The embryonic role of juvenile hormone in the firebrat, Thermobia domestica, reveals its function before its involvement in metamorphosis
Figures
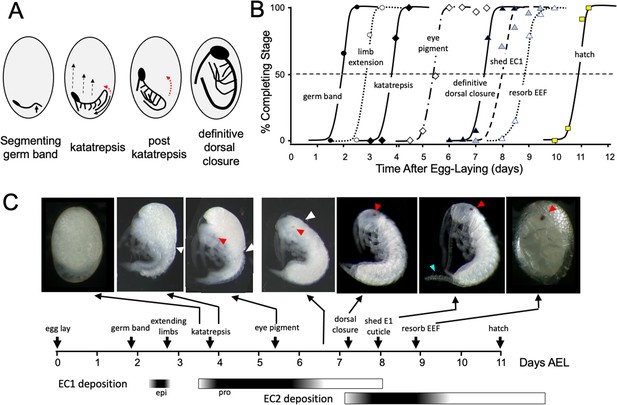
Timeline of Thermobia domestica embryogenesis at 37 °C.
(A) Diagrammatic representations of important events in embryogenesis. In the segmenting germ band, the arrow indicates the invagination of the mid-abdominal region into the yolk. By the onset of katatrepsis (Panfilio, 2008), dorsal closure along the abdomen (red arrow) has moved the abdomen ventrally (solid black arrow) and the contraction of the serosa pulls the sides of the embryo towards the anterior pole (dashed black arrows). After katatrepsis the expanding lateral edges of the embryo displace cells of the amnion, gradually zipping up the dorsal thoracic midline (red arrow) until definitive dorsal closure is accomplished. (B) Summary of the progression of embryonic development at various times after egg deposition. The interception of each stage with the 50% line (dashed) is the basis for the embryonic timeline in (C). EEF: extraembryonic fluid. (C) Timeline of embryonic development based on (B). Micrographs show the appearance of embryos at the indicated times; first and last embryos are covered by the egg chorion. White triangle: progression of dorsal closure; red triangle: eye pigmentation; blue triangle: expanded cerci after shedding of the E1 cuticle. Times of cuticle deposition based on Konopová and Zrzavý, 2005.
-
Figure 1—source data 1
The percent of embryos attaining a particular developmental ‘milestone’ as a function of the time after egg laying at 37 °C.
Age of each sample calculated from the midpoint of its 12 hr egg collection.
- https://cdn.elifesciences.org/articles/92643/elife-92643-fig1-data1-v1.docx
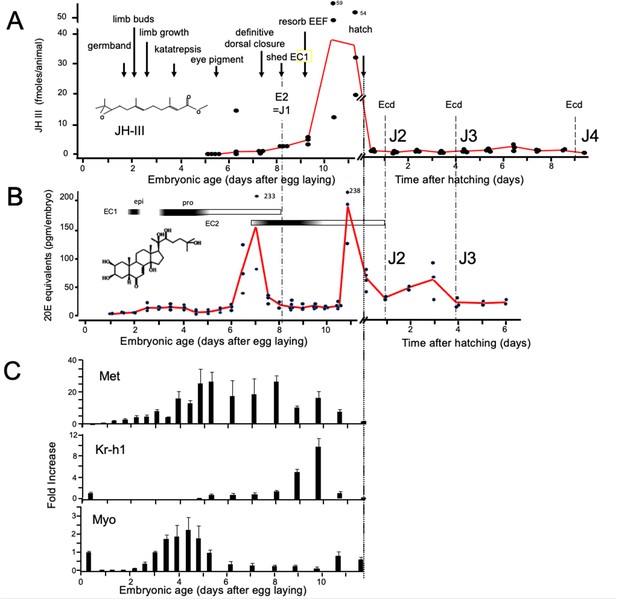
Titers of hormones and hormone related gene transcripts during embryogenesis of Thermobia domestica.
