The MODY-associated KCNK16 L114P mutation increases islet glucagon secretion and limits insulin secretion resulting in transient neonatal diabetes and glucose dyshomeostasis in adults
Figures

Kcnk16L114P neonates exhibit loss of glucose-stimulated Ca2+ entry and insulin secretion leading to transient neonatal hyperglycemia and death.
(A) Schematic of the Kcnk16 L114P mouse line generation in the C57BL/6J background and the C57BL/6J:CD-1 (ICR) mixed background. (B) χ2 analysis of the F1 progeny from C57BL/6J and heterozygous Kcnk16 L114P (L/P) crossings genotyped at weaning on postnatal day 21 (P21). (C) Blood glucose measurements of male wildtype (WT; black; N=8), heterozygous Kcnk16 L114P (L/P; green; N=16), and homozygous Kcnk16 L114P (P/P; blue; N=9) mice on P4. (D) Plasma insulin measurements performed on P4 in WT (N=4), Kcnk16 L114P (L/P; N=4), and Kcnk16 L114P (P/P; N=5) neonates. (E, F) In vitro glucose-stimulated insulin secretion from P4 mouse islets stimulated with 2.8 mM glucose (G) or 20 mM G, respectively (WT; N=4, Kcnk16 L114P (L/P); N=8, and Kcnk16 L114P (P/P); N=6). (G) Pancreas weight/ body weight measurements of P4 male mice (WT; N=6, Kcnk16 L114P (L/P); N=14, and Kcnk16 L114P (P/P); N=9). (H) Representative immunostaining images of pancreas sections from P0 WT and Kcnk16 L114P (L/P) mice (N=3 mice/genotype), stained against insulin (INS; green), glucagon (GCG; red), somatostatin (SST; magenta), and Hoechst (blue); scale bar = 100 μm. (I, J) Average islet area and area fraction of hormone staining per islet quantified using Fiji ImageJ software in P0 mouse pancreas sections. (K) Representative glucose-stimulated Ca2+ influx traces from P4 mouse islets sequentially stimulated with 2 mM glucose (G), 7 mM G, 20 mM G, and 20 mM G with 30 mM KCl (WT; N=6, Kcnk16 L114P (L/P); N=9, and Kcnk16 L114P (P/P); N=7). (L–N) Average area under the curve (AUC) analysis of normalized Ca2+ during 7 mM G, 20 mM G, and 20 mM G+30 mM KCl stimulations. (O) Survival curve for Kcnk16 L114P (P/P) mice treated with (N=6) or without (N=6) insulin (Lantus insulin glargine; 0.2 U/kg/day) starting at P0 up until death or weaning. Data are presented as mean ± SEM. Data were analyzed using Student’s t-test, one-way ANOVA, and two-way ANOVA as appropriate. *p<0.05, **p<0.01, ***p<0.001, and ****p<0.0001.
Generation of Kcnk16 L114P model and assessment of neonatal glucose homeostasis and lethality.
(A) Targeted region of Kcnk16 exon 3 using CRISPR/Cas9 leading to the introduction of HinfI restriction enzyme site (red arrows) and CTG to CCA mutation in codon 337 corresponding to p.TALK-1 L114P (green arrows). (B) PCR confirmation of the male founder Kcnk16 L114P (L/P) mouse using HinfI restriction digestion. (C) χ2 analysis of the F1 progeny from B6; CD-1 Kcnk16 L114P (L/P) crosses. (D) Body weight measurements of male (left) and female (right) wildtype (WT; black), heterozygous Kcnk16 L114P (L/P; green), and homozygous Kcnk16 L114P (P/P; blue) mice on P4. (E) Blood glucose measurements of female mice on P4. (F) Pancreas weight/ body weight measurements of P4 female mice. Data are presented as mean ± SEM. Data were analyzed using Student’s t-test or one-way ANOVA. *p<0.05, and ****p<0.0001.
