Drosophila SUMM4 complex couples insulator function and DNA replication control
Figures
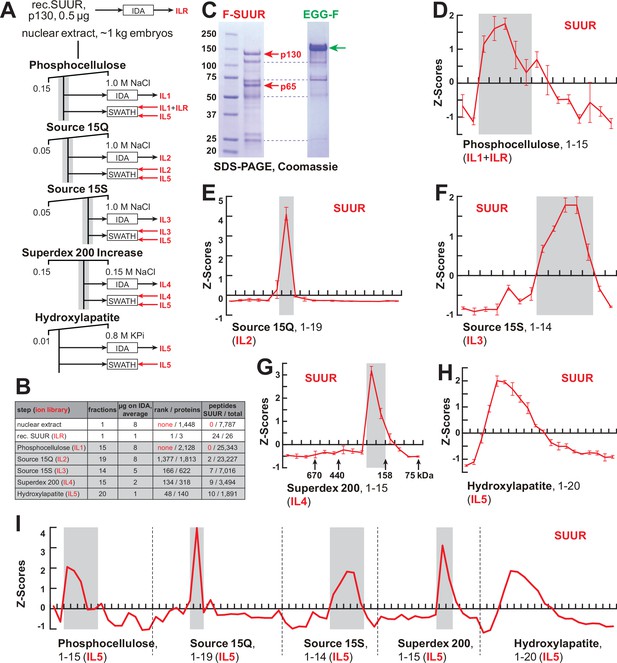
FPLC fractionation and MS-Enabled Rapid protein Complex Identification (MERCI) quantification of native SUUR.
(A) Schematic of FPLC purification of the native form of SUUR using MERCI approach. ILR, ion library obtained by information-dependent acquisitions (IDA) of recombinant FLAG-SUUR; IL1-5, ion libraries obtained by IDA of FPLC fractions from chromatographic steps 1–5. KPi, potassium phosphate, pH 7.6. (B) Representation of SUUR in ion libraries ILR and IL1-5 (Supplementary file 1). Total number of identified proteins and the confidence rank of SUUR among them as well as the total number of detected peptides (95% confidence) and the number of SUUR-specific peptides are shown. (C) Recombinant FLAG-SUUR expressed in Sf9 cells. Identities of eight most prominent bands were determined by mass-spectroscopy. p130 and p65 correspond to full-length and C-terminally truncated FLAG-SUUR, respectively (red arrows). Other bands represent common Sf9-specific contaminants purified by FLAG chromatography (blue dashed lines), cf. purified EGG-F (green arrow). Molecular mass marker bands are indicated (kDa). (D–H) SWATH quantitation profiles of SUUR fractionation across individual FPLC steps. Ion libraries (IL) used for SWATH quantitation are shown at the bottom of each panel. Z-scores across indicated column fractions are plotted; error bars, standard deviations (N = 3). Gray rectangles, fraction ranges used for the next FPLC step; in (G), black arrows, expected peaks of globular proteins with indicated molecular masses in kDa. (I) SWATH quantitation profiles of SUUR fractionation across five FPLC steps. IL5 ion library was used for SWATH quantification.
-
Figure 1—source data 1
FPLC column parameters (Figure 1A).
The following FPLC column parameters were used for partial purification of native SUMM4. HEG: 25 mM HEPES, pH 7.6, 0.1 mM EDTA, 10% glycerol, 0.02% NP-40, 1 mM DTT, 1 mM benzamidine, 0.4 mM PMSF; 10 mM KPi: 10 mM potassium phosphate, pH 7.6, 10% glycerol, 1 mM DTT, 1 mM benzamidine, 0.4 mM PMSF; 0.8 M KPi: 800 mM potassium phosphate, pH 7.6, 10% glycerol, 1 mM DTT, 1 mM benzamidine, 0.4 mM PMSF; cv, column volume.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig1-data1-v2.docx
-
Figure 1—source data 2
Recombinant proteins expressed in Sf9 cells and purified by FLAG affinitychromatography.
Lane 1, protein size marker; lane 2, FLAG-SUUR, 72 hr infection of Sf9 cells; lane 3, FLAG-SUUR, 60 hr infection of Sf9 cells; lane 4, XNP-FLAG (Emelyanov et al., 2010), 72 hr infection of Sf9 cells; lane 5, XNP-FLAG, 60 hr infection of Sf9 cells; lane 6, EGG-FLAG, 72 hr infection of Sf9 cells; lane 7, EGG-FLAG, 60 hr infection of Sf9 cells. Prep amounts equivalent to ~20 ml Sf9 culture were loaded in each lane. Cropped images encompassing lanes 1–2 and 6 (open boxes, dashed red line) were used for Figure 1C.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig1-data2-v2.zip
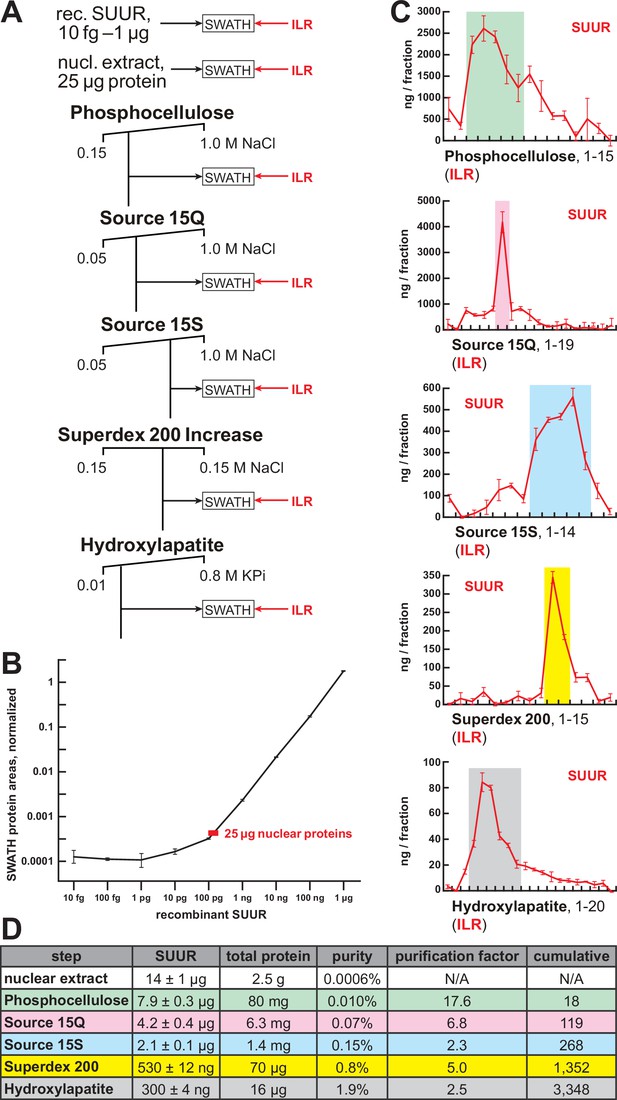
Quantification of SUUR in chromatographic fractions.
(A) Schematic of SWATH quantification of recombinant SUUR, nuclear extract (starting material) and FPLC fractions for SUUR using ion library ILR. (B) SUUR titration curve obtained by SWATH quantitation of 10 fg to 1 µg recombinant FLAG-SUUR in the presence of 25 µg E. coli lysate; both axes are logarithmic (log10). Red rectangle, SUUR quantification in 25 µg nuclear extract; error bars, standard deviations (N = 3). (C) SWATH quantitation profiles of SUUR fractionation across individual FPLC steps. Ion library ILR was used for SWATH quantification, and relative amounts were converted to estimated ng SUUR per fraction. Error bars, standard deviations (N = 3); colored boxes, peak fractions of SUUR. (D) SUUR purification by FPLC. Total protein was measured by BCA assay, and SUUR was measured as in (C). Relative purity, purification factor in each step and cumulative purification factor are shown.
