Major patterns in the introgression history of Heliconius butterflies
eLife assessment
This important study revises the evolutionary history of Heliconius butterflies, a well-established model system for understanding speciation in the presence of gene flow between species. Using a convincing statistical phylogenetic approach that relies on the multispecies coalescent, the authors reconstruct the evolution of the lineage, including the timing of speciation events and the history of gene flow. The new phylogeny will be of interest to all researchers working on Heliconius butterflies, and the phylogenetic approach to investigators aiming to understand the history of lineages that have experienced extensive gene flow.
https://doi.org/10.7554/eLife.90656.3.sa0Important: Findings that have theoretical or practical implications beyond a single subfield
- Landmark
- Fundamental
- Important
- Valuable
- Useful
Convincing: Appropriate and validated methodology in line with current state-of-the-art
- Exceptional
- Compelling
- Convincing
- Solid
- Incomplete
- Inadequate
During the peer-review process the editor and reviewers write an eLife Assessment that summarises the significance of the findings reported in the article (on a scale ranging from landmark to useful) and the strength of the evidence (on a scale ranging from exceptional to inadequate). Learn more about eLife Assessments
Abstract
Gene flow between species, although usually deleterious, is an important evolutionary process that can facilitate adaptation and lead to species diversification. It also makes estimation of species relationships difficult. Here, we use the full-likelihood multispecies coalescent (MSC) approach to estimate species phylogeny and major introgression events in Heliconius butterflies from whole-genome sequence data. We obtain a robust estimate of species branching order among major clades in the genus, including the ‘melpomene-silvaniform’ group, which shows extensive historical and ongoing gene flow. We obtain chromosome-level estimates of key parameters in the species phylogeny, including species divergence times, present-day and ancestral population sizes, as well as the direction, timing, and intensity of gene flow. Our analysis leads to a phylogeny with introgression events that differ from those obtained in previous studies. We find that Heliconius aoede most likely represents the earliest-branching lineage of the genus and that ‘silvaniform’ species are paraphyletic within the melpomene-silvaniform group. Our phylogeny provides new, parsimonious histories for the origins of key traits in Heliconius, including pollen feeding and an inversion involved in wing pattern mimicry. Our results demonstrate the power and feasibility of the full-likelihood MSC approach for estimating species phylogeny and key population parameters despite extensive gene flow. The methods used here should be useful for analysis of other difficult species groups with high rates of introgression.
Introduction
Introgression among species has been reported in a wide range of organisms, including humans and Neanderthals (Kuhlwilm et al., 2016), Darwin’s finches (Lamichhaney et al., 2018), felids (Li et al., 2016; Li et al., 2019), canids (Gopalakrishnan et al., 2018), horses (Jónsson et al., 2014; Gaunitz et al., 2018), living and extinct elephants (Palkopoulou et al., 2018), cichlid fishes (Malinsky et al., 2018), malaria mosquitoes (Fontaine et al., 2015; Small et al., 2020), Drosophila fruit flies (Suvorov et al., 2022), sunflowers (Rieseberg et al., 2003; Whitney et al., 2010), maize (Calfee et al., 2021), and yeast (Leducq et al., 2016). Introgression is now widely recognized as an important process that can facilitate adaptation and speciation. While previous studies have confirmed the prevalence of gene flow, they give only a fragmented understanding of introgression because they mostly rely on approximate methods based on simple data summaries such as genome-wide site pattern counts or estimated gene trees. Those methods have limited power and cannot infer certain modes of gene flow or estimate most population parameters characterizing the process of species divergence and gene flow.
Recent advances make it possible to model genomic evolution under the multispecies coalescent (MSC) framework and use full-likelihood methods to estimate the species tree and quantify introgression (Wen and Nakhleh, 2018; Zhang et al., 2018; Flouri et al., 2020). Analyses of both simulated and real data demonstrate that this full-likelihood MSC approach is efficient, accurate, and robust to moderate levels of model violation (Huang et al., 2022; Thawornwattana et al., 2023). A major advantage over approximate methods is the ability to estimate parameters of the species tree and introgression events among branches precisely, including the strength and direction of introgression as well as species divergence times, introgression times, and effective population sizes. Current approximate methods are mostly unable to estimate these parameters or infer gene flow between sister lineages (Jiao et al., 2021; Mirarab et al., 2021).
Neotropical butterflies of the genus Heliconius have become a model system for understanding introgression (Heliconius Genome Consortium, 2012; Martin et al., 2013; Kozak et al., 2021; Cicconardi et al., 2023). Previous phylogenomic studies have demonstrated introgression among closely related species, but investigations of gene flow deeper among lineages were only partially successful: the various different methods yielded different phylogenies and introgression scenarios (Kozak et al., 2021; Cicconardi et al., 2023), perhaps due to methodological artifcts.
Recently, we demonstrated deep-level introgression and hybrid speciation in the erato-sara clade of Heliconius (Edelman et al., 2019; Thawornwattana et al., 2022). Here, we focus on the more complex melpomene crown group, or ‘melpomene-silvaniform’ group, which includes the melpomene-cydno-timareta clade, and ‘silvaniform’ species that are mostly Müllerian mimics of Ithomiini models, together with the related Heliconius besckei and H. elevatus (Brown, 1981). The melpomene-silvaniform species frequently hybridize today, and laboratory crosses demonstrate some interfertility across the entire group (Mallet et al., 2007). Thus, extensive gene flow is likely, making estimation of the true species phylogeny difficult. We also examine the overall species divergence and deeper introgression history of the entire genus. By using full-likelihood analysis of whole-genome data, we overcome limitations of approximate methods to obtain a robust estimate of the species tree, with major introgression events between branches quantified in terms of direction, strength, and timing. We use subsets of species to represent each major clade and answer specific questions of introgression, which helps to keep the computation manageable. Our species phylogeny and introgression history provide a more parsimonious explanation for evolution of key traits in Heliconius than those previously inferred using approximate methods (Jay et al., 2018; Kozak et al., 2021; Cicconardi et al., 2023). We find evidence of extensive autosomal gene flow across the melpomene-silvaniform group and show how trees based on the Z chromosome most likely represent the true species phylogeny. The so-called silvaniform species appear to be paraphyletic, contrary to previous findings based on approximate methods (Zhang et al., 2016; Kozak et al., 2021). As well as improving the understanding of diversification and gene flow in the genus Heliconius, we believe our approach provides useful pointers for studying species phylogeny with complex patterns of introgression in other taxa.
Results
Ancestral gene flow at the base of Heliconius phylogeny
We first establish phylogenetic relationships among six major clades of Heliconius using blocks of 200 loci (well-spaced short segments) across the genome, with each block spanning 1–3 Mb. We select a representative species from each major clade (H. burneyi, H. doris, H. aoede, H. erato, H. sara), three species from the melpomene-silvaniform group (H. melpomene, H. besckei, H. numata), and one outgroup species (Eueides tales) (Supplementary file 1a–c). Species tree search in each block was carried out under the MSC model using the Bayesian program bpp (see ‘Methods’). Although each block was assumed to lack gene flow, different trees in each block across the genome are likely to result from introgression. Unlike a windowed concatenated tree approach, our MSC method takes into account incomplete lineage sorting (ILS) and uncertainty in gene trees. Coding and noncoding loci were analyzed separately.
Two major patterns of species relationships were found at the base of the genus Heliconius: scenario 1 (erato-early), with the erato-sara clade diverging first, is supported by ~75% of genomic blocks, while scenario 2 (aoede-early), with H. aoede diverging first, is supported by ~20% of blocks (Figure 1A; see Supplementary file 1d for genome-level summaries and Supplementary file 1e for chromosome-level summaries). Using a different reference genome or more stringent filtering yielded similar fractions of the same species trees across the genome (Figure 1—figure supplements 1 and 2, Supplementary file 1d–g). In both scenarios, there is uncertainty concerning (i) the branching order of H. doris and H. burneyi, with H. doris diverging first being the most common relationship in both scenarios, and (ii) the branching order within the melpomene-silvaniform clade, with both H. besckei + H. numata and H. melpomene + H. numata being nearly equally common across the genome (Figure 1B and C). To focus on the deep divergences, we constructed simplified summaries of inferred species trees with H. besckei, H. numata, and H. melpomene grouped together (‘BNM’ in Figure 1A; also see Figure 1—figure supplement 2 and Supplementary file 1d and e). Summaries of full species trees are provided in Figure 1—figure supplement 1 and Supplementary file 1f and g; posterior probabilities of these local blockwise trees are generally low, with the median probability for each maximum a posteriori (MAP) tree <0.7, reflecting both limited information from only 200 loci and the challenge of resolving short branches in the species trees.
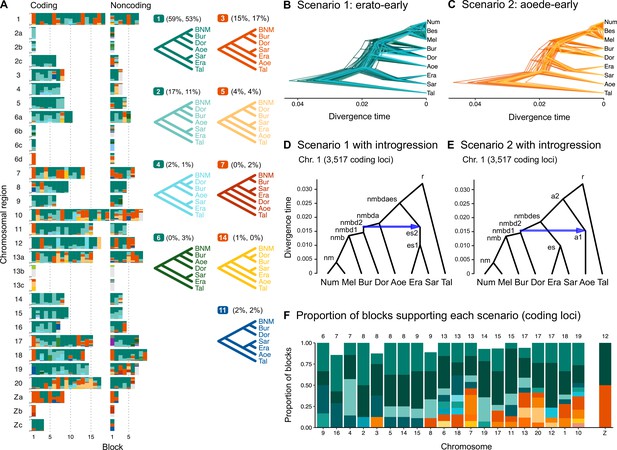
Ancestral gene flow at the base of Heliconius.
(A) Posterior probabilities of species trees for major Heliconius clades inferred from bpp analysis of 200-locus blocks (each spanning 1–3 Mb) across the genome under the multispecies coalescent (MSC) model with no gene flow. The y-axis shows the posterior probability of species trees and ranges from 0 to 1. Colors correspond to the nine most common maximum a posteriori (MAP) trees, summarized by lumping species in the melpomene-silvaniform clade (Bes, Num, Mel) into a single tip (BNM); see Figure 1—figure supplement 1 for full trees. Proportions of coding and noncoding blocks with each tree as a MAP tree are shown in parentheses. (B, C) Two scenarios of early divergence of Heliconius: (B) erato-early versus (C) aoede-early. Each tree is a MAP tree from a block having the MAP tree supporting one of these scenarios, with estimates of branch lengths (posterior means). (D, E) Phylogenetic and introgression histories estimated under an MSC model with introgression (MSC-I) corresponding to the two scenarios based on coding loci in chromosome 1 (3517 coding loci; see Figure 1—figure supplements 5 and 6). (F) Proportions of trees of scenarios 1 or 2 in each chromosome in order of increasing number of loci (used as a proxy for chromosome length). The Z chromosome (chr. 21) is placed at the right end. Number of blocks is shown on top of each bar. Tal: Eueides tales; Mel: H. melpomene; Bes: H. besckei; Num: H. numata; Bur: H. burneyi; Dor: H. doris; Aoe: H. aoede; Era: H. erato; Sar: H. sara.
