Mega-scale movie-fields in the mouse visuo-hippocampal network
Figures
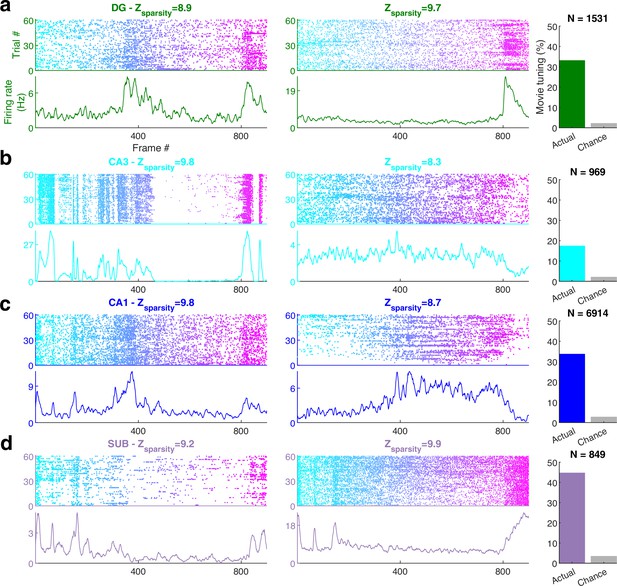
Movie frame selectivity in hippocampal neurons.
(a) Raster plots of two different dentate gyrus (DG) neurons as a function of the movie frame (top) over 60 trials, and the corresponding mean firing rate response (bottom). These two cells had significantly increased activity in specific segments of the movie. Z-scored sparsity indicating strength of modulation is indicated above. 33.1% of dentate neurons were significantly modulated by the movie (right, green bar), far greater than chance (gray bar). Total active, broad spiking neurons for each brain region indicated at top (Ntuned /Ncells = 506/1531). (b) Same as (a), for CA3 (168/969, 17.3%), (c) CA1 (2326/6914, 33.6%), and (d) subiculum (379/849, 44.6%) neurons.
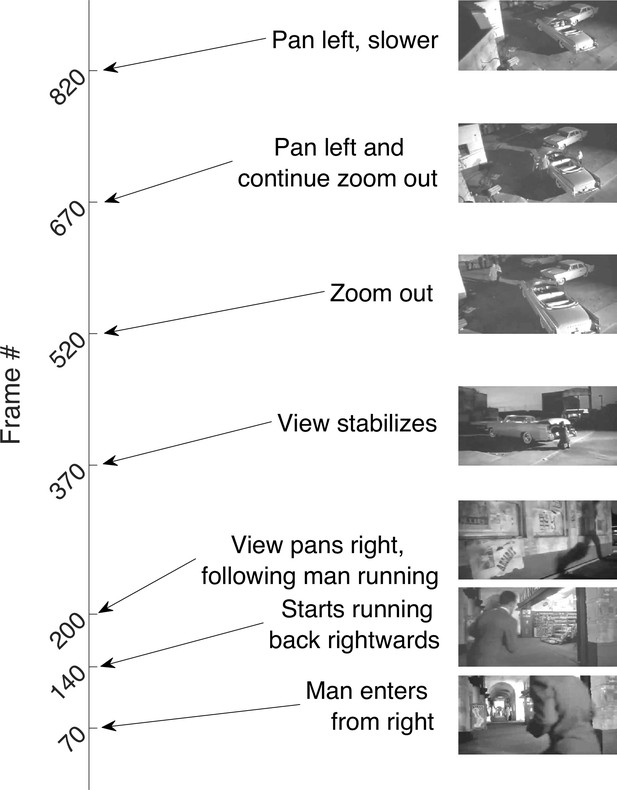
The movie.
The 30-s long, silent, black-and-white, isoluminant movie with frame numbers denoting key episodes in this continuous segment.
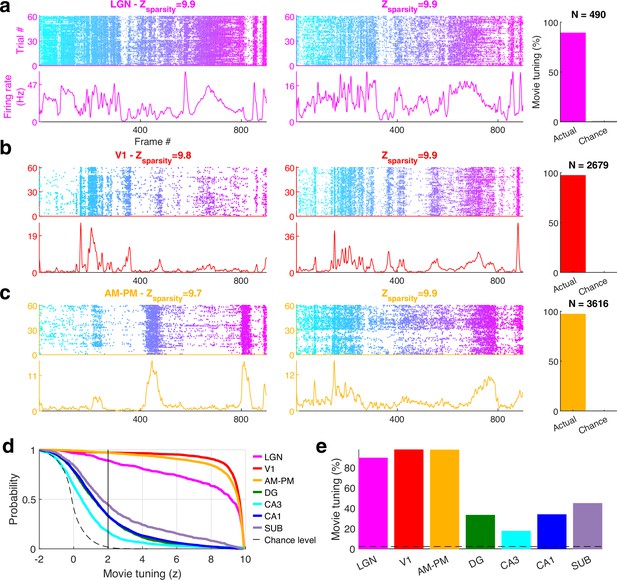
Movie selectivity across brain areas.
(a) Similar to Figure 1, representative single cells from lateral geniculate nucleus (LGN) showing selective movie responses. Strength of modulation, quantified by z-scored sparsity is indicated above. The total number of broad spiking cells used (N) and the fraction selective are shown by the bar chart on the right. (b) Same as that (a), for V1 and (c) for higher visual areas AM–PM. (d) Cumulative distribution of movie selectivity across all broad spiking cells, including significantly (z > 2 vertical black line, see Methods) tuned cells. The largest prevalence of selectivity in broad spiking neurons was seen in primary visual cortex (V1, 97.3%, 2606 out of 2679) and least in CA3 hippocampus (17.3%, 168 out of 969). (e) All brain regions analyzed showed far greater selectivity than the chance level (dashed gray line). There was a clear difference in the strength of movie tuning between visual and hippocampal areas.
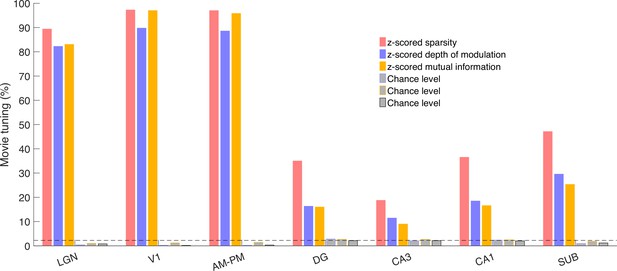
Multiple metrics show significant and comparable movie tuning.
The percentage of movie-tuned cells, deemed as z-scored metric >2, were significantly greater than chance levels (p < 4.9 × 10−11), using either sparsity or depth of modulation or mutual information as the metric (see Methods for metric definitions). Sparsity yielded higher movie tuning than depth of modulation across all brain regions (p < 1.8 × 10−3), putatively because it captures multi-peaked tuning better than depth of modulation, which only relies on the largest and smallest firing rate responses. Similarly, z-scored mutual information led to greater tuning than chance levels (p < 4.9 × 10−11), but lesser than that with the sparsity metric (p < 1.3 × 10−5).
