Origin of wiring specificity in an olfactory map revealed by neuron type–specific, time-lapse imaging of dendrite targeting
Figures
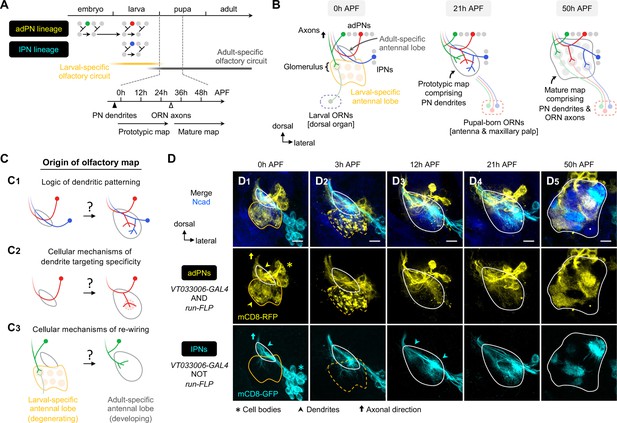
Organization and development of the adult olfactory circuit in Drosophila.
(A, B) Timeline (A) and schematic illustration (B) of Drosophila olfactory circuit development. Green, red, and blue circles denote the birth of embryonic-born anterodorsal projection neuron (adPN), larval-born adPN, and larval-born lPN, respectively. At the onset of metamorphosis, the larval-specific olfactory circuit degenerates; larval olfactory receptor neurons (ORNs) die while embryonic-born adPNs prune their larval-specific processes and re-extend new processes into the adult-specific olfactory circuit. In the adult-specific olfactory circuit, projection neuron (PN) dendrites extend first and form a prototypic map. This is followed by an extension of ORN axons and synaptic partner matching between cognate PN dendrites and ORN axons to form a mature map. Solid and open arrowheads in A indicate onset of innervation for PN dendrites and ORN axons, respectively. (C) Overview of this study investigating the logic of dendritic patterning (C1; see Figures 3 and 4) as well as cellular mechanisms of dendrite targeting specificity (C2; see Figures 6 and 7) and re-wiring (C3; see Figure 8) that contribute to the developmental origin of the adult Drosophila olfactory map. (D) Staining of fixed brains at indicated stages showing dendrite development of adPNs (VT033006+ run+ ; labeled in yellow) and lPNs (VT033006+ run–; labeled in cyan). As run-FLP is expressed before 0 h APF in adPN but not lPN neuroblasts, we can use it to label adPNs and lPNs with two distinct colors using an intersectional reporter (see Materials and methods for the genotype). Yellow arrowheads in (D1) mark larval- and adult-specific dendrites of adPNs in larval- and adult-specific antennal lobes, respectively. Cyan arrowheads in (D3) denote specific targeting of lPN dendrites at the opposite ends of the dorsomedial-ventrolateral axis. (D1): N=12; (D2): N=7; (D3): N=17; (D4): N=10; (D5): N=12. Common notations in this study: Unless otherwise indicated, all images in this and subsequent figures are partial z projections of confocal stacks of representative images. N indicates the number of antennal lobes imaged. Antennal lobe neuropils are revealed by N-Cadherin (Ncad; in blue) staining. Adult-specific (developing) antennal lobe is outlined with a white solid line. Larval-specific antennal lobe is outlined with an orange line (dashed line used to denote the degeneration stage) and is distinguished from the developing antennal lobe by the more intense nc82 staining as shown in Figure 1—figure supplement 1 (nc82 channel not shown here). Asterisks (*) indicate PN cell bodies, which are outside the antennal lobe neuropil (and sometimes appear on top because of the z-projections). Arrowheads mark PN dendrites. Arrows mark PN axons projecting towards higher olfactory centers (see Figure 1—figure supplement 2 for PN axons at their targets in the mushroom body and lateral horn). h APF: hours after puparium formation; h ALH: hours after larval hatching. DL: dorsolateral; DM: dorsomedial; VM: ventromedial; VL: ventrolateral. Scale bar = 10 µm.
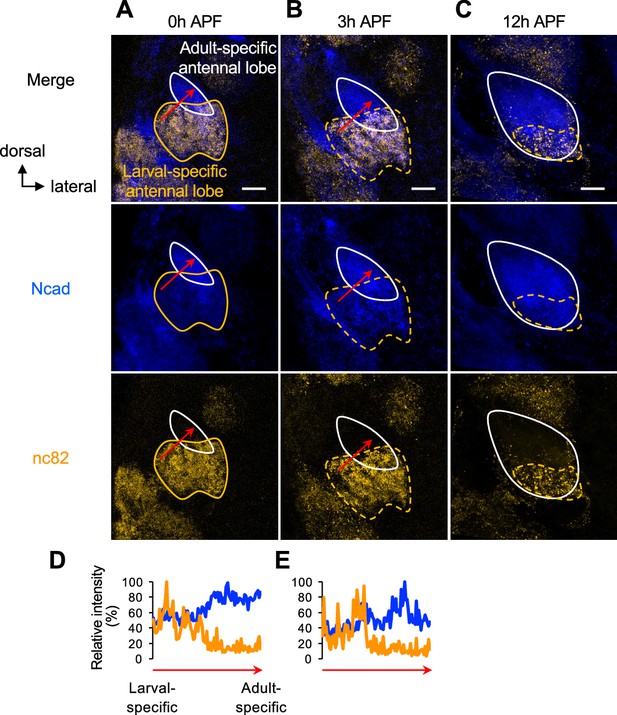
Visualization of larval- and adult-specific antennal lobes by co-staining of Ncad and nc82.
(A–C) N-Cadherin (Ncad) and nc82 staining of antennal lobes are shown in Figures 1D1—3. Adult-specific antennal lobes characteristic of strong Ncad and weak nc82 staining are outlined by a white solid line. Larval-specific antennal lobes characteristic of weak Ncad and strong nc82 are outlined by an orange solid/dashed lines. Note that larval-specific antennal lobes are more anterior to adult-specific antennal lobes, and thus appear to overlap in these z-projections. See sample size in Figure 1 legend. (D, E) Quantification of the relative intensity (%) of Ncad and nc82 staining from larval- to adult-specific antennal lobes at 0 hr APF (D) and 3 hr APF (E) (red arrows in D and E correspond to those in A and B, respectively). See Figure 1 legend for common notations.
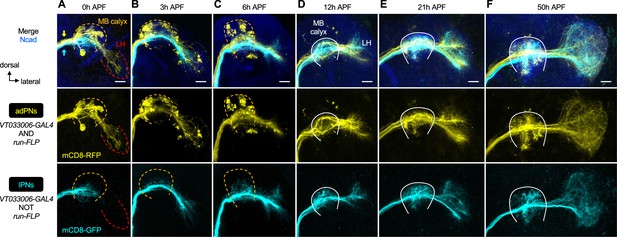
Projection neuron (PN) axon development across pupal stages.
(A–F) Staining of fixed brains at indicated stages showing axon development of anterodorsal projection neurons (adPNs) (VT033006+ run+ ; labeled in yellow) and lPNs (VT033006+ run–; labeled in cyan). Yellow and cyan arrows in A indicate the segregation of adPN and lPN axons along the inner antennocerebral tract. MB: mushroom body; LH: lateral horn. MB calyx (where PN axons and Kenyon cells of the mushroom body form synapses) and LH neuropils (where PN axons form synapses with their postsynaptic target neurons) are outlined as follows. In A–C, an orange dashed line denotes the degeneration of larval-specific MB calyx. In A, larval-specific LH located more ventrally is outlined by a red dashed line. In D–F, the developing adult-specific MB calyx is outlined by a white solid line and the adult-specific LH is to the right of the calyx. (A): N=4; (B): N=1; (C): N=4; (D): N=3; (E): N=4; (F): N=2. See Figure 1 legend for common notations.
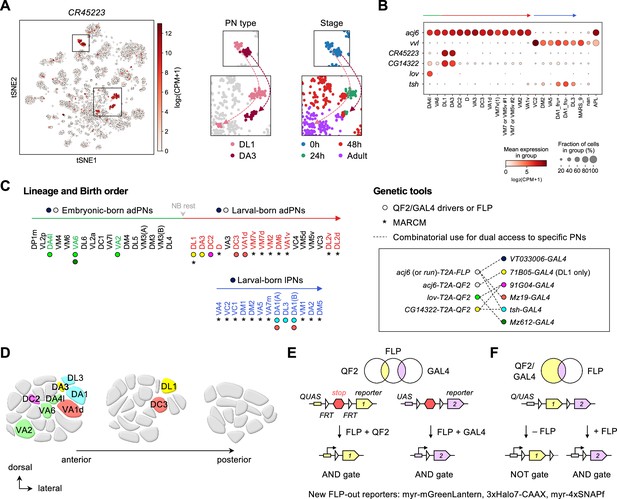
Expanded genetic toolkit for dual-color, type-specific labeling of projection neurons (PNs).