Scenarios 1 and 2 are related via ancestral gene flow. If scenario 1 (Figure 1B) represents the true species tree, gene flow between the erato-sara clade and the common ancestor of H. doris, H. burneyi, and the melpomene-silvaniform clade would lead to trees of scenario 2, with reduced estimated divergence time of the erato-sara clade. Similarly, if scenario 2 (Figure 1C) is the true tree, gene flow between the aoede clade and the common ancestor of H. doris, H. burneyi, and the melpomene-silvaniform clade would lead to trees of scenario 1, with reduced estimated divergence time of H. aoede. This expected reduction in divergence time as a result of introgression is not apparent in our species tree estimates, partly due to a short internal branch separating H. aoede and the erato-sara clade in both scenarios (Figure 1B and C). To assess which scenario fits the data better, we calculated the Bayes factor under the MSC model with introgression (MSC-I) implemented in bpp, with H. besckei excluded to simplify the model (Figure 1D and E; 'etales-8spp' dataset in Supplementary file 1a). The Bayes factor provides mixed evidence, with different chromosomes either strongly supporting alternative scenarios or not significant at 1% level; however, the Z chromosome supports scenario 2 (Supplementary file 1h). In scenario 2, divergence of H. aoede was estimated to be older than that of the erato-sara clade in scenario 1 while root age estimates were comparable, further supporting the hypothesis that H. aoede most likely diverged before the erato-sara clade (Figure 1—figure supplements 3 and 4, Supplementary file 1i and j). This is because younger divergence may be explained by introgression whereas older divergence more likely represents true time of divergence. In scenario 2, introgression from the doris-burneyi-melpomene clade was estimated as unidirectional into H. aoede with a high probability (~75%), occurring shortly before the divergence of H. doris (Figure 1—figure supplements 3 and 6, Supplementary file 1j). Surprisingly, under scenario 1 we also find strong unidirectional introgression into the erato-sara clade with a high introgression probability (~65%) (Figure 1—figure supplements 3 and 5, Supplementary file 1i).
Two additional pieces of evidence support scenario 2. First, species trees of scenario 2 are most common in the Z chromosome (chr. 21) and tend to be more common in longer chromosomes, which have lower recombination rates per base pair (Figure 1F). Conversely, species trees of scenario 1 are more common in shorter chromosomes, which have higher recombination rates. If we assume that regions of low recombination tend to exhibit less introgression, with the Z chromosome usually most resistant to gene flow in Heliconius (Appendix 1—figure 1, Figure 1—figure supplement 7, Supplementary file 1k), as suggested by previous studies (Edelman et al., 2019; Martin et al., 2019), the association of species trees of scenario 2 with regions of low recombination suggests that scenario 2 more likely represents the true species relationships, whereas scenario 1 is a result of introgression. Second, we obtain a star tree whenever H. erato is assumed to diverge before H. aoede (scenario 1) under the MSC-with-migration (MSC-M or isolation-with-migration; IM) model of three species that allows for gene flow between two ingroup species since their divergence but does not allow gene flow with the outgroup species (Appendix 1—figure 2). Star trees are commonly observed when the assumed bifurcating tree is incorrect (Dalquen et al., 2017). By contrast, assuming H. aoede diverges before H. erato (scenario 2) always leads to a bifurcating tree.
Four small inversion regions (~100–400 kb) had been identified previously to be differentially fixed between the melpomene group and the erato-sara clade (Seixas et al., 2021): 2b, 6b, 6c, 13b, and 21b. We were able to extract only a small number of loci (<100; see Supplementary file 1c) from each region. While species tree estimates are more uncertain (Figure 1—figure supplements 1 and 2, Supplementary file 1d–g), the 13b region (~360 kb with respect to H. erato demophoon reference) consistently shows a unique pattern in which H. doris and H. burneyi cluster with the erato-sara clade instead of the melpomene-silvaniform clade. This suggests ancient introgression of the inversion from the erato-sara clade into H. doris and H. burneyi (Seixas et al., 2021).
In conclusion, we detect some hitherto unrecognized introgression among the deepest branches within Heliconius sensu lato. The aoede-early scenario coupled with these deep introgression events may have led to some of the morphological and ecological evidence previously used in support of the erection the subgenera Neruda (for H. aoede and allies [Turner, 1976]) as well as Laparus (for H. doris [Turner, 1968]) that appeared to conflict with more recent molecular genetic data.
Major introgression patterns in the melpomene-silvaniform clade
We next focus more closely on the melpomene-silvaniform clade (including H. besckei, H. numata, and H. melpomene, represented by ‘BNM’ in Figure 1A). This is one of the most phylogenetically difficult groups of Heliconius due to ongoing hybridization and extensive gene flow involving most members of the group (Mallet et al., 2007). Previous studies have inferred conflicting introgression scenarios in this clade (Martin et al., 2013; Zhang et al., 2016; Jay et al., 2018; Edelman et al., 2019; Cicconardi et al., 2023). We compiled a multilocus dataset from high-quality genomic data comprising eight (out of 15) species representing all major lineages within the clade: H. melpomene, H. cydno, H. timareta, H. besckei, H. numata, H. hecale, H. elevatus, and H. pardalinus (Supplementary file 1l). Our analysis below confirms widespread introgression within this clade.
We identify four major species relationships from blockwise estimates of species trees under the MSC model without gene flow (Figure 2A, Figure 2—figure supplement 1, Supplementary file 1m and n): (a) autosome-majority (trees 1–3), (b) autosome-variant (trees 5–7), (c) the Z chromosome (chr. 21; tree 4), and (d) chromosome 15 inversion region (15b; tree 24). The pattern is highly similar between coding and noncoding loci. The first three relationships (trees 1–9) account for >90% of the blocks, with a well-supported pardalinus-hecale clade ((H. pardalinus, H. elevatus), H. hecale) and a cydno-melpomene clade ((H. timareta, H. cydno), H. melpomene). They differ in the position of H. numata and in the relationships among the three species in the cydno-melpomene clade. We first focus on the three scenarios relating to different positions of H. numata: (a) H. numata sister to the pardalinus-hecale clade + the cydno-melpomene clade, (b) H. numata sister to the pardalinus-hecale clade, and (c) H. numata sister to H. besckei. The Z-chromosome tree (i.e. tree 4 in scenario c) is the MAP tree with a high posterior probability in almost all blocks of the Z chromosome (median probability of 1; Figure 2A, Supplementary file 1m), with H. besckei + H. numata diverging first, followed by a split between the pardalinus-hecale clade and the cydno-melpomene clade. A similar distinction between the autosome-majority trees and the Z-chromosome tree was also found by Zhang et al., 2016. All four scenarios a–d confirm paraphyly of the silvaniform species (H. besckei, H. numata, and the pardalinus-hecale clade), consistent with some recent phylogenomic studies (Zhang et al., 2016; Massardo et al., 2020; Cicconardi et al., 2023). Monophyly of the silvaniforms was suggested in concatenation/sliding-window analysis (Heliconius Genome Consortium, 2012; Kozak et al., 2015; Kozak et al., 2021; Zhang et al., 2021), but this conclusion may suffer from a failure to account for deep coalescence (Edwards et al., 2016).
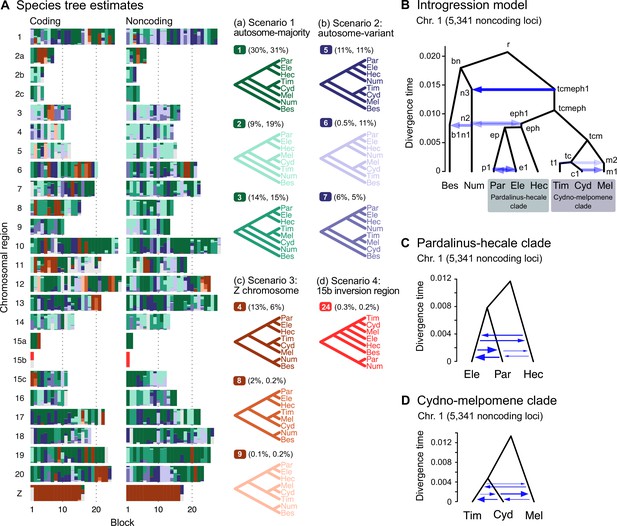
Major introgression events in the melpomene-silvaniform clade.
(A) Blockwise estimates of species trees of the melpomene-silvaniform clade inferred from 200-locus blocks across the genome under the multispecies coalescent (MSC) model with no gene flow using bpp (see Supplementary file 1l for data, Supplementary file 1m and n and Figure 2—figure supplement 1 for full results). Maximum a posteriori (MAP) trees are labeled in decreasing order of frequency among blocks. Proportions of coding and noncoding blocks with each tree as a MAP tree are shown in parentheses. (B) The multispecies coalescent with introgression (MSC-I) model events that can explain the three major groups of trees in (A). Branch lengths are based on posterior means of divergence/introgression times estimated from 5341 noncoding loci on chromosome 1 (Supplementary file 1q). Each internal node is given a label, which is used to refer to a population above the node, for example, the population between nodes r and bn is referred to as branch bn. Each horizontal arrow represents a unidirectional introgression event, for example, the arrow from tcmeph1 to n3 represents tcmeph→Num introgression at time τtcmeph1 = τn3 with probability φtcmeph1→n3. (C) Continuous migration (IM) model for the pardalinus-hecale clade, allowing bidirectional gene flow among the three species. (D) Multispecies coalescent with migration (MSC-M) model for the cydno-melpomene clade. For (C) and (D), branch lengths are based on estimates from noncoding loci in chromosome 1 (Figure 2—figure supplement 6, Supplementary file 1s–u), and arrow sizes are proportional to estimated migration rate (M = Nm). Bes: H. besckei; Num: H. numata; Par: H. pardalinus; Ele: H. elevatus; Hec: H. hecale; Tim: H. timareta; Cyd: H. cydno; Mel: H. melpomene.
Our species tree search under the MSC (Figures 1A and 2A) accounts for ILS but does not account for gene flow. To approximate a fuller introgression history of this group, we construct a species tree model with introgression that can explain the four scenarios above. We use estimates of migration rates between each pair of species under the MSC-with-migration (MSC-M or IM) model of species triplets (3s analysis) to inform placement of introgression edges (Appendix 1—figure 2 , Figure 2—figure supplement 2). Our proposed model has six pairs of bidirectional introgression events (Figure 2B). We use the Z-chromosome tree (tree 4) as the backbone that most likely represents the true species tree, while conflicting trees result from historical introgression. Tree 4 is largely limited to the Z chromosome, which appears more resistant to gene flow in Heliconius (Zhang et al., 2016; Van Belleghem et al., 2018; Martin et al., 2019; Massardo et al., 2020; Thawornwattana et al., 2022). Consistent with this interpretation, we find no evidence of gene flow in the Z chromosome and high prevalence of gene flow on the autosomes based on the 3s analysis (Appendix 1—figure 2 , Figure 2—figure supplement 2, Supplementary file 1o and p). Thus, we include two introgression events (between nodes n3–tcmeph1 and n2–eph1 in Figure 2B) to explain alternative positions of H. numata in the autosomes (scenarios a and b). Next, to explain the secondary source of genealogical variation within the cydno-melpomene clades (i.e. among trees 1–3, trees 4/8/9 and trees 5–7), we add two further introgression events between H. melpomene and H. cydno (m1–c1), and between H. melpomene and H. timareta (m2–t1). We do not model H. cydno–H. timareta introgression (these species are allopatric). We also include introgression between H. besckei and H. numata (b1–n1) to relate the autosome trees to the Z-chromosome tree. Finally, sister species H. pardalinus and H. elevatus hybridize today in sympatric populations (Rosser et al., 2019), so we allow introgression between them (p1–e1). According to the 3s analysis, the rates of gene flow in this pair are among the highest (Figure 2—figure supplement 2). This sister-species introgression does not alter species trees (Figure 2A) because it does not change the topology.