-
Figure 1—figure supplement 1—source data 1
Original file for the DNA gel analysis in Figure 1—figure supplement 1B (PCR confirmation of the male founder Kcnk16 L114P (L/P) mouse using HinfI restriction digestion).
- https://cdn.elifesciences.org/articles/89967/elife-89967-fig1-figsupp1-data1-v1.tif
-
Figure 1—figure supplement 1—source data 2
Original file for the DNA gel analysis in Figure 1—figure supplement 1B (PCR confirmation of the male founder Kcnk16 L114P (L/P) mouse using HinfI restriction digestion) with bands and DNA ladder labeled.
- https://cdn.elifesciences.org/articles/89967/elife-89967-fig1-figsupp1-data2-v1.tif
Kcnk16 L114P male and female mice show transient neonatal hyperglycemia.
(A) Average blood glucose measurements of B6;CD-1 male mice at P1, P4, P10, P15, and P21 including: WT (black; P1 N=9, P4 N=27, P10 N=9, P15 N=9, P21 N=8), Kcnk16 L114P (L/P; green; P1 n=15, P4 N=28, P10 N=24, P15 N=14, P21 N=14), and Kcnk16 L114P (P/P; green; P1 n=5, P4 N=10, no P/P male mice survived past P4). (B) Average blood glucose measurements of B6;CD-1 female mice at P1, P4, P10, P15, and P21 including: WT (black; P1 N=8, P4 N=13, P10 N=8, P15 N=8, P21 N=8), Kcnk16 L114P (L/P; green; P1 n=24, P4 N=33, P10 N=24, P15 N=24, P21 N=24), and Kcnk16 L114P (P/P; green; P1 n=10, P4 N=16, P10 N=5, P15 N=4, P21 N=4). Data are presented as mean ± SEM. Data were analyzed using Student’s t-test; *p<0.05, **p<0.01, ***p<0.001, and ****p<0.0001.
Adult Kcnk16 L114P mice exhibit fasting hyperglycemia and glucose intolerance.
(A, B) Intraperitoneal glucose tolerance test (i.p. GTT) performed in 15-week-old and 18-week-old male mice following a 4 hr fast in response to 2 mg/g glucose injection (WT; black; N=8–10 and Kcnk16 L114P (L/P); green; N=9–10). (C) Average area under the curve (AUC) of the 2 hr GTT excursion profiles from ages 7 weeks up to 18 weeks in male mice. (D) Weekly body weight measurements of male WT; N=5 mice and Kcnk16 L114P (L/P); N=5 mice. (E, F) I.p. GTT performed in 15-week-old and 18-week-old female mice following a 4 hr fast in response to 2 mg/g glucose injection (WT; N=9–11 and Kcnk16 L114P (L/P); N=10–11). (G) Average AUC of the 2 hr GTT excursion profiles from ages 7 weeks up to 18 weeks in female mice. (H) Body weight measurements of female WT (black; N=4) and Kcnk16 L114P (L/P; blue; N=5) mice. Data are presented as mean ± SEM. Data were analyzed using Student’s t-test. *p<0.05, **p<0.01, and ***p<0.001.
Oral glucose tolerance is impaired in Kcnk16 L114P (L/P) mice in the B6;CD-1 background.
Oral glucose tolerance test (OGTT) performed in 4- (A) and 14-week-old (B) male mice following a 4 hr fast in response to oral gavage of 2 g/kg glucose. (C) Average area under the curve (AUC) of the 2 hr OGTT excursion profiles for male mice at 4, 5, 6, 7, and 14 weeks of age. OGTT performed in 4- (D) and 14-week-old (E) female mice following a 4 hr fast in response to oral gavage of 2 g/kg glucose. (F) Average AUC of the 2 hr OGTT excursion profiles for female mice at 4, 5, 6, 7, and 14 weeks of age. Data are presented as mean ± SEM; N=5 mice/cohort. Data were analyzed using Student’s t-test; *p<0.05.