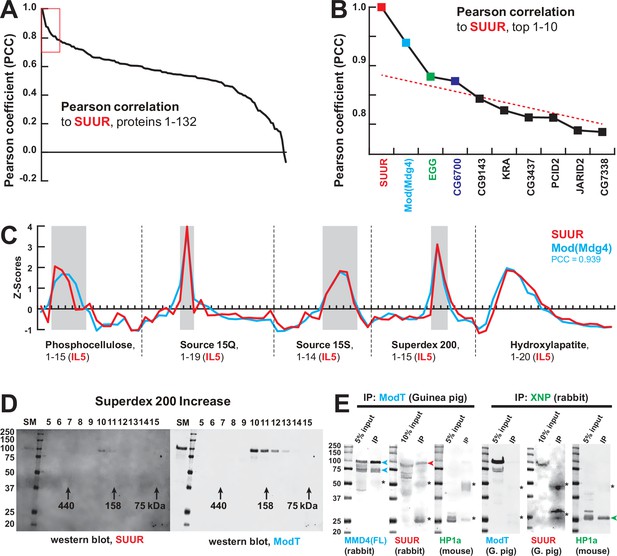
Identification of the SUMM4 complex by MS-Enabled Rapid protein Complex Identification (MERCI).
(A) Pearson correlation of fractionation profiles for individual 132 proteins to that of SUUR, sorted from largest to smallest. Red box, the graph portion shown in (B). (B) Top 10 candidate proteins with the highest Pearson correlation to SUUR. Red dashed line, trend line extrapolated by polynomial regression (n = 5) from the bottom 130 proteins. (C) SWATH quantitation profiles of SUUR (red) and Mod(Mdg4) (cyan) fractionation across five FPLC steps, Figure 1I. IL5 ion library was used for SWATH quantification. (D) Western blot analyses of Superdex 200 fractions with SUUR and ModT antibodies, Figure 1G. Molecular mass markers are shown on the left (kDa). (E) Co-IP experiments. SUUR (red arrowhead) co-purifies from nuclear extracts with Mod(Mdg4)-67.2 (cyan arrowheads) but not HP1a (green arrowhead). Anti-XNP co-IPs HP1a but not SUUR of Mod(Mdg4)-67.2. Asterisks, IgG heavy and light chains detected due to antibody cross-reactivity. Mod(Mdg4)-67.2(FL) antibody recognizes all splice forms of Mod(Mdg4).
-
Figure 2—source data 1
Western blots of chromatographic fractions.
Left panels, 700 nm channel (Odyssey Fc), rabbit anti-SUUR antibody and protein size marker; right panels, 800 nm channel (Odyssey Fc), guinea pig ModT antibody; top panels, hydroxylapatite fractions: starting material, flow-through, marker, fractions 1–12 (Figure 1H); bottom panels, Superdex 200 increase fractions: starting material, marker, fractions 5–15 (Figure 1G). Cropped images from bottom panels (open boxes, dashed red line) were used for Figure 2D.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig2-data1-v2.zip
-
Figure 2—source data 2
Co-IP of SUMM4 subunits.
(A, E) Westerns, 700 nm channel (Odyssey Fc), mouse anti-HP1a and protein size marker; (B) western, 800 nm channel (Odyssey Fc), rabbit anti-Mod(Mdg4)-FL; (C, G) Westerns, 700 nm channel (Odyssey Fc), protein sizemarker only; (D) Western, 800 nm channel (Odyssey Fc), rabbit anti-SUUR; (F) Western, 800 nm channel (Odyssey Fc), guinea pig ModT; (H) Western, 800 nm channel (Odyssey Fc), guinea pig anti-SUUR. Lanes 1, 5, 9, 12, 15, and 18, protein size marker; lanes 2, 6, 10, 13, 16, and 19, input (nuclear extract), 5 or 10%; lanes 3 and 7, IP with guinea pig ModT antibody #1; lanes 4 and 8, IP with guinea pig ModT antibody #2; lanes 11 and 17, IP with rabbit preimmune serum; lanes 14 and 20, IP with rabbit anti-XNP. Cropped images encompassing lanes 1–3, 5–7, 12–14, and 18–20 (open boxes, dashed red line) were used for Figure 2E.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig2-data2-v2.zip
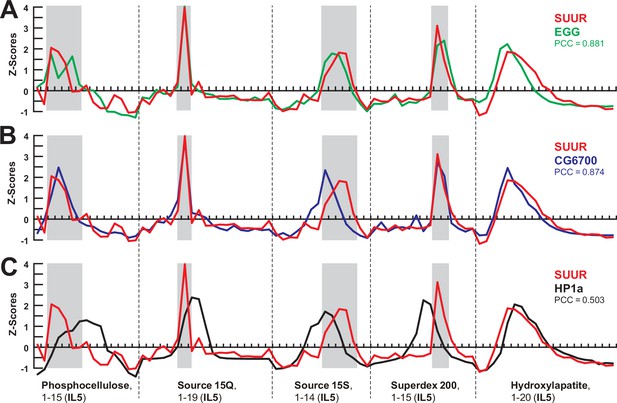
Comparisons of SWATH quantification profiles for protein fractionation.
(A–C) SWATH quantitation of SUUR (red), EGG (A, green), CG6700 (B, blue), and HP1a (C, black) fractionation profiles across five FPLC steps as in Figures 1I and 2C. Pearson coefficients (PCC) are shown (Figure 2A and B).
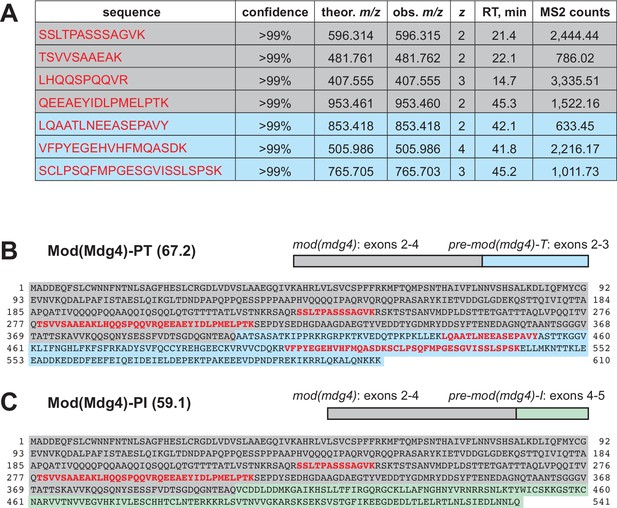
Identification of Mod(Mdg4)-67.2 as a subunit of the SUMM4 complex.
(A) Mod(Mdg4)-specific peptides from ion library IL5 (Supplementary file 1). Gray shading, peptides specific to the common part (coding exons 2–4) of Mod(Mdg4); cyan shading, peptides specific to polypeptide Mod(Mdg4)-67.2 encoded by pre-mod(mdg4)-T, exons 2–3. Peptide sequences, confidence levels, charges (z), theoretical and observed m/z, column retention times (RT), and total MS2 ion counts are shown. (B) Mod(Mdg4)-67.2 polypeptide sequence. The common part is shaded in gray, and splice form-specific part is shaded in cyan. Peptides from ion library IL5 (as in A) are highlighted in bold red. (C) Mod(Mdg4)-59.1 polypeptide sequence. The common part is shaded in gray, and splice form-specific part is shaded in light green.
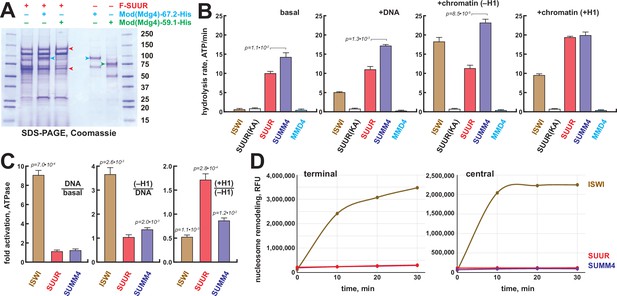
Biochemical activities of recombinant SUMM4.
