An essential periplasmic protein coordinates lipid trafficking and is required for asymmetric polar growth in mycobacteria
Figures
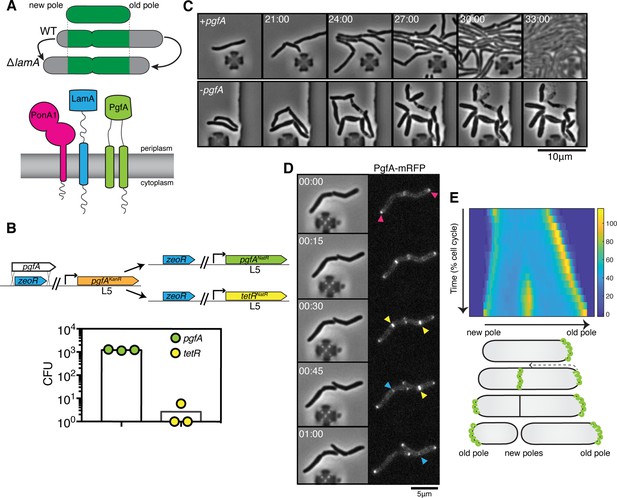
PgfA is an essential polar growth factor that localizes asymmetrically.
(A) Top: Graphical depiction of growth pattern in wild type (WT) and ΔlamA cells. Green = old cell wall material. Gray = new cell wall material. Bottom: LamA is a membrane protein that co-immunoprecipitates with PonA1, a bifunctional penicillin binding protein, and MSMEG_0317/PgfA, a protein of unknown function. (B) Schematic and results of allelic exchange experiment. Vectors with pgfA or without pgfA (tetR) were transformed into a strain whose only copy of pgfA was at the L5 integration site. Transformants carrying the incoming vectors were counted by colony forming units (CFU). (C) A strain whose only copy of PgfA is tetracycline inducible was imaged over time with (+pgfA) or without (-pgfA) anhydrotetracycline (ATC). Cells were loaded into a microfluidic device 18 hr after the removal of ATC (bottom) or a mock control (top). (D) Cells whose only copy of PgfA was fused to mRFP were imaged over time by phase and fluorescence microscopy in a microfluidic device with constant perfusion of media. (E) Top: Individual cells (N=25) were followed from birth to division and the fluorescence was measured from new to old pole. Each resulting kymograph was interpolated over cell length and time and then averaged together. Using this analysis, we find that PgfA is first at the old poles (pink triangles), partially re-localizes to the septum (yellow triangles) during cell division, and then disappears from the site of division before the next cell cycle (blue triangles) to establish asymmetry in the next generation. Bottom: A depiction of this localization pattern is shown as a cartoon.
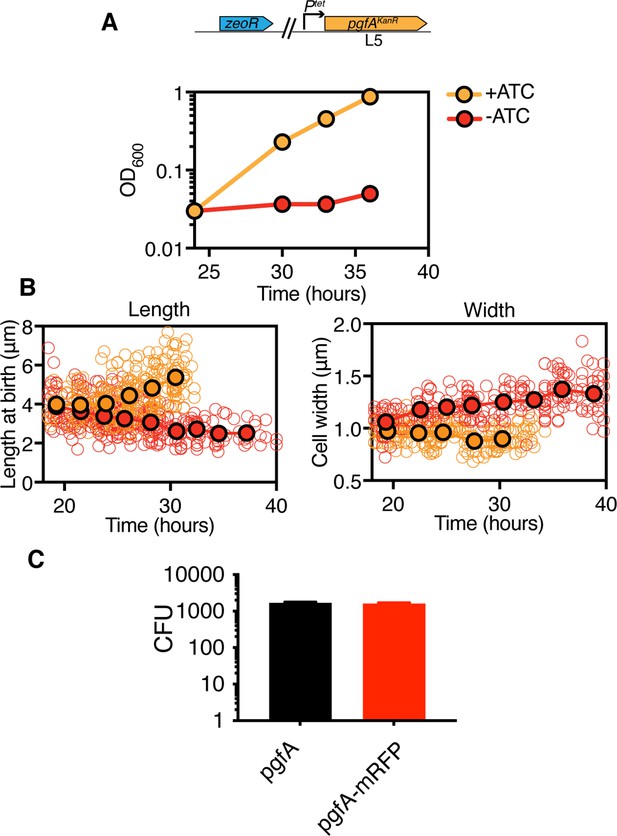
PgfA is essential and depletion leads to reductive cell division.
(A) Optical density over time of a strain whose only copy of pgfA is tetracycline inducible, with and without ATC (+/-ATC). Points are means of three biological replicates. Error bars are too small to be visible. Experiment was performed at least four times on separate days, and representative data are shown. ATC = anhydrotetracycline. Time on the x-axis corresponds to time after ATC was removed. (B) Birth lengths and cell widths over time of depletion (orange = +ATC; red = -ATC). Open circles represent individual cells (length: N=261 (length) and 253 (width) for +ATC; N=355 (length) and 254 (width) -ATC). Closed circles are not fit to the data but represent the mean of cells in 2.25 hr (length) or 2.75 hr (width) bins. The experiment was performed twice on separate days. In +ATC cultures, cell density became too high to measure length and width past ~33 hr. ATC = anhydrotetracycline (C) L5-allele swapping with pgfA or pgfA-mRFP expressed by the native promoter. Bars represent means of three biological replicates. Error bars indicate standard deviation.