(A) tSNE plot of PN single-cell transcriptomes, color-coded according to CR45223 expression level in [log2(CPM +1)], where CPM stands for transcript counts per million reads. Zoom-in of boxes in the tSNE plot (left) is shown on the right, and color-coded according to PN types and developmental stages. (B) Dot plot showing the expression of acj6, vvl, CR45223, CG14322, lov, and tsh in 0 hr APF PNs arranged according to their birth order and lineage (green: embryonic-born anterodorsal projection neuron (adPNs); red: larval-born adPNs; blue: larval-born lPNs). Unit of expression is [log2(CPM +1)] as in A. Data from panels A are B are from Xie et al., 2021. (C) Birth orders of adPNs and lPNs summarized by Lin et al., 2012; Yu et al., 2010 and genetic tools used to access them. Left: Accessible PN types are colored. Circles beneath the PN types denote QF2/GAL4 drivers used to access them. Asterisks beneath the PN types denote access by MARCM. Gray arrowhead marks neuroblast (NB) rest. Right: Genetic tools. Inset shows the combinatorial use of QF2/FLP and GAL4 (linked by dashed lines) for comparative analyses of dendrite development of two groups of PNs in the same animal. (D) Schematic of glomerular projections of QF2/GAL4-accessible PNs in the adult antennal lobe. Indicated glomeruli are color-coded based on the genetic tools used to access them. See the color code in C. (E, F) Schematic of intersectional logic gates for dual-color labeling of PNs. See Figure 2—figure supplement 2 for newly generated FLP-out reporters.
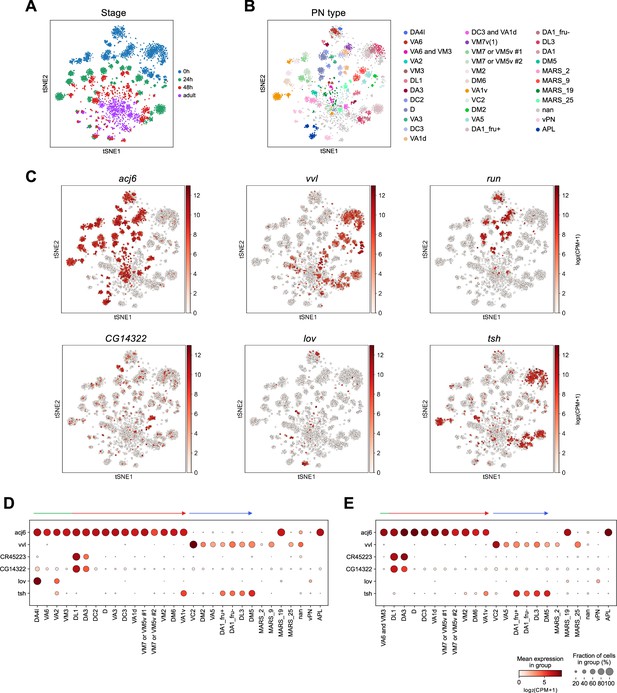
Expression of projection neuron (PN) marker genes across development.
(A–C) tSNE plots of PN transcriptomes, color-coded according to developmental stages (A), PN types (B), or the expression levels of the indicated gene [log2(CPM +1)] (C) using scRNA-seq data from Xie et al., 2021. (A) and (B) are reproduced from Figure 7A and B of Xie et al., 2021. (D, E) Dot plot showing the expression levels of acj6, vvl, CR45223, CG14322, lov, and tsh in PNs [log2(CPM +1)] at 24 hr APF (D) and 48 hr APF (E).
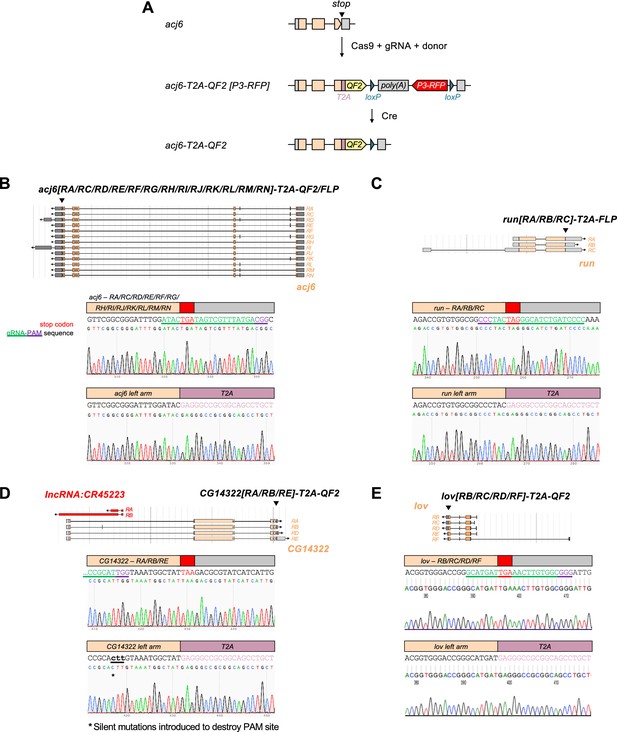
Generation of T2A-QF2/FLP transgenic flies by CRISPR/Cas9.
(A) Schematic of generation of transgenic driver lines by CRISPR/Cas9. acj6-T2A-QF2 is shown as an example. (B–E) Top: Transcripts of acj6 (B), run (C), CG14322 (D), and lov (E) visualized using FlyBase JBrowse. Bottom: Targeted insertion of T2A-QF2/FLP right before the stop codon of the endogenous gene. Stop codon and gRNA-PAM sequence are color-coded as indicated.
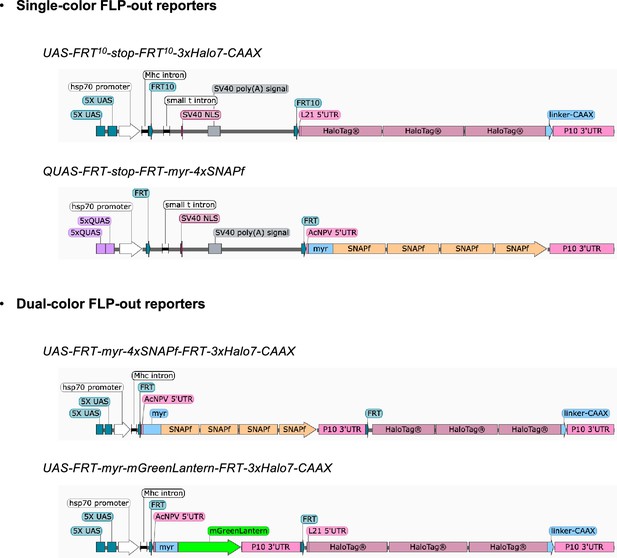
Design of single- and dual-color FLP-out reporters.
Images, created with SnapGene, show four newly generated Q/UAS-based single- and dual-color FLP-out reporters.
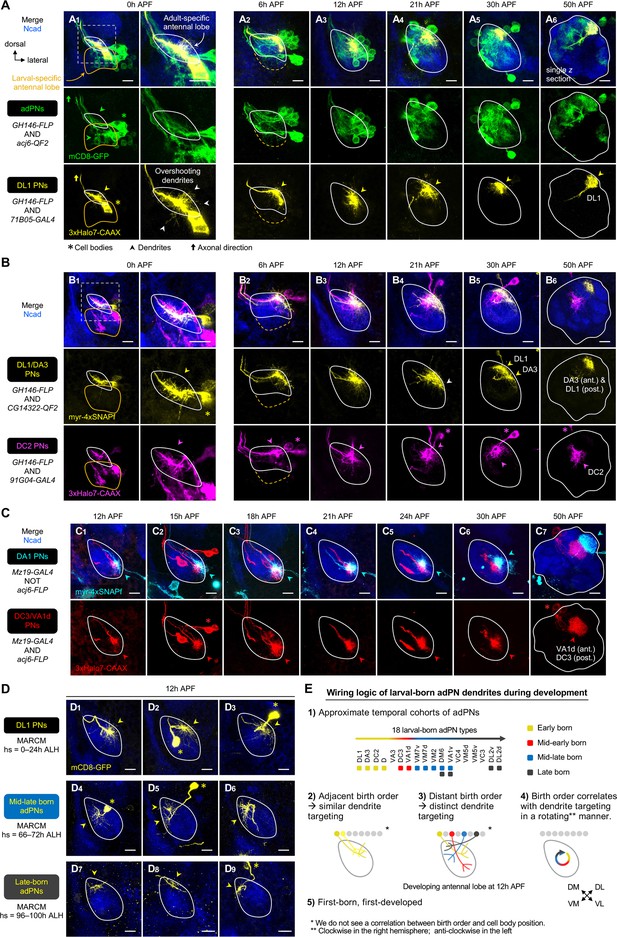
Birth order–dependent spatial patterning of anterodorsal projection neuron (adPN) dendrites in the developing antennal lobe.