(A) Recombinant SUMM4. Mod(Mdg4)-His6, 67.2 (p100, cyan arrowhead) and 59.1 (p75, green arrowhead) splice forms were co-expressed with FLAG-SUUR (red arrowheads, p130 and p65) or separately in Sf9 cells and purified by FLAG or Ni-NTA affinity chromatography. Mod(Mdg4)-67.2 forms a specific complex with SUUR. Identities of the 130, 100, 75, and 65 kDa protein bands from FLAG- and Ni-NTA-purified material were determined by mass spectroscopy. (B) ATPase activities of recombinant ISWI (brown bars), FLAG-SUUR (red bars), and SUMM4 (FLAG-SUUR + Mod(Mdg4)-67.2-His6, purple bars). Equimolar amounts of proteins were analyzed in reactions in the absence or presence of plasmid DNA or equivalent amounts of reconstituted oligonucleosomes,±H1. SUUR(KA) and MMD4, ATPases activities of K59A mutant of SUUR (gray bars) and Mod(Mdg4)-67.2-His6 (cyan bars). Hydrolysis rates were converted to moles ATP per mole protein per minute. All reactions were performed in triplicate (N=3), error bars represent standard deviations. p-Values for statistically significant differences are indicated (Mann–Whitney test). (C) DNA- and nucleosome-dependent stimulation or inhibition of ATPase activity. The activities were analyzed as in (B). Statistically significant differences are shown (Mann–Whitney test). (D) Nucleosome sliding activities by EpiDyne-PicoGreen assay (see ‘Materials and methods’) with 5 nM of recombinant ISWI, SUUR, or SUMM4. Reaction time courses are shown for terminally (6-N-66) and centrally (50-N-66) positioned mononucleosomes (Figure 3—figure supplement 2B–E). RFU, relative fluorescence units produced by PicoGreen fluorescence.
-
Figure 3—source data 1
Recombinant proteins expressed in Sf9 cells and purified by FLAG or Ni-NTA affinity chromatography.
Lanes 1 and 7, protein size marker; lane 2, FLAG-SUUR, FLAG-purified; lane 3, FLAG-SUUR + Mod(Mdg4)-67.2-His6, FLAG-purified; lane 4, FLAG-SUUR + Mod(Mdg4)-59.1-His6, FLAG-purified; lane 5, Mod(Mdg4)-67.2-His6, Ni-NTA-purified; lane 6, Mod(Mdg4)–-9.1-His6, Ni-NTA-purified. All proteins were purified 72 hr post-infection. Prep amounts equivalent to ~20 ml (FLAG-purified, lanes 2–4) or ~1 ml (Ni-NTA-purified, lanes 5 and 6) Sf9 cultures were loaded in each lane. Cropped image encompassing all lanes (open box, dashed red line) was used for Figure 3A.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig3-data1-v2.zip
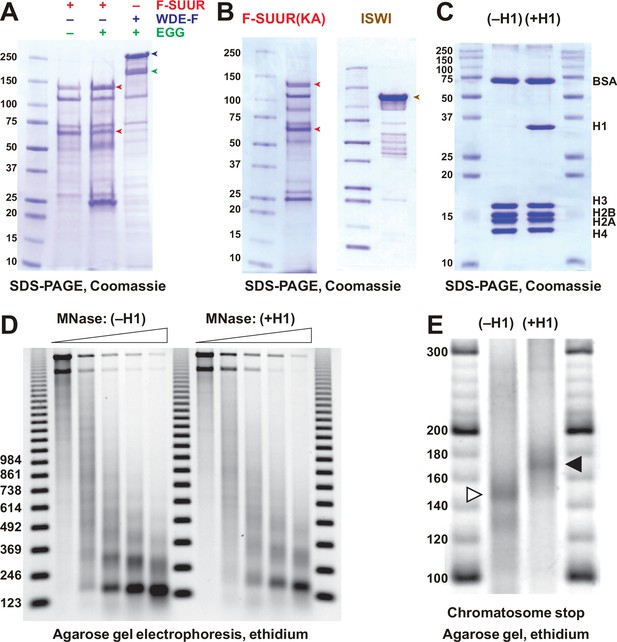
Recombinant proteins and biochemical substrates.
(A) Physical interactions of recombinant EGG, SUUR, and WDE. Untagged EGG (green arrowhead) was co-expressed with FLAG-SUUR (red arrowheads, p130 and p65) or WDE-FLAG (purple arrowhead) in Sf9 cells and purified by FLAG affinity chromatography. EGG forms a specific complex with WDE but not SUUR. Molecular mass markers (kDa) are shown on the left. (B) Recombinant FLAG-SUUR(K59A) expressed in Sf9 cells and ISWI expressed in E. coli. See legend to Figure 1C. (C) Protein composition of in vitro reconstituted chromatin. Oligonucleosomes prepared from plasmid DNA and core histones with (+H1) or without H1 (–H1) were analyzed by SDS-PAGE and Coomassie staining. Positions of BSA, H1, and core histone bands are indicated on the right. (D) Micrococcal nuclease (MNase) analysis of reconstituted chromatin. Partial digestion with five different dilutions of MNase was performed on H1-free (–H1) and H1-containing (+H1) oligonucleosomes. Deproteinated DNA fragments were analyzed by agarose gel electrophoresis and stained with ethidium. Note the increased nucleosome repeat length in (+H1) lanes consistent with H1 incorporation. Triangles at the top indicate increasing MNase concentrations; 123 bp ladder was used as a molecular mass marker. (E) Chromatosome stop assay. Oligonucleosomes assembled with or without H1 were subjected to partial MNase digestion, and DNA was analyzed by agarose gel electrophoresis and ethidium bromide staining. Positions of the core particle and chromatosome DNA are indicated by arrowheads. DNA fragment sizes in the 20 bp DNA ladder marker are shown.
-
Figure 3—figure supplement 1—source data 1
Recombinant proteins expressed in Sf9 cells and purified by FLAG or Ni-NTA affinity chromatography.
Lane 1, protein size marker; lane 2, FLAG-SUUR, FLAG- purified; lane 3, FLAG-SUUR + EGG untagged, FLAG-purified; lane 4, WDE-FLAG + EGG untagged, FLAG-purified; lane 5, EGG-FLAG, FLAG-purified; lane 6, EGG-FLAG + His6-SUUR, FLAG-purified; lane 7, WDE-FLAG + EGG untagged + His6-SUUR, FLAG-purified; lane 8, His6-SUUR, Ni-NTA-purified; lane 9, EGG-FLAG + His6-SUUR, Ni-NTA-purified; lane 10, WDE-FLAG + EGG untagged + His6-SUUR, Ni-NTA-purified. All proteins were purified 72 hr post-infection. Prep amounts equivalent to ~20 ml Sf9 cultures were loaded in each lane. Cropped image encompassing lanes 1–4 (open box, dashed red line) was used for Figure 3—figure supplement 1A.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig3-figsupp1-data1-v2.zip
-
Figure 3—figure supplement 1—source data 2
Recombinant proteins expressed in Sf9 or E. coli cells and purified by FLAG, Ni-NTA, or chitin affinity chromatography.
Lanes 1, 7, and 10, protein size marker; lane 2, FLAG-SUUR(K59A), FLAG-purified; lane 3, FLAG-SUUR(K59R), FLAG-purified; lane 4, FLAG-SUUR wild-type, FLAG-purified; lane 5, His6-SUUR wild-type, Ni-NTA-purified. All proteins were purified 72 hr post-infection. Prep amounts equivalent to ~20 ml Sf9 cultures were loaded in each lane. Lane 6, FLAG-ISWI (Sf9 ells), FLAG-purified, 2 µg; lane 8, ISWI untagged (E. coli), chitin-purified, 0.5 µg; lane 9, ISWI untagged (E. coli), chitin-purified, 1 µg; lane 11, ISWI untagged (E. coli), chitin-purified, 2 µg. Cropped images encompassing lanes 1–2 and 10–11 (open boxes, dashed red line) were used for Figure 3—figure supplement 1B.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig3-figsupp1-data2-v2.zip
-
Figure 3—figure supplement 1—source data 3
SDS-PAGE of salt dialyzed chromatin ±H1.