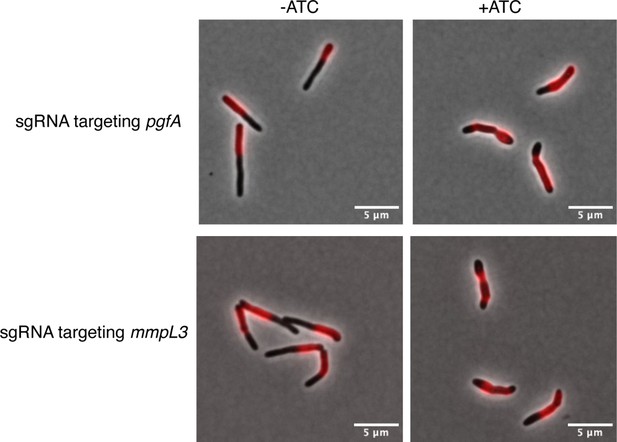
Cells depleted of PgfA and MmpL3 do not grow from their poles.
Cells from a log-phase culture were stained overnight with the fluorescent D-amino acid dye RADA at a final concentration of 2mM. The next morning, those cultures were split into two tubes, one of which received 100ng anhydrous tetracycline (aTC) to deplete either MmpL3 or PgfA. After growing for 5hr of depletion, all cells were washed of fluorescent dye, allowed to outgrow for 3hr, and imaged to visualize the amount of newly incorporated cell envelope.
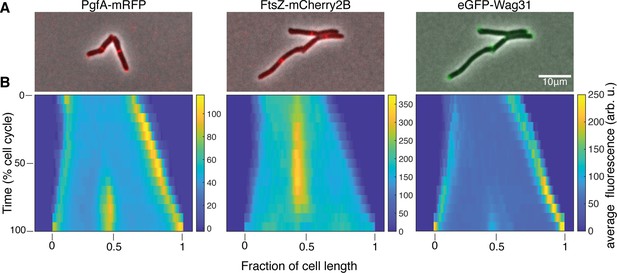
PgfA localizes to midcell and the poles.
(A) Merged phase contrast and fluorescence images of Msm cells expressing PgfA-mRFP, eGFP-Wag31, and FtsZ-mCherry2b. (B) Average fluorescent distributions over time (kymographs) from cells aligned from new to old poles and from birth to division (N=20 PgfA-mRFP; N=48 FtsZ-mCherry2B; N=20 eGFP-Wag31).
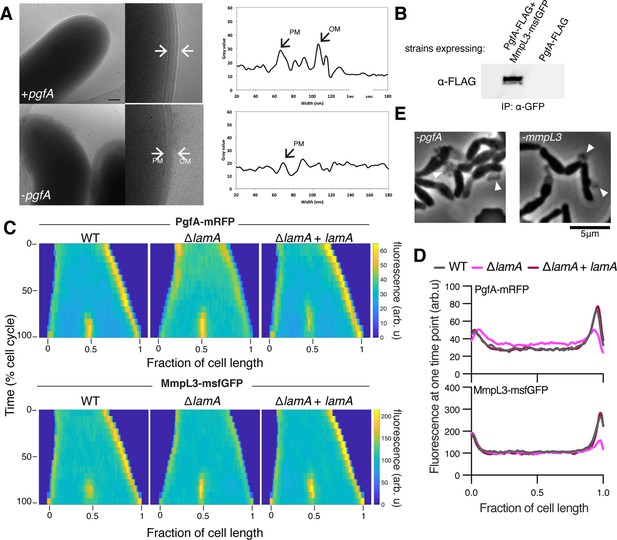
LamA recruits PgfA and MmpL3 to the old pole to build the mycomembrane.
(A) The cell envelope of a representative wild type Msm cell and a cell depleted of PgfA for 24 hr were visualized and measured by cryo-electron microscopy. PM = plasma membrane; OM = outer membrane. Scale bar = 100 nm. (B) The lysates from strains expressing PgfA-FLAG with or without MmpL3-msfGFP were immunoprecipitated (IP) using GFP-trap beads, and the elution was probed for the presence of PgfA-FLAG via anti-FLAG Western blot. (C) Fluorescent protein fusions to either PgfA or MmpL3 were imaged by time-lapse microscopy in wild type, ΔlamA, and complement cells (ΔlamA +pNative lamA) N=25. The resulting images were then processed as described in Figure 1D. (D) The same data as panel C are plotted at a single timepoint to show the decrease in old pole accumulation of MmpL3 and PgfA in the absence of lamA. (E) Cells were depleted using CRISPRi for pgfA or mmpL3 and a representative timepoint is shown. Cell wall material is excreted from the poles and septa during depletion (white triangles).
-
Figure 2—source data 1
Unprocessed anti-FLAG Western blot.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig2-data1-v2.zip
-
Figure 2—source data 2
Unprocessed and labeled anti-FLAG Western blot.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig2-data2-v2.zip

The cell envelopes of PgfA-depleted show outer membrane wall fraying at 24 hr (left) and 33 hr (right) of depletion.