The resultant MSC-I model is used to estimate species divergence times and effective population sizes for extant and ancestral species, and the intensity, timing, and direction of introgression. Consistent with scenario c representing the true species tree, we find least introgression on the Z chromosome (Figure 2—figure supplements 3 and 4, Supplementary file 1q and r). On the autosomes, there is substantial introgression from the pardalinus-hecale + cydno-melpomene clade into H. numata, and to a lesser extent, between H. numata and the pardalinus-hecale clade. These patterns match well with the frequencies of the two main autosomal relationships (scenarios a and b in Figure 2). Within the cydno-melpomene clade, introgression is predominantly unidirectional from H. cydno and H. timareta into H. melpomene. The H. pardalinus–H. elevatus pair shows ongoing extensive but variable introgression across the genome, with the introgression time estimated to be zero. See Appendix 1, section ‘Major introgression patterns in the melpomene-silvaniform clade inferred using 3s and BPP' for more details.
The age of the melpomene-silvaniform clade (τr) is estimated to be ~0.020 substitutions per site based on noncoding data (Figure 2B, Figure 2—figure supplement 4, Supplementary file 1q). This translates to ~1.7 (CI: 0.9, 3.8) million years ago (Ma), assuming a neutral mutation rate of 2.9 × 10–9 per site per generation (95% CI: 1.3 × 10–9, 5.5 × 10–9) and four generations per year (Keightley et al., 2015). This is not very different from a previous estimate of 3.7 (CI: 3.2, 4.3) Ma based on molecular clock dating (Kozak et al., 2015), which ignores ancestral polymorphism and is therefore expected to overestimate divergence time. Overall, our estimates of species divergence time tend to be precise and highly similar across the genome (Figure 2—figure supplements 3 and 4). The posterior means from coding and noncoding loci are strongly correlated, with τC ≈ bτNC where b varies between 0.4 and 0.6 (r2 > 0.95) in most chromosomal regions (Figure 2—figure supplement 5). The scale factor of b < 1 can be explained by purifying selection removing deleterious nonsynonymous mutations in coding regions of the genome (Shi and Yang, 2018). Present-day and ancestral population sizes (θ) are of the order of 0.01 (Figure 2—figure supplement 4B, Supplementary file 1q and r). For inbred individuals (chosen for sequencing to facilitate genome assembly) among our genomic data (H. melpomene, H. timareta, H. numata, and H. pardalinus; see Supplementary file 1a), θ estimates vary among chromosomes by orders of magnitude, with the inbred genome of H. melpomene having the lowest population size of ~0.002–0.004 on average. Adding more individuals should help stabilize estimates of θ, but should not affect estimates of age or introgression rates.
The introgression model of Figure 2B assumes that gene flow occurs in single pulses. This may be unrealistic if gene flow is ongoing. We thus employ the MSC-M model implemented in bpp to estimate migration rates (M = Nm) between all pairs of species in each of the pardalinus-hecale and cydno-melpomene clades (Figure 2C and D). The MSC-M model assumes continuous gene flow since lineage divergence at the rate of M migrants per generation (Flouri et al., 2023). The results concur with the introgression pulse model in suggesting high gene flow between H. pardalinus and H. elevatus (Figure 2—figure supplement 6A and B, Supplementary file 1s). There is also evidence of gene flow between H. hecale and H. pardalinus/H. elevatus at lower levels, with M < 0.1 in most chromosomes (Figure 2—figure supplement 6A, Supplementary file 1s). Allowing for continuous gene flow as well as gene flow involving H. hecale, we obtain slightly older estimates of both species divergence times (between H. pardalinus and H. elevatus, and between H. hecale and H. pardalinus + H. elevatus) (Figure 2—figure supplement 6A, Supplementary file 1s), than under the single-pulse introgression model (Supplementary file 1q and r). The cydno-melpomene clade shows a similar pattern of older divergence times with substantial gene flow between all three species although at smaller magnitudes (M ~0.01–0.1) (Figure 2—figure supplement 6C, Supplementary file 1u). For more discussion of parameter estimates, see ‘Appendix 1,’ section 'MSC-M model for pardalinus-hecale and cydno-melpomene clades.' We conclude that a model with continuous gene flow involving all three species may better describe the history of both the pardalinus-hecale clade and the cydno-melpomene clade.
In summary, we have identified substantial gene flow within the cydno-melpomene and pardalinus-hecale clades based on both pulse introgression (MSC-I) and continuous migration (MSC-M) models (Figure 2C and D,, Figure 2—figure supplement 6, Supplementary file 1q–u). Earlier genomic studies failed to quantify the intensity of gene flow (introgression probability or migration rate) or infer direction and timing of gene flow. Gene flow within the cydno-melpomene clade has been extensively studied at population/subspecies levels and at specific loci involved in wing pattern mimicry (Bull et al., 2006; Pardo-Diaz et al., 2012; Kronforst et al., 2013; Martin et al., 2013; Wallbank et al., 2016; Enciso-Romero et al., 2017; Martin et al., 2019), but gene flow involving other species has received less attention (Heliconius Genome Consortium, 2012; Wallbank et al., 2016; Zhang et al., 2016; Jay et al., 2018).
Complex introgression in the 15b inversion region (P locus)
A polymorphic series of tandem inversions on chromosome 15 is involved in switching mimicry color pattern in H. numata (Jay et al., 2018; Jay et al., 2022). The first inversion, P1 (~400 kb), is in the 15b region (also called the P locus), and is fixed in H. pardalinus and retained as a polymorphism in H. numata (Joron et al., 2011; Le Poul et al., 2014; Jay et al., 2018). Multiple introgression events are necessary to make the 15b tree (Figure 2A, scenario d, tree 24) compatible with either the Z chromosome tree or the autosomal trees (Figure 2A, scenarios a–c), suggesting a much more complex introgression history of this region than in the rest of the genome. This inversion contains the known wing patterning locus cortex (Jay et al., 2022), where it is maintained as a balanced polymorphism by natural selection (Joron et al., 2006; Nadeau et al., 2016; Van Belleghem et al., 2017). Another feature of this 15b region is that among the species without the inversion, the cydno-melpomene clade clusters with H. elevatus and is nested within the pardalinus-hecale clade (without H. pardalinus). This is contrary to the expectation based on the topologies in the rest of the genome (Figure 2A, scenarios a–c) that the cydno-melpomene clade would be sister to the pardalinus-hecale clade (without H. pardalinus). One explanation for this pattern is that introgression occurred between the common ancestor of the cydno-melpomene clade and either H. elevatus or the common ancestor of H. elevatus and H. pardalinus together with a total replacement of the non-inverted 15b in H. pardalinus by the P1 inversion from H. numata (Jay et al., 2018). We confirm and quantify this introgression below.
Using data from additional species (’silv_chr15' dataset in Supplementary file 1a; see ‘Methods’), we obtain a better resolution of species relationships along chromosome 15, although with some uncertainty within the inversion region due to small numbers of loci (Figure 3A, Supplementary file 1v). This analysis of independent data agrees with the Z chromosome tree (tree 24 in Figure 2A) and with (unrooted) trees obtained from concatenation analysis by Jay et al., 2018 (their Figures 2 and S1) and Jay et al., 2021 (their Figure S4), where H. numata with the inversion groups with H. pardalinus while H. numata without the inversion groups with its sister species, H. ismenius (Figure 3A, red trees). Outside the inversion region, H. numata with both inversion genotypes groups with H. ismenius as expected (Figure 3A, blue trees). Although this conclusion assumes that H. numata and H. ismenius are sister species while H. ismenius is not included in our species tree analysis of the melpomene-silvaniform clade (Figure 2), this sister relationship agrees with previous genomic studies of the autosomes and the sex chromosome (Zhang et al., 2016; Jay et al., 2021; Cicconardi et al., 2023; Rougemont et al., 2023).
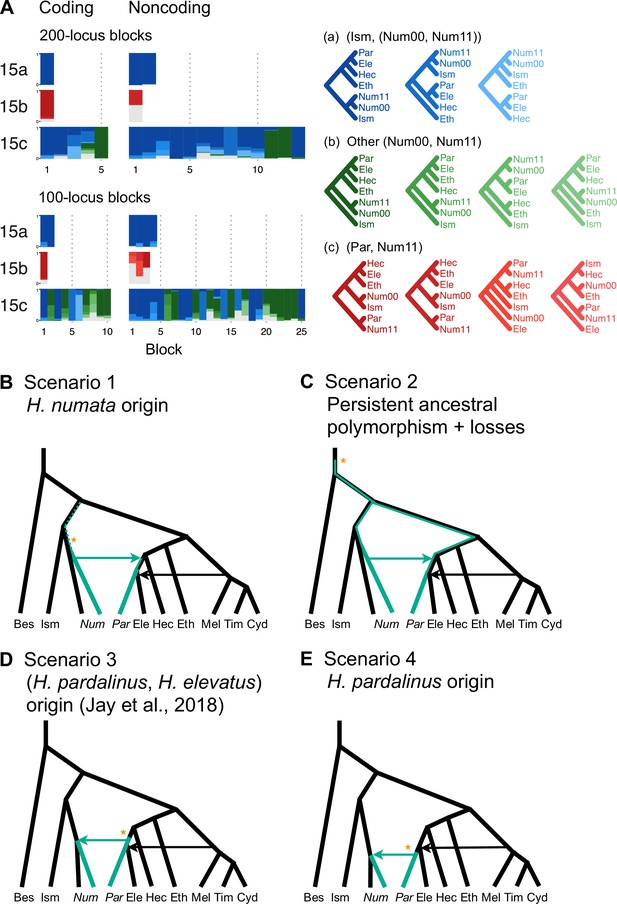
Introgression history of the chromosome 15 inversion region (15b).
(A) Blockwise estimates of species trees for the inversion (15b) and the remnant flanking regions (15a and 15c) of chromosome 15. Trees were inferred from 200-locus blocks and 100-locus blocks under the multispecies coalescent (MSC) model without gene flow using bpp (Supplementary file 1v). Tree legends are grouped by whether H. numata clusters with H. ismenius (blue), or H. numata with P1 inversion (Num11) clusters with H. pardalinus (red), or other relationships (green). (B–E) Four possible scenarios of the origin and introgression route of the P1 inversion. Star indicates the origin of the P1 inversion. Green lineages have the inversion, and green arrows indicates introgression of the inversion. Ism: H. ismenius; Num00: H. numata homozygous uninverted 15b (H. n. laura and H. n. silvana); Num11: H. numata homozygous for inversion P1 (H. n. bicoloratus); Eth: H. ethilla. See Figure 2 legend for codes for other species. For the direction of melpomene-cydno clade→H. elevatus, see Figure 3—figure supplement 3.
Given that H. numata is an early-diverging lineage of the melpomene-silvaniform clade (Figure 2B) and is polymorphic for P1 over large parts of its geographic distribution while H. pardalinus is fixed for this inversion (Joron et al., 2011; Jay et al., 2022), one parsimonious explanation is that the inversion originated either in H. numata after diverging from H. ismenius, or before the H. numata–H. ismenius split but was subsequently lost in H. ismenius, followed by introgression which introduced the inversion from H. numata into H. pardalinus, either before or after its divergence from H. elevatus (Figure 3B). If the introgression occurred before H. pardalinus–H. elevatus divergence, the lack of the inversion in H. elevatus can be explained by another introgression from the cydno-melpomene clade into H. elevatus, completely replacing the inversion with the original orientation (Figure 3B). This introgression route is reported in previous genomic studies, including at a different locus (optix gene, also involved in color pattern) in chromosome 18 as well as in the 15b region of chromosome 15 (Heliconius Genome Consortium, 2012; Wallbank et al., 2016). Under this scenario, we might expect the 15b region to be less diverse in H. pardalinus (recipient of P1) than in H. numata (donor of P1), with the magnitude depending on the duration between the origin of P1 and introgression into H. pardalinus. However, we do not see this reduced heterozygosity in our data (Figure 3—figure supplement 1), suggesting that the transfer likely occurred early, shortly after the formation of the inversion in H. numata, or shortly after the H. numata–H. ismenius split if the inversion originated earlier. This scenario is further supported by the 15b tree having similar times of divergence between H. ismenius and H. numata without the inversion, and between H. pardalinus and H. numata with the inversion (Figure 3—figure supplement 2). The topology of the 15b tree (Figures 2A and 3A) also indicates that the first split is between species with the inversion (H. numata and H. pardalinus) and those without the inversion. This suggests another possibility: inversion polymorphism could have existed earlier in the history of the melpomene-silvaniform clade but was subsequently lost in most species (Figure 3C). In this scenario, the ancestral polymorphism is maintained in H. numata while the inversion is fixed in H. pardalinus but is lost in other species. Introgression between H. numata and H. pardalinus is not required but could still occur.