Lanes 1 and 4, protein size marker; lane 2, oligonucleosomes, 2 µg DNA; lane 3, oligonucleosomes + H1, 2 µg DNA. Cropped image encompassing all lanes (open box, dashed red line) was used for Figure 3—figure supplement 1C.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig3-figsupp1-data3-v2.zip
-
Figure 3—figure supplement 1—source data 4
Micrococcal nuclease (MNase) digest of salt-dialyzed chromatin ± H1, 1.25% agarose gel, ethidium-stained.
Lanes 1, 7, and 13, 123-bp DNA ladder; lanes 2–6, oligonucleosomes, 250 ng DNA; lanes 8–12, oligonucleosomes + H1, 250 ng DNA; lanes 2 and 8, digested with 10-4 units MNase for 15 min at room temperature (RT); lanes 3 and 9, digested with 3•10-4 units MNase for 15 min at RT; lanes 4 and 10, digested with 10-3 units MNase for 15 min at RT; lanes 5 and 11, digested with 3•10-3 units MNase for 15 min at RT; lanes 6 and 12, digested with 10-2 units MNase for 15 min at RT. Cropped image encompassing all lanes (open box, dashed red line) was used for Figure 3—figure supplement 1D.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig3-figsupp1-data4-v2.zip
-
Figure 3—figure supplement 1—source data 5
Micrococcal nuclease (MNase) digest of salt-dialyzed chromatin ± H1, 3% agarose gel, ethidium-stained (chromatosome stop assay).
Lanes 1, 4, 5, 8, 9, 12, 13, and 16, 20-bp DNA ladder; lanes 2, 6, 10, and 14, oligonucleosomes; lanes 3, 7, 11, and 15, oligonucleosomes +H 1; lanes 2 and 3, digested with 5•10-3 units MNase for 15 min at room temperature (RT); lanes 6 and 7, digested with 1.5•10-2 units MNase for 15 min at RT; lanes 10 and 11, digested with 10-2 units MNase for 15 min at RT; lanes 14 and 15, digested with 3•10-2 units MNase for 15 min at RT; lanes 2, 3, 6, and 7, 125 ng DNA; lanes 10, 11, 14, and 15, 250 ng DNA. Cropped image (open box, dashed red line) was used for Figure 3—figure supplement 1E.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig3-figsupp1-data5-v2.zip
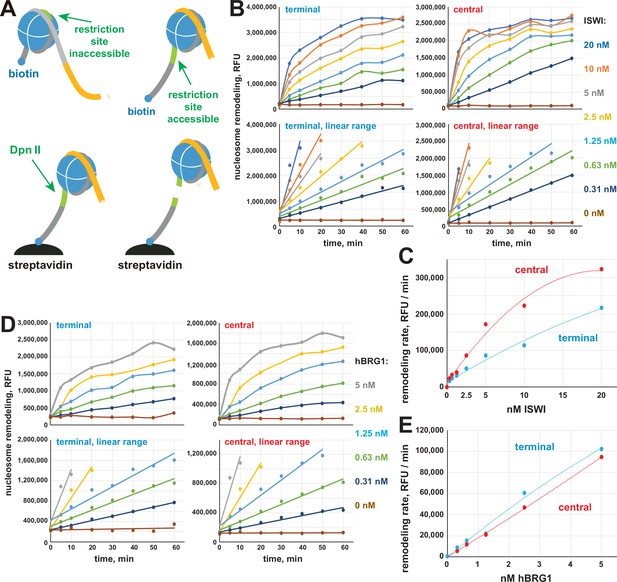
EpiDyne-PicoGreen biochemical assay.
(A) EpiCypher EpiDyne-PicoGreen assay design. EpiDyne nucleosomes encompass a restriction site shielded by the initial nucleosome position but exposed for Dpn II cleavage upon remodeling (sliding or displacement). Biotinylated substrates are immobilized on streptavidin magnetic beads. Digest by Dpn II releases the substrates from beads, and supernatant is quantified by PicoGreen (dsDNA detection reagent) fluorescence. (B) Titration of Drosophila ISWI remodeling activity using terminally (6-N-66) or centrally (50-N-66) positioned mononucleosomes. Early reaction time points were separately plotted to indicate linear ranges. RFU, relative fluorescence units. (C) Early remodeling rates for ISWI were calculated by linear regression analyses of data in respective linear ranges. ISWI exhibits a stronger remodeling activity with a centrally positioned nucleosome substrate. (D) Titration of human BRG1 remodeling activity. Data are presented as in (B). (E) Early remodeling rates for BRG1 were calculated and plotted as in (D). BRG1 does not exhibit a bias toward remodeling centrally or terminally positioned nucleosomes.
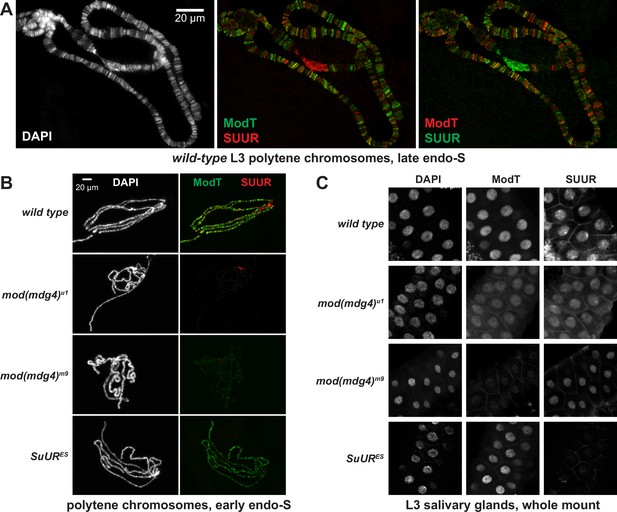
Spatiotemporal distribution of SUMM4 in vivo.
(A) Colocalization of SUUR and Mod(Mdg4)-67.2 in wild-type polytene chromosomes. Localization patterns of Mod(Mdg4)-67.2 and SUUR in L3 polytene chromosomes were analyzed by indirect immunofluorescence (IF) staining. The polytene spread fragment (3L and 3R arms) corresponds to a nucleus in late endo-S phase, according to PCNA staining (Figure 4—figure supplement 1A). Left panel, DAPI staining shows the overall chromosome morphology. Middle panel, ModT (green) and SUUR (red) signals overlap extensively in euchromatic arms. Right panel, a colocalization image with swapped red (ModT) and green (SUUR) channels is shown for comparison. Note the additional strong ModT IF loci that are SUUR-free as well as Mod(Mdg4)-67.2-free SUUR in pericentric 3LR. (B) SUUR loading into chromosomes during early endo-S phase is compromised in mod(mdg4) mutants. SuUR mutation does not appreciably change the distribution of Mod(Mdg4)-67.2. Endo-S timing was established by PCNA staining (Figure 4—figure supplement 3B). (C) Abnormal subcellular distribution of SUMM4 subunits in mod(mdg4) and SuUR mutants. L3 salivary glands were fixed and whole-mount-stained with DAPI, ModT, and SUUR antibodies. Whereas both polypeptides are mostly nuclear in wild-type, they are partially mis-localized to the cytoplasm in mod(mdg4)u1 mutant.
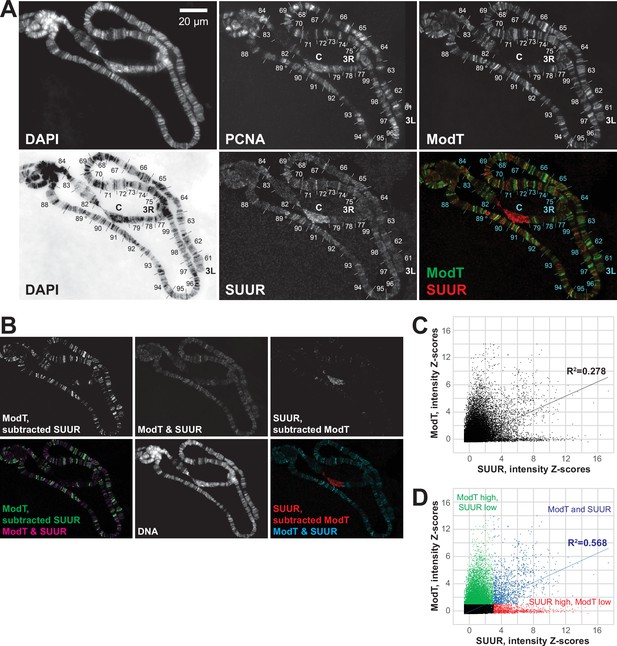
Spatial distribution of SUUR and Mod(Mdg4)-67.2 in polytene chromosomes and analyses of their colocalization.