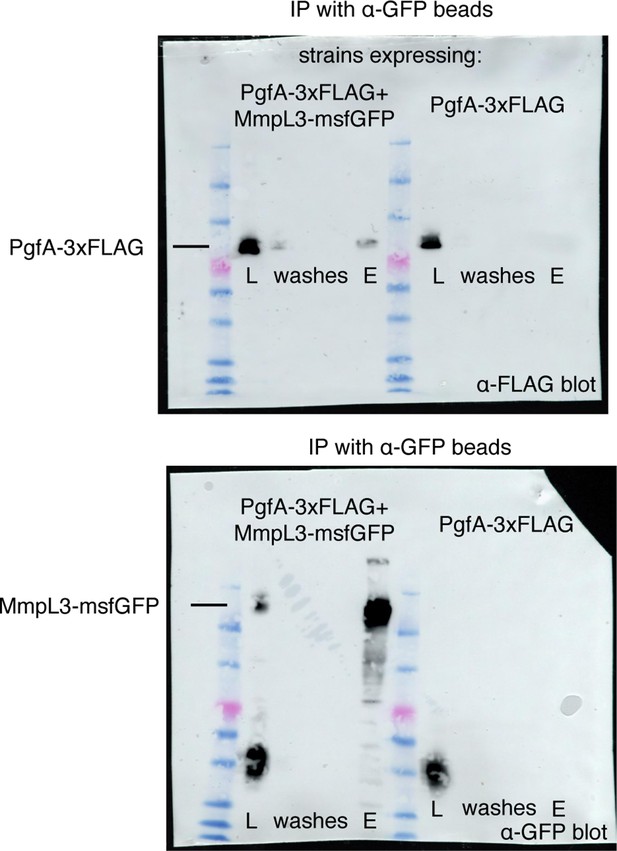
Full blots for the experiment in Figure 2B.
The indicated strains were lysed and incubated with anti-GFP beads. The lysates (L), washes, and elution (E), were run on two separate SDS-PAGE gels, transferred, and blotted with the indicated antibodies. The bands corresponding to PgfA-3× FLAG and MmpL3-msfGFP are shown.
-
Figure 2—figure supplement 2—source data 1
Unprocessed anti-FLAG Western blot.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig2-figsupp2-data1-v2.zip
-
Figure 2—figure supplement 2—source data 2
Unprocessed and labeled anti-FLAG Western blot.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig2-figsupp2-data2-v2.zip
-
Figure 2—figure supplement 2—source data 3
Unprocessed and labeled anti-GFP Western blot.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig2-figsupp2-data3-v2.zip
-
Figure 2—figure supplement 2—source data 4
Unprocessed and labeled anti-GFP Western blot.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig2-figsupp2-data4-v2.zip
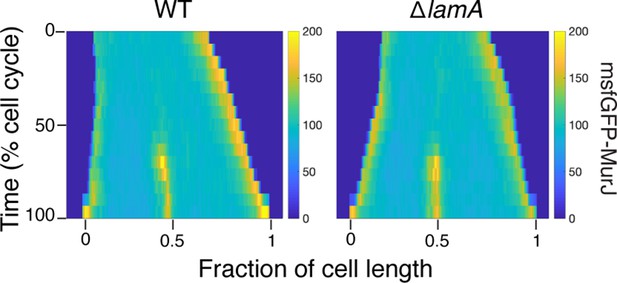
Kymographs of a fluorescent fusion to MurJ in wild type (WT) and in ΔlamA cells.
N=25.

An anti-strep Western blot of wild type (WT) Msm and Msm carrying an extra copy of MSMEG_0315-strep.
-
Figure 2—figure supplement 4—source data 1
Unprocessed anti-strep Western blot.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig2-figsupp4-data1-v2.zip
-
Figure 2—figure supplement 4—source data 2
Unprocessed and labeled anti-strep Western blot.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig2-figsupp4-data2-v2.zip
Phase time-lapse microscopy of cells carrying anhydrotetracycline (ATC)-inducible CRISRPi guides targeting either pgfA (left) or mmpL3 (right).
Exposure to ATC occurs at the beginning of the video.
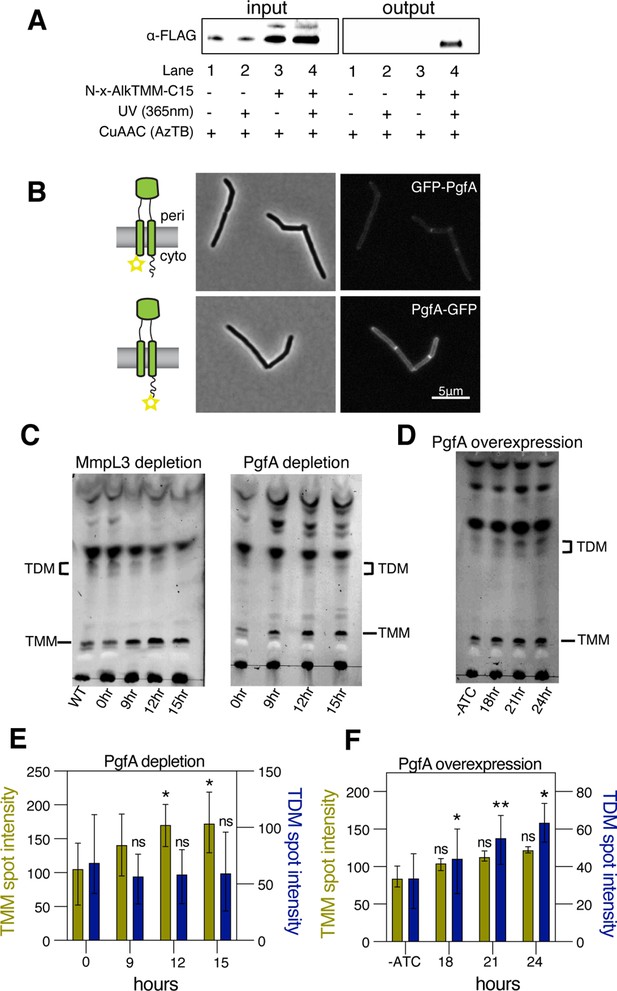
PgfA is a periplasmic protein that binds trehalose monomycolate (TMM) and is involved in TMM trafficking.