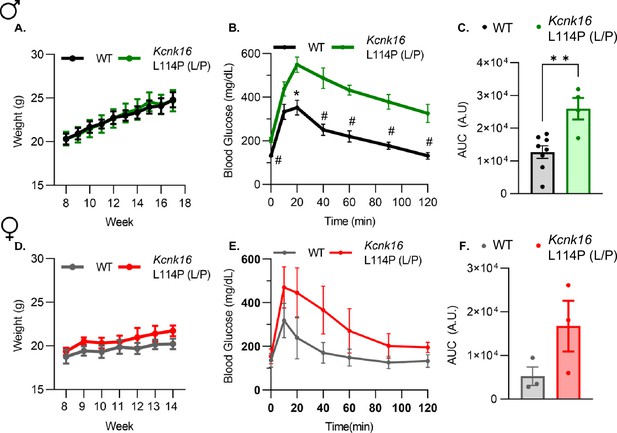
Glucose homeostasis is impaired in Kcnk16 L114P (L/P) mice in the B6 background.
(A) Body weight measurements of male WT (black; N=8) and Kcnk16 L114P (L/P; green; N=4) mice in the C57Bl/6J background. (B) Intraperitoneal glucose tolerance test (i.p. GTT) performed in 10-week-old male mice following a 4 hr fast in response to 2 mg/g glucose injection. (C) Average AUC of the 2 hr GTT excursion profiles. (D) Body weight measurements of female WT (gray; N=3) and Kcnk16 L114P (L/P; red; N=3) mice. (E) I.p. GTT performed in 11-week-old female mice following a 4 hr fast in response to 2 mg/g glucose injection. (F) Average AUC of the 2 hr GTT excursion profiles in (E). Data are presented as mean ± SEM. Data were analyzed using Student’s t-test; *p<0.05, **p<0.01, and #p<0.001.
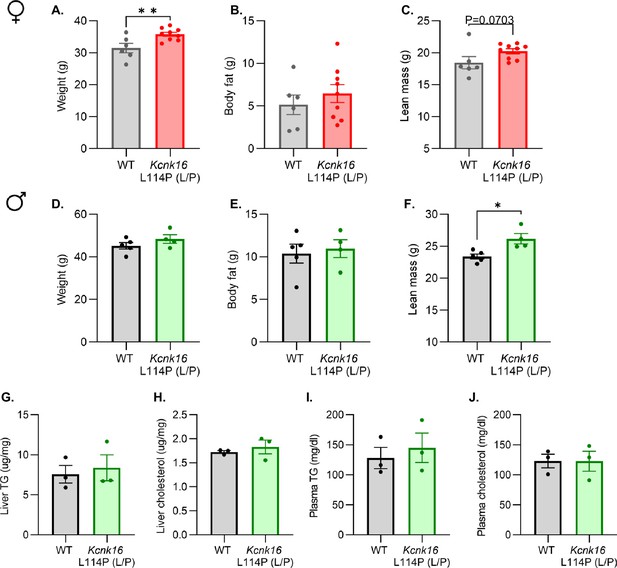
Body composition measurements and assessment of plasma and liver triglycerides and total cholesterol.
(A–F) Body composition analysis of male and female B6;CD-1 WT and Kcnk16 L114P (L/P) mice assessing weight (g), body fat (g), and lean mass (g) (N=5–9 mice/genotype). (G–J) Average liver and plasma cholesterol and triglyceride levels in male B6;CD-1 WT and Kcnk16 L114P (L/P) mice (N=3/genotype). Data are presented as mean ± SEM. Data were analyzed using Student’s t-test. *p<0.05, and **p<0.01.