(A) Confocal images of fixed brains at indicated stages showing dendrite development of adPNs (acj6+; labeled in green) and DL1 adPNs (71B05+; labeled in yellow). Right column of A1 shows a zoom-in of the dashed box. The labeling of acj6+ adPNs outlines the developing antennal lobe and is used in dual-color AO-LLSM imaging later (see Figure 7A–C). White arrowheads in (A1) mark dendrites overshooting the antennal lobe. (A1): N=14; (A2): N=12; (A3): N=14; (A4): N=6; (A5): N=4; (A6): N=4. (B) Confocal images of fixed brains at indicated stages showing dendrite development of DL1/DA3 adPNs (CG14322+; labeled in yellow) and DC2 adPNs (91G04+; labeled in magenta). As 91G04-GAL4 labels some embryonic-born projection neurons (PNs) from 0 to 6 hr APF, their neurites are found in the larval-specific antennal lobe (B1, 2). Right column of (B1) shows a zoom-in of the dashed box. White arrowhead in (B4) denotes the more ventrally targeted DL1/DA3 dendrites. (B1): N=6; (B2): N=5; (B3): N=12; (B4): N=4; (B5): N=7; (B6): N=2. (C) Confocal images of fixed brains at indicated stages showing dendrite development of DC3/VA1d adPNs (Mz19+ acj6+; labeled in red) and DA1 lPNs (Mz19+ acj6–; labeled in cyan). (C1): N=14; (C2): N=6; (C3): N=4; (C4): N=10; (C5): N=10; (C6): N=6; (C7): N=4. (D) Confocal images of single-cell MARCM clones (in yellow) of DL1 PNs (D1–3), mid-late larval-born adPNs (D4–6), and late larval-born adPNs (D7–9) in 12 hr APF pupal brains, generated by heat shocks (hs) at indicated times. Three biological samples are shown for each of the indicated adPN cohorts. D1–3: N=5; D4–6: N=4; D7–9: N=8. (E) Summary of wiring logic of larval-born adPN dendrites to form an olfactory map in the 12 hr APF developing antennal lobe. See Figure 1 legend for common notations.
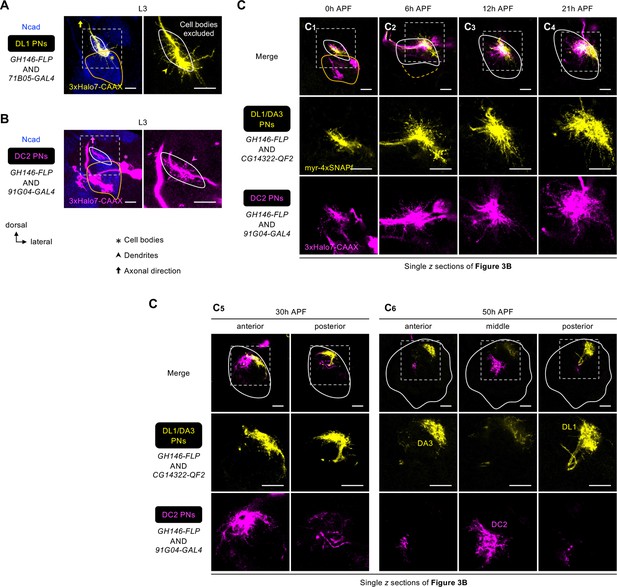
Dendrite development of early larval-born projection neurons (PNs).
(A) Dendritic extension of DL1 PNs (71B05+; labeled in yellow) across the developing antennal lobe at the wandering third instar larval stage (L3). Zoom-in of the dashed box shown on the right. N=3. (B) Dendritic extension of DC2 PNs (91G04+; labeled in yellow) across the developing antennal lobe at L3. Zoom-in of the dashed box shown on the right. N=8. (C) Single z sections of Figure 3B showing dendrite development of DL1/DA3 adPNs (CG14322+; labeled in yellow) and DC2 adPNs (91G04+; labeled in magenta). See Figure 1 legend for common notations.
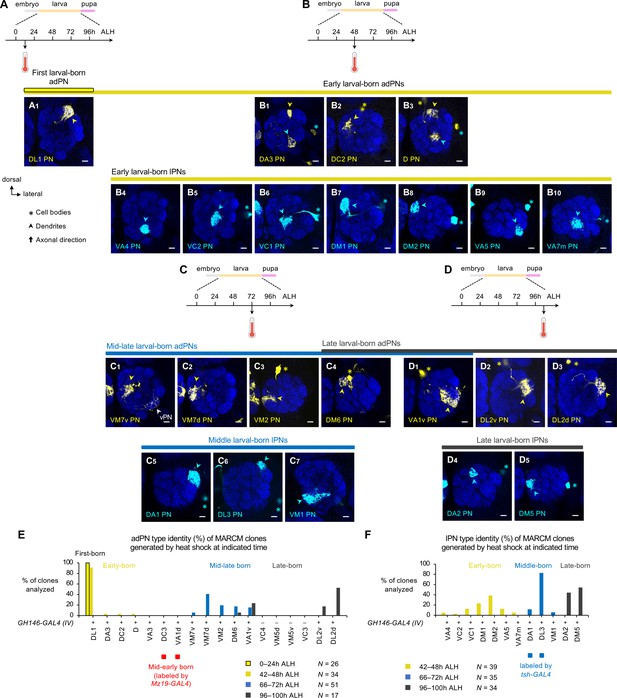
MARCM-labeled single-cell projection neurons (PNs) of indicated lineages in adult brains.
(A) MARCM clone of DL1 PN (in yellow) generated by heat shock at 0–24 hr after larval hatching (ALH). (B) MARCM clones of early larval-born PNs (B1–3: adPNs in yellow; B4–10: lPNs in cyan) generated by heat shock at 42–48 hr ALH. In (B1), a single-cell clone of the anterodorsal projection neuron (adPN) lineage and that of the lPN lineage, corresponding to DA3 PN (yellow arrowhead) and VA5 PN (cyan asterisk and arrowhead), were simultaneously generated. In (B3), single-cell adPN and lPN, corresponding to D PN (yellow asterisk and arrowhead) and VA7m PN (cyan asterisk and arrowhead), were simultaneously generated. (C) MARCM clones of mid-late larval-born PNs (C1–4 and D1: adPNs in yellow; C5–7: lPNs in cyan) generated by heat shock at 66–72 hr ALH. In (C1), the white arrowhead mark processes of vPN clone that do not belong to VM7v PN. (D) MARCM clones of late larval-born PNs (C4 and D1–3: adPNs in yellow; D4–5: lPNs in cyan) generated by heat shock at 96–100 hr ALH. (E) Percentage bar graph showing the adPN type identity of MARCM clones generated by heat shock at indicated times. Sample size N indicates the number of clones analyzed. (F) Percentage bar graph showing the lPN type identity of MARCM clones generated by heat shock at indicated times. Sample size N indicates the number of clones analyzed. See Figure 1 legend for common notations.
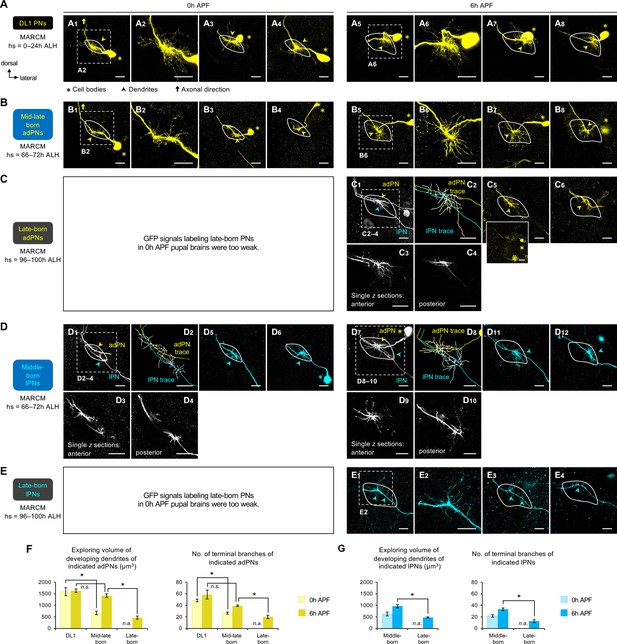
Dendrite development of DL1, middle larval-born, and late larval-born projection neurons (PNs) at early stages.
Adult-specific antennal lobes (revealed by high Ncad staining; not shown) are outlined by a white solid line. (A) MARCM clones of DL1 PNs (in yellow), generated by heat shock at 0–24 hr after larval hatching (ALH), in 0 hr APF (A1–4) and 6 hr APF (A5–8) pupal brains. (B) MARCM clones of mid-late larval-born adPNs (in yellow), generated by heat shock at 66–72 hr ALH, in 0 hr APF (B1–4), and 6 hr APF (B5–8) pupal brains. (C) MARCM clones of late larval-born anterodorsal projection neurons (adPNs) (in yellow), generated by heat shock at 96–100 hr ALH, in 6 hr APF pupal brains. In (C1), single-cell MARCM clones of adPN and lPN lineages were simultaneously labeled. (C2) show neurite tracing of adPN (in yellow) and lPN (in cyan) in (C1). Single z sections of (C1) are shown in C3–4. Small inset below (C5) reveals the cell body position. (D) MARCM clones of middle larval-born lPNs (in cyan), generated by heat shock at 66– 72hr ALH, in 0 hr APF (D1–6), and 6 hr APF (D7–12) pupal brains. In D1–4 and D7–10, single-cell adPN and lPN were simultaneously labeled. (D2) and (D8) shows neurite tracing of (D1) and (D7), respectively (adPN in yellow; lPN in cyan). Single z sections of (D1) are shown in D3–4, and those of (D7) are shown in D9–10. (E) MARCM clones of late larval-born lPNs (in cyan), generated by heat shock at 96–100 hr ALH, in 6 hr APF pupal brains. (F–G) Quantification of exploring volume of developing dendrites of indicated PNs: (F) adPNs; (G) lPNs at 0 h and 6 h APF (left). Quantification of the number of terminal branches of indicated PNs: (F) adPNs; (G) lPNs at 0 h and 6 h APF (right). Error bars, SEM; t-test; *p<0.05; n.s., p≥0.05. SEM, standard error of the mean; n.s., not significant; n.a., not applicable. Three antennal lobes are shown and 4–5 PNs analyzed for each of the indicated PN cohorts. Exact sample size N for quantitative analyzes are provided in (Figure 3—figure supplement 3—source data 1).