(A) Cells expressing PgfA-3× FLAG were cultured with N-x-AlkTMM-C15 (100 μM), UV-irradiated, and lysed. Lysates were reacted with azido-TAMRA-biotin reagent (AzTB) by Cu-catalyzed azide-alkyne cycloaddition (CuAAC ‘click’ reaction), and analyzed before (input) and after (output) avidin bead enrichment by anti-FLAG Western blot. The full analysis, including Coomassie and in-gel fluorescence scanning, is displayed in Figure 3—figure supplement 1. Data are representative of two independent experiments. (B) PgfA was fused to GFPmut3 at either its N- or C-terminus and integrated into Msm as the sole copy of PgfA. Cells carrying these fusions were imaged by fluorescence microscopy. Images are displayed on identical intensity scales to allow direct comparison. (C) Using CRISPRi, cells were depleted of MmpL3 or PgfA at the indicated timepoints and the cell envelopes were analyzed for TMM and trehalose dimycolate (TDM) by thin-layer chromatography (TLC). (D) Cells were induced to overexpress PgfA at the indicated timepoints, and their cell envelopes were analyzed by TLC for TMM and TDM. (E,F) Quantification of three biological replicates analyzed by TLC as shown in panels C and D. To determine statistical significance, a paired one-way ANOVA was performed that compared the intensity at the indicated timepoints to the intensity at time 0 or a no ATC control within the same replicate. *p<0.05; **p<0.005.
-
Figure 3—source data 1
Unprocessed MmpL3 depletion thin-layer chromatography (TLC).
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-data1-v2.zip
-
Figure 3—source data 2
Unprocessed and labeled MmpL3 depletion thin-layer chromatography (TLC).
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-data2-v2.zip
-
Figure 3—source data 3
Unprocessed PgfA depletion thin-layer chromatography (TLC).
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-data3-v2.zip
-
Figure 3—source data 4
Unprocessed and labeled PgfA depletion thin-layer chromatography (TLC).
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-data4-v2.zip
-
Figure 3—source data 5
Unprocessed PgfA overexpression thin-layer chromatography (TLC).
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-data5-v2.zip
-
Figure 3—source data 6
Unprocessed and labeled PgfA overexpression thin-layer chromatography (TLC).
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-data6-v2.zip

Full results of experiment present in main text Figure 3A.
Photoactivatable trehalose monomycolate (TMM) analog interacts with MSMEG_0317 in live Mycobacterium smegmatis. KG167 expressing 3× FLAG-tagged MSMEG_0317 was cultured with N-x-AlkTMM-C15 (100 μM), UV-irradiated, and lysed. Lysates were reacted with azido-TAMRA-biotin reagent (AzTB) by Cu-catalyzed azide-alkyne cycloaddition (CuAAC ‘click’ reaction), and analyzed before (input) and after (output) avidin bead enrichment. Input and output samples were analyzed by (A) Coomassie staining, (B) in-gel fluorescence scanning, and (C) anti-FLAG Western blot. Data are representative of two independent experiments. High-intensity band at low molecular weight in the Coomassie-stained gels represents lysozyme used in the cell lysis procedure.