(A) Colocalization of SUUR and Mod(Mdg4)-67.2 in wild-type polytene chromosomes. See legend to Figure 4A. 3L and 3R telomeres are marked; approximate boundaries of cytological regions are shown according to Lefevre, 1976; positions of intercalary heterochromatin regions 75C and 89E that are underreplicated and responsive to SuUR mutation are marked by circles. (B) The patterns of colocalization and independent loading of SUUR and Mod(Mdg4)-67.2 in wild-type polytene chromosomes. Subtracted and overlapping images were produced in ImageJ (‘Materials and methods’). Green, enriched Mod(Mdg4)-67.2 and low SUUR; red, enriched SUUR and low Mod(Mdg4)-67.2; magenta or cyan, overlapping enriched Mod(Mdg4)-67.2 and SUUR. (C) Quantification of the overlap between SUUR and Mod(Mdg4)-67.2 in wild-type polytene chromosomes (Figure 4A). Individual pixel intensities of anti-SUUR and anti-ModT IF signals are normalized to Z-scores and plotted on x- and y-axes, respectively (’Materials and methods’); they exhibit a weak positive correlation (R2 > 0.2). (D) Visually, the 2D plot (C) is split into four separate areas demarcated by ZModT = 1 and ZSUUR = 3. When pixels representing ModT-only and SUUR-only areas (green and red, respectively) are removed, the remaining pixels that are simultaneously enriched for Mod(Mdg4)-67.2 and SUUR (blue) exhibit a strong positive correlation (R2 > 0.5).

Alternative complex(es) of Mod(Mdg4)-67.2.
(A) Schematic of partial FPLC purification of an alternative complex of Mod(Mdg4)-67.2. Cyan boxes, fraction ranges used for the next chromatographic step. (B) Western blot analyses of Q Sepharose FF fractions with SUUR and ModT antibodies. SUUR and ~25% total Mod(Mdg4)-67.2 present in the starting material (SM) fractionate in the flow-through (FT, arrows), whereas Mod(Mdg4)-67.2 also fractionates as an additional, SUUR-free peak (cyan box). Molecular mass markers are as in Figure 2D. (C) Western blot analysis of Source 15S fractions with the ModT antibody. (D) Western blot analyses of Superose 6 fractions with the ModT antibody. Black arrows, expected peaks of globular proteins with indicated molecular masses in kDa.
-
Figure 4—figure supplement 2—source data 1
FPLC column parameters (Figure 4—figure supplement 2A).
The following FPLC column parameters were used for partial purification of an alternative complex of Mod(Mdg4)-67.2. HEG: 25 mM HEPES, pH 7.6, 0.1 mM EDTA, 10% glycerol, 0.02% NP-40, 1 mM DTT, 1 mM benzamidine, 0.4 mM PMSF; cv, column volume.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig4-figsupp2-data1-v2.docx
-
Figure 4—figure supplement 2—source data 2
Western blots of chromatographic fractions.
Left panels, 700 nm channel (Odyssey Fc), rabbit anti-SUUR antibody and protein size marker; right panels, 800 nm channel (Odyssey Fc), guinea pig ModT antibody; top panels, Q Sepharose FF fractions: starting material, flow-through, marker, fractions 1–10; bottom panels, Q Sepharose FF fractions: fractions 11–22, marker. Cropped and spliced images encompassing all panels (open boxes, dashed red line) were used for Figure 4—figure supplement 2B.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig4-figsupp2-data2-v2.zip
-
Figure 4—figure supplement 2—source data 3
Western blots of chromatographic fractions.
Left panels, 700 nm channel (Odyssey Fc), rabbit anti-SUUR antibody and protein size marker; right panels, 800 nm channel (Odyssey Fc), guinea pig ModT antibody; top panels, Source 15S fractions: starting material, flow-through, marker, fractions 1–9, empty, marker; bottom panels, Source 15S fractions: marker, empty, fractions 10–20, marker. Cropped images encompassing all panels (open boxes, dashed red line) were used for Figure 4—figure supplement 2C.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig4-figsupp2-data3-v2.zip
-
Figure 4—figure supplement 2—source data 4
Western blots of chromatographic fractions.
Left panel, 700 nm channel (Odyssey Fc), protein size marker; right panels, 800 nm channel (Odyssey Fc), guinea pig ModT antibody. Superose 6 fractions: starting material, marker, fractions 1–13. Cropped images from both panels (open boxes, dashed red line) were used for Figure 4—figure supplement 2D.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig4-figsupp2-data4-v2.zip
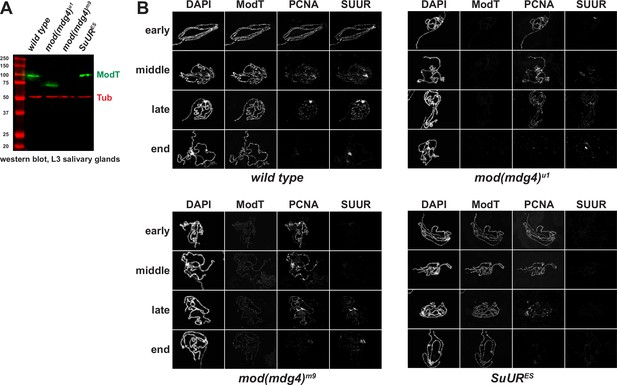
Spatiotemporal distribution of SUMM4 subunits in polytene chromosomes of mod(mdg4) and SuUR mutant alleles.
(A) Western blot analyses of lysates of whole salivary glands. L3 salivary glands from homozygous animals of indicated genotypes were probed with ModT (green) and β-tubulin antibodies (red, loading control). Mass marker sizes (kDa) are shown on the left. (B) Spatiotemporal distribution of SUUR in polytene chromosomes. See legend to Figure 4B. Although SUUR is not properly loaded into mod(mdg4) chromosomes during early endo-S phase (as in wild-type), its deposition partially recovers during late endo-S.
-
Figure 4—figure supplement 3—source data 1
Western blots of salivary gland lysates.