-
Figure 3—figure supplement 3—source data 1
Source data for Figure 3—figure supplement 3F and G.
- https://cdn.elifesciences.org/articles/85521/elife-85521-fig3-figsupp3-data1-v2.xlsx
3D rendering of z stacks of indicated projection neurons (PNs) in 12 hr APF antennal lobe.
This video shows a 3D rendering of z stacks with rotation along y-axis to visualize PN dendrites in the context of the antennal lobe in three dimensions. See Figure 3 for details.
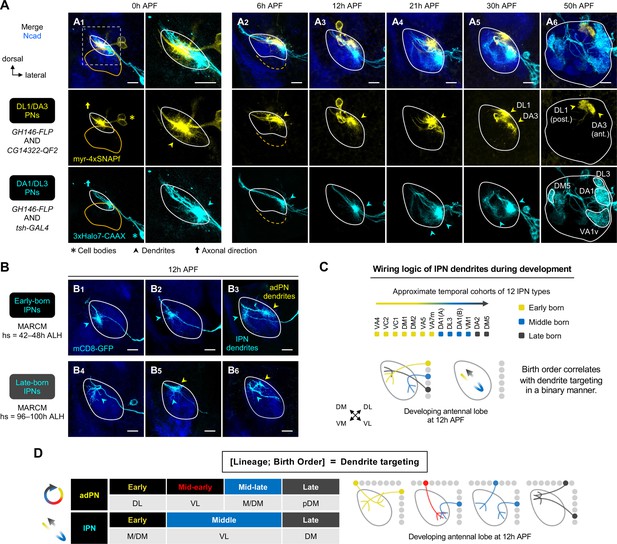
Birth order–dependent spatial patterning of lPN dendrites in the developing antennal lobe.
(A) Confocal images of fixed brains at indicated stages showing dendrite development of DL1/DA3 adPNs (CG14322+; labeled in yellow) and DA1/DL3 lPNs (tsh+; labeled in cyan). Right column of A1 shows a zoom-in of the dashed box. (A1): N=8; (A2): N=4; (A3): N=6; (A4): N=10; (A5): N=4; (A6): N=5. (B) MARCM clones (in cyan) of early (B1–3) and late (B4–6) larval-born lPNs in 12 hr APF pupal brains, generated by heat shocks (hs) at indicated times. In (B3), (B5), and (B6), single-cell clones of anterodorsal projection neuron (adPN) (yellow arrowheads) and lPN (cyan arrowheads) lineages were simultaneously labeled. Three biological samples are shown for each of the indicated lPN cohorts. B1–3: N=4; B4–6: N=6. (C) Summary of wiring logic of larval-born lPN dendrites to form an olfactory map in the 12 hr APF developing antennal lobe. (D) Summary of determination of dendrite targeting of larval-born PNs by lineage and birth order. See Figure 1 legend for common notations.
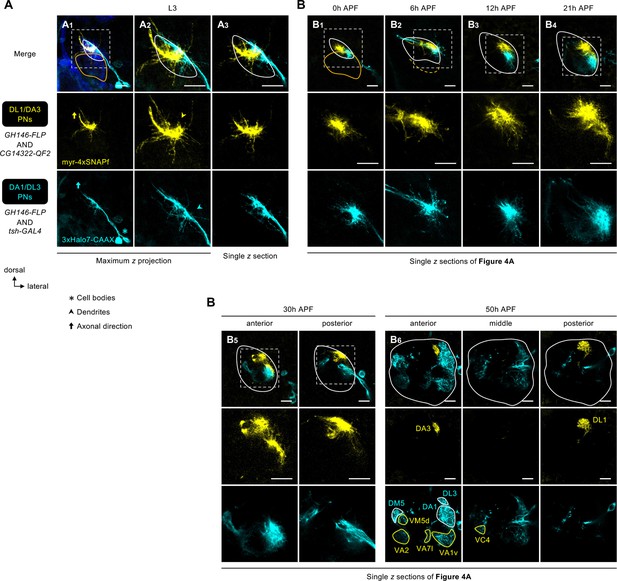
Dendrite development of DL1/DA3 and DA1/DL3 projection neurons (PNs).
(A) Dendritic extension of DL1/DA3 PNs (CG14322+; labeled in yellow) and DA1/DL3 PNs (tsh+; labeled in cyan) across the developing antenna lobe at the wandering third instar larval stage (L3). N=5. (B) Single z sections of Figure 4A showing dendrite development of DL1/DA3 PNs (CG14322+; labeled in yellow) and DA1/DL3 PNs (tsh+; labeled in cyan). In B6, glomeruli innervated by tsh+ anterodorsal projection neuron (adPN) and tsh+ lPN dendrites are outlined in yellow and cyan, respectively. See Figure 1 legend for common notations.
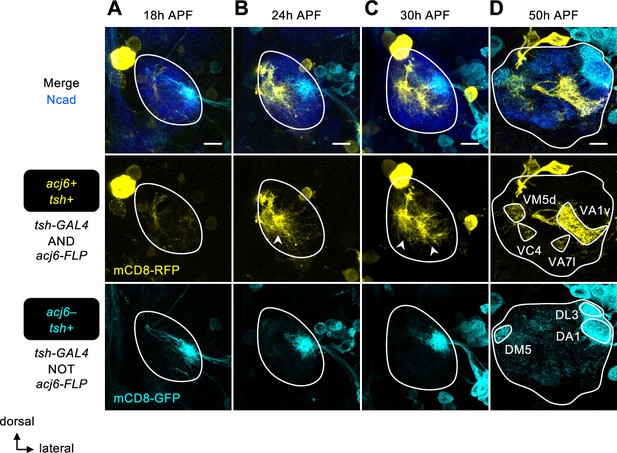
Expression patterns of tsh in the developing antennal lobe during mid-pupal stages.
(A) At 18 hr APF, tsh is only expressed in projection neurons (PNs) of the lateral linage (acj6–; in cyan). N=6. (B–D) From 24 hr APF onwards, tsh is expressed in some anterodorsal projection neurons (adPNs) (acj6+; in yellow), consistent with the transcriptome data (Figure 2B, Figure 2—figure supplement 1C–E). tsh-GAL4 seems to weakly label local interneurons at 50 hr APF (acj6–; in cyan). (B): N=6; (C): N=5; (D): N=4. See Figure 1 legend for common notations.
3D rendering of z stacks of indicated projection neurons (PNs) in 12 hr APF antennal lobe.
This video shows a 3D rendering of z stacks with rotation along y-axis to visualize PN dendrites in the context of antennal lobe in three dimensions. See Figure 4 for details.
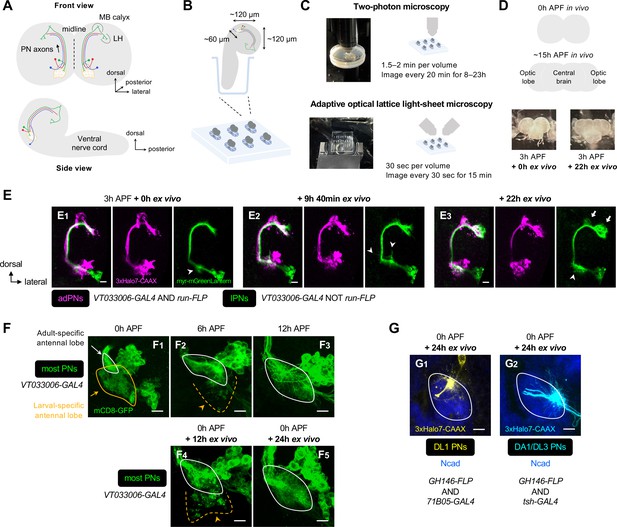
Establishment of an explant system for time-lapse imaging of olfactory map formation.
