Optogenetic induction of appetitive and aversive taste memories in Drosophila
Figures
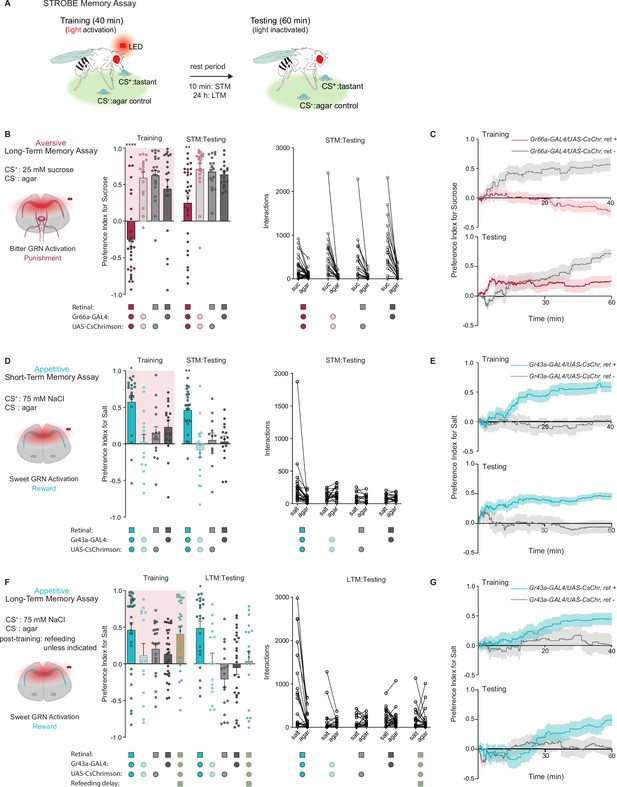
Gustatory receptor neurons (GRNs) produce punishment and reward signals capable of facilitating taste memory formation.
(A) Diagram outlining sip-triggered optogenetic behavioral enclosure (STROBE) memory paradigm. Training: 24 hr starved flies freely interact with a LED-activating tastant (CS+) and a non-LED-activating tastant (CS-) for 40 min. LED activation stimulates CsChrimson-expressing neurons. Testing: associative memory is measured by assessing a fly’s preference for the CS+ tastant compared to the CS- for 1 hr. In the short-term memory (STM) assay testing occurs 10 min after training. In the long-term memory (LTM) assay testing occurs 24 hr after training. (B) Aversive STM measured after pairing 25 mM sucrose (CS+) with bitter neuron optogenetic activation. Preference indices (left) and tastant interactions (right) for Gr66a>CsChrimson flies compared to controls during training and testing. The interaction numbers for individual flies are connected by lines. (C) Cumulative average preference indices over the course of training and testing in (B), (n=16–30). (D) Appetitive STM measured after pairing 75 mM NaCl (CS+) with sweet neuron optogenetic activation. Preference indices (left) and interactions (right) for Gr43a>CsChrimson flies compared to controls in the short-term memory assay. (E) Preference index of flies in (D) over time during training and testing (n=12–23). (F) Appetitive LTM measured after pairing of 75 mM NaCl (CS+) with sweet neuron optogenetic activation. Preference indices (left) and interactions (right) for Gr43a>CsChrimson flies compared to controls in the LTM assay. (G) Average preference index as a function of time for the training and testing in the LTM assay (n=14–30). All flies were starved for 24 hr prior to training. Preference index is mean ± SEM, Kruskal-Wallis with Dunn’s multiple comparison test: **p < 0.01, ****p < 0.0001.
-
Figure 1—source data 1
Raw data for Figure 1.