To reconcile the introgression history of 15b with the overall species tree, we add three additional bidirectional introgression events onto the main model in Figure 2B and assess their plausibility using bpp. We allow for (i) bidirectional introgression between the cydno-melpomene clade into H. elevatus, (ii) bidirectional introgression between H. numata and H. pardalinus, and (iii) introgression between H. besckei and the common ancestor of the cydno-melpomene + pardalinus-hecale clade (to account for H. besckei being clustered with other species that do not have the inversion; see Figure 2A). We consider five models (m1–m5) differing in the placements of introgression events (i) and (ii) either before or after the H. pardalinus–H elevatus split (Figure 3—figure supplement 3). Our results best support unidirectional introgression from H. numata into the common ancestor of H. pardalinus and H. elevatus, and from the common ancestor of the cydno-melpomene clade into H. elevatus shortly after its divergence from H. pardalinus (Figure 3—figure supplement 3, model m3). In other scenarios, estimated introgression times tend to collapse onto the H. pardalinus–H elevatus divergence time, suggesting that the introgression events were likely misplaced (Figure 3—figure supplement 3, Supplementary file 1w). Our finding that divergence of H. elevatus and introgression from the cydno-melpomene clade occurred almost simultaneously provides evidence for a hybrid speciation origin of H. elevatus resulting from introgression between H. pardalinus and the common ancestor of the cydno-melpomene clade (Rosser et al., 2019).
In an alternative scenario proposed by Jay et al., 2018, the inversion first originated in the common ancestor of H. pardalinus and H elevatus, and subsequently introgressed into some subspecies of H. numata, while the inversion in H. elevatus was completely replaced by introgression from the cydno-melpomene clade (Figure 3D). They used sliding-window gene tree topologies to support introgression of the inversion from H. pardalinus to H. numata shortly after its formation in the common ancestor of H. pardalinus and H elevatus (their Figures 4 and S4). By including H. ismenius and H. elevatus, sister species of H. numata and H. pardalinus, respectively, different directions of introgression should lead to different gene tree topologies. Clustering of (H. numata with the inversion, H. pardalinus) with H. numata without the inversion would suggest the introgression is H. numata → H. pardalinus while clustering of (H. numata with the inversion, H. pardalinus) with H. elevatus would suggest H. pardalinus → H. numata introgression. However, tree topologies supporting each direction of introgression were almost equally common within the inversion region, particularly in the first half of the inversion, undermining this argument. With high levels of ILS and introgression in the group, estimated gene trees need not reflect the true species relationships. Another variant of this scenario is that the inversion originated in H. pardalinus after its divergence from H. elevatus and was introgressed into some subspecies of H. numata (Figure 3E). We consider this scenario unlikely because the inversion appears to originated long before the H. pardalinus–H. elevatus split given deep divergence of lineages with and without the inversion (see 15b trees in Figures 2A and 3A).
Discussion
Approaches for estimating species phylogeny with introgression from whole-genome sequence data: Advantages and limitations
Our full-likelihood MSC approach yields several improvements over concatenation and other approximate methods for inferring species trees and introgression. First, we account for ILS due to coalescent fluctuations, so variation in inferred genealogical histories can be more firmly attributed to variation in gene flow across the genome. Second, we analyze the sequence data directly, rather than using estimated gene trees as data. This utilizes information in branch lengths of the gene trees while accommodating their uncertainty. We retain heterozygous sites in the alignments as unphased diploid sequences from each individual without collapsing them into a single randomly phased nucleotide as is common in other phylogenomic studies. Our approach allows multiple individuals per species to be included, leading to improved estimation of introgression parameters. Third, MSC models with pulse introgression (MSC-I) or continuous migration (MSC-M) allow direct estimation of key features of the direction, intensity, and timing of gene flow, and are applicable to gene flow between sister species. Widely used summary statistics such as D and fd do not estimate these parameters and cannot detect gene flow between sister lineages. Previous analyses using sliding-window concatenation or based on estimated gene trees were therefore liable to be less successful for estimating branching order in the melpomene-silvaniform clade, and because they may be misled by rapid speciation events coupled with extensive gene flow (Edelman et al., 2019; Massardo et al., 2020; Kozak et al., 2021).
There are several limitations in our approach. First, in the exploratory analysis of genomic blocks using MSC without gene flow (e.g. Figures 1A and 2A), we assume that one of the main topologies represents the true species tree and is related to other topologies via introgression. In general, it can be nontrivial to decide which topology most likely represents the true species tree, and how to add introgression edges onto the assumed species tree to explain other topologies. Here, we employ several strategies including a recombination-rate argument (Figure 1F), reduced introgression in the sex chromosome (Figure 2A), estimates of pairwise migration rates under an explicit model of gene flow (Appendix 1—figures 1 and 2), and using additional evidence from geographic distributions (Rosser et al., 2012) and records of natural hybrids (Mallet et al., 2007). These strategies could fail in taxa with more limited biological or distribution information, or when the sex chromosome (if available) is less unreliable as to species topology. Ideally, one would like to be able to estimate species phylogeny and introgression events simultaneously as part of a single unsupervised analysis, but this task remains computationally challenging. Software such as PhyloNet (Wen and Nakhleh, 2018) and starBEAST (Zhang et al., 2018) implement Bayesian inference of MSC with introgression but they are usually limited to small datasets of no more than 100 loci. Heuristics based on summary statistics may attempt to place introgression edges onto a species phylogeny estimated on the assumption of no gene flow (Malinsky et al., 2018; Suvorov et al., 2022; Cicconardi et al., 2023). However, these approaches do not make full use of sequence data and can be misled by incorrect initial species trees. Furthermore, they cannot convincingly infer direction of gene flow or gene flow between sister species.
Second, although our approach is more powerful than approximate methods, it is also more computationally intensive and does not scale well to analyses of large numbers of species (>20, say). In Heliconius, there are 47 species in six major clades. Our strategy has therefore been to analyze subsets of species representing major clades or representative species within each clade. We then combine the results into a larger phylogeny. One caveat is that not all species get explored at the same time, and some introgression events may be missed. Lastly, the MSC makes simplifying modeling assumptions such as neutral evolution, constant substitution rates on all branches, no recombination within each locus, and free recombination among loci, the implications of which have been discussed previously (Burgess and Yang, 2008; Thawornwattana et al., 2022). We note that although the MSC model assumes constant rate neutral evolution, estimated introgression probabilities (φ) are a combination of both actual introgression and subsequent positive and negative selection on introgressed loci, the extent of which is modulated by local recombination rate (Petry, 1983; Barton and Bengtsson, 1986).
In summary, we first explore major species relationships across genome blocks using a full-likelihood MSC approach. We then identify introgression events that explain the different local species trees by proposing introgression edges on a background species topology, with additional evidence from explicit models of gene flow. In addition, geographic distributions and the prevalence of natural hybrids can be employed to help with placement of introgression edges in a phylogeny. This process can result in several competing introgression models. We estimate parameters under each model using sequence data, evaluate the model fit, and perform model comparison. By breaking the problem into manageable parts in this way, our approach is computationally tractable.
An updated phylogeny of Heliconius
We have clarified phylogenetic relationships among major clades within Heliconius and quantified major introgression events, including introgression between sister species (which does not change tree topology). We summarize our three key findings in Figure 4. First, H. aoede is most likely the earliest-branching lineage of Heliconius, and an ancient introgression event led to its apparently closer relationship with the doris-burneyi-melpomene clade (Figure 1). We discuss an implication of this placement below. However, we emphasize the considerable level of uncertainty that remains, and having more high-quality genome data from H. aoede and its sibling species may help improve the phylogenetic resolution. Second, within the melpomene-silvaniform clade, we obtained a robust pattern in which the common ancestor of H. besckei, H. numata, and H. ismenius first split off from the rest, rendering the silvaniform species paraphyletic (Figure 2). This is contrary to a common belief that the silvaniforms are monophyletic (Heliconius Genome Consortium, 2012; Kozak et al., 2015; Jay et al., 2021; Kozak et al., 2021; Zhang et al., 2021). Third, the P1 inversion on chromosome 15 involved in wing pattern mimicry likely introgressed from H. numata into H. pardalinus based on species tree topologies (Figures 2 and 3) and direct modeling of introgression (Figure 3—figure supplement 3), although where and when it originated remain uncertain.
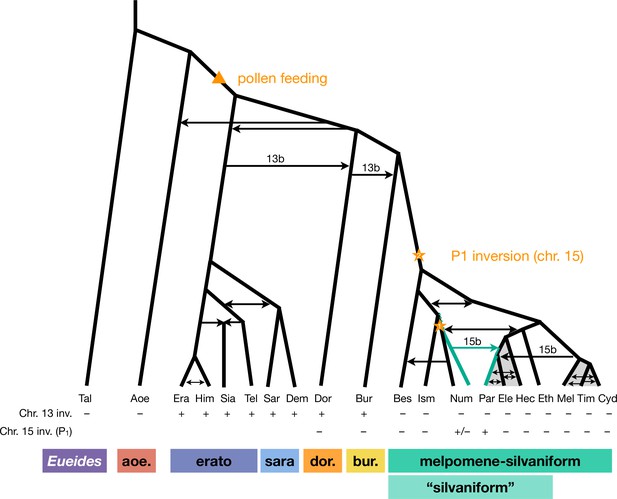
Revised phylogeny and introgression history of Heliconius.
Phylogeny of the erato-sara clade was previously estimated using a similar approach (Thawornwattana et al., 2022). Arrows represent introgression events. Introgression of chromosomal inversions are region-specific, indicated by a label (13b or 15b) above the arrow. Status of inversions 13b and 15b in each species is indicated in the two rows at the bottom, with + indicating inversion, – indicating standard orientation, and +/– inversion polymorphism. Gray shading represents a period of continuous gene flow. Triangle represents the origin of pollen feeding near the base of Heliconius. Star indicates a possible origin of the P1 inversion on chromosome 15, and green branches indicate lineages with the inversion (polymorphic in Num, fixed in Par). Him: H. himera; Sia: H. hecalesia; Tel: H. telesiphe; Dem: H. demeter; see Figures 1—3 legends for other species codes.
Overall, we conclude that the P1 inversion likely arose in H. numata or its close ancestor and became fixed in H. pardalinus via an introgression replacement event from the former species (Figure 3B), although a deeper origin followed by persistent ancestral polymorphism (Figure 3C) remains a possibility.