(A) Titer of juvenile hormone III (JH-III) during the last 60% of embryogenesis and the first eight days of juvenile life. The timing of various milestones of embryonic development are noted for this panel and those below. ecd: ecdysis; J#: start of second, third and fourth juvenile instars. (B) The ecdysteroid titer in 20-hydroxyecdysone (20E) equivalents during embryogenesis and the first six days of juvenile life. Dark bars indicate the approximate time of deposition of the first embryonic (EC1) epicuticle and procuticle, and the second (EC2) embryonic cuticle (based on Konopová and Zrzavý, 2005). epi: epicuticle deposition, pro: procuticle deposition. (C) The relative levels of transcripts of the JH receptor, Methoprene-tolerant (Met), the JH response gene Krüppel homolog 1 (Kr-h1), and the TGF-β family member Myoglianin (Myo) based on real-time PCR of timed embryos. Expression of each gene is related to the 12 hr timepoint which is given the value of 1. Each bar shows the mean (+/-S.D.) for three biological replicats.
-
Figure 2—source data 1
Age of samples extracted for Juvenile Hormone III measurements.
Age of samples from midpoint of 12 hr egg collections. E: embryonic age; J: age of juvenile; n.d.: below range of detectability.
- https://cdn.elifesciences.org/articles/92643/elife-92643-fig2-data1-v1.docx
-
Figure 2—source data 2
Age of samples extracted for ecdysteroid measurements (given as 20 hydroxyecdysone equivalents).
Age of samples from midpoint of 12 hr egg collections. Hatching occurred at 11.5 days after egg laying (AEL).
- https://cdn.elifesciences.org/articles/92643/elife-92643-fig2-data2-v1.docx
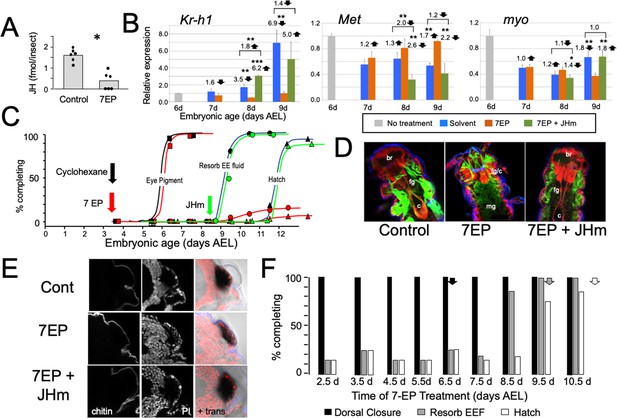
The effects of suppression of JH production on the embryonic development of Thermobia.
(A) Compared with solvent alone (Control), treatment of stage 2 juveniles with 7-ethoxyprecocene (7EP) resulted in a >75% reduction in JH-III levels as measured on the day after application. (B) The effect treating embryos at 6 d AEL with the cyclohexane solvent (blue) or 1 μg 7EP (orange) on their subsequent expression of Kr-h1, Met, and myo over the next 3 days as revealed by real-time PCR. Sub-groups of 7EP treated embryos were treated with 1 ng JHm at 7 d and measured over the next 2 days (green). Significant differences determined by t-test: *=p < 0.05; **=p < 0.01; ***=p < 0.001. Numbers above columns indicate fold upregulation (upward arrow) or downregulation (downward arrow) of the two hormone treatments relative to cyclohexane control, or to the effect of JHm rescue to 7EP treated embryos. Each based on three biological replicates. (C) A group of 20 embryos were treated with cyclohexane (black) or 1 μg 7EP (red) and their subsequent development monitored until hatching. Both groups started eye pigmentation at the same time, but resorption of the extraembryonic fluid and hatching were suppressed in the 7EP group. The latter two events were restored by treating embryos with the 1 ng JHm at 8.5 d AEL (green). (D) Confocal sections of the dorsal view of embryos treated as in ‘C’ and examined at the time of hatching of the controls. 7EP treatment prevented extension of the foregut (fg) and crop (c) and the posterior displacement of the midgut (mg). Normal gut development was restored with JHm treatment at 8.5d. br: brain; muscle (green); propidium iodide staining (red). (E) Pseudo-transmitted light and confocal sections showing cuticle and nuclei of the eye region of 10 d embryos that had been treated with cyclohexane (control) or 7EP at 5 d AEL. A subset of the latter was then given JHm on day 7.5. Control and JHm-treated embryos show local apolysis of the eye cuticle and expansion of the depth of the eye due to growth of the rhabdoms; embryos treated with 7EP alone failed to show this growth. PI: propidium iodide stain,+trans: pseudo transmitted light. (F) The relationship of the final phenotypes of embryos to the time of their treatment with 1 μg of 7EP. Arrows indicate the normal timing of the event. EEF: extraembryonic fluid.