- https://cdn.elifesciences.org/articles/81535/elife-81535-fig1-data1-v2.xlsx
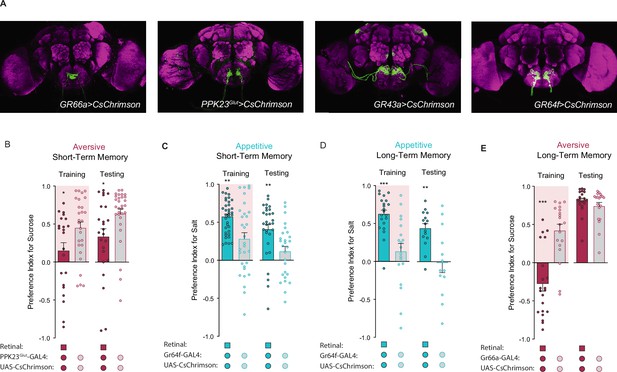
Gustatory receptor neuron (GRN) activation produces reward and punishment signals in taste memory formation.
(A) Projections of GR66a, Gr43a, PPK23Glut, and GR64f GRNs. (B) Aversive short-term memories (STM) measured following PPK23Glut>CsChrimson optogenetic activation paired with 25 mM sucrose (CS+) vs agar (CS-). Isogenetic flies not fed all-trans retinal serve as controls (n=22–30). (C, D) Appetitive STM (C) and long-term memories (LTM) (D) measured following Gr64f>CsChrimson optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) (n=28–36 in C and 13–21 in D). (E) Aversive LTM measured following Gr66a>CsChrimson optogenetic activation paired with 25 mM sucrose (CS+) vs agar (CS-) (n=19–21). All flies were starved for 24 hr prior to training. Preference indices are mean ± SEM, Mann-Whitney test: *p < 0.05, **p < 0.01, ***p < 0.001.
-
Figure 1—figure supplement 1—source data 1
Raw data for Figure 1—figure supplement 1.
- https://cdn.elifesciences.org/articles/81535/elife-81535-fig1-figsupp1-data1-v2.xlsx
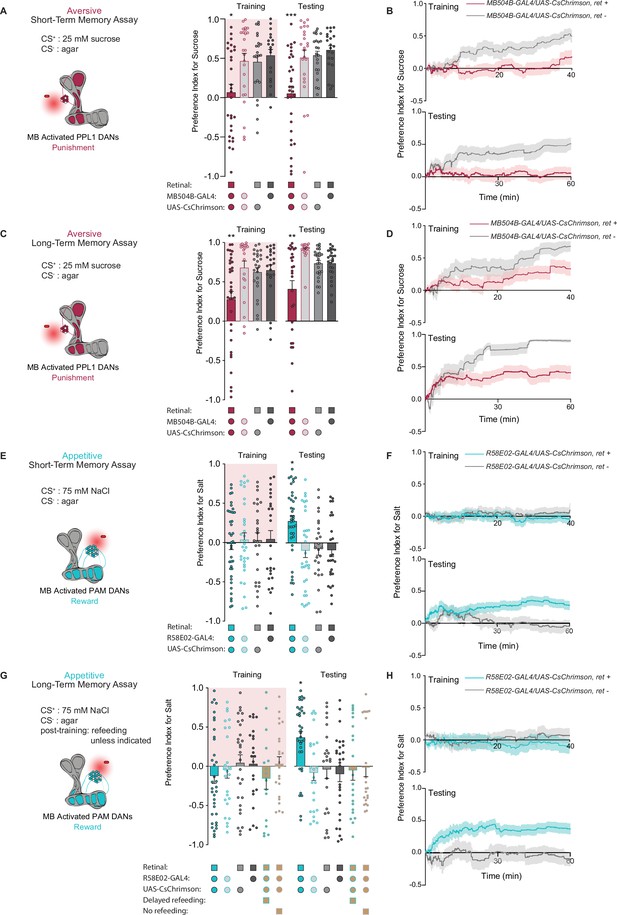
PPL1 and protocerebral anterior medial (PAM) neural activation is sufficient for the induction of short- and long-term taste memories.