Heliconius is an example of recent and rapid diversification with extensive gene flow occurring between closely related species as well as between more divergent lineages throughout its history at varying degrees. Since many species pairs of Heliconius show high degrees of sympatry today, there are plenty of opportunities for low levels of gene flow to occur whenever their ranges overlap (Rosser et al., 2015). This fact is evident in our estimates of pairwise migration rates among species in the melpomene-silvaniform clade (Appendix 1—figures 1 and 2). There are many documented natural hybrids (Mallet et al., 2007) and there is evidence for introgression between particular pairs of subspecies from genomic studies, most of which involve wing patterning loci (Wallbank et al., 2016; Enciso-Romero et al., 2017; Morris et al., 2020; Rougemont et al., 2023). We did not attempt to incorporate all possible introgression signals into our phylogeny but instead focused on major introgression events that explain major patterns of species tree variation across the genome using a representative subset of species. Thus our phylogeny in Figure 4 should be viewed as a simplified version of reality. One future direction is to model a low level of admixture among all lineages.
Implications for the evolution of pollen feeding
To illustrate how our updated phylogeny (Figure 4) can provide new insights into the evolution of Heliconius, we discuss an implication of our results for the evolution of pollen collection and pollen-feeding by adult Heliconius, a unique trait found in no other Lepidoptera (Gilbert, 1972). Our phylogeny indicates that the aoede clade, comprising H. aoede and three other species, is likely the earliest-branching lineage of Heliconius, followed by the erato-sara clade. This branching order is consistent with a phylogeny based on morphological and behavioral characters (Penz, 1999). Since the aoede clade is the only group of Heliconius that does not feed on pollen (Brown, 1981), the aoede-early scenario (Figure 1C) suggests that unique pollen feeding traits arose once after the divergence of the aoede clade (Figure 4). In contrast, the erato-early scenario (Figure 1B) is less parsimonious as it requires an additional loss of pollen feeding ability in the aoede clade (Cicconardi et al., 2023).
Turner, 1976 named the aoede clade as subgenus Neruda based on distinct morphology in all life stages, such as pupae that are more similar to those of Eueides (a sister genus of Heliconius), unique larva morphology, white eggs like those of Eueides (instead of yellow eggs in all other Heliconius), a different genus of larval host plant, Dilkea (Benson et al., 1975) (all other Heliconius species feed on Passiflora), and chromosome numbers of n = 21–31 which are variable and intermediate between Heliconius (n = 21 in most species) and Eueides (n = 31) (Brown et al., 1992). For these reasons, Brown, 1981 later raised Neruda from subgenus to genus.
Thus, morphological/behavioral/ecological characters tend to support early divergence of the aoede clade (Brown, 1972; Penz, 1999). Molecular phylogenetic work, on the other hand, seemed to support the erato-early scenario (Figure 1B; Brower and Egan, 1997; Beltrán et al., 2007; Kozak et al., 2015; Kozak et al., 2021; Cicconardi et al., 2023). Consequently, generic status of Neruda was revoked (Kozak et al., 2015). Our full-likelihood approach, which accounts for ILS as well as introgression, enables inference of a phylogeny that is more parsimonious with morphology and life history. This result parallels analysis of genomic data from African mosquitoes in the Anopheles gambiae species complex, in which coalescent-based likelihood analyses support species trees that are more parsimonious for chromosomal rearrangement data (Thawornwattana et al., 2018), while prior sliding-window and concatenation analysis favored trees that are less parsimonious (Fontaine et al., 2015).
Our likelihood analyses can thus be used to rescue generic or subgeneric status for Neruda. Nevertheless, there remains considerable uncertainty near the base of Heliconius. Further whole-genome data from H. aoede and related species in the aoede, burneyi and doris clades will likely improve resolution.
Methods
Whole-genome sequence data and genotyping
We obtained raw sequencing data from previous studies (Wallbank et al., 2016; Edelman et al., 2019; see Supplementary file 1a) and extracted multilocus data as described previously (Thawornwattana et al., 2022) as unphased diploid sequences, retaining heterozygous sites. Sequencing reads were mapped to each of two reference genomes (see below) using bwa mem v0.7.15 (Li, 2013) with default parameters, and then sorted using sambamba v0.6.8 (Tarasov et al., 2015). RealignerTargetCreator and IndelRealigner modules in GATK v3.8 were used to improve alignment around indels (McKenna et al., 2010; DePristo et al., 2011). We called genotypes of each individual using mpileup and call modules in bcftools v1.5 (Li et al., 2009) with the multiallelic-caller model (call -m) (Li, 2011), and set minimum base and mapping quality to 20. We retained high-quality genotype calls using bcftools filter using the following filters: (1) a minimum quality score (QUAL) of 20 at both variant and invariant sites; (2) for each genotype, a genotype quality score (GQ) ≥ 20 and a read depth (DP) satisfying max(meanDP/2, d) ≤ DP ≤ 2 meanDP, where d = 12 or 20 depending on the dataset, and meanDP is the sample-averaged read depth. This choice of d was used to retain a large number of loci while maintaining low genotype-calling error rate (Thawornwattana et al., 2022). For the Z chromosome in females (which are heterogametic in Heliconius), we halved the DP threshold (d). Finally, we excluded sites within five base pairs (bp) of indels.
Multilocus datasets
There are three main datasets in this study: (1) 'etales-9spp' contains eight species (H. melpomene, H. besckei, H. numata, H. burneyi, H. doris, H. aoede, H. erato, and H. sara), one diploid individual per species, representative of all six major clades of Heliconius plus one species from a sister genus Eueides (E. tales). We used two reference genomes (H. erato demophoon v1 and H. melpomene melpomene Hmel2.5; see http://ensembl.lepbase.org/index.html) for read mapping, and two choices of the minimum read depth cutoffs (12 and 20) to ensure high-quality genotypes, resulting in four datasets in total. All reference genomes were available from http://lepbase.org. (2) 'hmelv25-res' contains eight species (one individual per species) within the melpomene-silvaniform clade (H. melpomene, H. cydno, H. timareta, H. besckei, H. numata, H. hecale, H. elevatus, and H. pardalinus), mapped to Hmel2.5 reference. The DP threshold (d) for genotype calling was 20. (3) To understand the history of the 15b inversion region better, we also compiled a third multilocus dataset for chromosome 15 comprising the pardalinus-hecale clade, H. numata with and without the P1 inversion, as well as H. ismenius (sister species of H. numata) and H. ethilla (sister to the pardalinus-hecale clade) (Supplementary file 1a, ’silv_chr15' dataset; see below under 'Chromosome 15 inversion region: dataset and analysis') using publicly available data independent of our previous datasets used in this article (Jay et al., 2018; Jay et al., 2021).
We generated coding and noncoding multilocus datasets from each dataset (Supplementary file 1a) as follows (Appendix 1—figure 3). First, we extracted coordinates of coding and noncoding loci from the reference genome. In this study, loci refer to short segments of DNA that are far apart. The MSC model implemented in bpp assumes complete linkage within a locus and free recombination between loci. In simulations, species tree inference under MSC is found to be robust to within-locus recombination with recombination rates up to 10× the human rate (Zhu et al., 2022). Each coding locus coincided with a protein coding sequence (CDS) and had length at least 100 bp, whereas a noncoding locus can contain noncoding exons, introns, and intergenic regions, and had length 100–1000 bp. Since linkage disequilibrium in Heliconius species decays rapidly to background level within 10 kb (Heliconius Genome Consortium, 2012), we spaced loci ≥2 kb apart, each assumed approximately independent, to obtain sufficient data. We excluded as loci any repetitive regions annotated in the reference genome. We processed each locus by removing sites containing missing data: the locus was discarded if it consisted of >50% missing data. After filtering, we also discarded loci with 10 or fewer sites remaining. We obtained >10,000 loci in each dataset; see Supplementary file 1c and l for the number of loci. For the dataset with read depth cutoff of 12 aligned to the H. erato reference, we obtained about 19,000 noncoding loci and 48,000 coding loci. Note that we here obtain more coding than noncoding loci because noncoding loci were more difficult to align with the divergent Eueides outgroup (~7% divergent). Filtered noncoding loci were more conserved than coding loci for the same reason. The number of informative sites per locus was 10 for coding loci and 4 for noncoding loci on average. Average heterozygosity per site was about 0.43% for coding loci and 0.49% for noncoding loci, with H. besckei having the lowest heterozygosity (0.15–0.25%) and H. burneyi and E. tales having the highest heterozygosity (0.7–0.8%).
For the 'etales-9spp' dataset, we separated out inversion regions on chromosomes 2, 6, 13, and 21 into 2b, 6b, 6c, 13b, and 21b (with two adjacent inversions in chromosome 6; chromosome 21 is the Z [sex] chromosome) while flanking regions were denoted 2a, 2c, 6a, 6d, 13a, 13c, 21a, and 21c, resulting in 30 chromosomal regions in total. These inversions were first identified in a previous study (Seixas et al., 2021); see coordinates in Supplementary file 1b. We obtained >11,000 noncoding loci and >31,000 coding loci in the smallest dataset (aligned to the H. erato demophoon reference, d = 20) and >31,000 noncoding loci and >48,000 coding loci in the largest dataset (Hmel2.5 reference, d = 12); see Supplementary file 1c. The median number of sites was 100–130 depending on the dataset. The number of informative sites had median of 2–3 (range: 0–58) per locus for the noncoding loci and 4–5 (0–570) for the coding loci. Again, noncoding loci are underrepresented in our datasets and they tended to be more conserved.
For the 'hmvelv25-res' dataset, we split chromosomes 2 and 15 into inversion (2b and 15b) and flanking regions (2a, 2c, 15a, and 15c), resulting in 25 chromosomal regions in total; coordinates are in Supplementary file 1b. Although only the chromosome 15b inversion region has been hitherto identified in this melpomene-silvaniform clade, we wished to test whether the chromosome 2b inversion identified in the erato-sara clade was also present in this group. We obtained >80,500 noncoding loci and >73,200 coding loci for 'hmvelv25-res' (Supplementary file 1l). The median number of sites was 339 (range: 11–997) for noncoding loci and 147 (11–12,113) for coding loci. The median number of informative sites per locus was 6 (0–46) for noncoding loci and 2 (0–253) for coding loci.
Overview of analysis approach
We first used the MSC model without gene flow to explore variation in genealogical history across the genome. We then formulated MSC models with introgression (MSC-I) based on a parsimony argument to explain major patterns of genealogical variation. We estimated the direction, timing, and intensity of introgression under each MSC-I model, and assessed most likely introgression scenarios. For gene flow between closely related species that may be on-going, we also used an MSC model with continuous migration (isolation-with-migration; IM) to estimate rates and directionality of gene flow.
Species tree estimation under the MSC model without gene flow using bpp
We performed Bayesian inference of species trees under the MSC model without gene flow using bpp v4.4.0 (Yang and Rannala, 2014; Rannala and Yang, 2017; Flouri et al., 2018). This model accounts for gene-tree heterogeneity due to deep coalescence. Hence, the genome-wide variation in estimated genealogy is most likely due to differential gene flow. We grouped loci into blocks of 200 and estimated a posterior distribution of species trees for each block. This blockwise analysis allows us to explore genealogical variation along each chromosomal region and to choose models of introgression for estimation in later analysis. Blocks of coding and noncoding loci were analyzed separately.