Left panel, 700 nm channel (Odyssey Fc), mouse anti-tubulin antibody and protein size marker; right panels, 800 nm channel (Odyssey Fc), guinea pig ModT antibody. Lane 1, protein size marker; lane 2, L3 salivary glands, wild-type; lane 3, L3 salivary glands, mod(mdg4)u1; lane 4, L3 salivary glands, mod(mdg4)m9; lane 5, L3 salivary glands, SuURES. Cropped images from both panels (open boxes, dashed red line) overlayed in different colors (left panel, red; right panel, green) were used for Figure 4—figure supplement 3A.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig4-figsupp3-data1-v2.zip
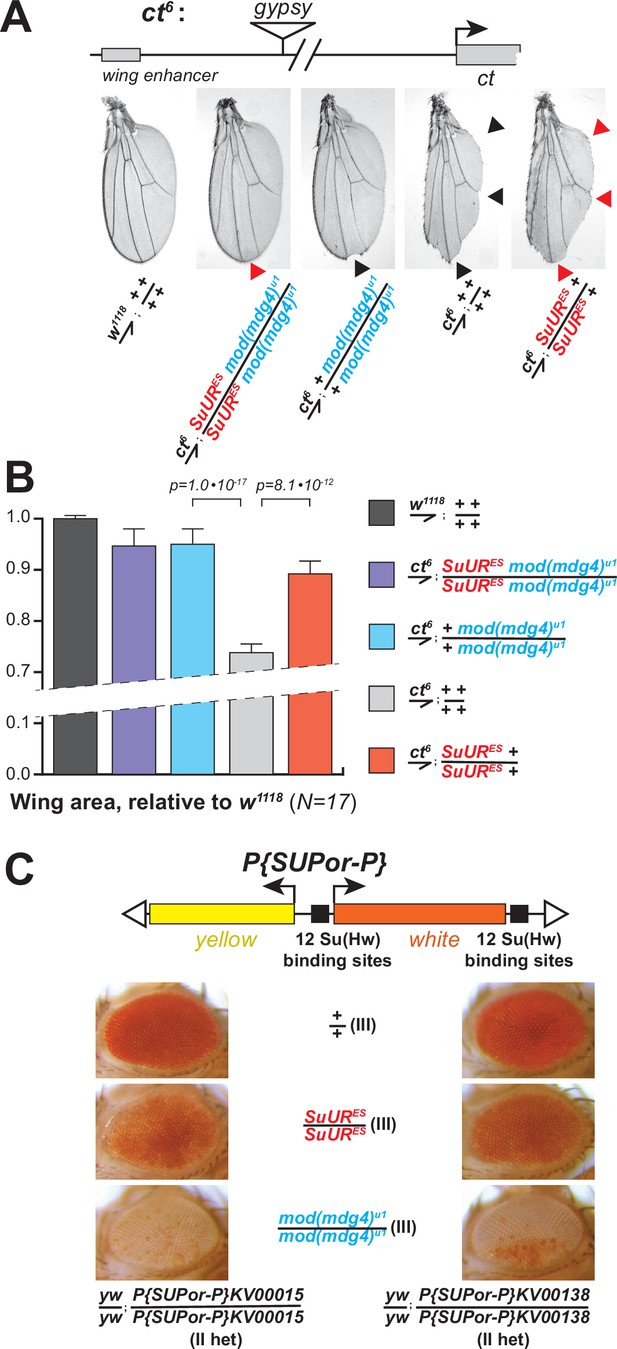
Biological functions of SUMM4 in the regulation of gene expression.
(A) SUMM4 subunits are required for the enhancer-blocking activity in ct6. Top: schematic diagram of the ct6 reporter system; the gypsy retrotransposon is inserted in between the wing enhancer and promoter of cut (Bag et al., 2019). Bottom left: the appearance of wild-type adult wing; bottom right: the appearance of ct6 adult wing in the wild-type background. SuURES and mod(mdg4)u1 alleles are recessive suppressors of the ct6 phenotype. Red and black arrowheads point to distinct anatomical features of the wing upon SuUR mutation. (B) Relative sizes (areas) of wings in adult male flies of the indicated phenotypes were measured (N=17) as described in ‘Materials and methods.’ p-Values for statistically significant differences are indicated (t-test). (C) SUMM4 subunits are required for the chromatin barrier activity of Su(Hw) binding sites. Top: schematic diagram of the P{SUPor-P} reporter system (Bellen et al., 2004); clustered 12 copies of gypsy Su(Hw) binding sites flanks the transcription unit of white. KV00015 and KV00138 are P{SUPor-P} insertions in pericentric heterochromatin of 2L. SuURES and mod(mdg4)u1 alleles are recessive suppressors of the boundary that insulates white from heterochromatin encroachment.
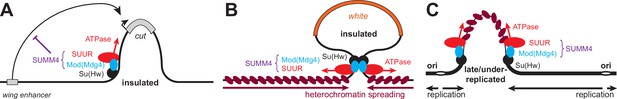
Schematic models for the biological functions of SUMM4 in the regulation of gene expression and DNA replication.
(A) Schematic model for the function of SUMM4 in blocking enhancer–promoter interactions in the ct6 locus. A gypsy mobile element inserted between wing enhancer and gene cut encompasses multiple Su(Hw) binding sites. (B) Schematic model for the function of SUMM4 in establishing a chromatin barrier in heterochromatin-inserted P{SUPor-P} elements. The reporter gene white is flanked on both sides by 12 copies of gypsy insulator element. (C) Schematic model for a putative function of SUMM4 in blocking/retardation of replication fork progression in intercalary heterochromatin domains. Black oval, Su(Hw) protein bound to a gypsy insulator element(s); cyan oval, Mod(Mdg4)-67.2 protein tethered to Su(Hw); red oval, SUUR protein associated with Mod(Mdg4)-67.2 in SUMM4 complex; brown ovals represent heterochromatin components; gray rectangles, gene cut and its upstream wing enhancer; orange rectangle, gene white.
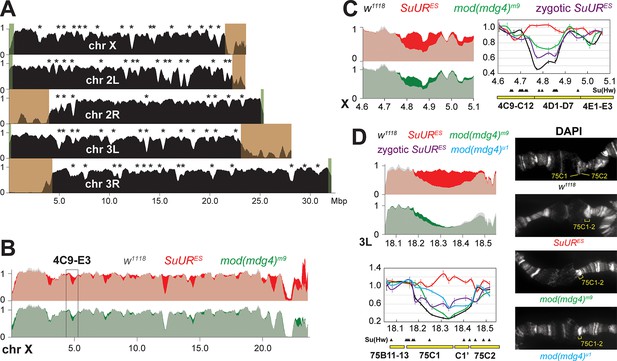
Biological functions of SUMM4 in the regulation of DNA replication.
(A) Genome-wide analyses of DNA copy numbers in Drosophila salivary gland cells (w1118 control). DNA from L3 salivary glands was subjected to high-throughput sequencing. DNA copy numbers (normalized to diploid embryonic DNA) are shown for chromosomes X, II, and III. Chromosome arms are indicated in white. Brown- and green-shades boxes, mapped pericentric and telomeric heterochromatin regions (Hoskins et al., 2015), respectively. Asterisks, positions of underreplicated domains (Table 1). Genomic coordinates in Megabase pairs are indicated at the bottom. (B) Analyses of DNA copy numbers in Drosophila salivary gland cells from wild-type and mutant alleles. Normalized DNA copy numbers are shown across the X chromosome. The control trace (w1118 allele) is shown as semitransparent light gray in the foreground; SuURES (homozygous null) and mod(mdg4)m9 (zygotic null from crosses of heterozygous parents) traces are shown in the background in red and green, respectively; their overlaps with w1118 traces appear as lighter shades of colors. Black box, 4C9-E3 cytological region. (C) Close-up view of DNA copy numbers in region 4C9-E3 from high-throughput sequencing data are presented as in (B). DNA copy numbers were also measured independently by real-time qPCR. The numbers were calculated relative to embryonic DNA and normalized to a control intergenic region. The X-axis shows chromosome positions (in Megabase pairs) of target amplicons. Black, w1118; red, SuURES (homozygous null); green, mod(mdg4)m9 (zygotic null from crosses of heterozygous parents); purple, SuURES (zygotic null from crosses of heterozygous parents). Error bars represent the confidence interval (N=9, see ‘Materials and methods’). Black arrowheads, positions of mapped Su(Hw) binding sites (Nègre et al., 2010). Yellow boxes show approximate boundaries of cytogenetic bands. (D) Close-up view of DNA copy numbers by high-throughput sequencing and by qPCR for region 75B11-C2 and DAPI-stained polytene chromosome segments around cytological regions 75B-75C. Yellow lines or brackets in DAPI images indicate positions of 75C1 and 75C2 bands (w1118 control) or fused 75C1-2 band (mutants); cyan, mod(mdg4)u1 (homozygous null); for other designations see (C).
-
Figure 7—source data 1
Primer sequences used for qPCR.
Genomic coordinates indicate full amplicons, including the length of each primer. Coordinates refer to the BDGP R6/dm3 assembly.