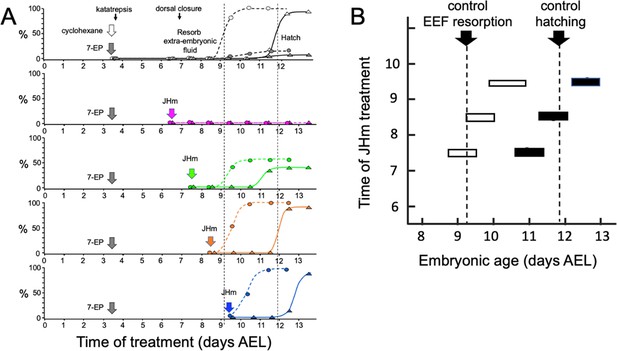
Time-course of the rescue of 7EP-imposed developmental arrest by treatment with JHm.
(A) Groups of about 20 embryos were treated with cyclohexane (controls) or 1 µg 7EP at d3.5 AEL and then monitored daily for the time of reabsorption of the extraembryonic fluid and hatching. The vertical dashed lines indicate the 50% time for these two developmental events in the control group. Replicate groups were also given JHm (1 ng pyriproxyfen) at the indicated times and their development followed to hatching. (B) Summary of the timing of resorption of the extraembryonic fluid (EEF, white bars) and of hatching (black bars) for 7EP-treated embryos, whose development was rescued by JHm treatment at the indicated times.
-
Figure 4—source data 1
The progression of embryonic development of embryos treated with solvent or 7-ethoxyprecocene (7EP) at 3.5 days of development.
Developmental rescue was attempted by subsequent treatment with a juvenile hormone mimic (pyriproxyfen) at the indicated developmental time thereafter. air: air appears between embryo and eggshell because of resorption of extraembryonic fluid; eye: appearance of eye pigment. Percent values are based on surviving embryos. *: these embryos blocked before dorsal closure because of the early timing of treatment with pyriproxyfen.
- https://cdn.elifesciences.org/articles/92643/elife-92643-fig4-data1-v1.docx
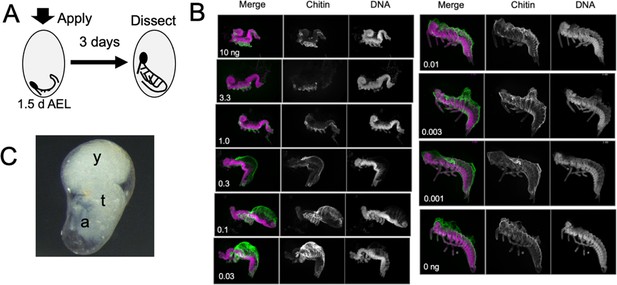
The response of Thermobia embryos to various doses of the JH mimic pyriproxyfen.
(A) Cartoon showing the time of JHm treatment versus the time of dissection. (B) Lateral projections of confocal stacks showing the appearance of typical embryos three days after treatment with the indicated dosage of pyriproxyfen. Embryos were stained for DNA (magenta) and chitin (green). The embryos were dissected away from the yolk which caused disruption in the dorsal thoracic and head regions of some embryos. (C) Micrograph of an embryo arrested in mid-katatrepsis. During its envelopment of the yolk, the contraction of the amnion segregated the yolk (y) from the embryo which hangs in the ventral half of the egg. t: thorax, a: abdomen.