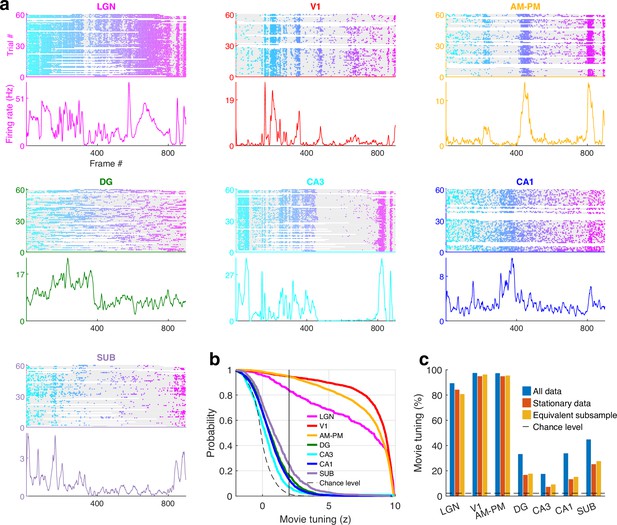
Movie tuning is intact during immobility.
(a) Similar to Figure 1, a representative cell from each of the seven brain regions showing significant modulation movie tuning using only the data when the mouse was immobile, while excluding the data when the mouse was running (stationary data, see Methods). (b) Fraction of selective neurons was significantly above chance in all brain regions, ranging from 94.7% in V1 up to 7.1% in CA3 in the stationary data. (c) To explicitly test the effect of running on movie selectivity, we compared the results in (b) with a random subsample of data, of equal duration as the stationary data, that included running and stationary, to control for the loss of data (see Methods). Prevalence of movie selectivity was not significantly different (Kolmogorov-Smirnov [KS] - test p > 0.05) in these two subsamples, except in CA1 (p = 0.03, 13.1% in stationary data, 15.0% in the equivalent subsample). Only sessions with at least 300 s of stationary data were used in this analysis to ensure sufficient statistical power. The reduction in fraction tuned neurons in (b) and (c) for ‘stationary data’, compared to ‘all data’ here and in Figure 1 and Figure 1—figure supplements 2 and 3 is because of the reduction in the amount of data, which directly reduces statistical significance.
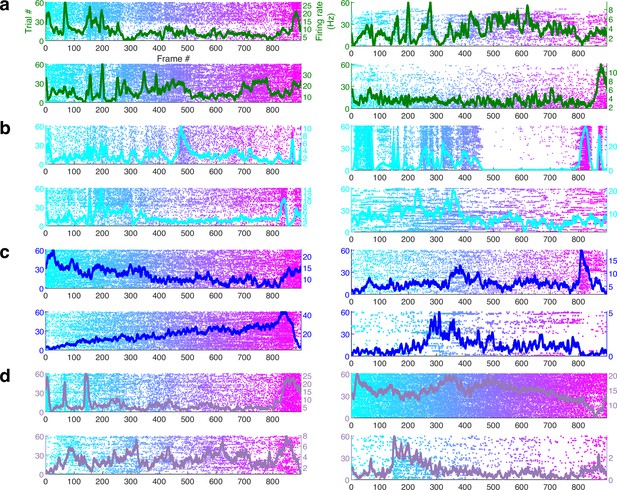
Simultaneously recorded hippocampal cells have different movie tuning.
Four simultaneously recorded and significantly movie-tuned cells each from (a) dentate gyrus, (b) CA3, (c) CA1, and (d) subiculum. Each cell shows different movie selectivity. Average responses are overlaid (on raster plots), and their color corresponds to the different brain regions, described in Figure 1 legend. This further demonstrates that hippocampal movie tuning is not an artifact of nonspecific variables that alter excitability.
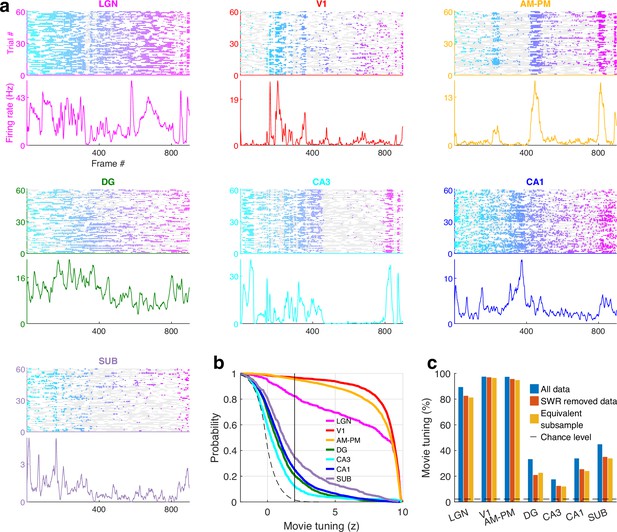
Movie tuning in unaffected by the removal of sharp-wave ripple (SWR) events.
(a) Similar to Figure 1—figure supplement 4, a representative cell from each brain region showing significant modulation movie tuning using data after removal of SWR events (14,371 cells from 20 sessions where SWR information was available, see Methods). (b) Fraction of selective neurons was significantly above chance in all brain regions, ranging from 96.7% in V1 up to 12.3% in CA3 in the SWR removed data. (c) To control the loss of data by the removal of SWR, we compared the results with movie tuning in an equivalent subsample of data. Prevalence of movie selectivity was not significantly different (KS-test p > 0.05) in these two subsamples, except in AM–PM (p = 0.02, 97.1% in SWR removed data, 94.6% in the equivalent subsample). As before (Figure 1—figure supplement 4), due to a reduction in the amount of data, a smaller number of neurons showed significant movie tuning in both SWR removed data as well as equivalent subsampled data.
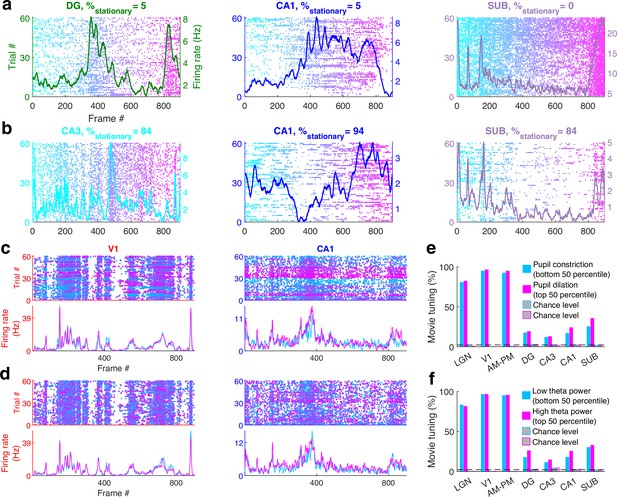
Movie tuning is comparable across sessions with or without prolonged stationary behavior, high or low pupil dilation or theta power.
