Post-ejaculatory inhibition of female sexual drive via heterogeneous neuronal ensembles in the medial preoptic area
Figures
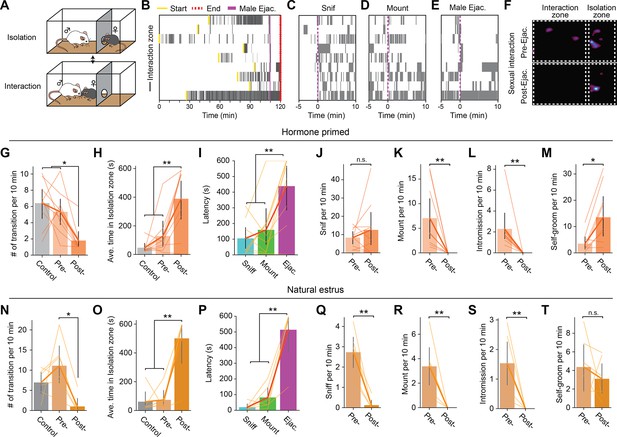
Female mice show decreased sexual motivation after male ejaculation.
(A) Schematic of female self-paced mating assay. The behavior apparatus was divided by a wall with a hole small enough for only the female subject to go through. The female subject was allowed to freely choose between ‘interaction’ with the male partner in the larger zone or ‘isolation’ in the smaller zone. Results using ovariectomized and hormone primed female mice are shown in B–M. Results using naturally estrus female mice are shown in N–T. (B) Raster plots of time spent in the interaction zone (gray). The onset of male ejaculation, start and the endpoint of the experiment is shown in yellow, blue, and red, respectively. (C–E) Raster plots of time spent in the interaction zone (gray) around the onset of the first female-to-male sniffing, male mounting, intromission and male ejaculation. (F) Representative heatmap showing the time spent in the behavior apparatus during a control trial or a sexual interaction trial. The sexual interaction trial was further divided into pre- and post- male ejaculation. (G–H) Behavior analysis for hormone primed animals. (G) Number of transitions between the zones per 10 min. RM ANOVA; F(2,16)=10.89, **p=0.001035889. Tukey’s HSD; Control vs Post, **p=0.0015. Control vs Pre, p=0.6284. Post vs Pre, *p=0.0142. (H) Average time spent in the isolation zone during the control trial, and pre- and post- male ejaculation. RM ANOVA; F(2,16)=14.34, **p=0.00027. Control vs Post, ***p=0.0004. Control vs Pre, p=0.5361. Post vs Pre, **p=0.0057. (I) Latency to return to the interaction zone after sniff, male mount or male ejaculation. RM ANOVA; F(2,16)=11.87, **p=0.00069052. Ejaculation vs Sniff, **p=0.0016. Ejaculation vs Mount, **p=0.0077. Sniff vs Mount, p=0.7951. (J) Number of female-to-male sniffing per 10 min during pre- and post- male ejaculation. p=0.65234375. (K) Number of male mounting per 10 min during pre- and post- male ejaculation. **p=0.00390625. (L) Number of intromission per 10 min during pre- and post- male ejaculation. **p=0.00390625. (M) Number of female self-grooming per 10 min during pre- and post- male ejaculation. *p=0.025061844. (N–T) Behavior analysis for naturally estrus animals. (N) Number of transitions between the zones per 10 min. RM ANOVA; F(2,10) = 7.54, *p=0.01007. Tukey’s HSD; Control vs Post, p=0.0871. Control vs Pre, p=0.2727. Post vs Pre, **p=0.0038. (O) Average time spent in the isolation zone during the control trial, and pre- and post- male ejaculation. RM ANOVA; F(2,10) = 15.71, ***p=0.00082. Control vs Post, ***p=0.0002. Control vs Pre, p=0.9862. Post vs Pre, ***p=0.0002. (P) Latency to return to the interaction zone after sniff, male mount, or male ejaculation. RM ANOVA; F(2,10) = 38.36, ***p=0.00002. Ejaculation vs Sniff, ***p=0. Ejaculation vs Mount, ***p=0. Sniff vs Mount, p=0.6201. (Q) Number of female-to-male sniffing per 10 min during pre- and post- male ejaculation. *p=0.03125. (R) Number of male mounting per 10 min during pre- and post- male ejaculation. *p=0.03125. (S) Number of intromission per 10 min during pre- and post- male ejaculation. *p=0.03125. (T) Number of female self-grooming per 10 min during pre- and post- male ejaculation. p=0.3452. All data are shown as mean ± 95% confidence interval and were analyzed by RM ANOVA with Tukey HSD post-hoc test (FWER = 0.05) (G–I and N–P) or Wilcoxon signed-rank test (J–M and Q–T). n=9 (D–K), n=6 (N–T). ∗p<0.05, ∗∗p<0.01, ∗∗∗p<0.001; ns, not significant.
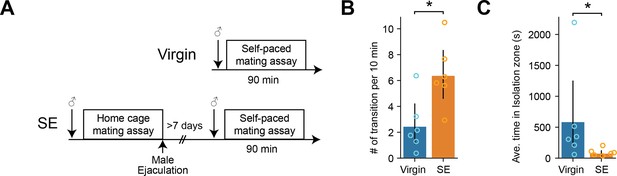
Sexual experience increase sexual motivation in female mice.
(A) Diagram showing the experiment design. Virgin animals were tested in a self-paced mating assay without any sexual experience. SE (sexually experienced) animals experienced male ejaculation in their home cage >7 d before the self-paced mating assay. (B) The number of entries in the isolation zone per 10 min between virgin animals and sexually experienced animals. *p=0.041125541. (C) The average time spent in the isolation zone between virgin animals and sexually experienced animals. *p=0.025974026. All data are shown as mean ± 95% confidence interval and were analyzed by Mann-Whitney U test (B and C). Virgin: n=6, SE: n=6. ∗p<0.05, ∗∗p<0.01, ∗∗∗p<0.001; ns, not significant.
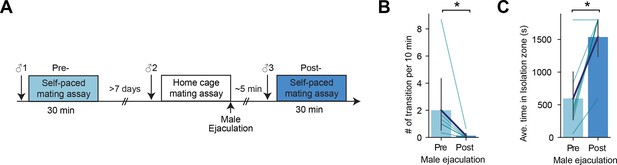
Reduction of sexual motivation after male ejaculation in female mice is not dependent on the state of the male mouse.
(A) Diagram showing the experiment design. First animals were put in a self-paced mating assay for 30 min (pre-ejaculation assay). After >7 d, the animals were put in a home cage mating assay until they experienced male ejaculation. Immediately after, they were put in a self-paced mating assay for 30 min (post-ejaculation assay). During each assay, the female mouse encountered a novel sexually active male mouse (male 1, 2, and 3). (B) The number of entries in the isolation zone per 10 min between pre- and post-ejaculation assays. *p=0.027281171. (C) The average time spent in the isolation zone between pre- and post-ejaculation assays. *p=0.027707849. All data are shown as mean ± 95% confidence interval and were analyzed by Wilcoxon signed-rank test (B and C). n=7. ∗p<0.05, ∗∗p<0.01, ∗∗∗p<0.001; ns, not significant.