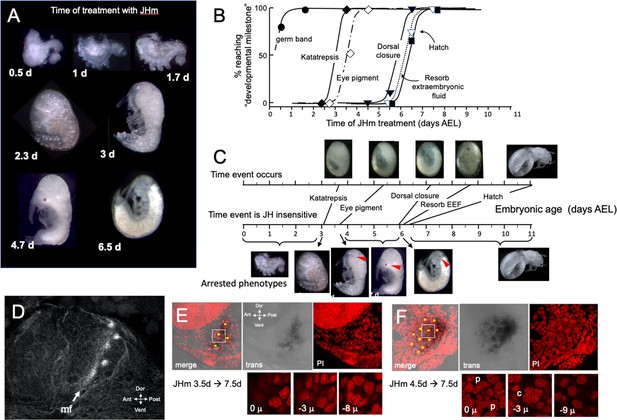
The response of Thermobia embryos to 1 ng JHm given at different times through embryogenesis.
(A) Examples of the terminal phenotypes of embryos resulting from JHm treatment at the indicated times [days after egg laying (AEL)]. Yolk was dissected away from the first three embryos. The remaining embryos had undergone provisional dorsal closure with the amnion enclosing the yolk mass. (B) Graph showing when each developmental ‘milestone’ was no longer suppressed by JHm treatment. (C) Comparison of when developmental milestones were no longer suppressed by JHm (based on Figure 1B) versus their normal time of occurrence. Red triangle in micrograph shows first appearance of eye pigment. (D) Confocal image of actin staining showing the state of patterning of the eye primordium at katatrepsis. The morphogenetic furrow (mf) had just started and had organized only the first few posterior ommatidia. (E, F). Lateral confocal and pseudo transmitted light images of the eye region of 7.5 d embryos that had been treated with JHm at (E) 3.5 d or (F) 4.5 d AEL. In (E) eye patterning arrested early resulting in only 3–4 proto-ommatidial clusters forming in the posterior margin of the eye primordium. In (F) the entire eye primordium was patterned. Yellow dots identify the center of each forming ommatidium. The insets below show various Z depths of the boxed cluster in the merged image, with ‘0 μ’ being at the surface. Nuclei of identifiable ommatidial cell types can only be recognized in (F). c: quartet of crystalline cone cells, p: crescent-shaped nuclei of the paired cells that secrete the cuticular lens.
-
Figure 6—source data 1
The relationship of the time of treatment with a JH mimic (1 ng pyriproxyfen) to when embryonic development subsequently stalled.
EEF: extraembryonic fluid; pre-dorsal closure was evident by dorsal closure to the neck region, but the posterior head capsule had not fully formed.
- https://cdn.elifesciences.org/articles/92643/elife-92643-fig6-data1-v1.docx
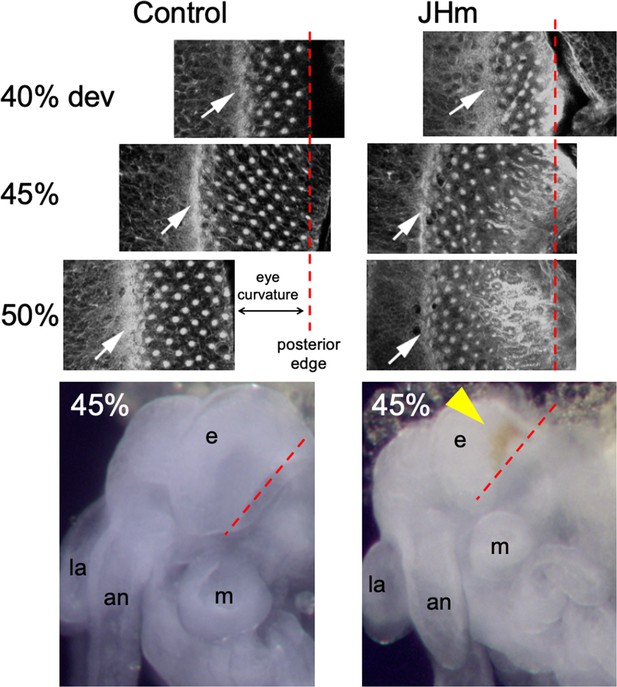
Confocal images showing the effects of treatment of day 1 Schistocerca gregaria embryos with a JHm (pyriproxyfen) versus cyclohexane (control).