(A) Schematic of the anatomical organization of the olfactory circuit in early pupal brain (0–3 hr APF). Green, red, and blue denote embryonic-born adPN, larval-born anterodorsal projection neuron (adPN), and larval-born lPN, respectively. MB: mushroom body; LH: lateral horn. (B) Schematic of explant culture system for early pupal brains. Wells created in the Sylgard plate from which brains were imbedded are shown in blue. (C) Schematic of explant culture and imaging system for early pupal brains. (D) Top: Schematic of morphological changes of brain lobes from 0 hr to ~15 hr APF during normal development. Bottom: Morphologies of a brain explant dissected at 3 hr APF and cultured for 0 hr ex vivo and cultured for 22 hr ex vivo. (E) Two-photon time-lapse imaging of adPNs (VT033006+ run+ ; labeled in magenta) and lPNs (VT033006+ run–; labeled in green) in pupal brain dissected at 3 hr APF and cultured for 0–22 hr ex vivo. Arrowheads mark dynamic but transient dendritic protrusions of lPNs in E1, 2, and extensive dendritic innervation of lPNs in (E3). Arrows in (E3) mark axonal innervation of lPNs in the mushroom body calyx and lateral horn. N=3. (F) Confocal images of antennal lobes labeled by VT033006+ projection neurons (PNs) (in green) at 0 hr (F1), 6 hr (F2), and 12 hr (F3) APF in vivo. Confocal images of antennal lobes labeled by VT033006+ PNs in pupal brains were dissected at 0 hr APF and cultured for 12 hr (F4) and 24 hr (F5) ex vivo. (F1): N=6; (F2): N=5; (F3): N=6; (F4): N=8; (F5): N=8. (G) Dendrite targeting regions of DL1 PNs (71B05+; in yellow; G1) and DA1/DL3 PNs (tsh+; in cyan; G2) in the antennal lobes in pupal brains dissected at 0 hr APF and cultured for 24 hr ex vivo. Antennal lobes are revealed by N-Cadherin (Ncad; in blue) staining. (G1): N=5; (G2): N=6. See Figure 1 legend for common notations.
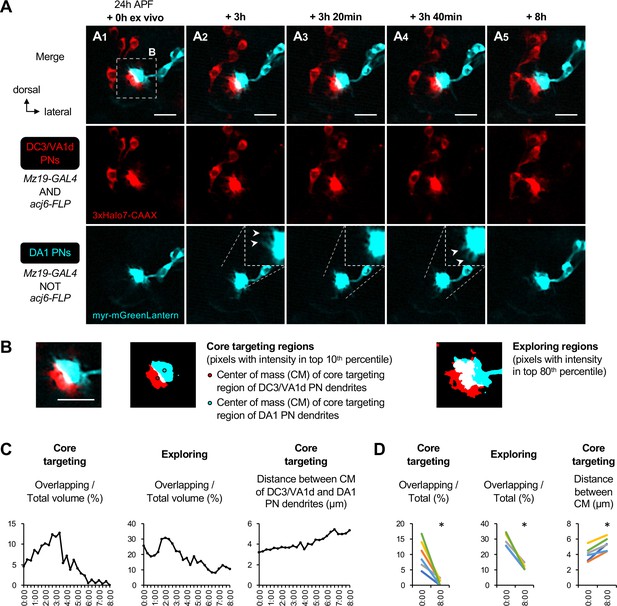
Dendritic segregation of DC3/VA1d adPNs and DA1 lPNs targeting neighboring proto-glomeruli.
(A) Two-photon time-lapse imaging of DC3/VA1d adPN (Mz19+ acj6+; labeled in red) and DA1 lPN (Mz19+ acj6–; labeled in red) dendrites in pupal brain dissected at 24 hr APF and cultured for 8 hr ex vivo. Insets in A2–4 shows the zoom-in. Arrowheads in A2–4 indicate the disappearance (compare A2 with A3) and extension (compare A4 with A3) of dendrites. (B) Core targeting region of projection neuron (PN) dendrites is defined using pixels with intensity in the top 10th percentile. Red and cyan circles mark the centers of mass of the core targeting regions of DC3/VA1d and DA1 PN dendrites, respectively. The exploring region of PN dendrites is defined using pixels with intensity in the top 80th percentile. (C) Left: Ratio of overlapping to total core targeting volume (in percentage) across the 8 hr imaging period. Middle: Ratio of overlapping to total exploring volume (in percentage) across the 8 hr imaging period. Right: Distance between centers of mass of DC3/VA1d and DA1 core targeting regions across the 8 hr imaging period. Sample size N=1. Timestamp 00:00 refers to HH:mm; H, hour; m, minute. (D) Left: Ratio of overlapping to total core targeting volume (in percentage) at 0 hr and 8 hr ex vivo. Middle: Ratio of overlapping to total exploring volume (in percentage) at 0 hr and 8 hr ex vivo. Right: Distance between centers of mass of DC3/VA1d and DA1 core targeting regions at 0 hr and 8 hr ex vivo. Sample size N=6 (see Figure 5—figure supplement 1—source data 1). Error bars, standard error of the mean; t-test; *p<0.05. Timestamp 00:00 refers to HH:mm; H, hour; m, minute.
-
Figure 5—figure supplement 1—source data 1
Source data for Figure 5—figure supplement 1C and D.
- https://cdn.elifesciences.org/articles/85521/elife-85521-fig5-figsupp1-data1-v2.xlsx
Two-photon time-lapse imaging of projection neuron (PN) development.
See Figure 5E for details. Timestamp 00:00:00 refers to HH:mm:ss; H, hour; m, minute; s, second.
Two-photon time-lapse imaging of projection neuron (PN) dendritic segregation.
See Figure 5—figure supplement 1 for details. Timestamp 00:00:00 refers to HH:mm:ss; H, hour; m, minute; s, second.
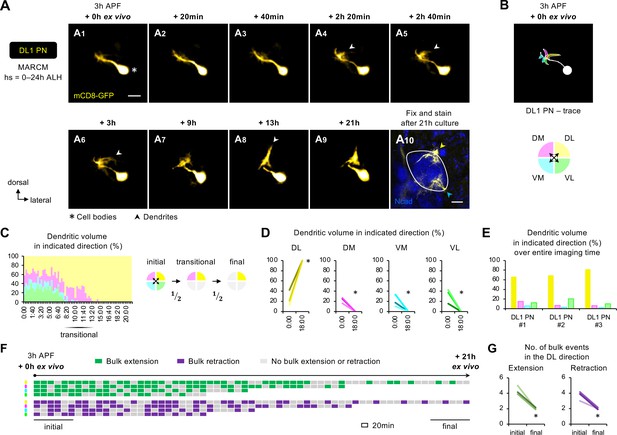
Two-photon time-lapse imaging reveals active dendrite targeting.
(A) Two-photon time-lapse imaging of MARCM-labeled DL1 projection neuron (PN) (pseudo-colored in yellow) in a brain dissected at 3 hr APF and cultured for 21 hr ex vivo (A1–9). Arrowheads in A4–6 denote protrusions of dendritic branches towards the dorsolateral direction. After 21 hr culture, the explant was fixed and immuno-stained for N-Cadherin (Ncad; in blue) to outline the developing antennal lobe (A10). Yellow and cyan arrowheads indicate DL1 PN dendrites and processes of other GH146+ cells, respectively. (B) Neurite tracing of DL1 PN at the beginning of live imaging (3 hr APF + 0 hr ex vivo). Dendrites are categorized based on the directions to which they extend and color-coded accordingly. (C) Left: Quantification of the percentage of dendritic volume in indicated direction during the time-lapse imaging period reveals a transitional phase during which dendrites were found in only two out of the four directions. Right: Schematic of the initial, transitional, and final phases during the course of targeting. ‘½’ denotes the reduction of available trajectory directions by half. Timestamp 00:00 refers to HH:mm; H, hour; m, minute. See Figure 6—source data 1. (D) Quantification of the percentage of DL1 PN dendritic volume in an indicated direction in 3 hr APF cultured brains at the beginning (0 hr ex vivo) and at/near the end of imaging (18 hr ex vivo). DL1 PN sample size = 3. t-test; *p<0.05. Timestamp 00:00 refers to HH:mm; H, hour; m, minute. (E) Quantification of the percentage of the sum of DL1 PN dendritic volume in indicated directions throughout the entire imaging time. DL1 PN sample size = 3. (F) Bulk dendrite dynamics of DL1 PN in Figure 6A. Each row represents bulk dendritic dynamics in the indicated direction (color-coded as in Figure 6B) across the 21 hr imaging period. Each block represents a 20 min window. Bulk extension (in green) and retraction (in magenta) events are defined as dendrites extending and retracting more than 2 μm between two consecutive time windows. The first and last six consecutive windows refer to the initial and final phases of imaging. (G) Quantification of the number of bulk extension and retraction events in the dorsolateral direction during the initial and final phases of imaging. DL1 PN sample size = 3. t-test; *p<0.05.
-
Figure 6—source data 1
Source data for Figure 6C–G and Figure 6—figure supplement 1D and E.
- https://cdn.elifesciences.org/articles/85521/elife-85521-fig6-data1-v2.xlsx
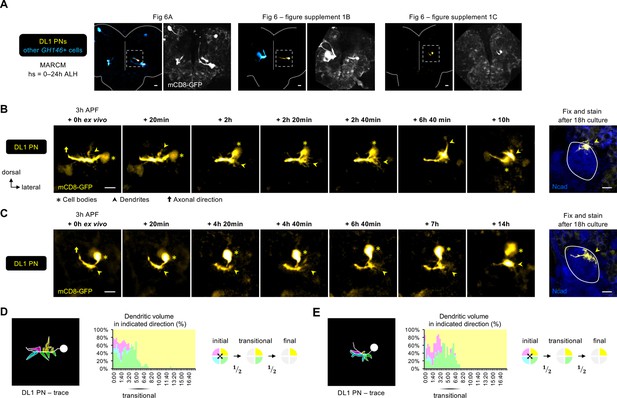
Two-photon time-lapse imaging of DL1 projection neuron (PNs).