(a) Three representative cells from sessions with rare stationary periods (% of time in stationary, indicated at the top) and mostly running behavior that showed significant movie tuning. (b) Similar to (a), three example cells from sessions with mostly running behavior showing significant movie tuning. Movie tuning persisted across all sessions. (c) One representative cell each from V1 and CA1, showing comparable movie tuning during dilated (magenta, high pupil area) or constricted (cyan, low pupil area) pupil. Each dot in the scatter (top) corresponds to one spike, and the color corresponds to the pupil area during that spike. Average movie responses for bottom 50 (cyan, pupil constriction) and top 50 (magenta, pupil dilation) percentiles are shown below. This separation based on 50 percentile ensures equal amount of data in both subsegments. (d) Similar to (c), showing similar movie tuning for data with high (magenta) and low (cyan) theta power. (e) Movie tuning in the top as well as bottom 50 percentile of pupil area data was significantly greater than their respective chance levels (p < 1.2 × 10−8). Top as well as bottom 50 percentile data did not have significantly different movie tuning prevalence for LGN, DG, and CA3 (p > 0.73, which could be because of smaller number of cells recorded in these brain regions), but dilated pupil corresponds to slightly greater tuning for other brain regions (p < 3.4 × 10−4). (f) Similar to (e), the movie tuning in high as well as low theta power data was significantly greater than chance levels (p < 5.0 × 10−10). Movie tuning was greater in data with high theta power for DG and CA1 (p < 2.1 × 10−6), but not significantly different for other brain regions (p > 0.07). Both subsegments had equal amounts of data to ensure fair comparison.
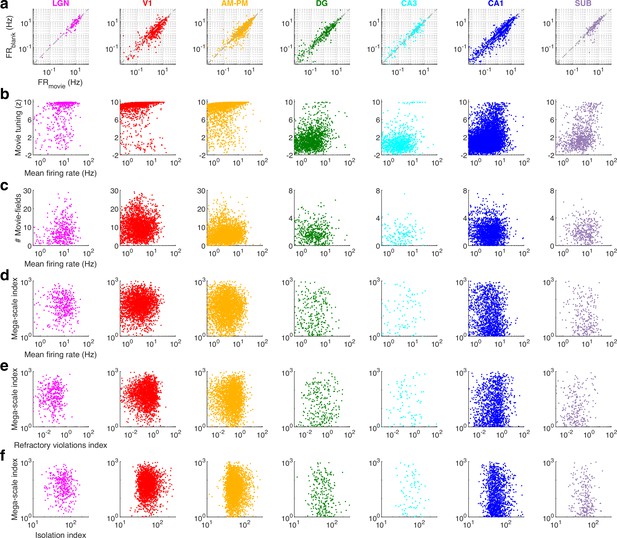
Movie presentation did not alter hippocampal firing rates and the mega-scale coding was unrelated to cluster quality.
(a) More than 50% of hippocampal place cells shut down during maze exploration (Ravassard et al., 2013). In contrast, there was no consistent pattern of neural activation or shutdown during the movie presentation in all brain areas. To make a more conservative estimate, this comparison was restricted to units whose firing rates did not differ by more than 20% across the two movie blocks. Furthermore, only the data when the animals were immobile was used to avoid confounding effects of running, and the firing rate threshold of 0.5 Hz was removed for this panel. (b) The amount of movie tuning was positively correlated with the mean firing rates of the neurons for all brain regions (r > 0.14, p < 4.2 × 10−10). (c) The number of movie-fields was uncorrelated with the mean firing rate of tuned cells in V1, DG, CA1, and SUB (p > 0.12), but positively correlated for LGN, AM–PM, and CA3 (r > 0.04, p < 0.01). Note the different y-scales for visual and hippocampal brain regions. Since the number of movie-fields is an integer, data along the y-axis were slightly jittered for better visualization. (d) The mega-scale index was only weakly correlated with the mean firing rate of a neuron in V1 (Pearson’s correlation coefficient r = 0.08, p = 7.3 × 10−5), CA1 (r = −0.14, p = 3.5 × 10−8) and subiculum (r = −0.14, p = 0.02), and was uncorrelated for other brain regions (p > 0.05). (e) The refractory violations index was uncorrelated with the mega-scale index (lower index means better cluster quality; Siegle et al., 2021; Hill et al., 2011) for all brain regions (p > 0.05). To remove the potential confounding effect of mean firing rates, we computed the partial correlation coefficient by factoring out the mean firing rate. (f) Similar to (c), the isolation index (greater isolation index means better cluster quality; Siegle et al., 2021; Schmitzer-Torbert et al., 2005) was uncorrelated with the mega-scale index for all brain regions (partial correlation coefficient, by factoring out the mean firing rate, p > 0.12). Factoring out the contribution of mean firing rate is necessary since the isolation index was typically positively correlated (the refractory violations index was typically negatively correlated) with the mean rate. The mega-scale index comparisons were restricted to movie active, tuned neurons with at least two movie peaks. Note-log spaced axes for (a–d), except y-axes of b and c.
Sequential movie.
The 30-s movie clip shown, along with the frame number indicated in the top right corner (updated every second, or every 30 frames). The same movie clip was shown in two blocks of 30 repeats each.
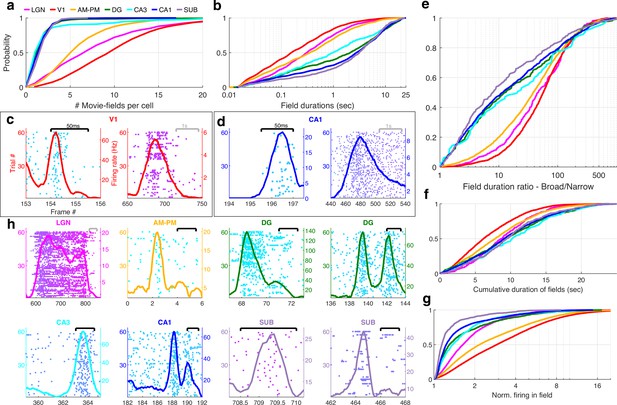
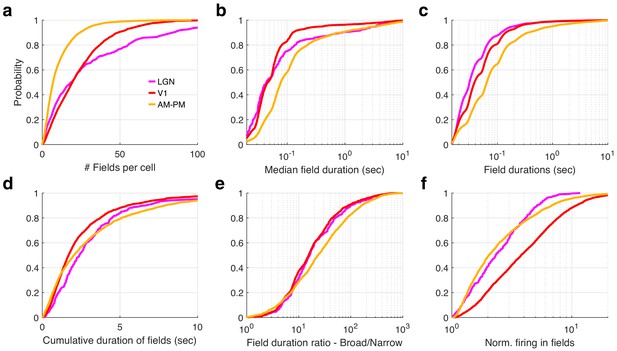
Multi-peaked, mega-scale movie-fields across all brain areas.