-
Figure 3—figure supplement 1—source data 1
Unprocessed Coomassie gel.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-figsupp1-data1-v2.zip
-
Figure 3—figure supplement 1—source data 2
Unprocessed in-gel fluorescence scan.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-figsupp1-data2-v2.zip
-
Figure 3—figure supplement 1—source data 3
Unprocessed anti-FLAG Western blot.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-figsupp1-data3-v2.zip
-
Figure 3—figure supplement 1—source data 4
Full uncropped gels with relevant bands labeled.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-figsupp1-data4-v2.zip
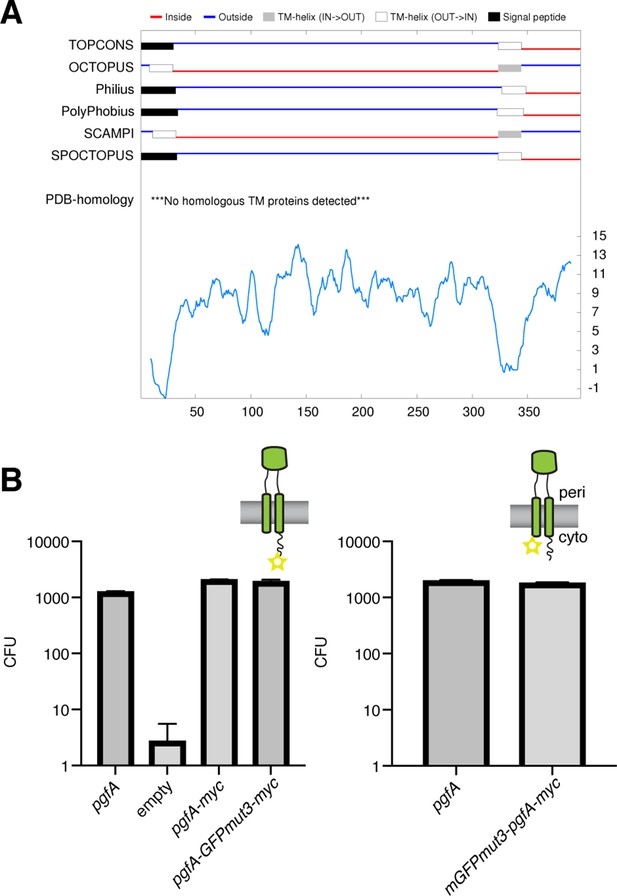
PgfA is a transmembrane protein with two cytoplasmic tails.
(A) Output from TOPCONS (https://topcons.cbr.su.se/), which aggregates the results of several transmembrane prediction algorithms. The input is MSMEG_0317, or PgfA. (B) Results of allele swapping with the indicated PgfA fusion proteins.
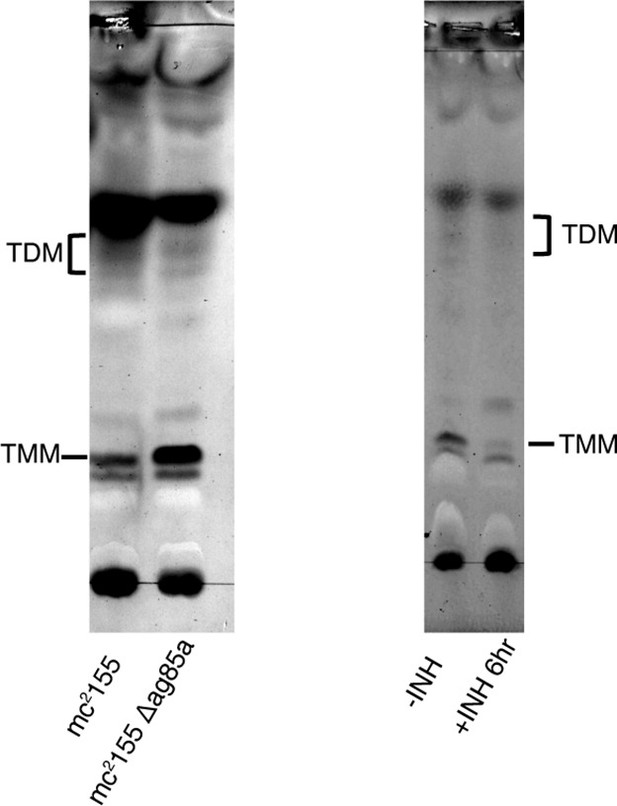
Control thin-layer chromatography (TLC) for TMM/TDM levels.
Left: Wild type (WT) Msm and Msm deleted for antigen85A (MSMEG_6398) were analyzed by thin-layer chromatography (TLC). Right: Msm was treated with 20 μg/ml isoniazid for 6 hr and analyzed by TLC.
-
Figure 3—figure supplement 3—source data 1
Unprocessed thin-layer chromatography (TLC) comparing wild type (WT) Msm to Msm Δantigen85A.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-figsupp3-data1-v2.zip
-
Figure 3—figure supplement 3—source data 2
Unprocessed and labeled thin-layer chromatography (TLC) comparing wild type (WT) Msm to Msm Δantigen85A.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-figsupp3-data2-v2.zip
-
Figure 3—figure supplement 3—source data 3
Unprocessed thin-layer chromatography (TLC) comparing wild type (WT) Msm +/-isoniazid.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-figsupp3-data3-v2.zip
-
Figure 3—figure supplement 3—source data 4
Unprocessed thin-layer chromatography (TLC) and labeled comparing wild type (WT) Msm +/-isoniazid.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig3-figsupp3-data4-v2.zip
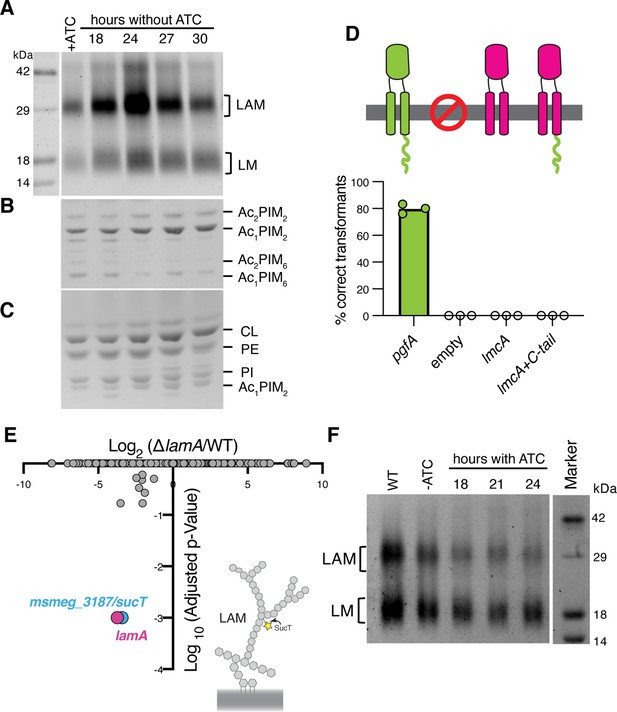
Lipomannan and lipoarabinomannan (LM/LAM) levels negatively correlate with abundance of PgfA.