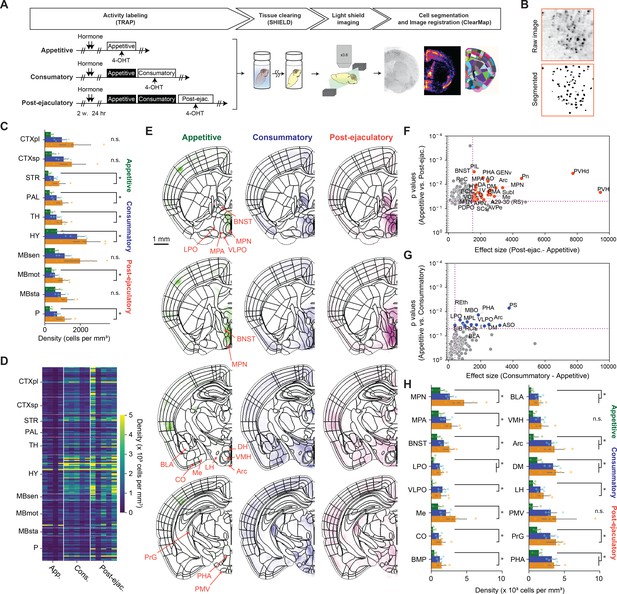
Brain-wide analysis of cells responding to male ejaculation in the female brain.
(A) Overview of the experiment pipeline. After the activity labeling using targeted recombination in active populations method (TRAP), brain tissue was collected and cleared using SHIELD. The cleared tissue was imaged using a light-sheet microscope. The dataset was registered to a reference brain atlas and the number of tdTomato + cells were quantified using ClearMap and ilastik. (B) Representative image showing cell segmentation. (C) Density of tdTomato + cells per mm3 in larger brain regions. CTXpl: Cortical plate, CTXsp: Cortical subplate, STR: Striatum, PAL: Pallidum, VERM: Vermal regions, HEM: Hemispheric regions, TH: Thalamus, HY: Hypothalamus, MBsen: Midbrain, sensory related, MBmot: Midbrain, motor-related, MBsta: Midbrain, behavioral state-related, P: Pons. (D) Heatmap showing density of tdTomato + cells per mm3 in subregions. Each column indicates data from one subject. Each row indicates data from one subregion. (E) Heatmap of average tdTomato density for each group from representative coronal planes. Scale bar, 1 mm. (F) Scatterplot showing difference of average tdTomato density and p-value after Tukey’s HSD post-hoc test between the post-ejaculatory group and the appetitive group. (G) Scatterplot showing difference of average tdTomato density and p-value after Tukey’s HSD post-hoc test between theconsummatory group and the appetitive group. (H) Density of tdTomato + cells per mm3 in a subset of subregions. The full list of brain regions is shown in Figure 1—figure supplement 2. MPN: Medial preoptic nucleus, MPA: Medial preoptic area, BNST: Bed nuclei of the stria terminalis, LPO: Lateral preoptic area, VLPO: Ventrolateral preoptic nucleus, Me: Medial amygdaloid nucleus, CO: cortical amygdaloid nucleus, BMP: Basomedial amygdaloid nucleus, posterior part, BLA: Basolateral amygdaloid nucleus, anterior part, BLP: Basolateral amygdaloid nucleus, posterior part, VMH: Ventromedial hypothalamic nucleus, Arc: Arcuate hypothalamic nucleus, DM: Dorsomedial hypothalamic nucleus, LH: Lateral hypothalamic area, PMV: Premamillary nucleus, ventral part, PrG: Pregeniculate nucleus of the prethalamus, PHA: Posterior hypothalamic area. All data are shown as mean ± 95% confidence interval and were analyzed by ANOVA test with Tukey’s HSD post-hoc test (FWER = 0.05) (C): FDR = 0.05, (I): FDR = 0.10. Detailed statistical values are shown in Supplementary file 2 and Supplementary file 3. Appetitive (green), n=4. Consummatory (blue), n=5. Post-ejaculatory (orange), n=5. ∗p<0.05, ns, not significant.

Brain-wide analysis of male ejaculation-responding cells in the female brain.
Average density heatmap of tdTomato + cells in the appetitive, consummatory and post-ejaculatory group. Appetitive, n=4. Consummatory, n=5. Post-ejaculator, n=5. Scale bar, 1 mm.
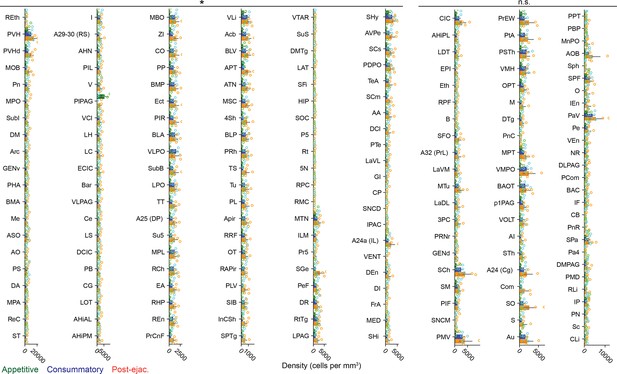
Brain-wide analysis of neurons active during post-ejaculatory period in the female brain.
Density of tdTomato + cells in each brain region from post-ejaculatory, consummatory, and appetitive group. Statistical significances are shown above each column. The regions are sorted from the top left to bottom right by the qvalue and then the difference in the density between post-ejaculatory group and appetitive group. Details of statistical values and full name of acronyms are shown in Supplementary file 3. Appetitive, n=4. Completion, n=5. Post-ejaculatory, n=5. ANOVA with Benjamini/Hochberg correction (FDR = 0.10), ∗q<0.10; ns, not significant.
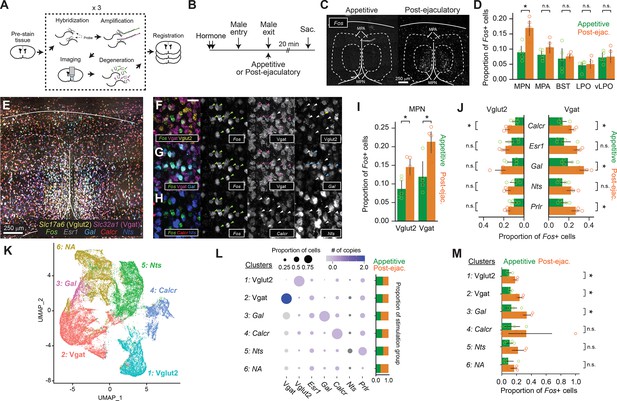
Male ejaculation responding cells are composed of a subset of excitatory and inhibitory cells in the medial preoptic area (MPOA).