(a) Distribution of the number of movie-fields per tuned cell (see Methods) in different brain regions (shown by different colors, top line inset, arranged in their hierarchical order). Hippocampal regions (blue-green shades) were significantly different from each other (KS-test p < 0.04), except DG–CA3. All visual regions were significantly different from each other (KS-test p < 7.0 × 10−11). All visual–hippocampal region pairwise comparisons were also significantly different (KS-test p < 1.8 × 10−44). CA1 had the lowest number of movie-fields per cell (2.0 ± 0.02, mean ± standard error of the mean [SEM]) while V1 had the highest (10.4 ± 0.1). (b) Distribution of the durations of movie-fields identified in (a), across all tuned neurons from a given brain region. These were significantly different for all brain region pairs (KS-test p < 7.3 × 10−3). The longest movie-fields were in subiculum (median ± SEM, here and subsequently, unless otherwise mentioned, 3169.9 ± 169.8 ms), and the shortest in V1 (156.6 ± 9.2 ms). (c) Snippets of movie-fields from an example cell from V1, with two of the fields zoomed in, showing 60× difference in duration. Black bar at top indicates 50 ms, and gray bar indicates 1 s. Each frame corresponds to 33.3 ms. Average response (solid trace, y-axis on the right) is superimposed on the trial wise spiking response (dots, y-axis on the left). Color of dots corresponds to frame numbers as in Figure 1. (d) Same as (c), for a CA1 neuron with 54× difference in duration. (e) The ratio of longest to shortest field duration within a single cell, i.e., mega-scale index, was largest in V1 (56.7 ± 2.2) and least in subiculum (8.0 ± 9.7). All visual–visual and visual–hippocampal brain region pairs were significantly different on this metric (KS-test p < 0.02). Among the hippocampal–hippocampal pairs, only CA3–SUB were significantly different (p = 0.03). (f) For each cell, the total duration of all movie-fields, i.e., cumulative duration of significantly elevated activity, was comparable across brain regions. The largest cumulative duration (10.2 ± 0.46 s, CA3) was only 1.66× of the smallest (6.2 ± 0.09 s) (V1). Visual–hippocampal and visual–visual brain region pairs’ cumulative duration distributions were significantly different (KS-test p < 0.001), but not hippocampal pairs (p > 0.07). (g) Distribution of the firing within fields, normalized by that in the shuffle response. All fields from all tuned neurons in a brain region were used. Firing in movie-fields was significantly different across all brain region pairs (KS-test, p < 1.0 × 10−7), except DG–CA3. Movie-field firing was largest in V1 (2.9 ± 0.03) and smallest in subiculum (1.14 ± 0.03). (h) Snippets of movie-fields from representative tuned cells, from lateral geniculate nucleus (LGN) showing a long movie-field (233 frames, or 7.8 s, panel 1), and from AM–PM and from hippocampus showing short fields (two frames or 66.6 ms wide or less).
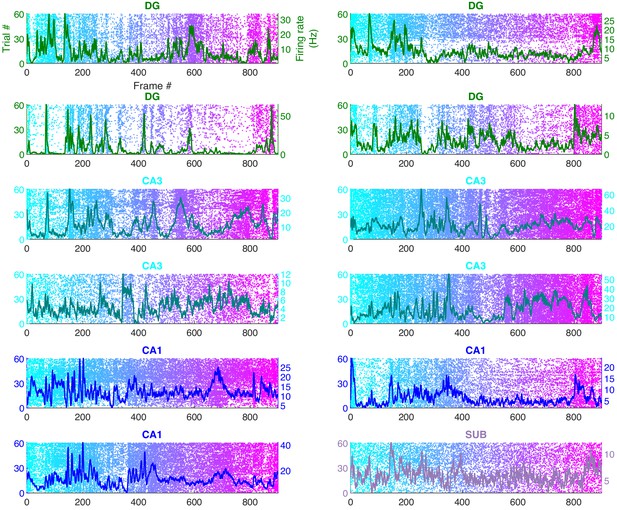
Few hippocampal neurons had greater than five movie-fields.
Only a handful of movie-tuned neurons from dentate gyrus (rows 1 and 2), CA3 (rows 3 and 4), CA1, and subiculum (bottom-right), had more than five distinct movie-fields. Similar format as Figure 1 and Figure 1—figure supplement 5. This is in contrast to the visual areas where a large number of movie-fields were common and the average number of movie-fields per cell was greater than 6 (Figure 2).
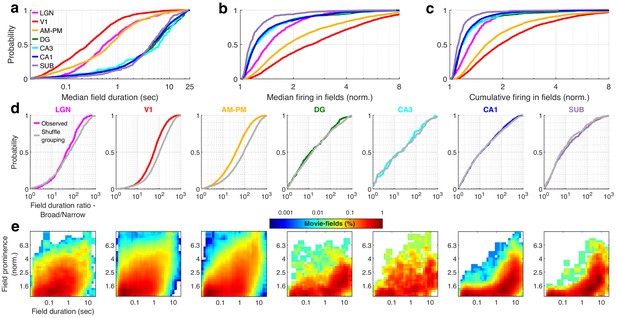
Mega-scale movie-coding within a single cell is smaller than the ensemble wide mega-scale index in visual, but not hippocampal areas.
(a) Distribution of median duration of movie-fields, computed across all fields of a single neuron. Median movie-field duration was significantly larger in all hippocampal areas compared to all visual areas (KS-test p < 7.1 × 10−31). Median field duration between DG–CA3 and DG–CA1 were not significantly different, but all other visual–visual and hippocampal–hippocampal region pairs were significantly different. (KS-test p < 0.04). CA3 had the largest median field duration (6.3 ± 0.48 s), and V1 had the smallest (0.27 ± 0.03 s). Surprisingly, lateral geniculate nucleus (LGN) movie-field durations (0.57 ± 0.13 s) were about twofold longer than V1 (p = 2.5 × 10−21); though both were smaller than those in the higher order brain areas (0.71 ± 0.05 s). (b) Firing in movie-fields, normalized by that in the shuffled response were used to obtain the median value from all fields of a neuron. This metric of median movie-field activation is significantly different across all brain region pairs (KS-test p < 3.4 × 10−5), except DG–CA3, CA3–CA1, and DG–CA1 pairs. The largest median movie-field activation was in V1 (2.5 ± 0.05), and the smallest in subiculum (1.13 ± 0.03). (c) Cumulative firing in movie-fields, normalized by that in the shuffle response, obtained by adding the activity within all fields of a neuron was significantly different across all brain region pairs (KS-test p < 3.0 × 10−7), except DG–CA3, CA3–CA1, and DG–CA1. V1 response was largest (1.93 ± 0.04), and subiculum was the smallest (1.11 ± 0.02). (d) For each brain region, the movie-field duration ratio was recalculated by randomly reassigning the cell ids to all the movie peaks from that brain region. Using this new assignment of movie peaks to a cell, we obtain the expected mega-scale index (largest/smallest peak duration) based on the ensemble behavior. The observed mega-scale index within a cell was smaller than expected from the ensemble in all the visual areas (KS-test p < 3.2 × 10−3, median was 77.5%, 56.2%, and 41.7% of chance for LGN, V1, and AM–PM, respectively). This was not the case in hippocampal regions (p > 0.23). Thus, individual cells in the visual, but not hippocampal areas sampled a subset of possible mega-scale coding values of the ensemble. (e) Histogram of movie-fields, binned for their durations (log-scaled) and their prominence (also log-scaled). The most prominent fields tended to be wider in most brain areas, and this effect was stronger in hippocampal regions, than visual. Note that the histogram color is also log-scaled.