(A) Aversive short-term memories (STM) measured following PPL1 neuron optogenetic activation paired with 25 mM sucrose (CS+) vs agar (CS-). Flies lacking retinal or one genetic element for expression of CsChrimson serve as controls (n=19–31). (B) Preference indices over time for the experiment shown in (A). (C) Aversive long-term memories (LTM) measured following PPL1 optogenetic activation paired with 25 mM sucrose (CS+) vs agar (CS-) (n=20–33). (D) Preference indices over time for the experiment shown in (C). (E) Appetitive STM measured following PAM neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) (n=25–38). (F) Preference indices over time for the experiment shown in (E). (G, H) Appetitive LTM measured following PAM neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) (n=17–35). Flies were refed with standard food for 1 hr directly after training unless otherwise indicated as delayed refeeding (8 hr after training) or no refeeding. (H) Preference indices over time for the experiment shown in (G). All flies were food deprived for 24 hr prior to the start of experimentation. Preference indices are mean ± SEM, Kruskal-Wallis with Dunn’s multiple comparison test: *p < 0.05, **p < 0.01, ***p < 0.001.
-
Figure 2—source data 1
Raw data for Figure 2.
- https://cdn.elifesciences.org/articles/81535/elife-81535-fig2-data1-v2.xlsx
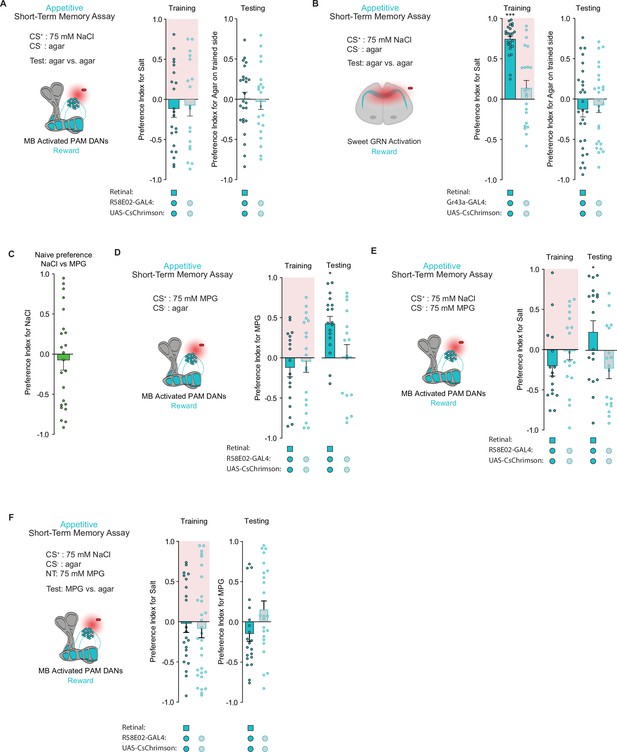
Taste memories are specific to the CS+.
(A) Appetitive short-term memories (STM) following protocerebral anterior medial (PAM) neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) and then measured as agar (CS-) vs agar (CS-) (n=21–25). (B) Appetitive STM following sweet neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) and then measured as agar (CS-) vs agar (CS-) (n=23–26). (C) Naïve preference between 75 mM NaCl and 75 mM monopotassium glutamate (MPG) (n=23). (D) Appetitive STM following PAM neuron optogenetic activation paired with 75 mM MPG (CS+) vs agar (CS-) (n=16–18). (E) Appetitive STM following PAM neuron optogenetic activation paired with 75 mM NaCl (CS+) vs 75 mM MPG (CS-) (n=16–19). (F) Appetitive STM following PAM neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) and then measured as preference for 75 mM MPG as a novel tastant (NT) vs agar (CS) (n=21–31). All flies were starved for 24 hr prior to training. Preference indices are mean ± SEM, Mann-Whitney test: *p < 0.05.
-
Figure 2—figure supplement 1—source data 1
Raw data for Figure 2—figure supplement 1.