(A) Brain sections went through multiplexed in situ RNA hybridization chain reaction (HCR) to identify cell type markers of MPOA and an immediate early gene Fos. Slc32a1 (Vgat), Slc17a6 (Vglut2), Esr1, Gal, Calcr, Nts, Prlr were used as cell type markers. (B) Schematic of tissue sampling for Fos expression analysis in the MPOA. Wild-type female subjects were administered with hormones before the experiment day. On the experiment day, male partners entered the female home cage. Male partners were removed immediately after ejaculation (‘post-ejaculatory’ group) or during the appetitive phase before they showed mounting (‘appetitive’ group). Brain tissue was harvested 20 min after the male exit. (C) Representative coronal section showing Fos mRNA expression in the MPOA of Appetitive and Post-ejaculatory group. Scale bar, 250 μm. (D) Proportion of Fos+ cells in each brain subregion. MPN: medial preoptic nucleus, MPA: medial preoptic area, BNST: bed nucleus of stria terminalis, LPO: lateral preoptic area, vLPO: ventral lateral preoptic area. BNST; t(8)=-0.369, p=1. LPO; t(8)=-0.3094, p=1. MPA; t(8)=-1.841, p=0.514. MPN; t(8)=-4.2709, *p=0.0136. VLPO; t(8)=-0.1751, p=1. (E) Representative coronal section showing expression of seven genes: Fos, Vgat, Vglut2, Esr1, Gal, Calcr, Nts. Scale bar, 250 μm. (F–H) Expanded image of area highlighted in yellow in (E). (F) Expression of Fos, Vgat, Vglut2. (G) Expression of Fos, Vgat and Gal. (H) Expression of Fos, Calcr, and Nts. Colored arrow indicates cells expressing given gene. Scale bar, 50 μm. (I) Proportion of Vgat or Vglut2 and Fos+ cells in the MPN of Appetitive and post-ejaculatory group. Vglut2; t(8)=-3.3885, *p=0.019. Vgat; t(8)=-3.5361, *p=0.0153. (J) Proportion of Vgat+ Fos + or Vglut2 + Fos+ and cell type marker expressing cells in the MPN of Appetitive and post-ejaculatory group. Vgat + Calcr+ ; t(8)=-3.6646, *p=0.0318. Vgat +Esr1+; t(8)=-3.1442, p=0.0686. Vgat +Gal+ ; t(8)=-4.0896, *p=0.0174. Vgat +Nts+ ; t(8)=-2.6382, p=0.149. Vgat +Prlr+ ; t(8)=-3.9487, *p=0.0212. Vglut2 +Calcr+ ; t(8)=-3.9487, *p=0.0146. Vglut2 +Esr1+; t(8)=-3.9487, p=0.8223. Vglut2 +Gal+ ; t(8)=-3.9487, p=0.1299. Vglut2 +Nts+ ; t(8)=-3.9487, p=0.1951. Vglut2 +Prlr+ ; t(8)=-3.9487, p=0.1038. (K) UMAP plot of MPN cells. (L) Disc plot showing proportion of gene expressing cell and the number of copies per cell for each gene and cluster. The proportion of cells from each group are shown on the right. Cluster Vglut2+; t(8)=-4.1631, *p=0.0189. Cluster Vgat+; t(8)=-5.9018, **p=0.0022. Cluster Gal+; t(8)=-3.9208, *p=0.0265. Cluster Calcr+; t(8)=-1.5438, p=0.9993. Cluster Nts+; t(8)=-2.1595, p=0.377. Cluster N.A.; t(8)=-1.8254, p=0.6322. (M) Proportion of Fos + cells from Appetitive and post-ejaculatory group in each cluster. All data are shown as mean ± 95% confidence interval and were analyzed by Student’s t-test with Bonferroni correction (D, E, K and N). appetitive group, n=5. post-ejaculatory group, n=5. ∗p<0.05, ∗∗p<0.01, ∗∗∗p<0.001; ns, not significant.
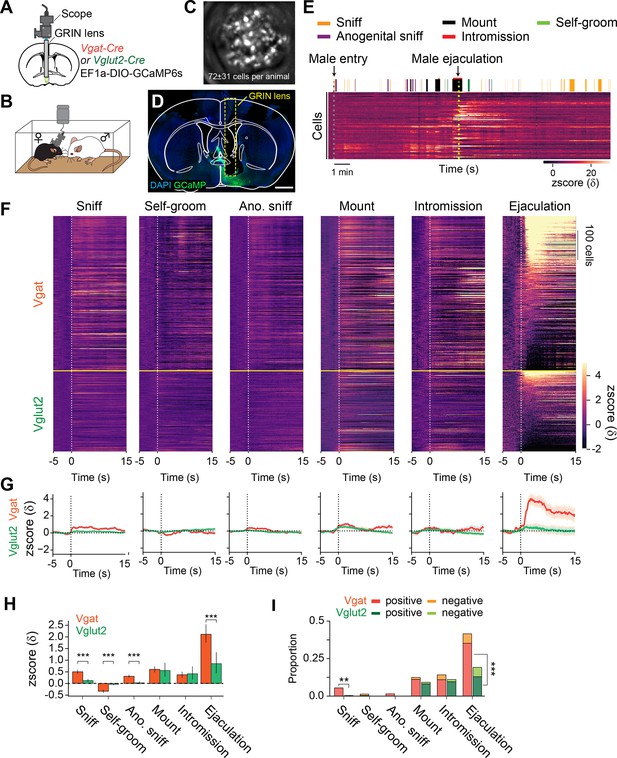
Male ejaculation signal is strongly represented in the MPOA of the female mice.