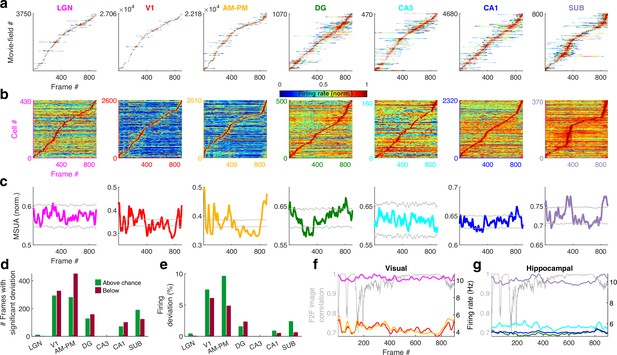
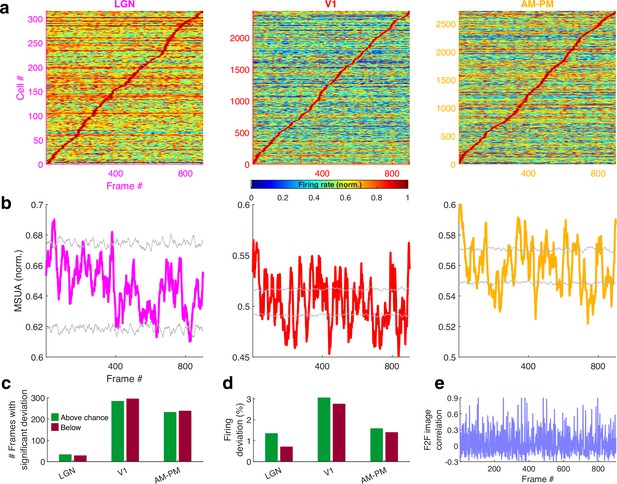
Population averaged movie tuning varies across brain areas.
(a) Stack plot of all the movie-fields detected from all tuned neurons of a brain region. Color indicates relative firing rate, normalized by the maximum firing rate in that movie-field. The movie-fields were sorted according to the frame with the maximal response. Note accumulation of fields in certain parts of the movie, especially in subiculum and AM–PM. (b) Similar to (a), but using only a single, tallest movie-field peak from each neuron showing a similar pattern, with pronounced overrepresentation of some portions of the movie in most brain areas. Each neuron’s response was normalized by its maximum firing rate. The average firing rate of non-peak frames, which was inversely related to the depth of modulation, was smallest (0.35× of the average peak response across all neurons) for V1, followed by AM–PM 0.37, leading to blue shades. Average non-peak responses were higher for other regions (0.57× the peak for LGN, CA3 – 0.61, DG – 0.62, CA1 – 0.64, and SUB – 0.76), leading to warmer off-diagonal colors. (c) Multiple single-unit activity (MSUA) in a given brain region, obtained as the average response across all tuned cells, by using maxima-normalized response for each cell from (b). Gray lines indicate mean ± 4*std response from the shuffle data corresponding to p = 0.025 after Bonferroni correction for multiple comparisons (see Methods). AM–PM had the largest MSUA modulation (sparsity = 0.01) and CA1 had the smallest (sparsity = 1.8 × 10−4). The MSUA modulation across several brain region pairs – AM&PM–DG, V1–CA3, DG–CA3, CA3–CA1, and CA1–SUB were not significantly correlated (Pearson correlation coefficient p > 0.05). Some brain region pairs, DG–LGN, DG–V1, AM&PM–CA3, LGN–CA1, V1–CA1, DG–SUB, and CA3–SUB, were significantly negatively correlated (r < −0.18, p < 4.0 × 10−7). All other brain region pairs were significantly positively correlated (r > 0.07, p < 0.03). (d) Number of frames for which the observed MSUA deviates from the z = ±4 range from (c), termed significant deviation. V1 and AM–PM had the largest positive deviant frames (289), and CA3 had the least (zero). Unlike CA3, the low number of deviant frames for LGN could not be explained by sample size, because there were more tuned cells in LGN than SUB. (e) Firing in deviant frames above (or below) chance level, as a percentage of the average response. Above chance level deviation was greater or equal to that below, for all brain regions except DG, with the largest positive deviation in AM–PM (9.3%), largest negative deviation in V1 (6.0%), and least in CA3 (zero each). (f) Total firing rate response of visual regions across tuned neurons. All regions had significant negative correlation (r < −0.39, p < 3.4 × 10−34) between the ensemble response and the frame-to-frame (F2F) image correlation (gray line, y-axis on the left) across movie frames. (g) Similar to (f), for hippocampal regions. CA3 response were not significantly correlated with the F2F correlation, dentate gyrus (r = 0.26, p = 4.0 × 10−15) and CA1 (r = 0.21, p = 1.5 × 10−10) responses were positively correlated, and subiculum response was negatively correlated (r = −0.44, p = 2.2 × 10−43). Note the substantially higher mean firing rates of LGN in (f) and subiculum neurons in (g) (colored lines closer to the top) compared to other brain areas.
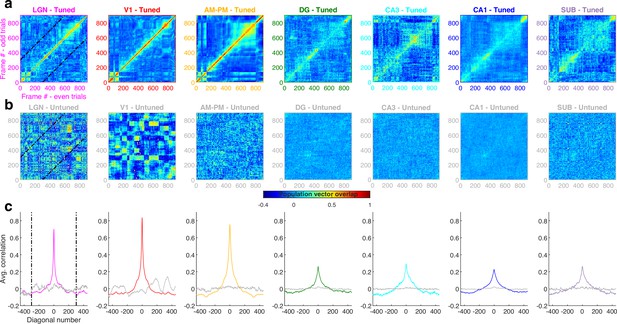
Population vector overlap is wider in hippocampus than visual areas.
(a) Population vector overlap between even and odd trials for the population of tuned neurons show highest overlap along the diagonal (i.e., for the same movie frame) for all brain regions. Each neuron’s response was normalized by its mean rate and the average response in even as well as odd trials was smoothed by a Gaussian window of two frames (66.6 ms, see Methods). Dashed black lines indicate the −300 and +300 frames away from the diagonal. Notice large correlations (close to unity, horizontal color bar) indicating stable responses. The correlations decay quickly to smaller values for the visual areas but more slowly for hippocampal areas, due to their broader movie-fields. (b) Same as (a), but for untuned neurons, resulting in a salt and pepper overlap pattern and low values of correlation, indicating lesser stability than the tuned neurons. Since the majority of cells in the visual areas were tuned, the untuned population was smaller, leading to more variable population vector overlap. (c) The average population vector overlap, computed across all frames, as a function of the number of movie frames away from the diagonal in (a). It had a large value in visual regions for the 0th diagonal (colored lines) indicating stable responses, whereas the untuned neuron population (gray lines) were unstable, with values near zero, or chance level. The highest population vector overlap in hippocampal regions was smaller than visual areas but persisted for more frames, due to their broader movie-fields (full width at half maximum of the peak −17.3 frames for LGN, 22.7 – V1, 39.0 – AM&PM, 49.8 – DG, 57.4 – CA3, 64.7 – CA1, and 59.2 – subiculum).