- https://cdn.elifesciences.org/articles/81535/elife-81535-fig2-figsupp1-data1-v2.xlsx
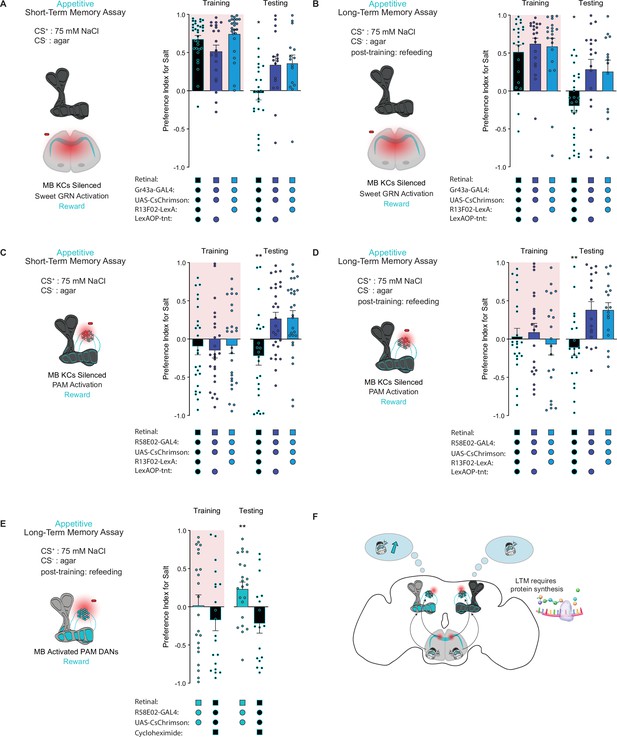
The mushroom body (MB) is required for the formation of short- and long-term taste memories.
(A, B) Appetitive short-term memories (STM) (A) and long-term memories (LTM) (B) measured following sweet taste neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) with Kenyon cells (KCs) silenced by expression of tetanus toxin (n=16–34 for STM and n=13–27 for LTM). Controls are missing one genetic element for KC silencing and therefore exhibit memory. (C, D) Appetitive STM (C) and LTM (D) measured following protocerebral anterior medial (PAM) neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) with KCs silenced by expression of tetanus toxin (n=24–28 for STM and n=17–23 for LTM). Controls are missing one genetic element for KC silencing and therefore exhibit memory. Memory assays when the MB is silenced, compared to controls. (E) Appetitive LTM measured following PAM neuron optogenetic activation with 75 mM NaCl (CS+) vs agar (CS-). Flies were either fed retinal or retinal plus cycloheximide (n=17–22). (F) Model of appetitive taste memory formation via gustatory receptor neuron (GRN)/PAM activation. All flies were starved for 24 hr prior to training. Preference indices are mean ± SEM, Kruskal-Wallis with Dunn’s multiple comparison test (A–D) or Mann-Whitney test (E): *p < 0.05, **p < 0.01.
-
Figure 3—source data 1
Raw data for Figure 3.