The MSC model without gene flow has two types of parameters: species divergence times (τ) and effective population sizes (θ = 4Nμ), both measured in expected number of mutations per site. For the 'etales-9spp' dataset (all four versions; Figure 1 used H. erato reference depth greater than 12, ‘minDP12’), we assigned a diffuse gamma prior to the root age τ0 ~ G(7, 200), with mean 0.035, and to all population sizes θ ~ G(4, 200), with mean 0.02. Given τ0, other divergence times were assigned a uniform-Dirichlet distribution (Yang and Rannala, 2010, eq. 2). For each block of loci, we performed 10 independent runs of MCMC, each with 2 × 106 iterations after a burn-in of 105 iterations, with samples recorded every 200 iterations. We assessed convergence by comparing the posterior distribution of species trees among the independent runs. Nonconvergent runs were discarded. The samples were then combined to produce the posterior summary such as the MAP tree. There were 1355 blocks in total from all four versions of the dataset (Supplementary file 1c), so there were 13,550 runs in total. Each run took about 20–30 hr.
Similarly, for the 'hmelv25-res' dataset, we used τ0 ~ G(4, 200), with mean 0.02, and population sizes θ ~ G(2, 200) for all populations, with mean 0.01. Each of the 10 independent runs of the MCMC took 1 × 106 iterations after a burn-in of 105 iterations, with samples recorded every 100 iterations. There were 7830 runs in total. Each run took about 15–20 hr.
Migration rate estimation under the MSC-M model for species triplets using 3s
To obtain more direct evidence of gene flow, we estimated migration rates between all pairs of Heliconius species in the 'etales-8spp' dataset under an MSC-with-migration (MSC-M or IM) model using the maximum likelihood program 3s v3.0 (Dalquen et al., 2017). The implementation in 3s assumes a species phylogeny of three species ((S1, S2), S3) with continuous gene flow between S1 and S2 since their divergence at constant rates in both directions, and requires three-phased haploid sequences per locus. Since our multilocus data were unphased diploid, we phased the data using PHASE v2.1.1 (Stephens et al., 2001) to obtain two haploid sequences per individual at each locus. At each locus, we then sampled three types of sequence triplets 123, 113, and 223 with probabilities 0.5, 0.25, and 0.25, respectively, where 113 means two sequences from S1 and one sequence from S3 chosen at random, etc. We used E. tales as the outgroup (S3) for all pairs (S1, S2). We analyzed coding and noncoding loci on the autosomes and three regions of the Z chromosome (chromosome 21) separately, each with 28 pairs among the eight Heliconius species. Additionally, we analyzed each autosomal region separately. This analysis was done with two choices of reference genome at read depth cutoff (d) of 12.
We fitted two models to each dataset: an MSC without gene flow (M0) and a bidirectional IM (M2). Model M0 has six parameters: two species divergence times (τ1 for S1–S2 divergence, τ0 for the root) and four population sizes (θ1 for S1, θ2 for S2, θ4 for the root, and θ5 for the ancestor of S1 and S2); there is no θ3 for S3 because there is at most one sequence from S3 per locus. Model M2 has two additional parameters: M12 and M21, where M12 = m12N2 is the expected number of migrants from S1 to S2 per generation, m12 is the proportion of migrants from S1 to S2 and N2 is the effective population size of S2. M21 is defined similarly. For each model, we performed 10 independent runs of model fitting and chose the run with the highest log-likelihood. We discarded runs with extreme estimates (close to boundaries in the optimization). We then compared models M0 and M2 via likelihood ratio test (LRT) using a chi-squared distribution with two degrees of freedom as a null distribution at a significance threshold of 1%. Adjusting this threshold to account for multiple testing did not change our conclusions because the LRT values were usually extreme, especially in the analysis of all autosomal loci (Supplementary file 1h). There were 31 (30 chromosomal regions + all of autosomal loci together) × 2 (coding and noncoding) × 28 (species pairs) × 2 (choices of reference genome) × 10 (replicates) = 34,720 runs in total. For our largest dataset with >46,000 loci, each run of fitting two models (M0 and M2) took 2–3 hr.
We tested between two competing scenarios (Figure 1B and C) to infer which was more likely by estimating the internal branch length (Δτ = τ0 – τ1) when either H. aoede or H. erato was used as an outgroup, with ingroup species representing the melpomene-silvaniform clade. To this end, we compiled another dataset similar to 'hmelv25-res' but included species from all six major clades of Heliconius (Supplementary file 1a, 'hmelv25-all' dataset). We followed the same procedure as described above. There were 55 pairs in total for each choice of the outgroup species. Estimates of internal branch length close to zero (with the resulting tree becoming star-shaped) suggest that the specified species tree ((S1, S2), S3) was likely incorrect. There were 26 (25 chromosomal regions + all autosomal loci together) × 2 (coding and noncoding) × 55 (species pairs) × 2 (choices of reference genome) × 10 (replicates) = 57,200 runs in total.
Parameter estimation under MSC-I using bpp
Given the species-tree models with introgression of Figure 1D and E, we estimated introgression probabilities (φ), species divergence times and introgression times (τ), and effective population sizes (θ) for each coding and noncoding dataset from each chromosomal region using bpp v4.6.1 (Flouri et al., 2020). We assumed that population sizes of source and target populations remained unchanged before and after each introgression event (thetamodel = linked msci). There were 25 unique parameters in total. We used the same prior distributions for τ and θ as in the MSC analysis without gene flow above, with root age τ0 ~ G(7, 200) and θ ~ G(4, 200) for all populations. We assigned a uniform prior U(0,1) to all introgression probabilities (φ). For each chromosomal region, we performed 10 independent runs of MCMC, each with 1 × 106 iterations after a burn-in of 105 iterations, with samples recorded every 100 iterations. We assessed convergence by comparing the posterior estimates among the independent runs. Non-convergent runs were discarded. Samples were then combined to produce posterior summaries. Multiple posterior peaks, if they existed, were recorded and processed separately. There were 30 (chromosomal regions) × 2 (coding and noncoding) × 2 (trees 1 and 3) × 10 (replicates) = 1200 runs in total. Each run took 200–300 hrs.
For the two MSC-I models of Figure 1D and E, we also estimated the marginal likelihood for each model using thermodynamic integration with 32 Gaussian quadrature points in bpp (Lartillot and Philippe, 2006; Rannala and Yang, 2017) and calculated Bayes factors to compare the two models (Supplementary file 1k). This was done for each coding and noncoding dataset from each chromosomal region. Since the estimates of marginal likelihoods can be noisy, occasionally with extreme outliers, we adjusted estimates of Bayes factor by fitting local quadratic polynomials with span of 0.4 to the difference in the mean log likelihood from the two models, using the loess function in R (Appendix 1—figure 4). Additionally, we checked for reliability of the Bayes factor estimates by performing replicate calculation of the Bayes factor for a few chromosomes (3, 4, 9, and 21a) and found the preferred model choice to be reliable. The replicate estimates of log Bayes factor were –8.44, 19.35, –6.65, and 51.45 for chromosomes 3, 4, 9, and 21a, respectively, while the raw estimates were –11.41, 19.63, –10.94, and 53.12 and the adjusted estimates were –2.15, 20.83, –8.84, and 37.80 (Supplementary file 1k).
The MSC-I model of Figure 2B has six pairs of bidirectional introgression events, with 12 introgression probabilities (φ), 13 species divergence times and introgression times (τ), and 15 population size parameters (θ), a total of 40 parameters. We assigned the same prior distributions to τ0 and θ as in the MSC analysis above, and φ ~ U(0,1) for all introgression probabilities. The MSC-I model of Figure 2C has three more bidirectional introgression edges, with nine additional parameters (six introgression probabilities and three introgression times). Other settings were the same as above. There were 25 (chromosomal regions) × 2 (coding and noncoding) × 10 (replicates) = 500 runs in total. Most runs took 20–40 d.
Parameter estimation under an MSC-M model using bpp
In the MSC-I model of Figure 2B, each of the pardalinus-hecale and cydno-melpomene clades has very recent estimated introgression times; thus gene flow may be ongoing. We therefore used the MSC-M model to estimate six continuous migration rates between the three species in each clade using bpp v4.6.1 (Flouri et al., 2023), assuming the species tree as in Figure 2. We assigned prior distributions τ0 ~ G(2, 200) for the root age (mean 0.01), θ ~ G(2, 500) for all populations (mean 0.004), and M ~ G(2, 10) for all migration rates (mean 0.2). The MCMC setup was the same as in the MSC-I analysis above. There were 25 (chromosomal regions) × 2 (coding and noncoding) × 10 (replicates) × 2 (pardalinus-hecale and cydno-melpomene clades) = 1000 runs in total. Each run took 120–150 hr.
Chromosome 15 inversion region: Dataset and analysis
To investigate the evolutionary history of the inversion region in chromosome 15 (15b region or P locus), we obtained genomic sequence data for six species (H. numata homozygous for the ancestral orientation, H. numata homozygous for the inversion, H. ismenius, H. pardalinus, H. elevatus, H. hecale, and H. ethilla), with two individuals per species, from previous studies (Jay et al., 2018; Jay et al., 2021; Supplementary file 1a, ’silv_chr15' dataset). We extracted a multilocus dataset using an improved pipeline compared with that described above (see 'Whole-genome sequence data and genotyping' and 'Multilocus datasets'). Here, a different filtering strategy used more complex, multi-stage genotyping to account for multiple individuals per species as follows. First, we removed Illumina adapters and trimmed low quality bases using trimmomatric v.0.39 (SLIDINGWINDOW:4:20 MINLEN:50). Next, sequencing reads were mapped to the H. melpomene melpomene reference assembly (Hmel2.5) using bwa-mem v.0.7.17 (Li, 2013). Duplicate reads were masked using MarkDuplicates (Picard) in GATK v.4.2.6.1 (Poplin et al., 2018). We jointly called genotypes on chromosome 15 of individuals from the same species (and the same inversion genotype) using mpileup and call modules in bcftools v1.17 (Li et al., 2009) with the multiallelic-caller model (call -m) (Li, 2011), and set the minimum base and mapping quality to 20. Only high-quality SNPs (QD score ≥2.0 and MQ score ≥40) were retained.
To obtain multilocus data, genomic coordinates of coding and noncoding loci were obtained from the reference genome as described above. Noncoding loci were 100–1000 bp in length and at least 2 kb apart. Coding loci were at least 100 bp in length (no maximum length limit) and at least 2 kb apart. To maximize information, minimum spacing was enforced for loci within the inversion region. We then extracted sequence alignments for each locus using the following procedure. All SNPs passing the quality filter were included. Constant sites were obtained from the reference sequence unless they were masked by one of the following criteria: (1) read depth below 20 (coded as '–') and (2) non-SNP variant or low-quality SNP (coded as 'N'). For each locus, we excluded sequences with >50% of sites missing ('–' or 'N'), and excluded sites with all missing data. We discarded loci with only a single sequence remaining after filtering. Finally, we grouped loci into three regions as before: one inversion region (15b) and two flanking regions (15a and 15c). We obtained 218, 95, and 960 coding loci and 424, 368, and 2446 noncoding loci in 15a, 15b, and 15c regions, respectively.
We performed blockwise estimation of species trees under the MSC model without gene flow as described earlier in blocks of 100 and 200 loci (see 'Species tree estimation under the MSC model without gene flow using bpp'). For the 200-locus blocks, there were 23 (7 coding blocks + 16 noncoding blocks) × 10 (replicates) = 230 runs; each run took about 60 hr. For the 100-locus blocks, there were 45 (13 coding blocks + 32 noncoding blocks) × 10 (replicates) = 450 runs; each run took about 30 hrs.