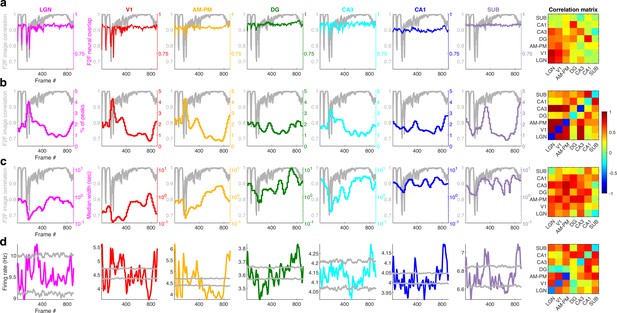
Movie-field properties strongly reflect the frame-to-frame correlation structure of the movie in the visual but not hippocampal areas.
(a) The adjacent movie frame (framen,framen+1) correlation coefficient, indicating the similarity of two consecutive frames, termed F2F image correlation, is shown in gray. Similarly, the correlation coefficient between the population vector of neural responses between adjacent frames was termed F2F neural correlation, computed separately for each brain region is shown in color. The relationship between F2F image, and F2F neural correlation across brain regions is shown in the matrix on the right. Diagonal entries indicate correlation between F2F image and F2F neural, with largest correlation for LGN (+0.82), followed by V1 (+0.75), CA3 (+0.56), DG (+0.52), AM&PM (+0.38), CA1 (+0.14), and SUB (+0.07). All correlations were significant (p < 1.1 × 10−3). Above diagonal entries indicate the correlation coefficient between brain region pairs. Below diagonal entries indicate the same but using partial correlations that factor out the F2F image correlation. All partial correlations were significant (p < 5.4 × 10−3), except V1–CA1 and AM&PM–CA3. (b) Histogram of the number of movie-field peaks (i.e., movie-field density) across all tuned neurons in a brain region, as a function of the movie frame. This distribution was significantly non-uniform (Chi-square goodness-of-fit test for uniform distribution, p < 3.8 × 10−6) for all brain regions. All distributions were significantly negatively correlated with F2F image correlation (p < 10−7). These correlations were much stronger in visual (LGN −0.77, V1 −0.73, AM–PM −0.71) areas than hippocampal areas (DG −0.23, CA3 −0.70, CA1 −0.18, SUB −0.27). The largest partial correlation after factoring out the F2F image correlation was between LGN–V1 and V1–CA3 (0.89) and the least between LGN–DG (0.16). All partial correlations were significant (p < 1.2 × 10−6). (c) Same as (b), but for the median duration of movie-fields. F2F image correlation shown in gray, with larger correlation between consecutive frames between frames 400 and 800 clearly reflected in larger movie-field durations in visual areas. All distributions were significantly non-uniform (Chi-square goodness-of-fit test, p < 10−100) and all distributions were significantly positively correlated with F2F image correlation (r > 0.24, p < 2 × 10−13), with greater values for visual areas (LGN +0.61, V1 +0.51, AM–PM +0.55) than hippocampal (DG +0.39, CA3 +0.58, CA1 +0.42, SUB +0.24). Note that the y-axes for the histogram are log-scaled and show larger median durations for hippocampal regions than visual. The largest correlation was between AM&PM–CA3 (0.81) and the least between CA3–SUB (−0.03). All partial correlations were significant (p < 4.4 × 10−4), except LGN–CA1 (p = 0.11) and CA3–SUB (p = 0.39). (d) Total firing rate across all broad spiking neurons in different brain regions, showing similar non-uniformity as Figure 3c. All brain regions had significantly negative correlation with the F2F image correlation (r < −0.08, p < 0.03), except DG, which was significantly positively correlated (r = 0.21, p = 2.4 × 10−10). The largest number of above chance (gray lines) deviations were seen for AM–PM (340 frames), and least for CA3 (57 frames), which could be due to the low cell count in CA3. Below chance level deviations were least common in LGN (25 frames), and most common in AM–PM (441 frames). The largest partial correlation among brain region pairs was between DG–CA1 (0.76) and the least between V1–DG (−0.1) after factoring out the F2F image correlation. All partial correlations were significant (p < 3.2 × 10−3), except LGN–DG (p = 0.81). Similar to Figure 3c–e.
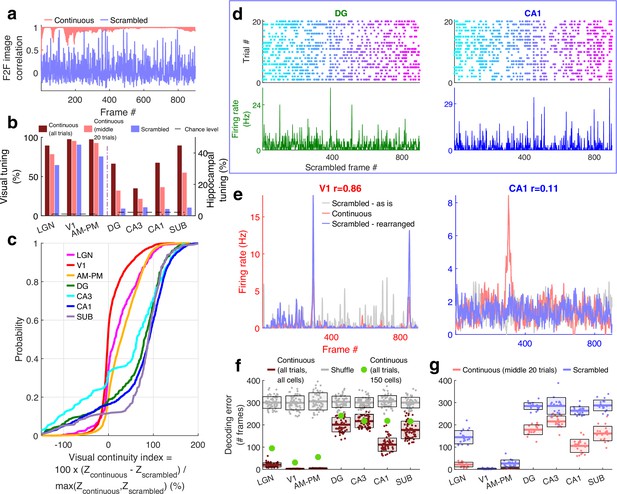
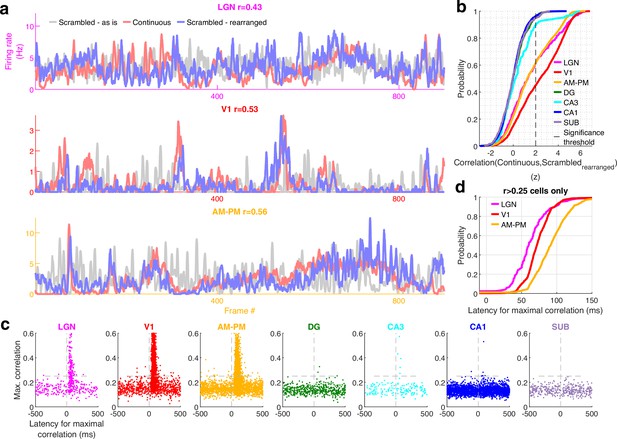
Larger reduction of selectivity in hippocampal than visual regions due to scrambled presentation.
