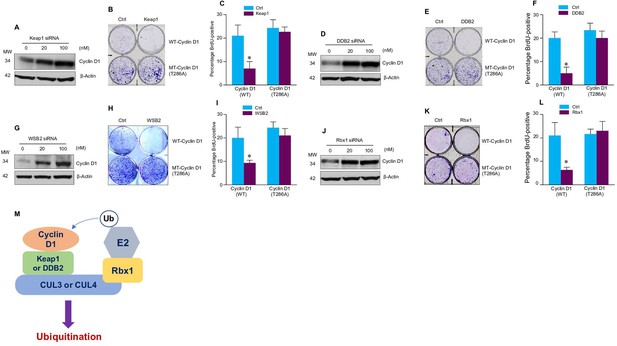
Cullin-associated E3 ligases affect cyclin D1 function.
(A, D, G, J) Keap1, DDB2, WSB2, or Rbx1 siRNAs were transiently transfected into human colon cancer cell line HCT-116 cells. Endogenous cyclin D1 protein levels were detected by the western blot using the anti-cyclin D1 antibody. (B, E, H, K) Cell proliferation assay. Keap1, DDB2, WSB2, or Rbx1 expression plasmids were transfected into HCT-116 cells which were stably transfected with wild-type (WT) or mutant cyclin D1 (T286). The cells were stained with crystal violet 5 days after Keap1, DDB2, WSB2, or Rbx1 transfection. (C, F, I, L) Bromodeoxyuridine (BrdU) incorporation assay. Keap1, DDB2, WSB2, or Rbx1 expression plasmid was transiently transfected into HCT-116 cells which were stably transfected with WT or mutant cyclin D1 (T286). 4 hr before the harvest of cells, the cells were treated with BrdU (20 μM). 48 hr after the transfection, BrdU incorporation assays were performed (n=3). Data were presented as means ± SD of three independent experiments. Statistical analyses were performed using two-way ANOVA followed by the Tucky’s post-hoc test, *P<0.05 (n=3). (M) A model showing that cullin-associated ubiquitin ligases are participated in the cyclin D1 proteolysis process.
-
Figure 2—source data 1
Numerical data obtained during experiments represented in Figure 2.
- https://cdn.elifesciences.org/articles/80327/elife-80327-fig2-data1-v2.xlsx
-
Figure 2—source data 2
Original western blot files for Figure 2.
- https://cdn.elifesciences.org/articles/80327/elife-80327-fig2-data2-v2.zip
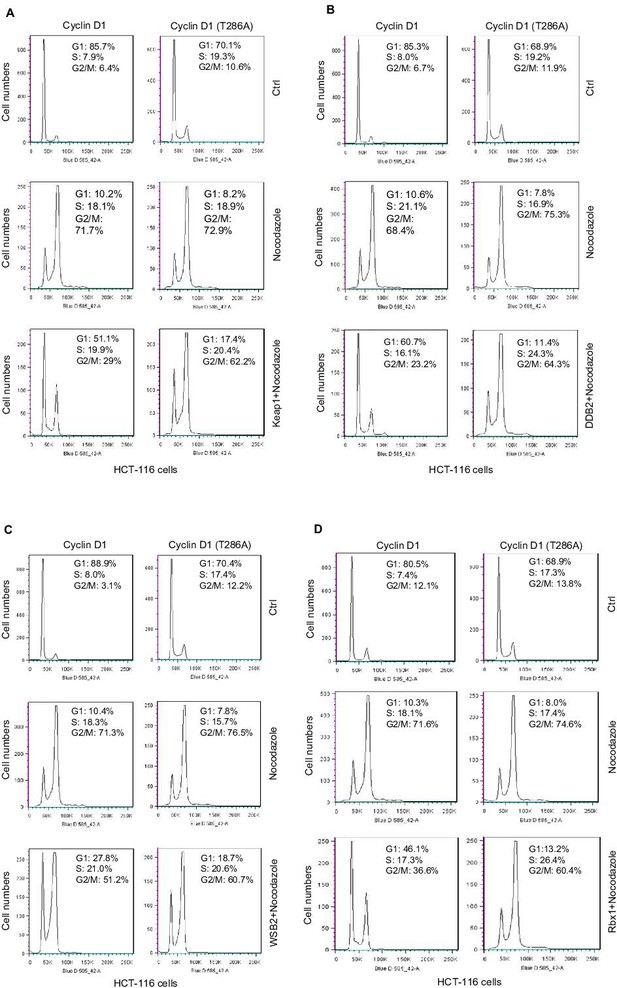
Fluorescence-activated cell sorting (FACS) analysis.
Keap1 (A), DDB2 (B), WSB2 (C) and Rbx1 (D) expression plasmids were transiently transfected into HCT-116 cells stably transfected with WT or mutant cyclin D1 (T286A). 48 hours after transfection, cells were synchronized through the treatment with nocodazle (8 μg/ml) for 16 hours. Then cell cycle progression was analyzed by FACS.
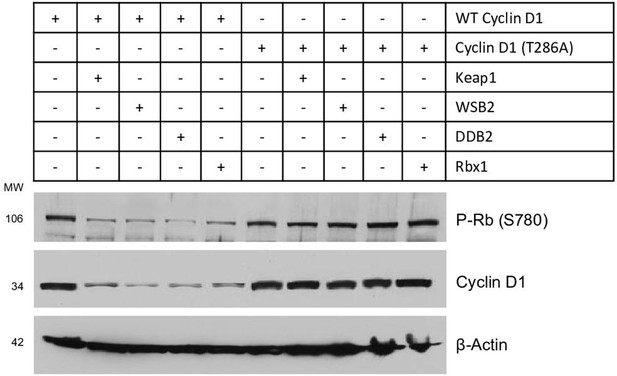
Cullin-associated E3 ligases decreased phospho-Rb expression.
WT or mutant cyclin D1 (T286A) expression plasmids were co-transfected with Keap1, WSB2, DDB2 or Rbx1 expression plasmids into HEK293 cells. 48 hours after transfection, changes in phospho-Rb and cyclin D1 protein levels were detected by the western blot analysis.

Cullin-associated E3 ligases couldn’t degrade K269R mutant cyclin D1.
WT or mutant cyclin D1 (K269R) expression plasmids were co-transfected with Keap1, DDB2, and AMBRA1 expression plasmids into HEK293 cells. 48 hours after transfection, changes in cyclin D1 protein levels were detected by the western blot analysis.

In vitro ubiquitination assay of cyclin D1 by Keap1, DDB2, WSB2, or AMBRA1.
In vitro ubiquitination of cyclin D1 by different Cullin-associated E3 ligases as indicated. Cyclin D1 and its ubiquitinated forms (bracket indicated) were detected by immunoblotting with an anti-cyclin D1 antibody. Mix: Mg-ATP, E1 (UAE), E2 (UBC9), Ubiquitin solution, IPP (Inorganic pyrophosphatase), DTT (DL-Dithiothreitol).