Confocal images show eye primordia of embryos stained with fluorescent phalloidin at various times in development showing the progression of ommatidia formation in the wake of the morphogenetic furrow (white arrow). By 50% of embryogenesis the furrow and posterior margin are no longer in the same plane of focus because of the pronounced curvature of the eye; the image has been shifted to accommodate the rows of posterior ommatidia missing from the image. The morphogenetic furrow arrests by 45% in the JHm treated embryos and the posterior rows of ommatidia show autofluorescence due to the premature appearance of screening pigment around each ommatidium. Bottom: at 45% of embryogenesis, premature pigment expression (yellow triangle) is evident in the eye of the JHm treated embryo but not in the control. an: antenna, e: eye, la: labrum, m: mandible. Red dashed line is the posterior border of the developing eye.
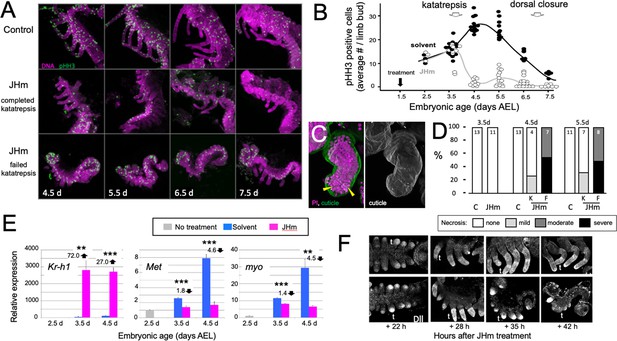
Effects of early JHm treatment on growth and patterning of the limbs.
(A). Z-stack projections showing the lateral view of embryos that were treated with either cyclohexane (Control) or 1 ng of pyriproxyfen in cyclohexane (JHm) on d1.5 AEL and then dissected and stained on the indicated developmental days. Propidium iodide (PI) (magenta) shows nuclei and anti-phosphohistone H3 (pHH3; green) shows dividing cells. The JHm series follows a subset of embryos that did not undergo katatrepsis (bottom) and a subset that did (middle). (B). Summary of the number of pHH3-positive cells in the developing limb buds of JHm and solvent-treated embryos through time. Each dot records the average number of pHH3-positive cells /limb from a single embryo. (C). Projected Z-stack through the limb of a JHm-treated embryos that did not undergo katatrepsis. The leg produced a robust cuticle (green; gray scale image) but then started necrosis as illustrated by the highly condensed PI-positive bodies (yellow triangles) in the distal leg. (D). The time course of necrosis in the limbs of control and JHm-treated embryos. Necrosis was pronounced in embryos that failed katatrepsis (F) but rather mild in those that completed katatrepsis (K). (E). Levels of hormone-related transcripts in embryos treated at 2.5 d AEL with solvent alone (blue bars) or JHm (1 ng pyriproxyfen; pink) and then examined over the following two days. The expression is related to that on the day of treatment (2.5 d; grey) which was set as 1. Kruppel homolog 1 (Kr-h1), Methoprene tolerant (Met), myoglianin (myo). Significant differences determined by t-test: *=p < 0.05; **=p < 0.01; ***=p < 0.001. Numbers above columns indicate fold upregulation (upward arrow) or downregulation (downward arrow) relative to cyclohexane control. Each based on three biological replicates. (F). Confocal images showing Distal-less (Dll) immunostaining at various times after treatment of embryos at 2 d AEL with solvent (C) or JHm. Embryos are shown from ventral view at 22 hr post-treatment, and then from lateral view thereafter. t: first thoracic leg.