(A) Schematic of in vivo calcium imaging from medial preoptic area (MPOA) Vgat and Vglut2 neurons. AAVDj EF1a-DIO-GCaMP6s was injected into the MPOA of Vgat-Cre or Vglut2-Cre female mice. A GRIN lens was placed above the MPOA following virus injection. (B) Calcium activity was imaged from a female subject during a free moving mating assay with a sexually experienced male partner. (C) Representative maximum projected image of the field of view during calcium imaging. Vgat: 87±28 cells, n=6. Vglut2: 68±42 cells, n=4. (D) Representative coronal section showing GCaMP6s expression and GRIN lens placement. (E) Representative raster plot showing behavior events during the mating assay and a heatmap showing calcium activity from cells imaged during the assay. Behaviors quantified: female-to-male sniffing (Sniff), female self-grooming (Self-groom), male-to-female anogenital sniffing (Ano. sniff), male mounting (Mount), male intromission (Intromission) and male ejaculation (Ejaculation). (F) Heatmap showing peri-event calcium activity around behavior onset. Cells are ordered by the average response after male ejaculation. (G) Line plot showing average calcium activity around behavior onset. Vgat neurons, blue. Vglut cells, orange. (H) Average calcium response magnitude following behavior onset per cell. Vgat-Cre: n=522 cells, Vglut2-Cre: n=271 cells. Sniff: t(791)=6.46, ***p=1.08E-09. Grooming: t(791)=-7.25, ***p=5.89E-12. Male anogenital sniffing: t(791)=6.64, ***p=3.41E-10. Mount: t(791)=0.34, p=1. Intromission: t(791)=-0.32, p=1. Ejaculation: t(791)=4.00, ***p=0.00042. (I) Proportion of cells which responded positively or negatively to behavior onset (>2δ). Positive: Sniff; Vgat 29/522 cells vs. Vglut2 1/271 cells, **p=0.00169. Grooming; Vgat 2/522 cells vs. Vglut2 0/271 cells, p=1. Male Anogenital Sniffing; Vgat 9/522 cells vs. Vglut2 0/271 cells, p=0.178. Mount; Vgat 58/522 cells vs. Vglut2 22/271 cells, p=1. Intromission; Vgat 57/522 cells vs. Vglut2 26/271 cells, p=1. Ejaculation; Vgat 184/522 cells vs. Vglut2 35/271 cells, ***p=1.52E-10. Negative: Sniff; Vgat 0/522 cells vs. Vglut2 0/271 cells, no statistic. Grooming; Vgat 5/522 cells vs. Vglut2 0/271 cells, p=0.636. Male Anogenital Sniffing; Vgat 0/522 cells vs. Vglut2 0/271 cells, no statistic. Mount; Vgat 7/522 cells vs. Vglut2 3/271 cells, p=1. Intromission; Vgat 17/522 cells vs. Vglut2 4/271 cells, p=0.831. Ejaculation; Vgat 33/522 cells vs. Vglut2 17/271 cells, p=1. All data are shown as mean ± 95% confidence interval and were analyzed by Student’s t-test with Bonferroni correction (H) and chi-square test with Bonferroni correction (I). ∗p<0.05, ∗∗p<0.01, ∗∗∗p<0.001; ns, not significant. Scale bar, 1 mm.

Lens placement and field of view of in vivo imaging experiment.
(A) Schematic illustration showing position of GRIN lens placement from each animal. Positions shown on the left hemisphere are from Vglut2 animals, the ones on the right hemisphere are from Vgat animals. (B) Field of view and region of interest (ROI) for each animal. The subject ID and the number of ROI in each FOV are shown above. Vgat-Cre: n=6, Vglut2-Cre: n=4. Scale bar, 1 mm.
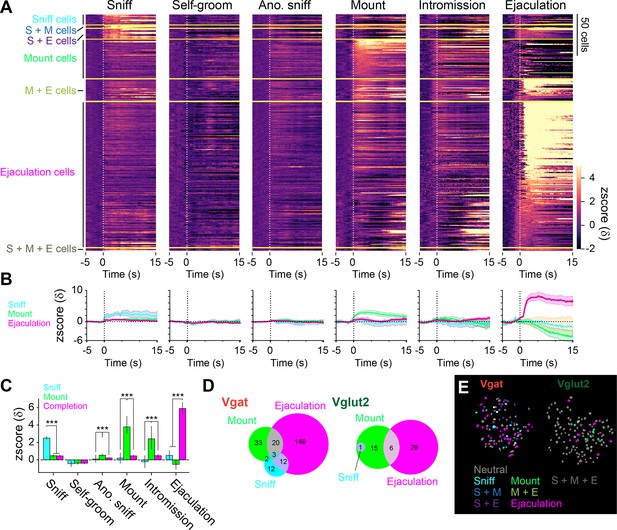
Heterogenous response properties to sexual behaviors in the medial preoptic area (MPOA).
(A) Heatmap showing peri-event calcium activity around behavior onset of cells that were categorized based on response magnitude. S: Sniff, M: Mount, E: Ejaculation. (B) Line plot showing average calcium activity around behavior onset. Sniff cells, blue. Mount cells, green. Ejaculation cells, magenta. (C) Average calcium response magnitude following behavior onset per cell in each cell types. Sniff: One-way ANOVA, F(2,235)=73.35, ***p=1.77E-25. Tukey’s HSD test, Ejaculation cells vs Mount cells, p=0.9996. Ejaculation cells vs Sniff cells, ***p=0. Mount cells vs Sniff cells, ***p=0. Grooming: One-way ANOVA, F(2,235)=0.09, p=0.915. Tukey’s HSD test, Ejaculation cells vs Mount cells, p=0.9723. Ejaculation cells vs Sniff cells, p=0.9236. Mount cells vs Sniff cells, p=0.9695. Male Anogenital Sniffing: One-way ANOVA, F(2,235)=12.34, ***p=8.01E-06. Tukey’s HSD test, Ejaculation cells vs Mount cells, ***p=0. Ejaculation cells vs Sniff cells, p=0.6794. Mount cells vs Sniff cells, **p=0.0048. Mount: One-way ANOVA, F(2,235)=68.32, ***p=4.1E-24. Tukey’s HSD test, Ejaculation cells vs Mount cells, ***p=0. Ejaculation cells vs Sniff cells, P=0.8726. Mount cells vs Sniff cells, ***p=0. Intromission: One-way ANOVA, F(2,235)=15.72, ***p=3.9E-07. Tukey’s HSD test, Ejaculation cells vs Mount cells, ***p=0. Ejaculation cells vs Sniff cells, p=0.5451. Mount cells vs Sniff cells, ***p=0.0009. Ejaculation: One-way ANOVA, F(2,235)=49.59, ***p=1.08E-18. Tukey’s HSD test, Ejaculation cells vs Mount cells, ***p=0. Ejaculation cells vs Sniff cells, ***p=0.0001. Mount cells vs Sniff cells, p=0.7295. (D) Venn diagram showing the number of cells with positive and negative response in each cell type. (>2δ). (E) Representative field of view showing spatial location of cells with different response properties to sexual behavior in Vgat (left) and Vglut2 (right) cells. All data are shown as mean ± 95% confidence interval and were analyzed by Student’s t-test with Bonferroni correction (C). Sniff cells: n=12 cells. Mount cells: n=48 cells. Ejaculation cells: n=178 cells. ∗p<0.05, ∗∗p<0.01, ∗∗∗p<0.001; ns, not significant.
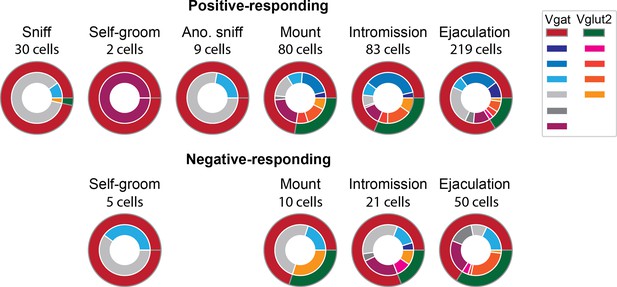
Distribution of subjects per cluster.
Cells that had significant positive or negative response (>2δ) to each behavior onset were categorized as positive- or negative-responding cells respectively. Outer ring indicates the mouse line Vgat or Vglut2. The inner ring indicates each subject. Vgat-Cre: n=6, Vglut2-Cre: n=4.