- https://cdn.elifesciences.org/articles/81535/elife-81535-fig3-data1-v2.xlsx
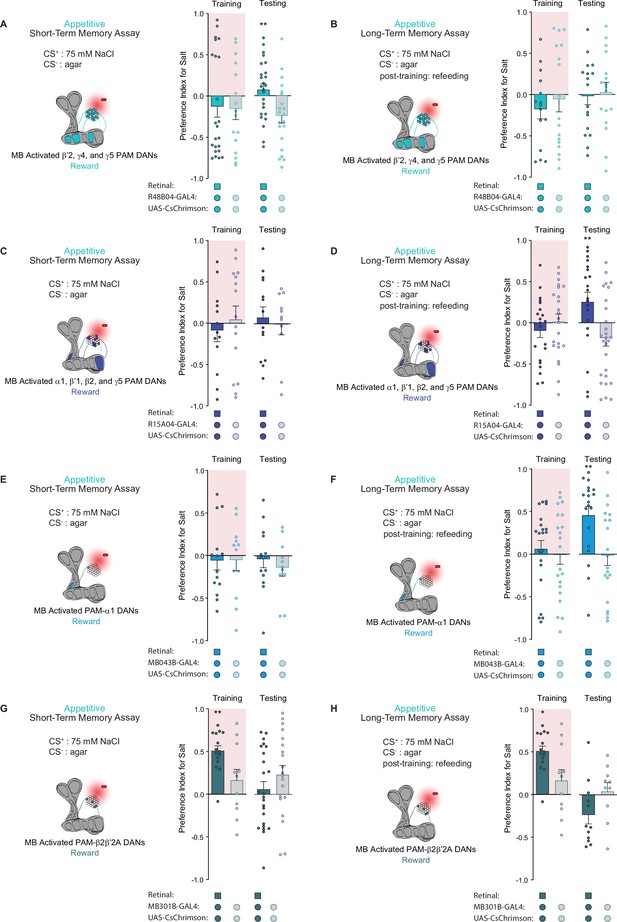
Distinct protocerebral anterior medial (PAM) subpopulations induce appetitive short- and long-term taste memories.
(A, B) Appetitive short-term memories (STM) (A) and long-term memories (LTM) (B) measured following β’2, γ4, and γ5 PAM neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) (n=21–28 for STM and n=15–17 for LTM). (C, D) Appetitive STM (C) and LTM (D) measured following α1, β’1, β2, and γ5 PAM neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) (n=11–15 for STM and n=20–27 for LTM). (E, F) Appetitive STM (E) and LTM (F) measured following PAM-α1 neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) (n=11–14 for STM and n=19–22 for LTM). (G, H) Appetitive STM (G) and LTM (H) measured following PAM-β2β′2a neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) (n=20–27 for STM and n=10–15 for LTM). All flies were starved for 24 hr prior to training. Preference indices are mean ± SEM, Mann-Whitney test: **p < 0.01.
-
Figure 4—source data 1
Raw data for Figure 4.
- https://cdn.elifesciences.org/articles/81535/elife-81535-fig4-data1-v2.xlsx
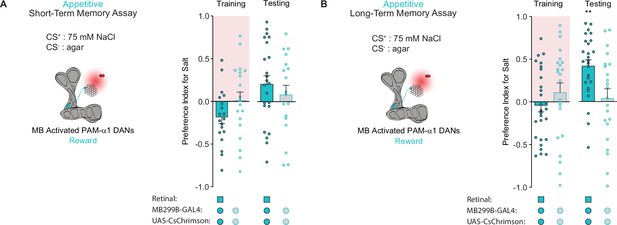
Activation of discrete protocerebral anterior medial (PAM) subpopulations induces distinct types of taste memories.
(A, B) Appetitive short-term memories (STM) (A) and long-term memories (LTM) (B) measured following PAM-α1 neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) (n=18–23 for STM and n=22–28 for LTM). All flies were starved for 24 hr prior to training. Preference indices are mean ± SEM, Mann-Whitney test: **p < 0.01 (n=18–26).
-
Figure 4—figure supplement 1—source data 1
Raw data for Figure 4—figure supplement 1.