(A) Two-photon images of MARCM-labeled DL1 PNs (pseudo-colored in yellow) and other GH146 + cells (pseudo-colored in blue) in pupal brains dissected at 3 hr APF. Zoom-in time-lapse images of the dashed boxes are shown in Figure 6A, Figure 6—figure supplement 1B, C. Background signals (in gray) are used to discern the orientation of DL1 PN in the brain. (B, C) Two-photon time-lapse imaging of MARCM-labeled DL1 PNs (pseudo-colored in yellow) in additional pupal brains dissected at 3 hr APF and cultured for 18 hr ex vivo. After culture, the explant was fixed and immune-stained for N-Cadherin (Ncad; in blue) to outline the developing antennal lobe. (D, E) Neurite tracing of DL1 PN (Figure 6—figure supplement 1B, C) at the beginning of live imaging (3 hr APF + 0 hr ex vivo) and quantification of the percentage of dendritic volume in indicated direction during the time-lapse imaging period. During the transitional period, dendrites are only found in the dorsolateral and ventrolateral directions. Timestamp 00:00 refers to HH:mm; H, hour; m, minute.
Two-photon time-lapse imaging of DL1 projection neuron (PN) dendrites.
See Figure 6A for details. Timestamp 00:00:00 refers to HH:mm:ss; H, hour; m, minute; s, second.
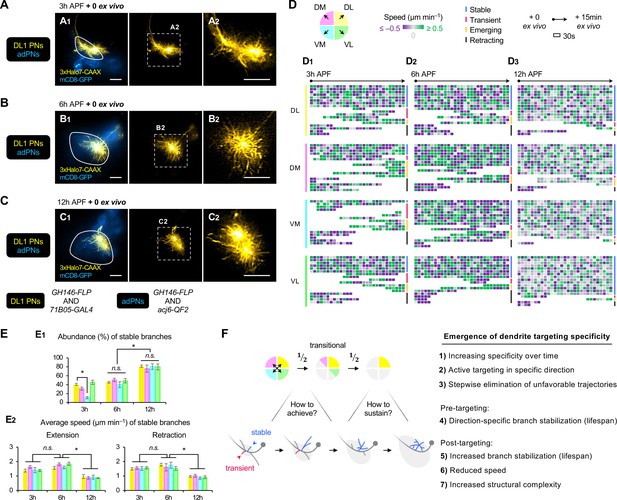
AO-LLSM time-lapse imaging reveals cellular mechanisms of dendrite targeting specificity.
(A–C) AO-LLSM imaging of DL1 projection neurons (PNs) (71B05+; labeled in yellow) and anterodorsal projection neurons (adPNs) (acj6+; labeled in blue) in cultured brains dissected at 3 hr (A), 6 hr (B), and 12 hr (C) APF. Zoom-in, single z-section images of (A1), (B1), and (C1) (outlined in dashed boxes) are shown in A2, B2 and C2, respectively. (D) Single dendritic branch dynamics of 3 hr (D1), 6 hr (D2), and 12 hr (D3) DL1 PNs shown in A–C. Terminal branches are analyzed and categorized based on the directions in which they extend. Their speeds are color-coded using purple-gray-green gradients (negative speeds, retraction; positive speeds, extension). Individual branches are also assigned into four categories: stable, transient, emerging, and retracting (color-coded on the right; see Figure 7—figure supplement 1A). Each block represents a 30s window. Each row represents individual branch dynamics across the 15 min imaging period. (E) Quantification of the abundance (in percentage) of DL1 PN stable branches in indicated direction at 3 hr, 6 hr, and 12 hr (E1). Average speed of DL1 PN stable branches in indicated direction at 3 hr, 6 hr, and 12 hr (E2). DL1 PN sample size: 3 hr=4; 6 hr=3; 12 hr=3. Error bars, SEM; t-test; One-way ANOVA; *p<0.05; n.s., p≥0.05. SEM, standard error of the mean; n.s., not significant. See Figure 7—source data 1. (F) Summary of mechanisms underlying the emergence of dendrite targeting specificity revealed by two-photon and AO-LLSM imaging of DL1 PN dendrites.
-
Figure 7—source data 1
Source data for Figure 7E.
- https://cdn.elifesciences.org/articles/85521/elife-85521-fig7-data1-v2.xlsx
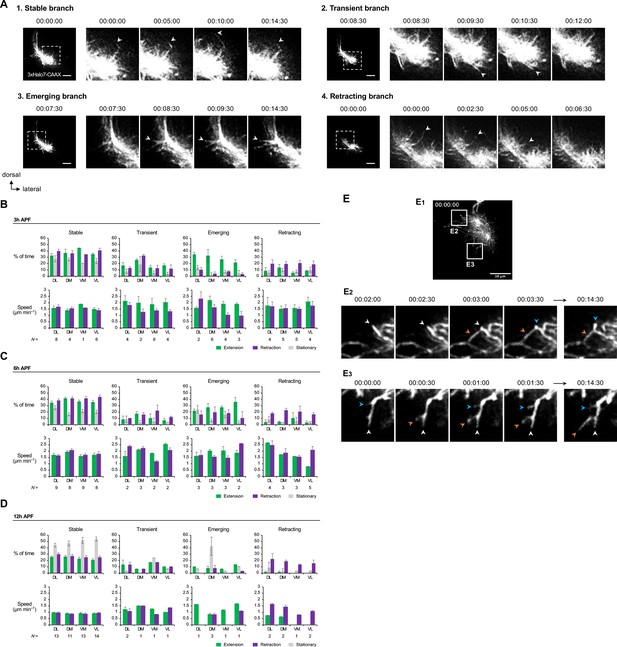
Analyses of DL1 projection neuron (PN) dendritic branches captured by AO-LLSM imaging.
(A) Categorization of branches into (1) stable, (2) transient, (3) emerging, and (4) retracting branches. Representative branches of each category are shown. Timestamp 00:00:00 refers to HH:mm:ss; H, hour; m, minute; s, second. (B–D) Top: Quantification of the percentage of time given branches spent on extending, retracting, and being stationary. Bottom: Extension/retraction speeds of DL1 PN stable, transient, emerging, and retracting branches in indicated directions at 3 hr APF (B), 6 hr APF (C), and 12 hr APF (D). Data are analyzed using DL1 PNs in Figure 7A–C. N indicates branch number. (E) Time-lapse AO-LLSM imaging of 12 hr APF DL1 PNs (E1; Figure 7C) reveals terminal branch arborization. (E2) and (E3) are selected time-lapse images of zoom-in of indicated boxes in (E1). White arrowheads point to the terminal branch of interest, and colored arrowheads point to secondary branches produced from the branch of interest. Timestamp 00:00:00 refers to HH:mm:ss; H, hour; m, minute; s, second.
AO-LLSM time-lapse imaging of 3 hr DL1 projection neuron (PN) dendrites.
See Figure 7A for details. Timestamp 00:00:00 refers to HH:mm:ss; H, hour; m, minute; s, second.
AO-LLSM time-lapse imaging of 6 hr DL1 projection neuron (PN) dendrites.
See Figure 7B for details. Timestamp 00:00:00 refers to HH:mm:ss; H, hour; m, minute; s, second.
AO-LLSM time-lapse imaging of 12 hr DL1 projection neuron (PN) dendrites.
See Figure 7C for details. Timestamp 00:00:00 refers to HH:mm:ss; H, hour; m, minute; s, second.
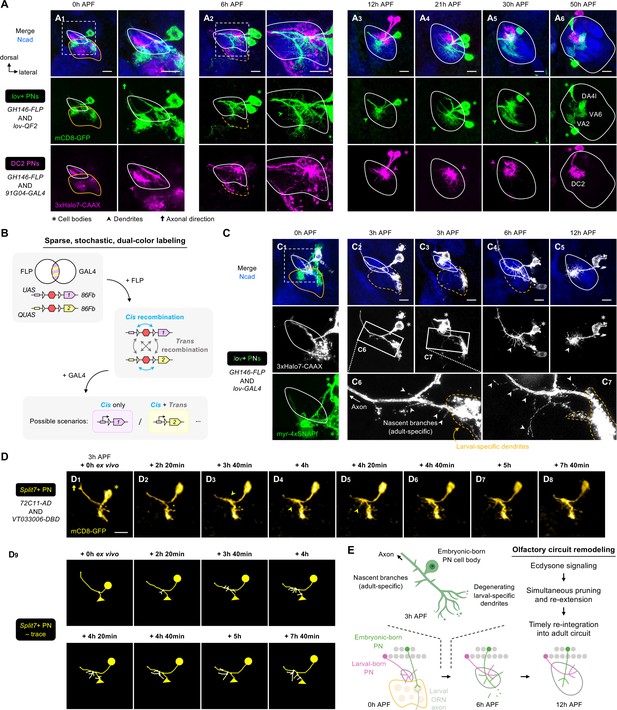
Embryonic-born projection neurons (PNs) timely participate in olfactory map formation via simultaneous pruning and re-extension.