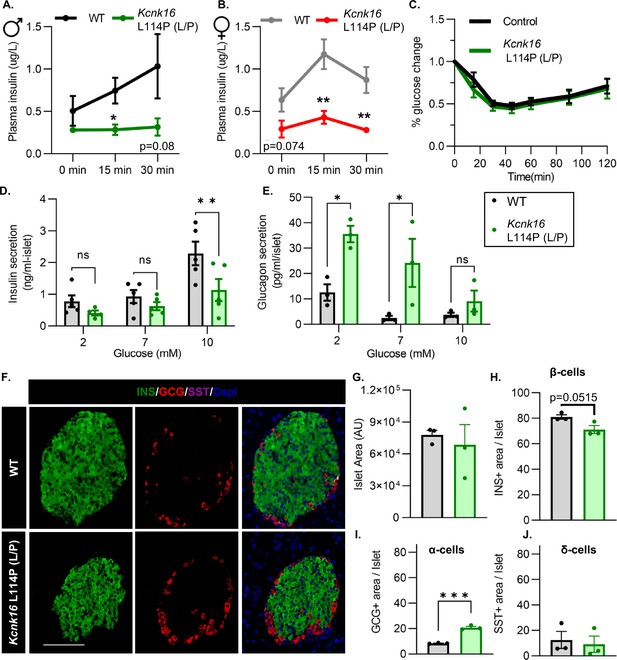
Adult Kcnk16 L114P mice show disrupted islet hormone secretion and islet composition.
(A, B) Plasma insulin levels in male (A) and female (B) WT and Kcnk16 L114P (L/P) mice following a 4 hr fast at the indicated time points before and after a 2 mg/g glucose injection. (C) Glucose (%) change in response to an intraperitoneal (i.p.) human insulin injection (0.75 UI/kg body weight) was measured as an indicator of insulin sensitivity in WT and Kcnk16 L114P male mice after a 4 hr fast. (D, E) In vitro insulin secretion (N=5 mice/genotype) and glucagon secretion (N=3 mice/genotype) from male mice at the specified glucose concentrations. (F) Representative immunostaining images of pancreas sections from WT and Kcnk16 L114P (L/P) male mice (N=3/genotype) stained against insulin (INS; green), glucagon (GCG; red), somatostatin (SST; magenta), and Hoechst (blue); scale bar = 100 μm. (G–J) Average islet area and area of hormone staining/islet for β-cells (INS), α-cells (GCG), and δ-cells (SST). Data are presented as mean ± SEM. Data were analyzed using Student’s t-test or two-way ANOVA as appropriate.*p<0.05, **p<0.01, and ***p<0.001.
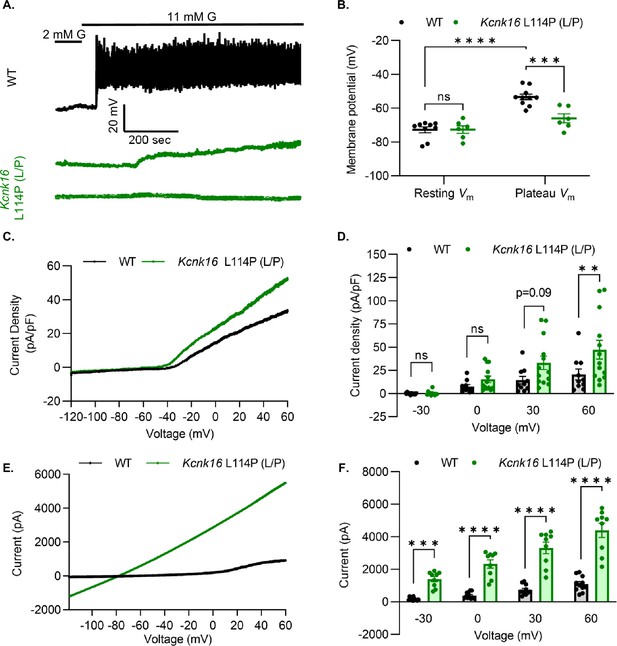
Kcnk16 L114P blunts β-cell glucose-stimulated electrical excitability and increases whole-cell two-pore domain K+ channel currents.