Appendix 1
Major introgression patterns in the melpomene-silvaniform clade inferred using 3s and BPP
To assess the possibility of introgression that might support the autosome-majority tree or the Z chromosome tree, we estimate pairwise gene flow rates under the MSC-M model between each pair of species in the melpomene clade as well as H. burneyi, H. doris, and H. erato using the program 3s (Zhu and Yang, 2012; Dalquen et al., 2017). This implementation allows for continuous bidirectional gene flow between two species since divergence. We performed maximum likelihood estimation and likelihood ratio tests (LRT) to determine if a allowing for bidirectional gene flow (model M2) is a better fit than without gene flow (model M0). An outgroup species was used to improve the power of the test but it was not involved in gene flow. We used H. aoede as an outgroup in all species pairs. We find gene flow to be prevalent in the autosomes but almost absent in the Z chromosome (Appendix 1—figure 2A). In particular, we find no strong evidence of gene flow in the Z chromosome between H. numata and H. besckei, which is required to explain the Z chromosome tree if the autosome-majority tree is the true species tree. Taken together, these results suggest that the Z chromosome tree represents the true species branching order, and autosomal trees are a result of extensive introgression throughout the history of this group. Using H. erato as an outgroup instead of H. aoede leads to the same conclusion (Appendix 1—figure 2), although the internal branch length becomes zero whenever one of the ingroup species is H. aoede. A full list of estimates from all species pairs is provided in Figure 2—figure supplement 2 and Supplementary files 1o and p.
Next, we formulated two models to capture major introgression events in the group using the Z chromosome tree as a base tree (Figure 2B and C). The first model is the main model used for most parts of the genome and had six pairs of bidirectional introgression (Figure 2B): two for explaining different positions of H. numata (nodes n3–tcmeph1 and n2–eph1, relating trees 1–3, tree 4 and trees 5–7), one for H. pardalinus–H. elevatus gene flow, which is ongoing in sympatric populations (p1–e1) (Rosser et al., 2019), two for gene flow involving H. melpomene with H. cydno (m1–c1) and with H. timareta (m2–t1) (explaining variation among trees 1–3 and trees 5–7), and one for the possibility of H. besckei and H. numata (b1–n1) as suggested by the relationship between the Z chromosome tree and autosome trees. To keep the model tractable, we excluded potential H. cydno–H. timareta introgression in the model because these species are presently allopatric and signals of gene flow between them are likely to be via H. melpomene. We also assumed that population size did not change after introgression. This model has 12 introgression probabilities (φ), 13 species divergence times and introgression times (τ), and 15 population size parameters (θ), a total of 40 parameters. The second model is for the chromosome 15 inversion region (15b), which had three additional pairs of introgression edges, with 9 additional parameters (Figure 3—figure supplement 3, model m1). We estimated those parameters for coding and noncoding loci of each chromosomal region using bpp (Flouri et al., 2020).
Our estimated introgression histories are largely consistent across the genome and between coding and noncoding regions (Figure 2—figure supplements 3 and 4, Supplementary file 1q and r). This pattern agrees with blockwise variation in species relationships (estimated without gene flow, Figure 2A). Gene flow was almost absent in the Z chromosome and consistent with our estimates under the MSC-M model.
The two introgression events involving H. numata that resolve majority autosomal relationships (trees 1–3, 5–7) and the Z chromosome tree (tree 4) were estimated to have high probabilities except for on the Z chromosome and in the chromosome 15b inversion region (Figure 2—figure supplement 4A). First, for the oldest introgression between H. numata and the common ancestor of the cydno-melpomene + pardalinus-hecale clade, the introgression is estimated to be unidirectional into H. numata, with probability (φn3←tcmeph1) ~ 0.85 in most parts of the genome while the introgression in the opposite direction was almost absent (φtcmeph1←n3 < 0.05). Second, the subsequent introgression between H. numata and the common ancestor of the pardalinus-hecale clade is estimated to be bidirectional in many chromosomes, with probabilities (φn2←eph1 and φeph1←n2) ~ 0.3 on average in both directions, although values of these probabilities varied considerably among chromosomes (Figure 2—figure supplement 4A).
Within the pardalinus-hecale clade, we estimate introgression probabilities between the sister species H. pardalinus and H. elevatus (φe1←p1 and φp1←e1) to be ~0.5 in both directions in most chromosomes, although there is some variation across the genome. The time of this introgression (τe1 or τp1) is essentially zero (Figure 2—figure supplement 4A, Supplementary file 1q and r), consistent with the fact that these two species still hybridize today (Mallet et al., 2007). In some chromosomes such as 13 and 21, posterior estimates of ~0.5 had wide intervals (Figure 2—figure supplement 4A). The uncertainty may be due to weak evidence for gene flow, even with non-zero posterior means.
Within the cydno-melpomene clade, both H. melpomene–H. cydno and H. melpomene–H. timareta introgression is estimated to be highly asymmetrical, with H. melpomene being the main recipient in both cases, and with the introgression probabilities varying widely across the genome (Figure 2—figure supplement 4A). The H. cydno → H. melpomene introgression is estimated to be strictly unidirectional, with introgression probability (φm1←c1) of ~0.35 in noncoding regions,~0.25 in coding regions, and zero in the opposite direction (φc1←m1). For H. melpomene–H. timareta, the introgression probability for H. timareta → H. melpomene (φm2←t1) is highly variable across the genome, with values ranging from close to 0 to almost 1; the opposite direction (φt1←m2) had low probabilities (<0.1) for most chromosomes.
Surprisingly, we found strictly unidirectional introgression from H. numata into H. besckei in all regions of the genome, including the Z chromosome, with probability (φb1←n1) around 0.3–0.4 in the noncoding regions and 0.5–0.6 in the coding regions. This is the only substantial introgression estimated in the Z chromosome, with φb1←n1 ≈ 0.3, which was still lower than most autosomes. The chromosome 15 inversion region (15b) has a unique history that is much more complex than the rest of the genome; results for 15b are discussed in the next section.
Species divergence time estimates are precise and highly similar across the genome (Figure 2—figure supplement 4A). The posterior means from coding and noncoding loci are strongly correlated, with τcoding ≈ x τnoncoding where x varies between 0.5 and 0.6 (r2 > 0.95) in most chromosomal regions (Figure 2—figure supplement 5). We estimate the age of the Melpomene clade (τr) to be ~0.020 substitutions per site based on noncoding data and ~0.013 based on coding data. Assuming a constant neutral mutation rate of 2.9 × 10–9 per site per generation (95% CI: 1.3 × 10–9, 1.5 × 10–9) and four generations per year (Keightley et al., 2015), we obtain a conservative estimate that the melpomene clade diverged about 1.7 (CI: 0.9, 3.8) Ma, which is comparable to a previous estimate of 3.7 (CI: 3.2, 4.3) Ma based on molecular clock dating (Kozak et al., 2015). We estimate present-day and ancestral population sizes (θ) to be of the order of 0.01, largely consistent among chromosomes (Figure 2—figure supplement 4B, Supplementary file 1q and r). For inbred reference individuals (H. melpomene, H. timareta, H. numata, and H. pardalinus; see Supplementary file 1a), the estimates (θMel, θTim, θNum, and θPar) tended to be much more variable, with some chromosomes having unusually low estimates in the order of 0.001 or lower. H. melpomene has the lowest population size estimate of about 0.002–0.004 on average. This pattern corresponds well to the fluctuating levels of heterozygosity among chromosomes as a result of inbreeding. Adding more individuals should help stabilize the estimates. Inbred individuals should be avoided in population genomic analyses.
MSC-M model for pardalinus-hecale and cydno-melpomene clades
In the pardalinus-hecale clade, we find the migration rates (MPar→Ele and MEle→Par) between H. pardalinus and H. elevatus to be high and largely asymmetrical, with M ≈ 2–4 migrants per generation in one direction and a lower rate (M < 1) in the opposite direction (Figure 2—figure supplement 6A), consistent with the introgression probability estimates (Figure 2—figure supplement 4A). However, we observe a label-switching problem in which different runs of model fitting on the same data converged to different posterior peaks that differed in the main direction of gene flow accompanied by a flip in the corresponding population size parameters. This near unidentifiability of M and θ appears to be partly associated with high migration rates and low genomic distinctiveness between H. pardalinus and H. elevatus. Since these two species appear to be virtually panmictic and to share effective population size over most of their genome, we also fitted an alternative model to these two species assuming all three populations (two present-day and one ancestral) share the same population size. Under this simpler model, the migration rates are still high (M ~ 1) but less extreme, and the unidentifiability issue largely disappears (Figure 2—figure supplement 6B, Supplementary file 1t). We also detect significant but lesser gene flow involving H. hecale, with migration rates MHec→Ele, MEle→Hec, MHec→Par, and MPar→Hec <0.1 in most chromosomes (Figure 2—figure supplement 6A). Divergence time estimates are more stable among chromosomes and are not affected by the unidentifiability issue. Under this MSC-M model, the divergence time between H. pardalinus and H. elevatus is τep ≈ 0.010 substitutions per site, and the divergence time between H. hecale and H. pardalinus + H. elevatus is τeph ≈ 0.013 substitutions per site based on noncoding data; coding data led to about half these values (Figure 2—figure supplement 6A, Supplementary file 1s). These estimates are slightly older than those obtained under a larger model assuming pulse introgression in Figure 2B are τep ≈ 0.0070 and τeph ≈ 0.0076 using noncoding data (τep ≈ 0.002 and τeph ≈ 0.003 using coding data) (Supplementary file 1q and r).
In the cydno-melpomene clade, estimated migration rates under the MSC-M model are below 0.1, generally much lower than those in the pardalinus-hecale clade, with large variation among chromosomes (Figure 2—figure supplement 6C, Supplementary file 1u). The label-switching problem is rare. Chromosomes with unusually low incoming migration rate estimates tend to be associated with low heterozygosity in inbred individuals (H. melpomene and H. timareta). For instance, the H. melpomene individual had almost no variation in chromosomes 2, 9, 10, and 18, and as a result, the migration rates MCyd→Mel and MTim→Mel (and θMel, θTim) are estimated about one or two orders of magnitude lower on those chromosomes compared with other chromosomes (Supplementary file 1u). Unlike the introgression model where introgression is largely from H. cydno and H. timareta into H. melpomene, there is no clear pattern of highly asymmetrical gene flow into H. melpomene. In this MSC-M model, we also allow for gene flow between H. timareta and H. cydno, which is estimated to be MCyd→Tim ≈ 0.1 and MTim→Cyd ≈ 0.04 migrants per generation in both coding and noncoding loci (Supplementary file 1u). Divergence time estimates are largely comparable across chromosomes. However, the estimates tend to be older than those obtained under the introgression model of Figure 2—figure supplement 4A. We obtained τtc ≈ 0.007 and τtcm ≈ 0.012–0.014 using coding or noncoding loci under the MSC-M model (Supplementary file 1u) while τtc ≈ 0.0025 and τtcm ≈ 0.007 using noncoding data and τtc ≈ 0.001 and τtcm ≈ 0.0043 using coding data (Supplementary file 1q and r).
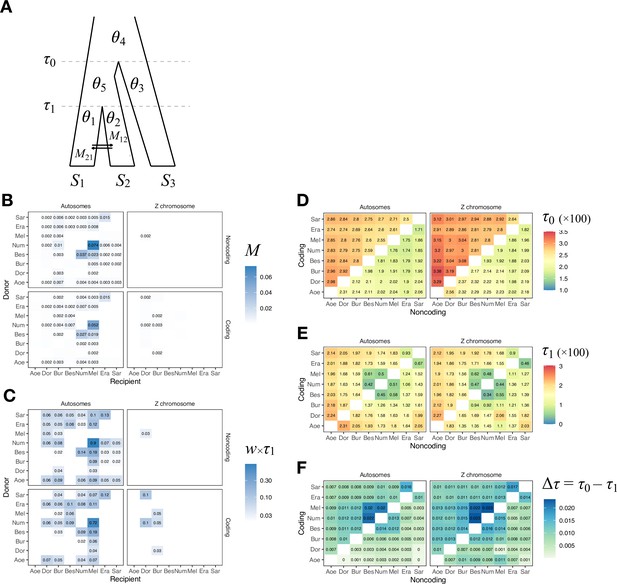
Isolation-with-migration (IM) analysis of the 'etales-9spp' dataset using 3s.