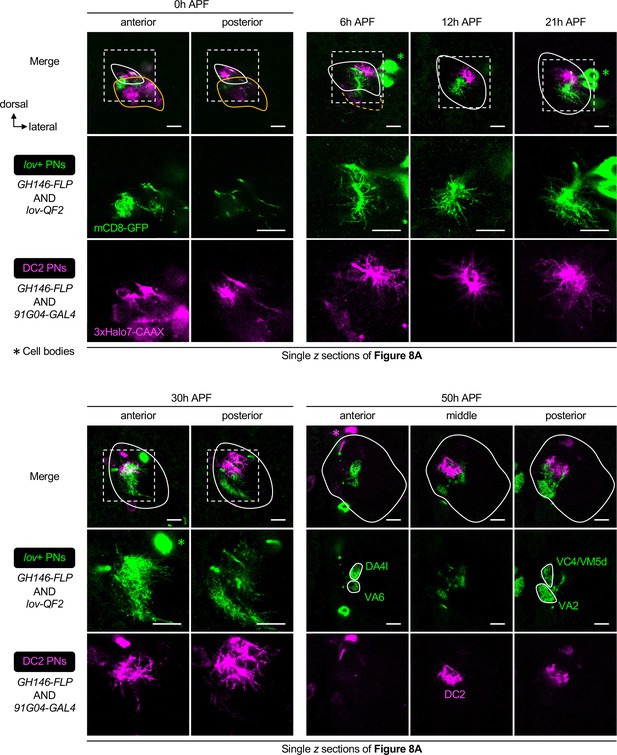
(A) Confocal images of fixed brains at indicated stages showing dendrite development of lov+ PNs (embryonic-born; labeled in green) and 91G04+DC2 PNs (larval-born; labeled in magenta). As 91G04-GAL4 also labels some embryonic-born PNs from 0 to 6 hr APF, their processes are found in the larval-specific antennal lobe (A1, 2). Right columns of A1, 2 show a zoom-in of the dashed boxes. Green arrowhead in (A2) indicates robust dendrite re-extension of embryonic-born PNs across the developing antennal lobe at 6 hr APF. (A1): N=6; (A2): N=12; (A3): N=9; (A4): N=12; (A5): N=9; (A6): N=5. (B) Schematic of the sparse, stochastic, and dual-color labeling strategy. In this strategy, the same cell has one copy of UAS-responsive conditional reporter 1 and one copy of QUAS-responsive reporter 2, both of which are integrated into the same 86Fb genomic locus (i.e. UAS-FRT-stop-FRT-reporter1/QUAS-FRT-stop-FRT-reporter2). FLP expression yields cis and trans recombination of FRT sites in a stochastic manner. Upon GAL4 expression, reporter 1 is expressed in cells with cis recombination, whereas reporter 2 is expressed only when cis and trans recombination events co-occur. (C) Sparse labeling of lov+ PNs (labeled in green; single-cell lov+ PNs in gray) at indicated developmental stages. (C6) and (C7) are zoom-in images of the rectangular boxes in (C2) and (C3), respectively. Arrowheads indicate nascent, adult-specific dendrites. Larval-specific dendrites are outlined by dashed orange lines. Arrows indicate axons projecting towards high brain centers. (C1): N=6; (C2–3): N=6; (C4): N=4; (C5): N=4. (D) Two-photon time-lapse imaging of a single embryonic-born PN (Split7+; pseudo-colored in yellow) in a brain dissected at 3 hr APF and cultured for 23 hr ex vivo. Arrowhead in (D3) denote the thickening of the main process. Arrowheads in D4, 5 denote dendritic protrusions dorsal to larval-specific dendrites. (D9) shows neurite tracing of the embryonic-born PN. Triangles in (D9) indicate the degenerating larval-specific dendrites. N=3. (E) Schematic summary of remodeling of embryonic-born PN dendrites. Following simultaneous pruning and re-extension, embryonic-born PNs timely integrate into an adult olfactory circuit and, together with larval-born PNs, participate in the prototypic map formation.
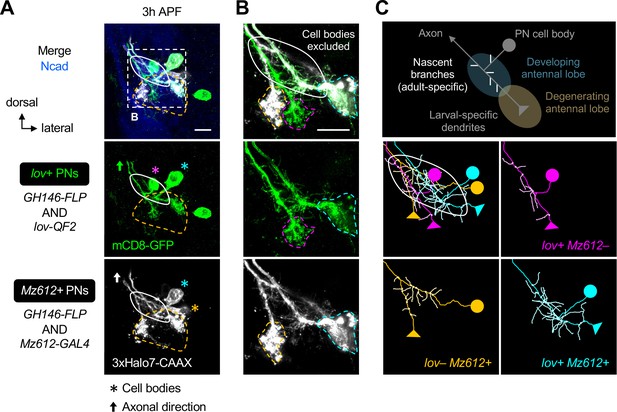
Dendrite re-extension of lov+ and Mz612+ embryonic-born projection neurons (PNs).
(A, B) Dendritic re-extension of lov+ (labeled in green) and Mz612+ (labeled in gray) PNs. (B) is the zoom-in of the dashed box in A. Magenta, cyan, and orange asterisks in A indicate cell bodies of lov+ Mz612– PN, lov+ Mz612+ PN, and lov– Mz612+ PN, respectively. Using the same color code, their larval-specific dendrites are outlined with dashed lines in B. N=4. (C) Schematic of co-existence of larval- and adult-specific dendrites of an embryonic-born PN (top row), and neurite tracing of the three embryonic-born PNs (middle and bottom rows).
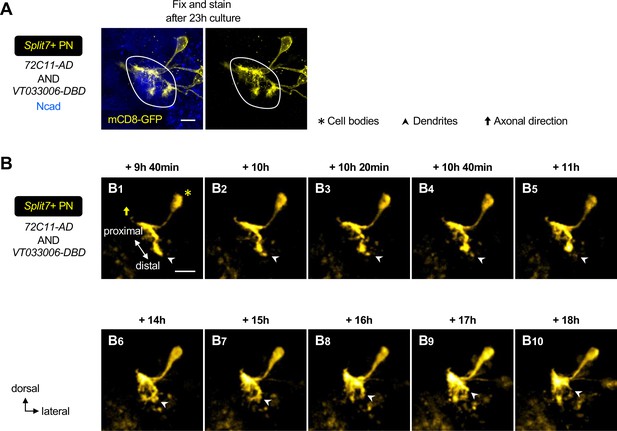
Two-photon time-lapse imaging of Split7+ projection neuron (PN) dendrites.
(A) Confocal image of Split7+ PNs (labeled in yellow; Figure 8D) after 23 hr culture. N-Cadherin (Ncad; in blue) staining outlines the developing antennal lobe. Split7-GAL4 is expressed in more than one PN type at later stages. (B) Two-photon time-lapse imaging of a Split7+ PN showing distal-to-proximal pruning of larval-specific dendrites.
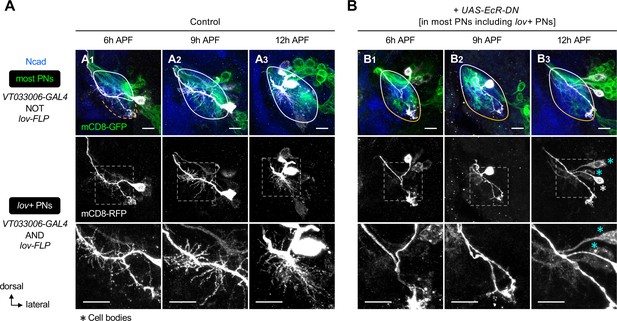
Dual requirement of ecdysone signaling in pruning and re-extension of embryonic-born projection neuron (PN) dendrites.
(A) Normal dendrite development seen in control lov+ embryonic-born PNs (VT033006+ lov+ ; labeled in gray). Other PNs (referred to as most PNs; VT033006+ lov–) are labeled in green. Bottom row shows a zoom-in of the dashed boxes. (B) Expression of a dominant negative form of ecdysone receptor (EcR-DN) in most PNs including lov+ PNs suppresses both pruning and re-extension of lov+ PN dendrites. Similar results were seen in multiple biological samples (N≥3 for each stage per genotype). Cyan asterisks in (B3) mark lov+ cells that had weak VT033006-GAL4 and thereby weak EcR-DN expression. These cells appeared to still elaborate dendrites, suggestive of a dose-dependent effect of EcR-DN. White asterisk in (B3) marks a lov+ cell with strong VT033006-GAL4 expression. The presumed fused larval- and adult-specific antennal lobes are outlined with white-orange gradient line.
3D rendering of z stacks of indicated projection neurons (PNs) in 12 hr APF antennal lobe.
This video shows a 3D rendering of z stacks with rotation along y-axis to visualize PN dendrites in the context of the antennal lobe in three dimensions. See Figure 8 for details.
Two-photon time-lapse imaging of Split7+ projection neuron (PN) dendrites.