(A) Representative perforated patch-clamp Vm recordings in response to 2 mM G and 11 mM G in islets from WT and Kcnk16 L114P (L/P) mice (N=6–10 islets from 5 mice/genotype). (B) Average resting islet Vm under 2 mM G and plateau islet Vm at 11 mM G. (C) Representative whole-cell two-pore domain K+ channel current density (pA/pF) recorded using a voltage ramp (–120 mV to +60 mV) at 11 mM G in β-cells from WT and Kcnk16 L114P (L/P) mice. (D) Average current density (pA/pF) at the specified membrane voltages during the voltage ramp recordings shown in panel C (N=9–13 cells/genotype). (E) Representative whole-cell two-pore domain K+ channel current (pA) recorded using a voltage ramp (–120 mV to +60 mV) in 11 mM G in HEK293T cells expressing either Kcnk16 WT or Kcnk16 L114P (L/P). (F) Average current (pA) at the specified membrane voltages during the voltage ramp recordings shown in panel E (N=9–11 cells/ group). Data are presented as mean ± SEM. Data were analyzed using two-way ANOVA. *p<0.05, **p<0.01, ***p<0.001, and ****p<0.0001.
Kcnk16 L114P attenuates β-cell glucose-stimulated electrical excitability in a heterogenous manner.
(A) Representative perforated patch-clamp Vm recording in response to 11 mM G and 11 mM G+30 mM K+ from a WT β-cell expressing GCaMP6s driven by an optimized RIP within an islet cluster (representative of N=11 islets from 5 mice). (B) Average resting Vm of GCaMP6s-positive WT and Kcnk16 L114P (L/P) β-cells within islet clusters in the presence of 2 mM G as well as plateau Vm in the presence of 11 mM G and 11 mM G+30 mM K+. (C) Percentage of GCaMP6s-positive WT and Kcnk16 L114P (L/P) β-cells displaying action potentials (APs) in response to 11 mM G and 11 mM G+30 mM K+. (D–F) Representative perforated patch-clamp Vm recordings in response to 11 mM G and 30 mM K+ illustrating cell-to-cell heterogeneity from Kcnk16 L114P (L/P) β-cells expressing GCaMP6s driven by an optimized RIP (representative of N=12 islets from 5 mice). Data are presented as mean ± SEM. Data were analyzed using two-way ANOVA; *p<0.05, **p<0.01, ***p<0.001, and ****p<0.0001.
Two-pore domain K+ channel currents in β-cells from homozygous Kcnk16 L114P (P/P) mice also exhibit a modest increase.
(A) Representative whole-cell two-pore domain K+ channel current density (pA/pF) recorded using a voltage ramp (–120 mV to +60 mV) in 11 mM G in islets from B6; CD-1 WT and Kcnk16 L114P (P/P) P4 neonates. (B) Average current density (pA/pF) measured at the specified membrane potentials (N=5 cells/genotype). Data are presented as mean ± SEM. Data were analyzed using two-way ANOVA. *p<0.05, and **p<0.01.

Kcnk16 L114P reduces glucose- and tolbutamide-stimulated islet Ca2+ entry and augments IP3-induced islet [Ca2+]ER release.
(A, B) Representative [Ca2+]c traces in islets from male (A) and female (B) WT and Kcnk16 L114P (L/P) mice in response to 2 mM G, 10 mM G, and 20 mM G. (C–F) Average normalized Ca2+ peak (C and E) and total area under the curve (AUC) (D and F) in response to the indicated glucose concentrations in islets from male and female WT and Kcnk16 L114P (L/P) mice (N=3–4 mice/genotype). (G) Representative [Ca2+]c traces in islets from WT and Kcnk16 L114P (L/P) male mice in response to 100 μM tolbutamide followed by 30 mM KCl stimulation. (H) Average AUC of normalized [Ca2+]c during 100 μM tolbutamide or 100 μM tolbutamide with 30 mM KCl stimulation (N=3 mice/genotype). (I) Representative [Ca2+]c traces in response to 100 μM acetylcholine in the absence of extracellular Ca2+ in islets from male WT and Kcnk16 L114P (L/P) mice. (J) Average AUC of normalized [Ca2+]c following 100 μM acetylcholine-stimulated [Ca2+]ER release (N=876 cells; WT, N=513 cells; Kcnk16 L114P (L/P)). Data are presented as mean ± SEM. Data were analyzed using Student’s t-test. *p<0.05, **p<0.01, ***p<0.001, and ****p<0.0001.