(a) Similarity between the visual content of one frame with the subsequent one, quantified as the Pearson correlation coefficient between pixel–pixel across adjacent frames for the continuous movie (pink) and the scrambled sequence (lavender), termed F2F image correlation. Similar to Figure 3g. For the scrambled movie, the frame number here corresponded to the chronological frame sequence, as presented. (b) Fraction of broad spiking neurons significantly modulated by the continuous movie (red) or the scrambled sequence (blue) using z-scored sparsity measures (similar to Figure 1, see Methods). For all brain regions, continuous movie generated greater selectivity than scrambled sequence (KS-test p < 7.4 × 10−4). (c) Percentage change in the magnitude of tuning between the continuous and scrambled movies for cells significantly modulated by either continuous or scrambled movie, termed visual continuity index. The largest drop in selectivity due to scrambled movie occurred in CA1 (90.3 ± 2.0%), and least in V1 (−1.5 ± 0.6%). Visual continuity index was significantly different between all brain region pairs (KS-test p < 0.03) and significantly greater for hippocampal areas than visual (8.2-fold, p < 10−100). (d) Raster plots (top) and mean rate responses (color, bottom) showing increased spiking responses to only one or two scrambled movie frames, lasting about 50 ms. Tuned responses to scrambled movie were found in all brain regions, but these were the least frequent in DG and CA1. (e) One representative cell each from V1 (left) and CA1 (right), where the frame rearrangement of scrambled responses resulted in a response with high correlation to the continuous movie response for V1, but not CA1. Pearson correlation coefficient values of continuous movie and rearranged scrambled responses are indicated on top. (f) Average decoding error for observed data (see Methods), over 60 trials for continuous movie (maroon), was significantly lower than shuffled data (gray) (KS-test p < 1.2 × 10−22). Solid line – mean error across 60 trials using all tuned cells from a brain region, shaded box – standard error of the mean (SEM), green dots – mean error across all trials using a random subsample of 150 cells from each brain region. Decoding error was lowest for V1 (30.9 frames) and highest in DG (241.2) and significantly different between all brain regions pairs (p < 1.9 × 10−4), except CA3–CA1, CA3–subiculum, and CA1–subiculum (p > 0.63). (g) Similar to (f), decoding of scrambled movie was significantly worse than that for the continuous movie (KS-test p < 2.6 × 10−3). Scrambled responses, in their ‘as is’, chronological order were used herein. Lateral geniculate nucleus (LGN) decoding error for scrambled presentation was 6.5× greater than that for continuous movie, whereas the difference in errors was least for V1 (1.04×). Scrambled movie decoding error for all visual areas and for CA1 and subiculum was significantly smaller than chance level (KS-test p < 2.6 × 10−3), but not DG and CA3 (p > 0.13). The middle 20 trials of the continuous movie were used for comparison with the scrambled movie since the scrambled movie was only presented 20 times. Middle trials of the continuous movie were chosen as the appropriate subset since they were chronologically closest to the scrambled movie presentation.
Scrambled movie elicits narrower but more movie-fields per cell than the continuous movie in all the visual regions.
(a) Cumulative distribution of the total number of fields per cell for the scrambled movie shows the largest number of fields in LGN (mean ± standard error of the mean [SEM], 31.8 ± 2.0), followed by V1 (24.0 ± 0.38) and last AM–PM (11.1 ± 2.1). All three brain regions were significantly different from each other (KS-test p < 2.0 × 10−5). (b) The median scrambled movie-field duration was shortest in LGN (43.9 ± 131.2 ms), intermediate in V1 (46.2 ± 24.8), and widest in AM–PM (77.6 ± 40.1 ms), and differences were significant (p < 7.0 × 10−4). This was much smaller than for the continuous movie (Figure 2). (c) Durations of fields for scrambled sequence across all fields of all neurons from a brain region. These were narrowest in LGN (31.3 ± 6.5 ms), followed by V1 (38.6 ± 0.2) and last AM–PM (64.3 ± 6.7). All differences were significant (KS-test p < 7.2 × 10−136). (d) Despite these differences, the cumulative duration of movie-fields was comparable across the three brain regions (1.69 ± 0.05 s for V1, 2.03 ± 0.07 for AM–PM, and 2.4 ± 0.2 for LGN), but significantly different (p < 1.7 × 10−5). Note the linear scale on the x-axis in this panel compared to the log-scale in other panels. (e) Ratio of field durations, i.e., mega-scale index, for the scrambled movie was smallest in V1 (15.5 ± 1.6), intermediate in LGN (16.3 ± 5.4), and largest in AM–PM (23.4 ± 2.1), and not significantly different between V1 and LGN (p = 0.28). V1–AM&PM and LGN–AM&PM were significantly different (p < 5.7 × 10−5). (f) Cumulative spiking activity, summed across all movie-fields of a given neuron was largest in V1 (3.8 ± 0.1), intermediate in LGN (2.3 ± 0.1), and smallest in AM–PM (2.0 ± 0.07), and significantly different between all brain region pairs (p < 0.02).
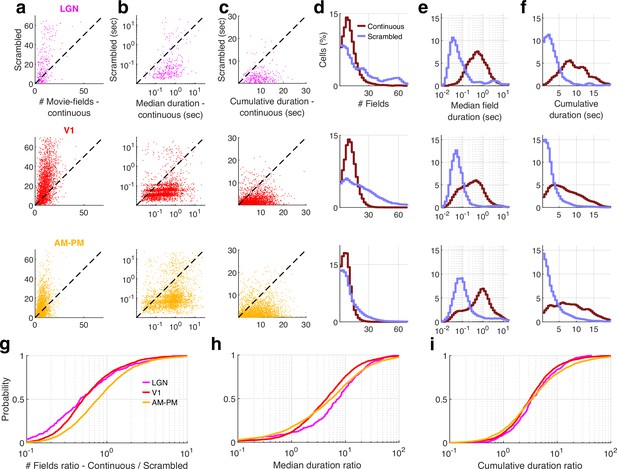
Cell by cell comparison of continuous versus scrambled movie responses.
Data for only those visual area neurons that were significantly modulated by both the continuous and scrambled movie were used. (a) The number of movie-fields per cell for the continuous movie was significantly smaller than that for scrambled sequence in all brain areas (LGN – continuous mean ± standard error of the mean [SEM] = 10.7 ± 0.42, scrambled = 31.8 ± 2.0, KS-test p = 2.0 × 10−23, V1 −10.8 ± 0.11 vs. 24.0 ± 0.38, KS-test p = 3 .7 × 10−210, AM&PM −6.9 ± 0.07, vs. 11.1 ± 0.21, KS-test p = 1.3 × 10−57). Data are additionally scattered by a small random number for the ease of visualization. (b) Median duration of movie-fields for a cell was significantly larger for continuous movie, compared to scrambled sequence in all visual regions. (LGN continuous = 0.46 ± 0.08 s, scrambled = 0.04 ± 0.13 s, KS-test p = 7.1 × 10−65, V1 −0.25 ± 0.03 vs. 0.04 ± 0.02 s, KS-test p < 10−150, AM&PM 0.65 ± 0.04 vs. 0.08 ± 0.04 s, KS-test p < 10−150). (c) Cumulative duration of all movie-fields for a cell was significantly larger for continuous movie, compared to scrambled sequence in all visual regions (LGN continuous = 8.9 ± 0.23 s, scrambled = 2.4 ± 0.19 s, KS-test p = 3.2 × 10−69, V1 −6.1 ± 0.09 vs. 1.69 ± 0.05 s, KS-test p = 3.3 × 10−296, AM–PM 7.8±0.1 s, vs. 2.0 ± 0.07, KS-test p = 9.0 × 10−318). (d) Histogram of number of fields per cell, for continuous and scrambled movies. (e) Logarithmically spaced histogram of median field durations was significantly different between continuous and scrambled sequence. (f) Similar to (e), histogram of cumulative duration of movie-fields for each cell. (g) The ratio of number of fields per cell between continuous and scrambled movies was biased to smaller than unity values for all brain regions, with the largest bias for LGN (0.46 ± 0.08), intermediate for V1 (0.5 ± 0.04), and least for AM–PM (0.77 ± 0.05). (h) The median field duration ratio was biased to values greater than unity, with the largest bias for LGN (7.4 ± 1.4), least for V1 (4.5 ± 0.68), and intermediate for AM–PM (5.5 ± 0.82). (i) The cumulative field duration ratio was also biased to values greater than unity, with similar biases for LGN (3.37 ± 0.36), V1 (3.1 ± 0.3), and AM–PM (3.3 ± 0.67).