See Figure 8D for details. Timestamp 00:00:00 refers to HH:mm:ss; H, hour; m, minute; s, second.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent (D. melanogaster) | GH146-FLP | DOI: 10.1038/nn.2442 | ||
| Genetic reagent (D. melanogaster) | QUAS-FRT-stop-FRT-mCD8-GFP | DOI: 10.1016 /j.cell.2010.02.025 | ||
| Genetic reagent (D. melanogaster) | UAS-mCD8-GFP | DOI: 10.1016 /s0896-6273(00)80701–1 | ||
| Genetic reagent (D. melanogaster) | UAS-mCD8-FRT-GFP-FRT-RFP | DOI: 10.1016 /j.neuron.2014.06.026 | ||
| Genetic reagent (D. melanogaster) | VT033006-GAL4 | DOI: 10.1101/198648 | ||
| Genetic reagent (D. melanogaster) | Mz19-GAL4 | DOI: 10.1242/dev.00896 | ||
| Genetic reagent (D. melanogaster) | 91 G04-GAL4 | DOI: 10.1016 /j.celrep.2012.09.011 | ||
| Genetic reagent (D. melanogaster) | Mz612-GAL4 | DOI: 10.1242/dev.01614 | ||
| Genetic reagent (D. melanogaster) | 71B05-GAL4 | DOI: 10.1016 /j.celrep.2012.09.011 | ||
| Genetic reagent (D. melanogaster) | Split7-GAL4 | DOI: 10.7554/eLife.63450 | FlyLight:SS01867 | |
| Genetic reagent (D. melanogaster) | QUAS-FLP | DOI: 10.1016 /j.cell.2010.02.025 | ||
| Genetic reagent (D. melanogaster) | UAS-EcR.B1-ΔC655.F645A | DOI: 10.1242/dev.00205 | ||
| Genetic reagent (D. melanogaster) | tsh-GAL4 | Bloomington Drosophila Stock Center | BDSC:3040 | |
| Genetic reagent (D. melanogaster) | lov-GAL4 | Bloomington Drosophila Stock Center | BDSC:3737 | |
| Genetic reagent (D. melanogaster) | UAS-mCD8-GFP, hs-FLP; FRTG13, tub-GAL80;; GH146-GAL4 | DOI: 10.1016 /s0896-6273(00)80701–1 | ||
| Genetic reagent (D. melanogaster) | FRTG13, UAS-mCD8-GFP | DOI: 10.1016 /s0896-6273(00)80701–1 | ||
| Genetic reagent (D. melanogaster) | UAS-FRT10-stop-FRT10-3xHalo7-CAAX | this paper | on either II or III chromosome; see Materials and methods | |
| Genetic reagent (D. melanogaster) | UAS-FRT-myr-4xSNAPf-FRT-3xHalo7-CAAX | this paper | on III chromosome; see Materials and methods | |
| Genetic reagent (D. melanogaster) | UAS-FRT-myr-mGreenLantern-FRT-3xHalo7-CAAX | this paper | on II chromosome; see Materials and methods | |
| Genetic reagent (D. melanogaster) | QUAS-FRT-stop-FRT-myr-4xSNAPf | this paper | on III chromosome; see Materials and methods | |
| Genetic reagent (D. melanogaster) | run-T2A-FLP | this paper | on X chromosome; see Materials and methods | |
| Genetic reagent (D. melanogaster) | acj6-T2A-FLP | this paper | on X chromosome; see Materials and methods | |
| Genetic reagent (D. melanogaster) | acj6-T2A-QF2 | this paper | on X chromosome; see Materials and methods | |
| Genetic reagent (D. melanogaster) | CG14322-T2A-QF2 | this paper | on III chromosome; see Materials and methods | |
| Genetic reagent (D. melanogaster) | lov-T2A-QF2 | this paper | on II chromosome; see Materials and methods | |
| Antibody | chicken polyclonal anti-GFP | Aves Lab | RRID:AB_10000240; Aves Lab:GFP-1020 | (1:1000) |
| Antibody | rabbit polyclonal anti-DsRed | TaKaRa | RRID:AB_10013483; TaKaRa:632496 | (1:500) |
| Antibody | rat monoclonal anti-Cadherin DN | Developmental Studies Hybridoma Bank | RRID:AB_528121; DSHB:DN-Ex#8 | (1:30) |
| Antibody | mouse monoclonal anti-Bruchpilot | Developmental Studies Hybridoma Bank | RRID:AB_2314866; DSHB:nc82 supernatant | (1:30) |
| Recombinant DNA reagent | pBPGUw-HACK-QF2 | Addgene | RRID:Addgene_80276 | |
| Recombinant DNA reagent | pU6-BbsI-chiRNA | Addgene | RRID:Addgene_45946 | |
| Recombinant DNA reagent | pUAS-3xHalo7-CAAX | Addgene | RRID:Addgene_87646 | |
| Recombinant DNA reagent | pUAS-myr-4xSNAPf | Addgene | RRID:Addgene_87637 | |
| Recombinant DNA reagent | pcDNA3.1-mGreenLantern | Addgene | RRID:Addgene_161912 | |
| Recombinant DNA reagent | p5XQUAS | Addgene | RRID:Addgene_24349 | |
| Recombinant DNA reagent | p10xQUAS-CsChrimson | Addgene | RRID:Addgene_163629 | |
| Recombinant DNA reagent | pUAS-FRT10-stop-FRT10-3xHalo7-CAAX | this paper | backbone from pUAS-3xHalo7-CAAX; see Materials and methods | |
| Recombinant DNA reagent | pUAS-FRT-myr-4xSNAPf-FRT-3xHalo7-CAAX | this paper | backbone from pUAS-3xHalo7-CAAX; see Materials and methods | |
| Recombinant DNA reagent | pUAS-FRT-myr-mGreenLantern-FRT-3xHalo7-CAAX | this paper | backbone from pUAS-3xHalo7-CAAX; see Materials and methods | |
| Recombinant DNA reagent | pUAS-myr-mGreenLantern | this paper | backbone from pUAS-myr-4xSNAPf; see Materials and methods | |
| Recombinant DNA reagent | pQUAS-FRT-stop-FRT-myr-4xSNAPf | this paper | backbone from p5XQUAS; see Materials and methods | |
| Chemical compound, drug | SYLGARD 184 Silicone Elastomer Kit | DOW | DOW:2646340 | |
| Chemical compound, drug | Schneider’s Drosophila Medium | ThermoFisher Scientific | ThermoFisher Scientific:21720001 | |
| Chemical compound, drug | Fetal Bovine Serum | ThermoFisher Scientific | ThermoFisher Scientific:16140071 | used at 10% |
| Chemical compound, drug | Human recombinant insulin | ThermoFisher Scientific | ThermoFisher Scientific:12585014 | used at 10 µg/mL |
| Chemical compound, drug | Penicillin-Streptomycin | ThermoFisher Scientific | ThermoFisher Scientific:15140122 | (1:100) |
| Chemical compound, drug | Ascorbic acid | Sigma | Sigma:A4544 | used at 50 mg/mL in water |
| Chemical compound, drug | 20-hydroxyecdysone | Sigma | Sigma:H5142 | used at 20 µM and 2 µM |
| Chemical compound, drug | JF503-cpSNAP | DOI: 10.1038/nmeth.4403; DOI: 10.1021/jacsau.1c00006 | (1:1000); gift from Dr. Luke Lavis | |
| Chemical compound, drug | JF646-Halo | DOI: 10.1038/nmeth.4403; DOI: 10.1021/jacsau.1c00006 | (1:1000); gift from Dr. Luke Lavis | |
| Chemical compound, drug | JFX650-SNAP | DOI: 10.1038/nmeth.4403; DOI: 10.1021/jacsau.1c00006 | (1:1000); gift from Dr. Luke Lavis | |
| Chemical compound, drug | JFX554-Halo | DOI: 10.1038/nmeth.4403; DOI: 10.1021/jacsau.1c00006 | (1:10000); gift from Dr. Luke Lavis | |
| Chemical compound, drug | JF635-Halo | DOI: 10.1038/nmeth.4403; DOI: 10.1021/jacsau.1c00006 | (1:1000); gift from Dr. Luke Lavis | |
| Chemical compound, drug | JF570-Halo | DOI: 10.1038/nmeth.4403; DOI: 10.1021/jacsau.1c00006 | (1:5000); gift from Dr. Luke Lavis | |
| Chemical compound, drug | Sulforhodamine 101 | Sigma | Sigma:S7635 | used at 1 µM |
| Software, algorithm | ZEN | Carl Zeiss | RRID:SCR_013672 | |
| Software, algorithm | ImageJ | National Institutes of Health | RRID:SCR_003070 | |
| Software, algorithm | Python Programming Language | Python | RRID:SCR_008394 | http://www.python.org/ |
Additional files
-
Supplementary file 1
Sample variability among individual brains.
A supplemental table describing the biological and technical variations we observed among individual brain samples, and measures we took to minimize them, if possible.
- https://cdn.elifesciences.org/articles/85521/elife-85521-supp1-v2.docx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/85521/elife-85521-mdarchecklist1-v2.pdf