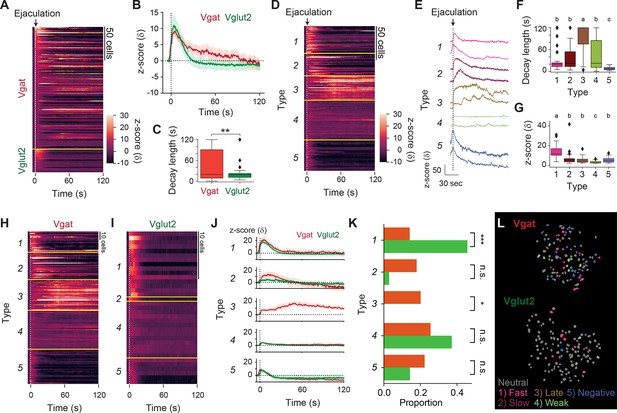
Medial preoptic area (MPOA) inhibitory neurons display prolonged activity late after the onset of male ejaculation in female mice.
(A) Heatmap showing peri-event calcium activity around male ejaculation onset for male ejaculation-responding cells in a longer time scale. (B) Line plot showing average calcium activity of Vgat and Vglut2 population around behavior onset. (C) The decay length for male ejaculation-responding cells from Vgat and Vglut2 population. Vgat-Cre: n=184 cells. Vglut2-Cre: n=35 cells. t(217)=3.039653717, **p=0.002659548. (D–G) Cluster-based analysis of the activity pattern of the male ejaculation-cells. (D) Heatmap showing peri-event calcium activity around male ejaculation onset sorted by cluster. (E) Representative calcium activity traces of cells from each cluster. (F) The decay length for male ejaculation-responding cells from each cluster. Further details of statistical tests are shown in Supplementary file 1. (G) Average z-scored response for male ejaculation-responding cells from each cluster. Further details of statistical tests are shown in Supplementary file 1. (H–K) Cluster-based analysis of the activity pattern of the male ejaculation-cells in Vgat and Vglut2 population. (H–I) Heatmap showing peri-event calcium activity of Vgat (H) and Vglut2 (I) cells around male ejaculation onset sorted by cluster. (J) Line plot showing average calcium activity of Vgat and Vglut2 cells in each cluster around behavior onset. (K) Proportion of Vgat and Vglut2 cells in each cluster over Vgat and Vglut2 cells in all clusters. Statistical results are shown next to the cluster name. Type1; Vgat 26/184 cells vs Vglut2 16/35 cells, ***p=6.80E-05. Type2; Vgat 33/184 cells vs Vglut2 1/35 cells, p=0.119810823. Type3; Vgat 37/184 cells vs Vglut2 0/35 cells, *p=0.018. Type4; Vgat 47/184 cells vs Vglut2 13/35 cells, p=0.792188347. Type5; Vgat 41/184 cells vs Vglut2 5/35 cells, p=1. (L) Representative field of view showing spatial location of cells with different response properties after male ejaculation in Vgat (left) and Vglut2 (right) cells. All data are shown as mean ± 95% confidence interval and were analyzed by Student’s t-test (C), ANOVA with post-hoc Tukey HSD test (F and G), and chi-square test with Bonferroni correction (K). Type 1: n=42 cells, Vgat-Cre: n=26 cells, Vglut2: n=16 cells. Type 2: n=34 cells, Vgat-Cre: n=33 cells, Vglut2: n=1 cells. Type 3: n=37 cells, Vgat-Cre: n=37 cells, Vglut2: n=0 cells. Type 4: n=60 cells, Vgat-Cre: n=47 cells, Vglut2: n=13 cells. Type 5: n=46 cells, Vgat-Cre: n=41 cells, Vglut2: n=5 cells. (C and K) ∗p<0.05, ∗∗p<0.01, ∗∗∗p<0.001; ns, not significant. (F and G) a vs. b: p<0.001. b vs. c: p<0.05. a vs. c: p<0.001.
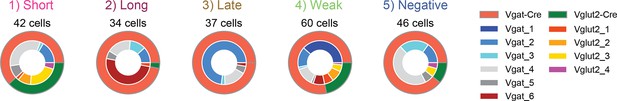
Distribution of subjects per cluster.
Cells that had significant positive or negative response (>2δ) to each behavior onset were categorized as positive- or negative-responding cells, respectively. Outer ring indicates the mouse line Vgat or Vglut2. The inner ring indicates each subject. Vgat-Cre: n=6, Vglut2-Cre: n=4.
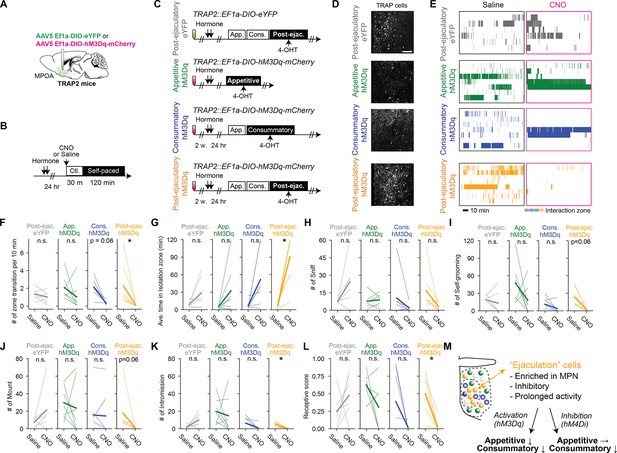
Activation of a subpopulation of medial preoptic area (MPOA) labeled following mating is sufficient to suppress female sexual behavior.