Islets from Kcnk16 L114P (L/P) mice on the B6 background also exhibit blunted glucose-stimulated Ca2+ entry.
(A) Representative glucose-stimulated [Ca2+]c influx traces in islets from male WT and Kcnk16 L114P (L/P) mice in the C57BL/6J background in response to 2 mM G, 10 mM G, and 20 mM G (N=3 mice/genotype). (B) Average total area under the curve (AUC) in response to the indicated glucose concentrations in islets from male WT and Kcnk16 L114P (L/P) mice. Data are presented as mean ± SEM. Data are analyzed using Student’s t-test. *p<0.05.
Kcnk16 L114P (L/P) islets exhibit prolonged glucose-stimulated phase 0 [Ca2+]ER uptake and show a complete absence of Ca2+ oscillations.
(A) Average relative [Ca2+]c at 2 mM G in islets from B6; CD-1 WT and Kcnk16 L114P (L/P) mice (N=4 mice/genotype). (B) Percent islets that exhibit the corresponding phase 0 response (drop in [Ca2+]c) length (s) in WT and Kcnk16 L114P (L/P) mice. (C) Representative glucose-stimulated [Ca2+]c oscillations recorded at 9 mM G in islets from WT and Kcnk16 L114P (L/P) mice. (D) Total number of islets analyzed for [Ca2+]c oscillations vs. the number of islets that exhibited [Ca2+]c oscillations from WT and Kcnk16 L114P (L/P) mice (N=3 mice/genotype). Data are presented as mean ± SEM. Data are analyzed using Student’s t-test. ns=non-significant.

Kcnk16 L114P islets exhibit altered expression of genes involved in β-cell identity and function, ion channel activity, hormone activity, inflammatory signaling, and extracellular matrix interaction pathways.
(A) Heatmap of a selected gene subsets showing differential gene expression in WT and Kcnk16 L114P islets. Normalized expression levels were scaled and centered by rows. (B) Volcano plot displays genes differentially expressed between WT and Kcnk16 L114P samples. Differentially expressed genes are defined by FDR <0.05 and log2FC (≥1). (C) Dotplot represents the top 10 most significantly (FDR <0.05) altered Gene Ontology (Molecular Function). GeneRatio represent (count of enriched genes)/(count of genes in the GO term). The color represents FDR-adjusted p-values and the size of the dot represents the number of genes that are significant from the experimental dataset. (D) qRT-PCR validation of the gene expression differences in WT and Kcnk16 L114P samples for the selected genes observed through bulk RNA sequencing. *p<0.05, and **p<0.01.
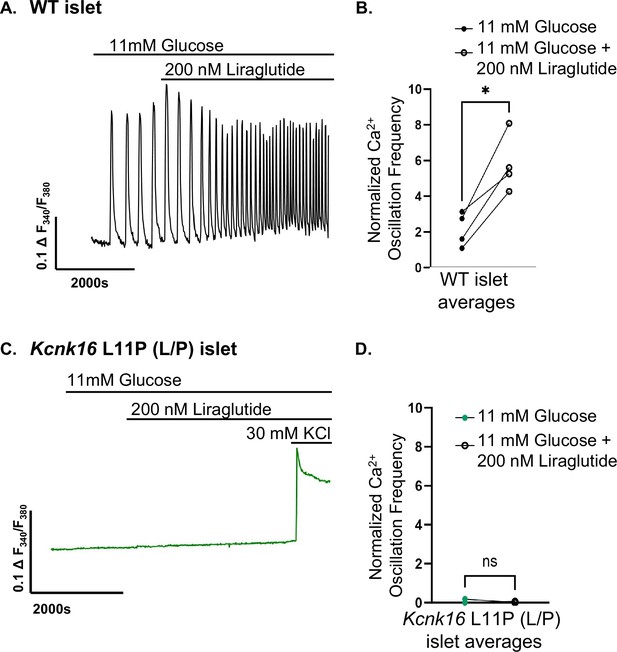
Liraglutide increased Ca2+ oscillation frequency in WT islets but does not impact Kcnk16 L114P (L/P) mouse islet Ca2+ handling.