- https://cdn.elifesciences.org/articles/81535/elife-81535-fig4-figsupp1-data1-v2.xlsx
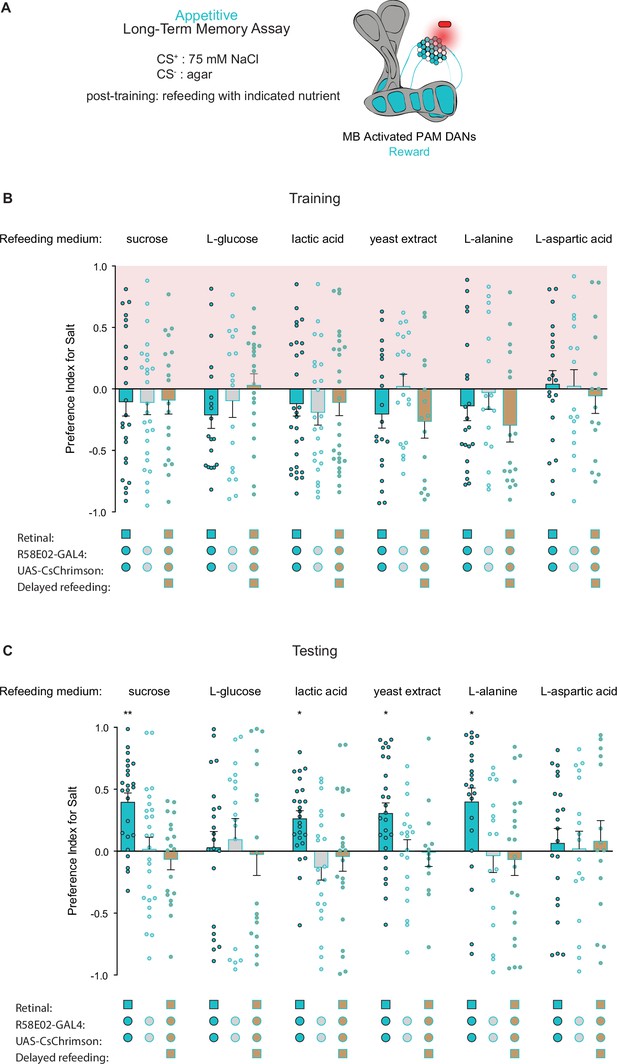
Caloric food sources are required for the formation of associative long-term taste memories.
(A) Schematic of the conditions and mushroom body (MB) compartments innervated by broad protocerebral anterior medial (PAM) driver R58E02-Gal4. (B, C) Training (B) and testing (C) of appetitive long-term memories (LTM) measured following PAM neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) (n=13–28). Flies were fed the indicated compounds for 1 hr immediately after training or after an 8 hr delay where indicated. All flies were starved for 24 hr prior to training. Preference indices are mean ± SEM, Kruskal-Wallis with Dunn’s multiple comparison test: *p < 0.05, **p < 0.01.
-
Figure 5—source data 1
Raw data for Figure 5.
- https://cdn.elifesciences.org/articles/81535/elife-81535-fig5-data1-v2.xlsx
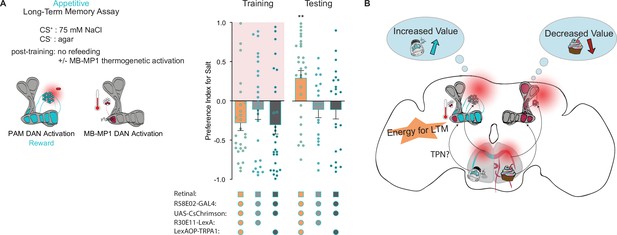
Mushroom body (MB)-MP1 neuron activation post training replaces energy signal required for the formation of long-term memories (LTM).
(A) Appetitive LTM measured following protocerebral anterior medial (PAM) neuron optogenetic activation paired with 75 mM NaCl (CS+) vs agar (CS-) (n=18 = 29). Flies were transferred to 29°C for 1 hr immediately after training to thermogenetically activate MB-MP1 using R30E11>TRPA1. Controls are lacking one genetic element for MB-MP1 thermogenetic activation. Graphic of timeline followed for the LTM taste assay with thermogenetic activation of MB-MP1 neurons. All flies were starved for 24 hr prior to training. Preference indices are mean ± SEM, Kruskal-Wallis with Dunn’s multiple comparison test: **p < 0.01.
-
Figure 6—source data 1
Raw data for Figure 6.
- https://cdn.elifesciences.org/articles/81535/elife-81535-fig6-data1-v2.xlsx