(A) Schematic of selective labeling of MPOA neurons using TRAP2. AAV5 EF1a-DIO-hM3Dq-mCherry or AAV5 EF1a-DIO-eYFP as control was injected into the MPOA of the female TRAP2 subjects. After incubation of the virus, the female subjects went through procedures to targeted recombination in active populations method (TRAP) cells in MPOA (further described in C). (B) Schematic of pharmaco-genetic activation of MPOA neurons during a female self-paced mating assay. Female subjects were primed with hormone before the experiment day. On the experiment day, subjects were injected i.p. with clozapine-N-oxide (CNO) to activate TRAP cells, or saline as control. 30 min after the injection, the male partner was placed into the apparatus and the animal behavior was recorded for 2 hr. (C) After incubation of the virus, the female subjects were administered with hormones, then received an ejaculation from the male partner or only interacted without mating. Immediately after, the subjects were administrated with 4-hydroxytamoxifen (4-OHT). (D) Representative image showing TRAP cells MPN from post-ejaculatory-eYFP, appetitive-hM3Dq and post-ejaculatory-hM3Dq group. Further histological analysis is shown in Figure 7—figure supplement 1. Scale bar, 100 μm. (E) Raster plots of time spent in the interaction zone the female self-paced mating assay (post-ejaculatory-eYFP: gray. appetitive-hM3Dq: green, consummatory-hM3Dq: blue, post-ejaculatory-hM3Dq: orange). (F) Number of zone transitions per 10 min. Post-ejaculatory-hM3Dq; *p=0.03125. Post-ejaculatory-eYFP; p=1. Appetitive-hM3Dq; p=0.875. Consummatory-hM3Dq; p=0.0625. (G) Average time in isolation zone. Post-ejaculatory-hM3Dq; *p=0.03125. Post-ejaculatory-eYFP; p=1. Appetitive-hM3Dq; p=0.875. Consummatory-hM3Dq; p=0.125. (H) Number of female-to-male sniff. Post-ejaculatory-hM3Dq; p=0.11197. Post-ejaculatory-eYFP; p=0.375. Appetitive-hM3Dq; p=1. Consummatory-hM3Dq; p=0.4594. (I) Number of female self-grooming. Post-ejaculatory-hM3Dq; p=0.0625. Post-ejaculatory-eYFP; p=1. Appetitive-hM3Dq; p=1. Consummatory-hM3Dq; p=0.552. (J) Number of male mount. Post-ejaculatory-hM3Dq; p=0.0625. Post-ejaculatory-eYFP; p=0.875. Appetitive-hM3Dq; p=1. Consummatory-hM3Dq; p=1. (K) Number of male intromission. Post-ejaculatory-hM3Dq; *p=0.03125. Post-ejaculatory-eYFP; p=0.625. Appetitive-hM3Dq; p=1. Consummatory-hM3Dq; p=0.2716. (L) Receptive score (number of mount episodes with intromissions/number of all mount episodes). Post-ejaculatory-hM3Dq; p=0.03125. Post-ejaculatory-eYFP; p=1. Appetitive-hM3Dq; p=0.625. Consummatory-hM3Dq; p=0.2716. (M) Schematic illustration summarizing how MPOA neurons suppress female sexual motivation after male ejaculation. All data are shown by thin lines for individual subjects and thick lines for mean. The data were analyzed by Wilcoxon rank sum test with Bonferroni correction (F–L). post-ejaculatory-eYFP: n=6, appetitive-hM3Dq: n=7, consummatory-hM3Dq: n=8. post-ejaculatory-hM3Dq: n=8. ∗p<0.05, ∗∗p<0.01, ∗∗∗p<0.001; ns, not significant.
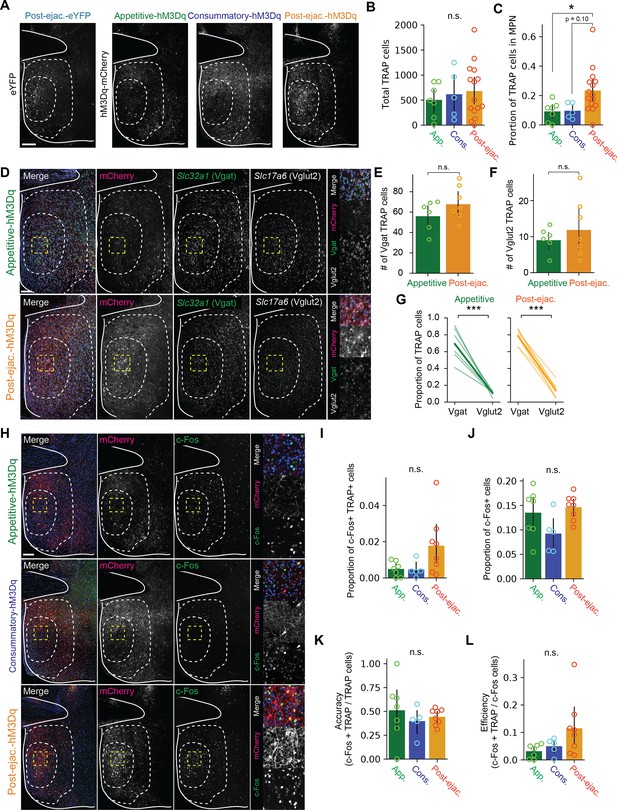
Histological analysis of targeted recombination in active populations method (TRAP) labeling.
(A) Histological analysis of the distribution of TRAP- labeled cells. Representative coronal section showing eYFP or hM3Dq-mCherry expression in the MPOA. Scale bar, 100 μm. (B–C) Data from post-ejaculatory-eYFP and post-ejaculatory-hM3Dq were grouped into post-ejaculatory group. Appetitive group, n=7. Consummatory group, n=5. post-ejaculatory group, n=14. (B) Number of TRAP labeled cells. One-way ANOVA, F(2,24)=0.3072, p=0.7383. (C) The proportion of TRAP labeled cells in the MPN. One-way ANOVA, F(2,24)=4.3335, *p=0.0247. Tukey HSD; Appetitive vs Consummatory, p=0.9975. Appetitive vs Post-ejaculatory, *p=0.0471. Consummatory vs Post-ejaculatory, p=0.0984. (D) In situ RNA hybridization analysis of the excitatory and inhibitory marker gene expression in the MPN. appetitive-hM3Dq and post-ejaculatory-hM3Dq groups were used for analysis. Representative coronal section showing mCherry, Slc32a1 (Vgat) and Slc17a6 (Vglut2) expression in the MPOA. Enlarged image indicated by the yellow box, is shown on the right. Scale bar, 100 μm. (E–G) Number of Vgat TRAP labeled cells in the medial preoptic area nucleus (MPN) (E) t(11)=-1.3299, p=0.2105, number of Vglut2 TRAP labeled cells in the MPN (F), t(11)=-0.7917, p=0.4453 and the proportion of TRAP labeled cells that express Vgat or Vglut2 (G) Appetitive, t(10)=6.6154, **p=0.0024. Post-ejaculatory; t(12)=13.9477, ***p=1.69E-05. Appetitive group, n=6. Post-ejaculatory group, n=7. (H) Antibody analysis of c-Fos expression after reactivation of male ejaculation ensemble. Representative coronal section showing mCherry and c-Fos expression in the MPOA. Enlarged image indicated by the yellow box, is shown on the right. Scale bar, 100 μm. (I–L) proportion of c-Fos + and TRAP labeled cells in the MPN (I) One-way ANOVA, F(2,17) = 3.2569, p=0.0635. Tukey HSD; Appetitive vs Consummatory, p=0.9991. Appetitive vs Post-ejaculatory, p=0.0942. Consummatory vs Post-ejaculatory, p=0.1268, proportion of c-Fos + cells in the MPN (J) One-way ANOVA, F(2,17)=3.1285, p=0.0697. Tukey HSD; Appetitive vs Consummatory, p=0.1724. Appetitive vs Post-ejaculatory, p=0.8445. Consummatory vs Post-ejaculatory, p=0.0631, the accuracy score (proportion of c-Fos +TRAP labeled cells among all TRAP labeled cells) (K) One-way ANOVA, F(2,17)=0.4666, p=0.6349 and the efficiency score (proportion of c-Fos +TRAP labeled cells among all c-Fos + cells) (L) One-way ANOVA, F(2,17)=2.8313, p=0.0868. Tukey HSD; Appetitive vs Consummatory, p=0.9028. Appetitive vs Post-ejaculatory, p=0.0882. Consummatory vs Post-ejaculatory, p=0.2666. Appetitive group, n=6. Consummatory group, n=5. Post-ejaculatory group, n=8. All data are shown by thin lines for individual subjects and thick lines for mean. The data were analyzed by ANOVA test with Tukey’s HSD post-hoc test (FWER = 0.05) (B, C, I, J, K, L) Student’s t-test (E, F) or Student’s relative t-test (H). ∗p<0.05, ∗∗p<0.01, ∗∗∗p<0.001; ns, not significant.