In a strain whose only copy of pgfA is anhydrotetracycline (ATC) inducible, (A) LM/LAM, (B) non-polar and polar PIMs, (C) and plasma membrane lipids were analyzed by (A) SDS-PAGE and (B–C) thin-layer chromatography (TLC) during PgfA depletion (without ATC). Cell pellets were normalized by wet weight to account for differences in cell density. To avoid changes in LM/LAM due to cell density, at the indicated timepoints, half of the cultures were taken for lipid analysis, and replaced with fresh media. Two other biological replicates are shown in Figure 4—figure supplement 1. CL, cardiolipin; PE, phosphatidylethanolamine; PI, phosphatidylinositol. (D) Results of exchanging the indicated pgfA and Corynebacterium glutamicum NCgl2760 alleles for a wild type copy of pgfA. Correct transformants are positive for the incoming selective marker and negative for the outgoing (KanR, NatS). (E) Transposon insertions in ΔlamA compared to wild type (WT). The only two genes with significantly different number of insertions (reduced) were lamA itself and sucT, a gene that codes for a protein known to modify LAM and arabinogalactan. (F) In WT cells and those carrying an ATC-inducible copy of pgfA on a multi-copy plasmid, cell-associated LM/LAM were extracted, separated via SDS-PAGE, and visualized by ProQ Emerald with and without ATC at the indicated timepoints.
-
Figure 4—source data 1
Unprocessed lipomannan and lipoarabinomannan (LM/LAM) SDS-PAGE during PgfA depletion.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-data1-v2.zip
-
Figure 4—source data 2
Unprocessed PIM thin-layer chromatography (TLC) during PgfA depletion.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-data2-v2.zip
-
Figure 4—source data 3
Unprocessed plasma membrane thin-layer chromatography (TLC) during PgfA depletion.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-data3-v2.zip
-
Figure 4—source data 4
Unprocessed, lipomannan and lipoarabinomannan (LM/LAM) SDS-PAGE, PIM, and plasma membrane thin-layer chromatography (TLC) during PgfA depletion, with relevant bands labeled.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-data4-v2.zip
-
Figure 4—source data 5
Results of Tn-seq experiment comparing insertions in wild type and ΔlamA.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-data5-v2.xlsx
-
Figure 4—source data 6
Unprocessed lipomannan and lipoarabinomannan (LM/LAM) SDS-PAGE during PgfA overexpression.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-data6-v2.zip
-
Figure 4—source data 7
Unprocessed and labeled lipomannan and lipoarabinomannan (LM/LAM) SDS-PAGE during PgfA overexpression.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-data7-v2.zip
Two additional biological replicates of lipomannan and lipoarabinomannan (LM/LAM) analysis during PgfA depletion.
(A) As in main text Figure 4A, LM/LAM and other lipids were extracted and separated by SDS-PAGE and thin-layer chromatography (TLC) in cells expressing (+ATC) and depleted for (-ATC) PgfA. Two biological replicates are shown. (B) For the same replicates optical density was measured during depletion. To avoid changes in LM/LAM due to cell density, at the indicated timepoints, half of the cultures were taken for lipid analysis and replaced with fresh media.
-
Figure 4—figure supplement 1—source data 1
Unprocessed lipomannan and lipoarabinomannan (LM/LAM) SDS-PAGE during PgfA overexpression.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-figsupp1-data1-v2.zip
-
Figure 4—figure supplement 1—source data 2
Unprocessed PIM thin-layer chromatography (TLC) during PgfA overexpression, left-hand side.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-figsupp1-data2-v2.zip
-
Figure 4—figure supplement 1—source data 3
Unprocessed PIM thin-layer chromatography (TLC) during PgfA overexpression, right-hand side.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-figsupp1-data3-v2.zip
-
Figure 4—figure supplement 1—source data 4
Unprocessed plasma membrane thin-layer chromatography (TLC) during PgfA overexpression, left-hand side.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-figsupp1-data4-v2.zip
-
Figure 4—figure supplement 1—source data 5
Unprocessed plasma membrane thin-layer chromatography (TLC) during PgfA overexpression, right-hand side.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-figsupp1-data5-v2.zip
-
Figure 4—figure supplement 1—source data 6
Full uncropped gels with relevant bands labeled.
- https://cdn.elifesciences.org/articles/80395/elife-80395-fig4-figsupp1-data6-v2.zip
Corynebacterial LmcA cannot functionally replace PgfA in mycobacteria.