(A) Representative islet Ca2+ recording from a WT islet in 11 mM glucose treated with 200 nM liraglutide. (B) Average Ca2+ oscillation frequency of WT islets in 11 mM glucose and in 11 mM glucose with 200 nM liraglutide (LIRA, N=57 islets from 4 mice). (C) Representative islet Ca2+ recording from a Kcnk16 L114P mice islet in 11 mM glucose treated first with 200 nM liraglutide and then with 30 mM KCl. (D) Average Ca2+ oscillation frequency of Kcnk16 L114P islets in 11 mM glucose and in 11 mM glucose with 200 nM liraglutide (LIRA, N=80 islets from 3 mice). Data are presented as mean ± SEM. Data were analyzed using paired Student’s t-test. *p<0.05.
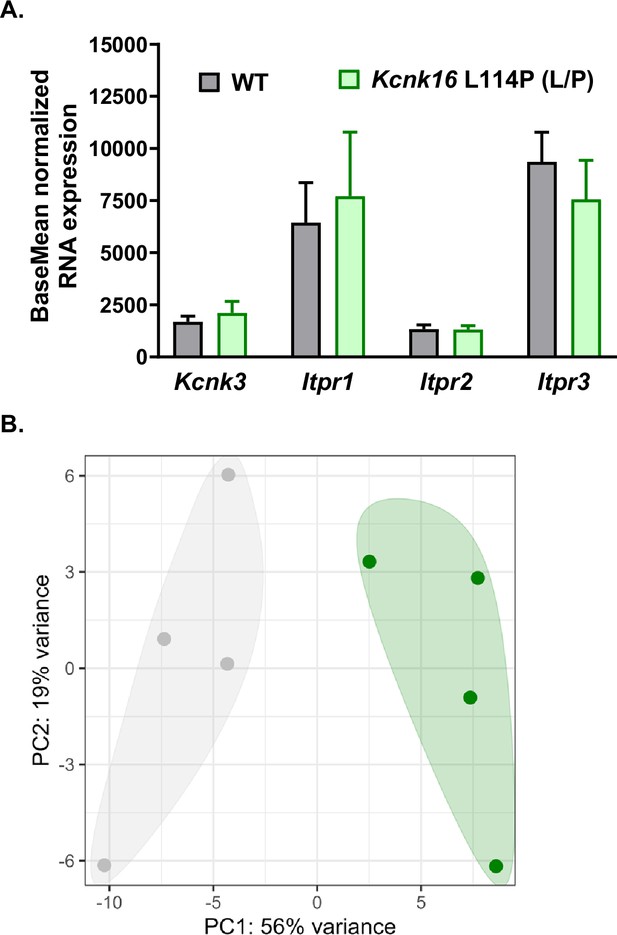
RNA sequencing showed equivalent expression of a subset of genes encoding proteins known to control [Ca2+]ER in WT and Kcnk16 L114P (L/P) male islets.
(A) Average normalized RNA levels (baseMean) of Kcnk3, Itpr1, Itpr2, and Itpr3 from WT islets (gray bars, N=4) and Kcnk16 L114P (L/P) (green bars, N=4). Data are presented as mean ± SEM. Data were analyzed with paired Student’s t-test.(B) Principal component analysis (PCA) showing clustering of WT (gray) and Kcnk16 L114P (L/P; green) islet RNA samples.
Additional files
-
Supplementary file 1
All mouse primer sequences utilized for qRT-PCR.
- https://cdn.elifesciences.org/articles/89967/elife-89967-supp1-v1.docx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/89967/elife-89967-mdarchecklist1-v1.docx