- https://cdn.elifesciences.org/articles/81828/elife-81828-fig7-data1-v2.docx
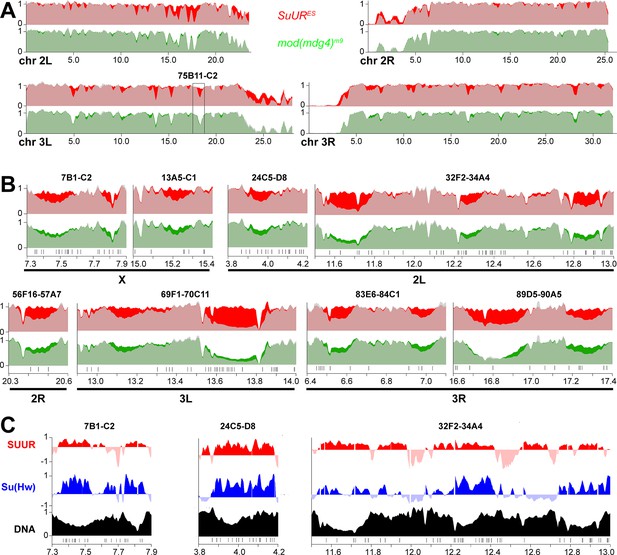
Biological functions of SUMM4 in the regulation of underreplication.
(A) Genome-wide analyses of DNA copy numbers in Drosophila salivary gland cells in chromosome arms 2L, 2R, 3L, and 3R. The data were obtained and presented as for the X chromosome (Figure 7B). Black box, 75B11-C2 cytological region. (B) Close-up view of DNA copy numbers by high-throughput sequencing for additional genomic regions. Approximate cytogenetic locations are indicated at the top of each panel. Short vertical bars at the bottom, positions of mapped Su(Hw) binding sites (Nègre et al., 2010). See legend to Figure 7C and D for other designations. (C) Sample plots of DamID profiles for SUUR (red) and Su(Hw) (purple), log2 enrichment over Dam-only control (Filion et al., 2010). Positive values are plotted in dark colors and negative values in light colors for contrast. DNA copy numbers in salivary gland cells (black) indicate underreplicated intercalary heterochromatin domains. Vertical bars, Su(Hw) binding sites (Nègre et al., 2010).
Tables
Underrepli cated domains and suppression of underreplication in (UR) SUMM4 subunit mutant alleles.
Domains of UR in euchromatic arms of polytene chromosomes were called in w1118 as described in ‘Materials and methods.’ Their genomic coordinates, approximate cytological location (‘Cyto band’), and average DNA copy numbers (‘<CN>’) in homozygous w1118, SuURES, and mod(mdg4)m9 L3 larvae are shown. <CN> numbers were normalized to the average DNA copy numbers across euchromatic genome. UR percent recovery levels were calculated as (<CN> mut –<CN>w1118) / (1 – <CN >w1118); negative numbers indicate increased UR. UR p-values were calculated using the DESeq2 package by averaging the Wald test p-values of each 5 kbp bin significantly different than the w1118 signal. UR was called as suppressible by a mutant if p<0.01; p-values for regions that exhibit a statistically significant recovery of UR are shown in bold blue. Averages of <CN> across all called underreplicated domains and averages of percent Recovery across all suppressible underreplicated domains (‘<Recovery>’, bottom row) were adjusted for each underreplicated domain length; calculation errors = standard deviations.
| N | Chromosome coordinates | Length | UR, w1118 | UR, SuURES | UR, mod(mdg4)m9 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Arm | Left | Right | Cyto band | <CN> | <CN> | Recovery (%) | p-Value | <CN> | Recovery (%) | p-Value | ||
| 1 | X | 2,950,001 | 3,140,000 | 3C3-C7 | 190,000 | 0.51 | 0.93 | 86 | 0.58 | 14 | 1.1E-02 | |
| 2 | X | 4,710,001 | 4,900,000 | 4C15-D5 | 190,000 | 0.56 | 0.96 | 92 | 0.81 | 57 | ||
| 3 | X | 4,965,001 | 5,070,000 | 4E1-E2 | 105,000 | 0.72 | 0.86 | 50 | 0.80 | 28 | 1.4E-02 | |
| 4 | X | 6,415,001 | 6,525,000 | 6A1-B1 | 110,000 | 0.71 | 0.90 | 65 | 0.80 | 29 | ||
| 5 | X | 7,335,001 | 7,560,000 | 7B1-B4 | 225,000 | 0.65 | 0.98 | 95 | 0.79 | 40 | ||
| 6 | X | 7,750,001 | 7,865,000 | 7B7-C1 | 115,000 | 0.64 | 0.94 | 84 | 0.84 | 55 | ||
| 7 | X | 8,880,001 | 9,005,000 | 8B5-C2 | 125,000 | 0.73 | 0.86 | 50 | 0.76 | 9 | ||
| 8 | X | 9,405,001 | 9,555,000 | 8D12-E7 | 150,000 | 0.72 | 0.91 | 67 | 0.85 | 47 | ||
| 9 | X | 11,170,001 | 11,325,000 | 10A10-B3 | 155,000 | 0.67 | 0.84 | 53 | 0.78 | 35 | ||
| 10 | X | 12,040,001 | 12,430,000 | 11A2-A10 | 390,000 | 0.38 | 0.97 | 94 | 0.42 | 6 | ||
| 11 | X | 13,950,001 | 14,100,000 | 12D1-E1 | 150,000 | 0.69 | 0.72 | 10 | 1.0E-02 | 0.73 | 14 | 1.4E-02 |
| 12 | X | 14,290,001 | 14,565,000 | 12E7-F1 | 275,000 | 0.51 | 0.94 | 87 | 0.69 | 36 | ||
| 13 | X | 17,925,001 | 18,030,000 | 16F3-F5 | 105,000 | 0.67 | 0.99 | 98 | 0.90 | 68 | ||
| 14 | X | 20,000,001 | 20,105,000 | 19A4-B1 | 105,000 | 0.79 | 1.12 | 157 | 0.82 | 12 | ||
| 15 | X | 20,525,001 | 21,020,000 | 19D2-E7 | 495,000 | 0.50 | 0.97 | 93 | 0.51 | 2 | ||
| 16 | X | 21,630,001 | 22,450,000 | 20A5-C1 | 820,000 | 0.04 | 0.32 | 29 | 0.06 | 2 | ||
| 17 | X | 22,550,001 | 22,995,000 | 20C2-F3 | 445,000 | 0.48 | 0.81 | 64 | 0.74 | 51 | ||
| 18 | 2L | 3,920,001 | 4,025,000 | 24D1-D4 | 105,000 | 0.63 | 0.93 | 81 | 0.80 | 46 | ||
| 19 | 2L | 4,585,001 | 4,790,000 | 25A2-A5 | 205,000 | 0.66 | 0.99 | 98 | 0.78 | 36 | ||
| 20 | 2L | 5,400,001 | 5,510,000 | 25E1-E4 | 110,000 | 0.82 | 0.99 | 95 | 0.90 | 45 | ||
| 21 | 2L | 6,155,001 | 6,320,000 | 26B9-C2 | 165,000 | 0.74 | 1.08 | 130 | 0.88 | 54 | ||
| 22 | 2L | 9,030,001 | 9,150,000 | 29F8-30A2 | 120,000 | 0.76 | 0.98 | 93 | 0.95 | 79 | ||
| 23 | 2L | 11,535,001 | 11,795,000 | 32F2-33A1 | 260,000 | 0.44 | 0.90 | 83 | 0.57 | 24 | ||
| 24 | 2L | 12,215,001 | 12,340,000 | 33D3-E1 | 125,000 | 0.58 | 0.86 | 66 | 0.75 | 40 | ||
| 25 | 2L | 12,765,001 | 12,970,000 | 33F5-34A3 | 205,000 | 0.55 | 0.91 | 79 | 0.73 | 40 | ||
| 26 | 2L | 14,685,001 | 15,010,000 | 35B4-B8 | 325,000 | 0.41 | 0.88 | 80 | 0.54 | 23 | ||