(A) Model specification in 3s. There are three types of parameters: divergence times (τ1, τ0), effective population sizes (θ1, θ2, θ4, and θ5) and migration rates (M12, M21). (B) Maximum-likelihood estimates (MLEs) of the pairwise migration rates (M12, M21), with donor species on the y-axis and recipient species on the x-axis. We used Eueides tales as the outgroup (S3 in A). Top and bottom quadrants are results from noncoding and coding loci, respectively. Left quadrants show results from all autosomal loci. Right quadrants show results from all Z chromosome loci (21a + 21b + 21c). For the number of loci, see Supplementary file 1c (H. erato reference, minDP (d) = 12). For pairs without displayed numbers, the likelihood ratio test (LRT) was not significant at 0.1%, thereby failing to reject a null model of zero gene flow. (C) Mutation-scaled migration rates (w12 = 4M12/θ2 and w21 = 4M21/θ1) as a measure of expected admixture fraction in the recipient genome. (D) Root age (τ0). Estimates from coding loci are shown in the upper triangle while estimates from noncoding loci are in the lower triangle. (E) Divergence time of the ingroup species pair (τ1). (F) Internal branch length (Δτ = τ0 – τ1). Full results are in Supplementary file 1k and Figure 1—figure supplement 7.
Maximum likelihood estimates (MLEs) of migration rates (M), divergence times (τ0, τ1), and the internal branch length (Δτ = τ0 – τ1) from 3s analysis of the 'hmelv25-all' dataset, using (A) H. aoede or (B) H. erato as an outgroup.
See legend to Appendix 1—figure 1.
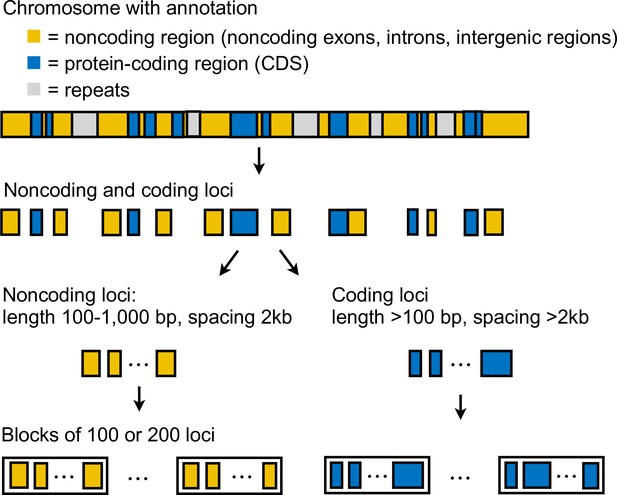
Generation of multilocus data.
Coordinates of coding and noncoding loci were obtained from a reference genome with an annotation of coding sequences (CDS).
Raw (black) and adjusted (red) estimates of the difference in the mean log likelihood from the two models (y-axis).
Raw and adjusted estimates of the Bayes factor are in Supplementary file 1h.
Data availability
All multilocus alignment datasets generated in this study (see Supplementary file 1a, c and l) and scripts for reproducing main analyses are available in Zenodo at https://doi.org/10.5281/zenodo.8415106.
-
ZenodoMajor patterns in the introgression history of Heliconius butterflies.https://doi.org/10.5281/zenodo.8415106
-
NCBI BioProjectID PRJNA532398. Genomic architecture and introgression shape a butterfly radiation.
-
NCBI BioProjectID PRJNA471310. Whole genome sequence data from silvaniform Heliconius individuals.
-
NCBI BioProjectID PRJEB40136. Heliconius numata individual whole genome shotgun sequencing dataset.
References
-
The barrier to genetic exchange between hybridising populationsHeredity 57 (Pt 3):357–376.https://doi.org/10.1038/hdy.1986.135
-
Do pollen feeding, pupal-mating and larval gregariousness have a single origin in Heliconius butterflies? Inferences from multilocus DNA sequence dataBiological Journal of the Linnean Society 92:221–239.https://doi.org/10.1111/j.1095-8312.2007.00830.x
-
Coevolution of plants and herbivores: Passion flower butterfliesEvolution; International Journal of Organic Evolution 29:659–680.https://doi.org/10.1111/j.1558-5646.1975.tb00861.x
-
Cladistic analysis of Heliconius butterflies and relatives (Nymphalidae: Heliconiiti): a revised phylogenetic position for Eueides based on sequences from mtDNA and a nuclear geneProceedings of the Royal Society of London. Series B 264:969–977.https://doi.org/10.1098/rspb.1997.0134
-
The biology of Heliconius and related GeneraAnnual Review of Entomology 26:427–457.https://doi.org/10.1146/annurev.en.26.010181.002235
-
Implementing and testing the multispecies coalescent model: A valuable paradigm for phylogenomicsMolecular Phylogenetics and Evolution 94:447–462.https://doi.org/10.1016/j.ympev.2015.10.027
-
Species tree inference with BPP using genomic sequences and the multispecies coalescentMolecular Biology and Evolution 35:2585–2593.https://doi.org/10.1093/molbev/msy147
-
A Bayesian implementation of the multispecies coalescent model with introgression for phylogenomic analysisMolecular Biology and Evolution 37:1211–1223.https://doi.org/10.1093/molbev/msz296
-
Interspecific Gene Flow Shaped the Evolution of the Genus CanisCurrent Biology 28:3441–3449.https://doi.org/10.1016/j.cub.2018.08.041
-
Inference of gene flow between species under misspecified modelsMolecular Biology and Evolution 39:12.https://doi.org/10.1093/molbev/msac237
-
Association mapping of colour variation in a butterfly provides evidence that a supergene locks together a cluster of adaptive lociPhilosophical Transactions of the Royal Society of London. Series B, Biological Sciences 377:20210193.https://doi.org/10.1098/rstb.2021.0193
-
Estimation of the spontaneous mutation rate in Heliconius melpomeneMolecular Biology and Evolution 32:239–243.https://doi.org/10.1093/molbev/msu302
-
Rampant genome-wide admixture across the Heliconius radiationGenome Biology and Evolution 13:evab099.https://doi.org/10.1093/gbe/evab099
-
Computing Bayes factors using thermodynamic integrationSystematic Biology 55:195–207.https://doi.org/10.1080/10635150500433722
-
Evolution of dominance mechanisms at a butterfly mimicry supergeneNature Communications 5:5644.https://doi.org/10.1038/ncomms6644
-
The sequence alignment/map format and SAMtoolsBioinformatics 25:2078–2079.https://doi.org/10.1093/bioinformatics/btp352
-
Recombination-Aware phylogenomics reveals the structured genomic landscape of hybridizing cat speciesMolecular Biology and Evolution 36:2111–2126.https://doi.org/10.1093/molbev/msz139
-
Whole-genome sequences of Malawi cichlids reveal multiple radiations interconnected by gene flowNature Ecology & Evolution 2:1940–1955.https://doi.org/10.1038/s41559-018-0717-x
-
Multispecies coalescent: Theory and applications in phylogeneticsAnnual Review of Ecology, Evolution, and Systematics 52:247–268.https://doi.org/10.1146/annurev-ecolsys-012121-095340
-
Higher level phylogeny for the passion-vine butterflies (Nymphalidae, Heliconiinae) based on early stage and adult morphologyZoological Journal of the Linnean Society 127:277–344.https://doi.org/10.1111/j.1096-3642.1999.tb00680.x
-
The effect on neutral gene flow of selection at a linked locusTheoretical Population Biology 23:300–313.https://doi.org/10.1016/0040-5809(83)90020-5
-
Efficient Bayesian species tree inference under the multispecies coalescentSystematic Biology 66:823–842.https://doi.org/10.1093/sysbio/syw119
-
Testing historical explanations for gradients in species richness in heliconiine butterflies of tropical AmericaBiological Journal of the Linnean Society 105:479–497.https://doi.org/10.1111/j.1095-8312.2011.01814.x
-
Geographic contrasts between pre- and postzygotic barriers are consistent with reinforcement in Heliconius butterfliesEvolution; International Journal of Organic Evolution 73:1821–1838.https://doi.org/10.1111/evo.13804
-
Subtle introgression footprints at the end of the speciation continuum in a clade of Heliconius butterfliesMolecular Biology and Evolution 40:msad166.https://doi.org/10.1093/molbev/msad166
-
A new statistical method for haplotype reconstruction from population dataAmerican Journal of Human Genetics 68:978–989.https://doi.org/10.1086/319501
-
Sambamba: Fast processing of NGS alignment formatsBioinformatics 31:2032–2034.https://doi.org/10.1093/bioinformatics/btv098
-
Coalescent analysis of phylogenomic data confidently resolves the species relationships in the Anopheles gambiae species complexMolecular Biology and Evolution 35:2512–2527.https://doi.org/10.1093/molbev/msy158
-
Inferring the direction of introgression using genomic sequence dataMolecular Biology and Evolution 40:msad178.https://doi.org/10.1093/molbev/msad178
-
Adaptive radiation and convergence in subdivisions of the butterfly genus Heliconius (Lepidoptera: Nymphalidae)Zoological Journal of the Linnean Society 58:297–308.https://doi.org/10.1111/j.1096-3642.1976.tb01000.x
-
Complex modular architecture around a simple toolkit of wing pattern genesNature Ecology & Evolution 1:52.https://doi.org/10.1038/s41559-016-0052
-
Unguided species delimitation using DNA sequence data from multiple LociMolecular Biology and Evolution 31:3125–3135.https://doi.org/10.1093/molbev/msu279
-
Bayesian inference of species networks from multilocus sequence dataMolecular Biology and Evolution 35:504–517.https://doi.org/10.1093/molbev/msx307
-
Maximum likelihood implementation of an isolation-with-migration model with three species for testing speciation with gene flowMolecular Biology and Evolution 29:3131–3142.https://doi.org/10.1093/molbev/mss118
Article and author information
Author details
Funding
Biotechnology and Biological Sciences Research Council (BB/T003502/1)
- Ziheng Yang
Biotechnology and Biological Sciences Research Council (BB/X007553/1)
- Ziheng Yang
Biotechnology and Biological Sciences Research Council (BB/R01356X/1)
- Ziheng Yang
Natural Environment Research Council (NE/X002071/1)
- Ziheng Yang
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
This study was supported by Harvard University (YT, FS, and JM), and by Biotechnology and Biological Sciences Research Council grants (BB/T003502/1, BB/X007553/1 and BB/R01356X/1) and Natural Environment Research Council grant (NSFDEB-NERC NE/X002071/1) to ZY
Version history
- Preprint posted:
- Sent for peer review:
- Reviewed Preprint version 1:
- Reviewed Preprint version 2:
- Version of Record published:
Cite all versions
You can cite all versions using the DOI https://doi.org/10.7554/eLife.90656. This DOI represents all versions, and will always resolve to the latest one.
Copyright
© 2023, Thawornwattana et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 2,437
- views
-
- 256
- downloads
-
- 16
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Citations by DOI
-
- 4
- citations for umbrella DOI https://doi.org/10.7554/eLife.90656
-
- 2
- citations for Reviewed Preprint v1 https://doi.org/10.7554/eLife.90656.1
-
- 10
- citations for Version of Record https://doi.org/10.7554/eLife.90656.3