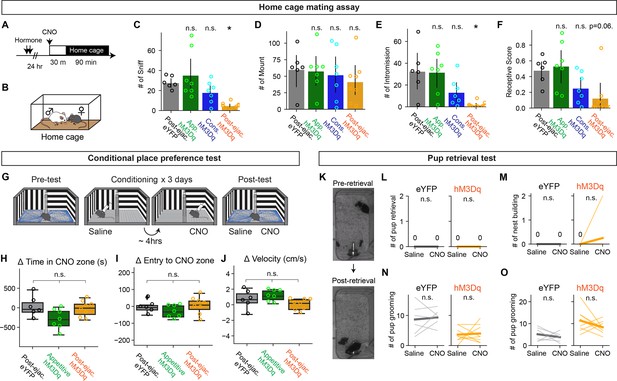
MPOApost-ejaculatory cells supress female sexual behavior in home cage mating assay but not other behaviors.
(A) Schematic of pharmaco-genetic activation of medial preoptic area (MPOA) neurons during a home cage mating assay. Female subjects were primed with hormone before the experiment day. On the experiment day, subjects were injected i.p. with clozapine-N-oxide (CNO) to activate targeted recombination in active populations method (TRAP) cells 30 min before the experiment. Animal behavior was recorded for 90 min. (B) The male partner was placed into the home cage of the female subject. (C–F) Number of female-to-male sniff (C) Post-ejaculatory-eYFP vs Appetitive-hM3Dq, p=1. vs Consummatory-hM3Dq, p=0.2581. vs Post-ejaculatory-hM3Dq, **p=0.0072, male mounting (D) Post-ejaculatory-eYFP vs Appetitive-hM3Dq, p=1. Post-ejaculatory-eYFP vs Consummatory-hM3Dq, p=1. Post-ejaculatory-eYFP vs Post-ejaculatory-hM3Dq, p=0.4116, male intromission (E) Post-ejaculatory-eYFP vs Appetitive-hM3Dq, p=1. Post-ejaculatory-eYFP vs Consummatory-hM3Dq, p=0.3042. Post-ejaculatory-eYFP vs Post-ejaculatory-hM3Dq, *p=0.0165 and the receptive score (number of mounting episodes with intromissions over all mounting episodes) (F), Post-ejaculatory-eYFP vs Appetitive-hM3Dq, p=1. Post-ejaculatory-eYFP vs Consummatory-hM3Dq, p=0.1888. Post-ejaculatory-eYFP vs Post-ejaculatory-hM3Dq, p=0.0626. (G) Schematic of conditional place preference (CPP) assay to evaluate the valence of activation of MPOA neurons. A two-chamber behavior apparatus, one room with vertical stripes and one room with horizontal stripes. On the first day, animals were placed in the behavior apparatus to test their initial preference to each room for 30 min (Pre-test). On the conditioning days, the animals were placed in one side of the room where they were i.p. injected with either saline as control or CNO. CNO was injected 4 hr after the saline injection. On the last day, the animals were placed in the behavior apparatus to test their preference 30 min (Post-test). (H–J) The difference in the time spent in the CNO zone (Time spent in the CNO zone in Post-test – Pre-test) (H) Post-ejaculatory-eYFP vs Appetitive-hM3Dq, p=0.2203. Post-ejaculatory-eYFP vs Post-ejaculatory-hM3Dq, p=1. Appetitive-hM3Dq vs Post-ejaculatory-hM3Dq, p=1, the difference in the number of CNO zone entries (Entries in Post-test – Pre-test) (I) Post-ejaculatory-eYFP vs Appetitive-hM3Dq, p=1. Post-ejaculatory-eYFP vs Post-ejaculatory-hM3Dq, p=1. Appetitive-hM3Dq vs Post-ejaculatory-hM3Dq, p=1 and the difference in velocity of the animal (Velocity in Post-test–Pre-test) (J) Post-ejaculatory-eYFP vs Appetitive-hM3Dq, p=1. Post-ejaculatory-eYFP vs Post-ejaculatory-hM3Dq, p=0.8472. Appetitive-hM3Dq vs Post-ejaculatory-hM3Dq, p=0.8472. (K) Representative image of pup retrieval test. Female subject was i.p. injected with saline as control or CNO in their home cage. 30 min after injection, 3x-7w-old pup was placed in the corner of the home cage. Animal behavior was examined for 10 min. (L–O) The number of pup retrievals (L) eYFP, P=1, hM3Dq, p=1, the number of nest building (M) eYFP, p=1, hM3Dq, p=0.635, the number of pup grooming (N) eYFP, p=1, hM3Dq, p=1 and the number of pup sniffing (O) eYFP, p=1, hM3Dq, p=0.391. All data are shown by thin lines for individual subjects and thick lines for mean. Appetitive-hM3Dq, consummatory-hM3Dq and post-ejaculatory-hM3Dq group were tested against post-ejaculatory-eYFP group by Mann–Whitney U test with Bonferroni correction (C–F, H–J) and Wilcoxon rank sum test (L–O) with Bonferroni correction. post-ejaculatory-eYFP: n=6, appetitive-hM3Dq: n=7, consummatory-hM3Dq: n=7. post-ejaculatory-hM3Dq: n=8. ∗p<0.05, ∗∗p<0.01, ∗∗∗p<0.001; ns, not significant.
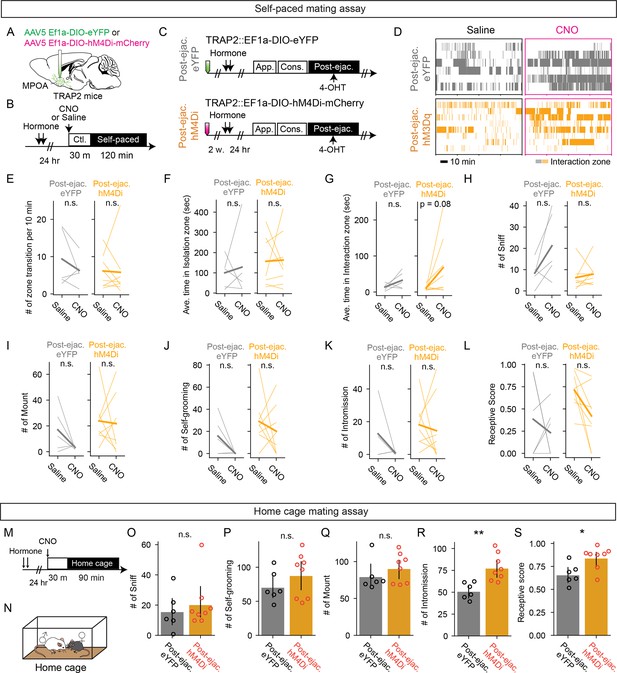
MPOApost-ejaculatory cells is not necessary for sexual motivation but for sexual receptivitiy.