Multiple single-unit activity (MSUA) across all movie-tuned neurons in a brain region shows greater modulation than chance for the scrambled sequence in all visual areas.
(a) Stack plot of tuned responses to the scrambled movie presentation from each brain region, sorted according to the frame with peak response. Each firing rate profile is normalized by the peak response causing the diagonal to be unity. The average firing rate of non-peak frames (similar to Figure 3b, legend) was smallest (0.50× of the average peak response across all neurons) for V1, followed by AM&PM – 0.56 and largest for LGN – 0.65. (b) Colored trace – average response, across all tuned responses from (a), gray trace – chance level, z = ±4, corresponding to the p = 0.025 level after Bonferroni correction. (c) Number of frames for which the observed response exceeds (or falls below) z = ±4 cutoff from (b), called significantly deviant frames. V1 had the largest number of positive (279 frames) and negative (297) deviant frames, similar to the continuous movie (Figure 3d, and 289 positive and 324 negative). AM–PM had intermediate (225 and 235) deviant frames for the scrambled movie, which was lower than the continuous movie (285 and 454). LGN had the least number of significantly deviant frames (31 and 29), larger than the continuous movie (12 and 0). (d) Firing rate deviation above chance levels, corresponding to the significant frames, as identified in (c), normalized by the mean rate of the MSUA. Largest deviation was observed in V1 (above – 3.1 and below – 2.7%), and least in LGN (1.1% and 0.45%). Compare with Figure 3. (e) Frame-to-frame (F2F) image correlation, from Figure 4a for comparison. This had no structure and values hovered around zero (unlike the continuous movie) and was not significantly correlated with the MSUA responses in (b), for any of the brain regions (Pearson correlation coefficient LGN p = 0.71, V1 p = 0.06, AM–PM p = 0.21). Despite this, the MSUA shows significant modulation.
Latency of responses to the scrambled-sequence corresponds to the anatomical hierarchy of visual areas.
(a) Average response for one representative cell from each visual region, that had high similarity between the continuous movie and the rearranged scrambled sequence responses (see Methods). Gray response in background corresponds to the chronological scrambled sequence. (b) Cumulative histogram of z-scored correlation between continuous and scrambled-rearranged tuning responses (see Methods). Dotted black line indicates significance threshold of z > 2. (c) The latency at which continuous and scrambled-rearranged responses were maximally correlated showed high values (heuristically above 0.25) in a short range of positive latencies for LGN, V1, and AM–PM neurons. This analysis was restricted to neurons tuned in continuous as well as scrambled movies. Similar analysis for hippocampal regions resulted in almost no correlations above 0.25. (d) Cumulative histogram of latencies, when the continuous and scrambled-rearranged responses were maximally correlated, was the smallest for LGN (59.5 ± 4.6 ms), and largest for higher visual areas, AM–PM (91.6 ± 1.6 ms). Hippocampal regions were excluded, owing to lack of data with correlation above 0.25.
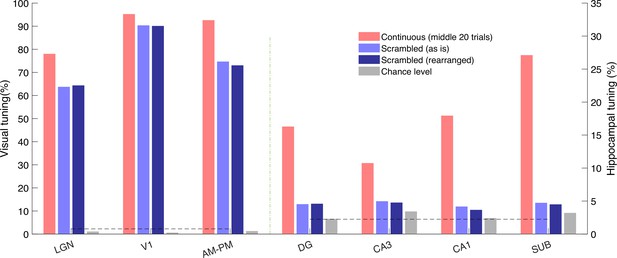
Movie tuning in hippocampal neurons remains near chance level even after rearranging scrambled movie frames.
Histogram showing percentage of tuned cells for movie presentation in the continuous (red), scrambled order, taken as is (light blue), or the scrambled order but rearranged (dark blue). Movie tuning was significantly higher for the continuous presentation (p < 3.5 × 10–3) than the scrambled as is condition or scrambled rearranged condition (p < 2.6 × 10–6), in all brain regions. Movie tuning for the scrambled presentation taken as is, or after rearrangement was not significantly different for all brain regions (p > 0.08), except LGN (p = 1.3 × 10–5) and V1 (p = 0.001), although the prevalence of tuning was comparable (63.7% and 64.3% – LGN and 90.1% and 90.0% – V1).
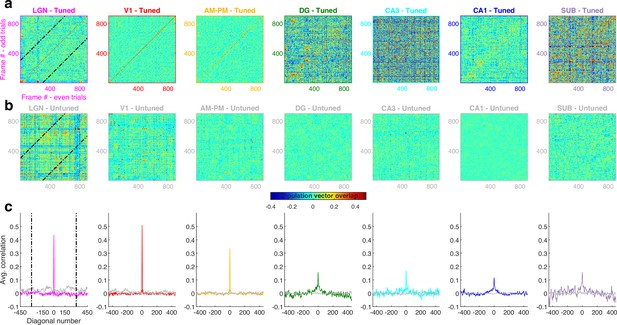
Population vector overlap was narrower for the scrambled compared to the continuous movie.
(a) Population vector overlap between even and odd trials for tuned neurons showing higher overlap along the diagonal for all brain regions. Black lines indicate the −300 and +300 diagonal, whereas the main diagonal is the 0th diagonal. (b) Same as (a) but for untuned neurons, resulting in a salt and pepper overlap without higher correlation around the diagonal. (c) The average overlap along diagonals had a large value in visual regions for the 0th diagonal, which was not true for the untuned neuron population. Average correlation in hippocampal regions was broader and lesser in magnitude compared to visual regions. Similar to Figure 3—figure supplement 1. Full width at half maximum of the peak – 4.4 frames for LGN, 4.8 – V1, 5.2 – AM&PM, 7.6 – DG, 5.7 – CA3, 10.8 – CA1, and 15.1 – subiculum, even though consecutive frames in the scrambled presentation were largely uncorrelated.
Scrambled movie.
Frames from the sequential video clip (Figure 1—video 1) were presented in a scrambled sequence, with the same sequence repeated 20 times (2 blocks of 10 trials each). Frame numbers in the scrambled sequence are indicated in the top right corner.