(A) Results of exchanging the indicated pgfA alleles for a wild type copy of pgfA. Bars represent medians. (B) Cell morphology analysis of a surviving transformant. Lines represent medians. (C) Results of exchanging the indicated pgfA alleles for a wild type copy of pgfA. Bars represent medians.
PgfA is sufficient to restore old pole growth in ΔlamA cells.
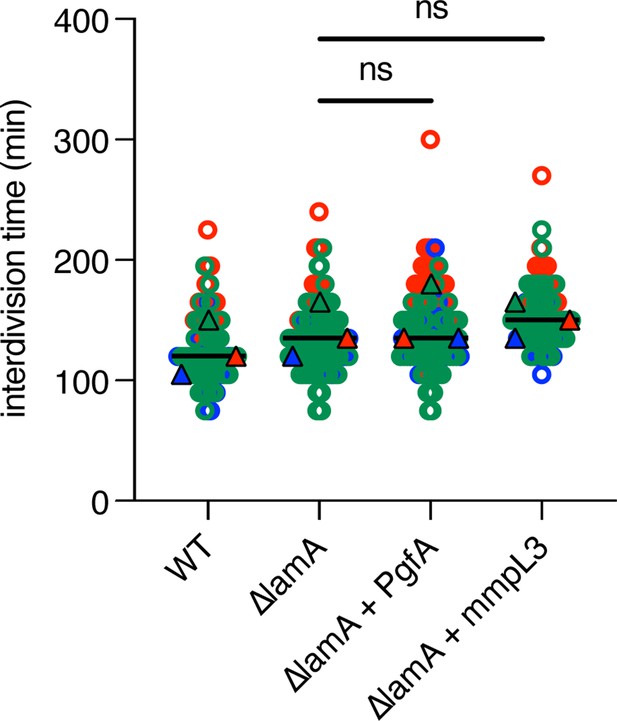
(A) Cells were stained with amine-reactive dye, loaded into a microfluidic device, and imaged while perfusing dye-free media. By following the amount of unlabeled cell wall material, growth at the old and new poles was measured over time. The amount of growth incorporated per cell cycle is compared across wild type (WT) cells, ΔlamA cells, and ΔlamA cells expressing a second copy of pgfA or mmpL3 driven by a strong promoter. To determine statistical significance, this procedure was repeated three times on separate days with independently grown cultures (N=3) (colors represent the replicates). For each replicate, a median was calculated (solid circles). A paired one-way ANOVA was performed comparing the medians of the replicates (aka a ‘super plot’). All strains were compared against ΔlamA. Solid lines represent the means of the solid circles. *p<0.05; **p<0.005. (B) Cells were stained with a fluorescent D-alanine analog, HADA, fixed, and imaged by fluorescence microscopy. MicrobeJ was used to segment the cells and measure intensity profiles along the long axis of the cell. Profiles were then aligned by the brightest pole and ordered by length as a proxy for cell cycle. (N=194 WT; N=259 ΔlamA; N=335 ΔlamA +lamA) (C) A model for asymmetric growth. PgfA is recruited by LamA to the old pole, where, together with other proteins, it functions to organize the mycobacterial cell envelope and promote fast cell elongation at one side of the cell.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mycobacterium smegmatis) | Mycobacterium smegmatis mc2155 | Wild type Mycobacterium smegmatis | ||
| Strain, strain background (Mycobacterium smegmatis) | KG26 | This work | mc2155 ∆MSMEG_0317 L5::pTetO-MSMEG_0317 | Parental swap strain |
| Strain, strain background (Mycobacterium smegmatis) | HR342 | Rego et al., 2017 | mc2155 ∆MSMEG_4265 | LamA knockout strain |
| Strain, strain background (Mycobacterium smegmatis) | KG97 | This work | mc2155 ∆MSMEG_0317 L5::pTetO-MSMEG_0317-myc / pTetR | Strain used for the anhydrotetracyline-inducible depletion of PgfA |
| Strain, strain background (Mycobacterium smegmatis) | KG81 | This work | mc2155 ∆MSMEG_0317 pL5::pNative-MSMEG_0317 -mRFP-myc | Strain used for determining PgfA localization |
| Strain, strain background (Mycobacterium smegmatis) | HR388 | Rego et al., 2017 | mc2155 L5::pG-MCK-ptb21-eGFP-wag31 tweety::ptb21-ftsZ-mCherry2B | Strain used to determine localization of FtsZ and Wag31 |
| Strain, strain background (Mycobacterium smegmatis) | CMG184 | This work | mc2155 tw::ptb21 mmpL3TB-msfGFP | Strain used to determine localization of MmpL3 |
| Strain, strain background (Mycobacterium smegmatis) | CMG530 | This work | mc2155 ∆MSMEG_0317 L5::pTetO-MSMEG_0317-3x- FLAG tw::ptb21-mmpL3TB-msfGFP | Strain used for co-immunoprecipitation of MmpL3 and PgfA |
| Strain, strain background (Mycobacterium smegmatis) | KG167 | This work | mc2155 ∆MSMEG_0317 L5::pTetO-MSMEG_0317-3x-FLAG | Control strain used for co-immunoprecipitation of MmpL3 and PgfA |
| Strain, strain background (Mycobacterium smegmatis) | CMG184 | This work | mc2155 tw::ptb21-mmpL3TB-msfGFP | Strain used to determine localization of MmpL3 |
| Strain, strain background (Mycobacterium smegmatis) | CMG481 | This work | mc2155 ∆lamA::zeo L5:: pNative-lamA-strep tw:: ptb21-mmpL3TB-msfGFP | Strain used to determine localization of MmpL3 in a ∆lamA background with LamA complemented under its native promoter |