(A) Schematic of selective labeling of medial preoptic area (MPOA) neurons using TRAP2. AAV5 EF1a-DIO-hM4Di-mCherry or AAV5 EF1a-DIO-eYFP as control was injected into the MPOA of the female TRAP2 subjects. After incubation of the virus, the female subjects went through procedures to targeted recombination in active populations method (TRAP) cells in MPOA (further described in C). (B) Schematic of pharmaco-genetic inhibition of MPOA neurons during a female self-paced mating assay. Female subjects were primed with hormone before the experiment day. On the experiment day, subjects were injected i.p. with clozapine-N-oxide (CNO) to inhibit TRAP cells, or saline as control. 30 min after the injection, the male partner was placed into the apparatus and the animal behavior was recorded for 2 hr. (C) After incubation of the virus, the female subjects were administered with hormones, then received an ejaculation from the male partner. Immediately after, the subjects were administrated with 4-hydroxytamoxifen (4-OHT). (D) Raster plots of time spent in the interaction zone the female self-paced mating assay (Post-ejaculatory-eYFP: gray. Post-ejaculatory-hM3Dq: orange). (E–L) Number of zone transitions per 10 min (E), eYFP, p=0.4375. hM4Di, p=1, average time in isolation zone (F), eYFP, p=1. hM4Di, p=1, average time in interaction zone (G), eYFP, p=0.3125. hM4Di, p=0.15625, number of female-to-male sniff (H), eYFP, p=0.1592. hM4Di, p=0.9917, female self-grooming (I), eYFP, p=0.2883. hM4Di, p=1, male mount (J), eYFP, p=0.4456. hM4Di, p=1, male intromission (K), eYFP, p=0.4456. hM4Di, p=1. and the receptive score (number of mount episodes with intromissions/number of all mount episodes) (L), eYFP, P=1. hM4Di, p=0.1094. (M) Schematic of pharmaco-genetic inhibition of MPOA neurons during a home cage mating assay. Female subjects were primed with hormone before the experiment day. On the experiment day, subjects were injected i.p. with CNO to activate TRAP cells 30 min before the experiment. Animal behavior was recorded for 90 min. (N) The male mating partner was placed into the home cage of the female subject. (O–R) Number of female-to-male sniff (O), p=0.6969, male mounting (P), p=0.4003, male intromission (Q) **p=0.0045 and the receptive score (number of mounting episodes with intromissions over all mounting episodes) (R) *p=0.01998. All data are shown by thin lines for individual subjects and thick lines for mean. The data were analyzed by Wilcoxon rank sum test (E–L) with Bonferroni correction. Post-ejaculatory-eYFP: n=6, post-ejaculatory -hM4Di: n=8, or by Mann-Whitney U test (O–R). Post-ejaculatory -eYFP: n=6, post-ejaculatory -hM4Di: n=8 ∗p<0.05, ∗∗p<0.01, ∗∗∗p<0.001; ns, not significant.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mus musculus) | Mus musculus with name C57BL/6 J | https://www.jax.org/strain/000664 | RRID:IMSR_JAX:000664 | |
| Strain, strain background (Mus musculus) | Mus musculus with name Slc32a1tm2(cre)Lowl (vgat-cre) | https://www.jax.org/strain/016962 | RRID:IMSR_JAX:016962 | |
| Strain, strain background (Mus musculus) | Mus musculus with name Slc17a6tm2(cre)Lowl (vglut2-cre) | https://www.jax.org/strain/016963 | RRID:IMSR_JAX:016963 | |
| Strain, strain background (Mus musculus) | Mus musculus with name Fostm2.1(icre/ERT2)Luo/J (Fos2a-cre or TRAP2) | https://www.jax.org/strain/030323 | RRID:IMSR_JAX:030323 | |
| Strain, strain background (AAV5) | AAV5 hSyn-DIO-hM4D(Gi)-mCherry | https://www.addgene.org/44362/ | Catalog #: 44362-AAV5 | lot #: v178820 |
| Strain, strain background (AAV5) | AAV5 hSyn-DIO-hM3D(Gq)-mCherry | https://www.addgene.org/44361/ | Catalog #: 44361-AAV5 | lot #: v141469 |
| Strain, strain background (AAV5) | AAV5-Ef1a-DIO-eYFP | UNC Vector Core | lot #: av4802B | |
| Strain, strain background (AAVDj) | AAVDj EF1a-DIO-GCaMP6s | UNC Vector Core | lot#: av78310 | |
| Software, algorithm | Python 3.7 (Anaconda Distribution) | https://www.anaconda.com/ | ||
| Software, algorithm | R 4.0.4 | https://cran.r-project.org/ | ||
| Software, algorithm | Seurat3.1.1 | https://github.com/satijalab/seurat/releases/tag/v3.1.1; Hao et al., 2024; satijalab, 2019 | ||
| Software, algorithm | DeepLabCut 2.2 | https://github.com/DeepLabCut/DeepLabCut; Nath et al., 2019; Lauer et al., 2025 | ||
| Software, algorithm | SimBA 1.31 | https://github.com/sgoldenlab/simba; Goodwin et al., 2024; Nilsson and sgoldenlab, 2025 | ||
| Chemical compound, drug | Easyindex | LifeCanvas Technologies | ||
| Chemical compound, drug | SHIELD Kit | LifeCanvas Technologies | ||
| Chemical compound, drug | Delipidation Buffer | LifeCanvas Technologies | ||
| Chemical compound, drug | Conduction Buffer | LifeCanvas Technologies |
Additional files
-
Supplementary file 1
All results from statistical analysis, except for ones related to Figure 2, are included in Supplementary file 1.
- https://cdn.elifesciences.org/articles/91765/elife-91765-supp1-v1.xlsx
-
Supplementary file 2
Related to Figure 2C.
One-way ANOVA test with Benjamini/Hochberg correction (FDR = 0.05).
- https://cdn.elifesciences.org/articles/91765/elife-91765-supp2-v1.xlsx
-
Supplementary file 3
Related to Figure 2I, Figure 2—figure supplement 2.
One-way ANOVA test with Benjamini/Hochberg correction (FDR = 0.10) followed by post-hoc Tukey’s HSD test (FWER = 0.05).
- https://cdn.elifesciences.org/articles/91765/elife-91765-supp3-v1.xlsx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/91765/elife-91765-mdarchecklist1-v1.docx