| 27 | 2L | 15,295,001 | 15,735,000 | 35D1-D4 | 440,000 | 0.49 | 0.76 | 53 | 0.54 | 9 | ||
| 28 | 2L | 15,770,001 | 15,900,000 | 35D4-D6 | 130,000 | 0.54 | 0.87 | 71 | 0.68 | 31 | ||
| 29 | 2L | 15,925,001 | 16,240,000 | 35D6-F1 | 315,000 | 0.29 | 0.90 | 87 | 0.38 | 12 | ||
| 30 | 2L | 16,925,001 | 17,375,000 | 36B4-C7 | 450,000 | 0.23 | 0.89 | 85 | 0.26 | 4 | ||
| 31 | 2L | 17,515,001 | 18,100,000 | 36C10-E4 | 585,000 | 0.34 | 0.87 | 80 | 0.36 | 2 | ||
| 32 | 2L | 18,160,001 | 18,300,000 | 36E6-F2 | 140,000 | 0.67 | 0.99 | 97 | 0.90 | 69 | ||
| 33 | 2L | 20,110,001 | 20,290,000 | 38C1-C4 | 180,000 | 0.48 | 0.69 | 41 | 0.46 | –5 | ||
| 34 | 2L | 20,485,001 | 20,620,000 | 38C8-D1 | 135,000 | 0.77 | 0.98 | 93 | 0.99 | 97 | ||
| 35 | 2L | 21,400,001 | 21,550,000 | 39D3-E2 | 150,000 | 0.10 | 0.15 | 5 | 0.14 | 3 | ||
| 36 | 2L | 21,805,001 | 22,125,000 | 40A4-E4 | 320,000 | 0.53 | 0.94 | 87 | 0.54 | 1 | ||
| 37 | 2R | 4,875,001 | 5,050,000 | 41C4-D1 | 175,000 | 0.35 | 0.86 | 78 | 0.34 | –1 | ||
| 38 | 2R | 5,410,001 | 5,535,000 | 41F1-F3 | 125,000 | 0.58 | 0.79 | 50 | 0.52 | –13 | ||
| 39 | 2R | 6,290,001 | 6,505,000 | 42A14-B1 | 215,000 | 0.13 | 0.50 | 42 | 0.14 | 1 | ||
| 40 | 2R | 13,620,001 | 13,760,000 | 50B6-C3 | 140,000 | 0.63 | 0.95 | 88 | 0.78 | 41 | ||
| 41 | 2R | 20,355,001 | 20,540,000 | 56F17-57A5 | 185,000 | 0.56 | 0.92 | 83 | 0.71 | 35 | ||
| 42 | 2R | 21,830,001 | 21,945,000 | 58A2-A4 | 115,000 | 0.72 | 0.95 | 83 | 0.71 | –3 | 2.2E-02 | |
| 43 | 2R | 23,145,001 | 23,320,000 | 59D1-D6 | 175,000 | 0.62 | 1.04 | 110 | 0.67 | 13 | ||
| 44 | 3L | 4,840,001 | 5,100,000 | 64C1-C5 | 260,000 | 0.38 | 0.92 | 87 | 0.40 | 3 | ||
| 45 | 3L | 5,385,001 | 5,510,000 | 64C15-D3 | 125,000 | 0.51 | 0.88 | 76 | 0.73 | 45 | ||
| 46 | 3L | 6,290,001 | 6,485,000 | 65A11-B3 | 195,000 | 0.52 | 0.89 | 77 | 0.71 | 38 | ||
| 47 | 3L | 9,180,001 | 9,300,000 | 67A1-A7 | 120,000 | 0.67 | 0.97 | 90 | 0.73 | 20 | 1.0E-02 | |
| 48 | 3L | 10,000,001 | 10,195,000 | 67D3-D10 | 195,000 | 0.62 | 0.97 | 93 | 0.79 | 44 | ||
| 49 | 3L | 13,085,001 | 13,220,000 | 70A1-A2 | 135,000 | 0.66 | 1.01 | 104 | 0.89 | 66 | ||
| 50 | 3L | 13,550,001 | 13,855,000 | 70B6-C4 | 305,000 | 0.26 | 0.95 | 94 | 0.39 | 18 | ||
| 51 | 3L | 15,175,001 | 15,500,000 | 71B7-D3 | 325,000 | 0.39 | 0.94 | 89 | 0.46 | 10 | ||
| 52 | 3L | 17,115,001 | 17,240,000 | 73F1-74A1 | 125,000 | 0.71 | 1.02 | 106 | 0.84 | 45 | ||
| 53 | 3L | 18,175,001 | 18,525,000 | 75B11-75D2 | 350,000 | 0.45 | 0.87 | 76 | 0.47 | 4 | ||
| 54 | 3L | 20,555,001 | 20,695,000 | 77D1-77E3 | 140,000 | 0.60 | 1.02 | 106 | 0.84 | 61 | ||
| 55 | 3R | 6,060,001 | 6,310,000 | 83D2-E4 | 250,000 | 0.70 | 0.92 | 72 | 0.63 | –22 | 1.0E-02 | |
| 56 | 3R | 6,495,001 | 6,635,000 | 83F1-84A1 | 140,000 | 0.53 | 0.96 | 91 | 0.71 | 39 | ||
| 57 | 3R | 6,915,001 | 7,055,000 | 84B1-B2 | 140,000 | 0.64 | 0.93 | 80 | 0.82 | 49 | ||
| 58 | 3R | 7,550,001 | 7,785,000 | 84D9-84E2 | 235,000 | 0.44 | 0.80 | 65 | 0.51 | 12 | ||
| 59 | 3R | 10,450,001 | 10,660,000 | 86B6-C4 | 210,000 | 0.55 | 0.98 | 97 | 0.66 | 25 | ||
| 60 | 3R | 10,910,001 | 11,140,000 | 88C15-86D4 | 230,000 | 0.45 | 0.94 | 89 | 0.46 | 2 | ||
| 61 | 3R | 12,050,001 | 12,165,000 | 87A5-B1 | 115,000 | 0.63 | 0.96 | 88 | 0.81 | 49 | ||
| 62 | 3R | 12,745,001 | 12,935,000 | 87C8-D4 | 190,000 | 0.67 | 0.89 | 68 | 0.60 | –21 | 1.1E-02 | |
| 63 | 3R | 14,935,001 | 15,055,000 | 88D8-D10 | 120,000 | 0.70 | 0.88 | 61 | 0.84 | 47 | ||
| 64 | 3R | 16,670,001 | 16,970,000 | 89D6-E5 | 300,000 | 0.40 | 0.92 | 87 | 0.47 | 10 | ||
| 65 | 3R | 17,160,001 | 17,355,000 | 89F1-90A2 | 195,000 | 0.62 | 0.94 | 84 | 0.86 | 64 | ||
| 66 | 3R | 20,085,001 | 20,290,000 | 92C4-E1 | 205,000 | 0.61 | 0.81 | 53 | 0.71 | 26 | ||
| 67 | 3R | 20,340,001 | 20,525,000 | 92E4-E12 | 185,000 | 0.58 | 0.96 | 91 | 0.79 | 50 | ||
| 68 | 3R | 22,110,001 | 22,295,000 | 94A2-A4 | 185,000 | 0.61 | 0.93 | 83 | 0.76 | 39 | ||
| 69 | 3R | 28,005,001 | 28,295,000 | 98B7-C3 | 290,000 | 0.40 | 0.91 | 85 | 0.60 | 32 | ||
| 70 | 3R | 28,370,001 | 28,480,000 | 98C5-D2 | 110,000 | 0.73 | 0.98 | 94 | 0.91 | 66 | ||
| UR domains: <Length> : 216 ± 64 kbp Average <CN> across all UR domains: 0.49 ± 0.08 | Suppressed UR domains: <Length> : 217 ± 64 kbp <Recovery> : 78 ± 11% | Suppressed UR domains: <Length> : 225 ± 67 kbp <Recovery> : 26 ± 9% | ||||||||||
Additional files
-
Supplementary file 1
Protein identities and peptide spectral data (ion libraries) obtained by information-dependent acquisitions (IDA) for FPLC fractions (IL1-5, Figure 1A) and recombinant SUUR (ILR, Figure 1C).
- https://cdn.elifesciences.org/articles/81828/elife-81828-supp1-v2.xlsx
-
Supplementary file 2
Raw data of SWATH acquisitions for FPLC fractions (Figure 1A) quantified using ion library IL5 (Supplementary file 1) and an example of protein purification profile analyses (hydroxylapatite step, Figure 1A and H).
- https://cdn.elifesciences.org/articles/81828/elife-81828-supp2-v2.xlsx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/81828/elife-81828-mdarchecklist1-v2.pdf