| Strain, strain background (Mycobacterium smegmatis) | CMG557 | This work | mc2155 L5::pTetO-MSMEG_0317-myc | Strain used for measuring polar growth in a PgfA merodiploid background |
| Strain, strain background (M. smegmatis) | KG175 | This work | mc2155 ∆lamA L5::pTetO- MSMEG_0317-myc | Strain used for measuring polar growth in a PgfA merodiploid and LamA knockout background |
| Strain, strain background (Mycobacterium smegmatis) | CMG585 | This work | mc2155 L5::pTetO-mmpL3TB-myc | Strain used for measuring polar growth in an MmpL3 merodiploid background |
| Strain, strain background (Mycobacterium smegmatis) | CMG587 | This work | mc2155 ∆lamA L5:: pTetO-mmpL3TB-myc | Strain used for measuring polar growth in an MmpL3 merodiploid and LamA knockout background |
| Strain, strain background (Mycobacterium smegmatis) | KG154 | This work | mc2155 ∆lamA ∆MSMEG_0317 L5::pNative-MSMEG_0317 -mRFP-myc | Strain used for PgfA localization in LamA knockout background |
| Strain, strain background (Mycobacterium smegmatis) | KG157 | This work | mc2155 ∆lamA ∆MSMEG_0317 L5::pNative-MSMEG_0317 -mRFP-myc L5::pNative-lamA-strep | Strain used for PgfA localization in a background where lamA was complemented in LamA knockout |
| Strain, strain background (Mycobacterium smegmatis) | CMG206 | This work | mc2155 tw::ptb21-msfGFP-MurJTB | Strain used for localization of MurJ |
| Strain (Mycobacterium smegmatis) | CMG199 | This work | mc2155 ∆lamA tw::ptb21-msfGFP-murJTB | Strain used for localization of MurJ in a LamA knockout background |
| Strain, strain background (Mycobacterium smegmatis) | CMG485 | This work | mc2155 ∆lamA::zeo L5::pNative-LamA-strep tw::ptb21-msfGFP-MurJTB | Strain used for localization of MurJ in a LamA knockout background where LamA was complemented under its native promoter |
| Strain, strain background (Mycobacterium smegmatis) | KG244 | This work | mc2155 L5::pLJR962-mmpL3-sgRNA | CRISPR-inducible MmpL3 depletion strain |
| Strain, strain background (Mycobacterium smegmatis) | KG289/ CMG549 | This work | mc2155 L5::pLJR962- MSMEG_0317-sgRNA tw::pNative- MSMEG_0315-strep | CRISPR-inducible PgfA depletion strain |
| Strain, strain background (Mycobacterium smegmatis) | KG47 | This work | mc2155 ∆MSMEG_0317 L5::pTetO-MSMEG_0317-mGFPmut3-myc | C-terminal GFP tagged PgfA strain |
| Strain, strain background (Mycobacterium smegmatis) | KG73 | This work | mc2155 ∆MSMEG_0317 L5::pTetO-mGFP-mut3- MSMEG_0317-myc | N-terminal GFP tagged PgfA strain |
| Strain, strain background (Mycobacterium smegmatis) | KG167 | This work | mc2155 ∆MSMEG_0317 L5::pTetO- MSMEG_0317-3xFLAG | Strain carrying MSMEG_0317-3×-FLAG, used for TMM pulldown |
| Strain, strain background (Mycobacterium smegmatis) | KG60 | This work | mc2155 /pTetOR- MSMEG_0317-myc | Strain carrying tetracycline inducible overexpression plasmid for PgfA |
| Strain, strain background (Mycobacterium smegmatis) | KG286 | This work | mc2155 Δmsmeg_0317 L5::pTetO- MSMEG_0317-ΔC | Strain carrying msmeg_0317 without c-terminal cytoplasmic tail |
| Strain, strain background (Mycobacterium smegmatis) | HR404 | This work | mc2155 ΔMSMEG_6398 | Antigen 85A KO strain used in TLC control experiments |
| Recombinant DNA reagent | KG147 (plasmid) | This work | DH5α / pL5-pTetO-Ncgl2760-myc | Plasmid carrying corynebacterial ncgl2760 gene |
| Recombinant DNA reagent | KG284 (plasmid) | This work | DH5α / pL5-pTetO-Ncgl2760-MSMEG_0317-C-term | Plasmid carrying corynebacterial ncgl2760 gene fused to cytosolic C-terminal of msmeg_0317 gene |
| Recombinant DNA reagent | CMG541 (plasmid) | This work | Dh5a / pTweety-pNative MSMEG_0315-strep | Plasmid carrying mycobacterial gene MSMEG_0315 with a C-terminal strep tag |
Additional files
-
Supplementary file 1
A list of primers and oligos used in this study.
- https://cdn.elifesciences.org/articles/80395/elife-80395-supp1-v2.xlsx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/80395/elife-80395-mdarchecklist1-v2.docx