-
Figure 7—source data 1
The effects of early treatment with a JH mimic (1 ng pyriproxyfen) on the subsequent proliferative activity in the embryonic limb buds indicated by the number of limb cells expressing phosphohistone H3 (pPH3).
N is the number of embryos scored for each treatment.
- https://cdn.elifesciences.org/articles/92643/elife-92643-fig7-data1-v1.docx
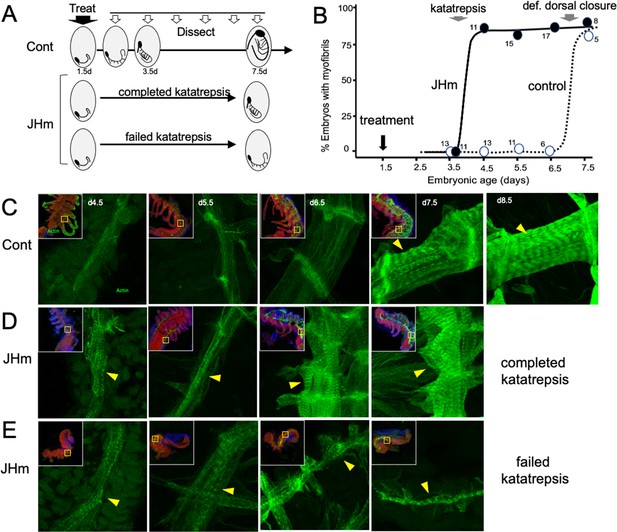
The effects of JHm in inducing early differentiation of Thermobia embryos.
(A) Schematic showing the time of treatment with solvent or 1 ng pyriproxyfen (JHm) at 1.5 d AEL and the subsequent times of dissection and staining of the embryos. (B) Quantitation of the effects of JHm treatment on the appearance of striated myofibrils in the developing embryonic muscles. Myofibrils normally appear around the time of definitive dorsal closure, but JHm treatment at 1.5 d AEL advances their appearance by three days. Numbers are embryos examined per point. (C–E). Confocal optical sections showing F-actin staining in developing longitudinal muscles of control (C) and JHm-treated (D, E) embryos that were examined at the indicated days AEL. Yellow triangles indicate striations of the myofibrils. Insets show low power views of embryos with the magnified region boxed. green: F-actin shown by phalloidin binding, red: propidium iodide. JHm-treated examples are embryos that underwent katatrepsis (D) and ones that failed to complete katatrepsis (E).
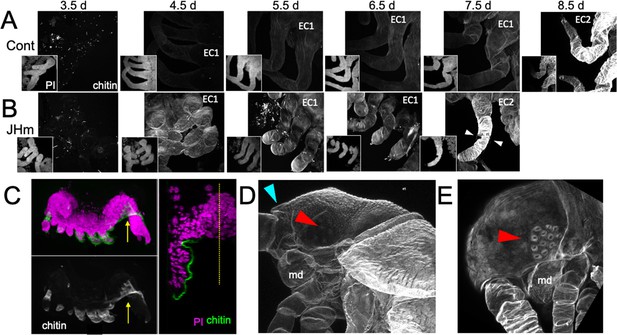
Effects of 1 ng pyriproxyfen (JHm) on the production of E1 and E2 cuticles.
(A, B) Projected confocal Z-stacks of embryos treated with solvent (Control) or JHm at 1.5 d AEL and showing chitin staining of the developing legs at various times thereafter. The insets are propidium iodide (PI) staining of the same stack to show the form of the limbs on each day. In both groups no chitin was evident at 3.5 d AEL, when only epicuticle is present (dots are due to surface debris). In control embryos, the procuticle of the first embryonic cuticle (EC1) (starting at 4.5 d AEL) stains weakly for chitin, while the second embryonic cuticle (EC2) shows strong chitin staining (8.5d). JHm-treated embryos show enhanced chitin staining of their EC1 and they produce their EC2 a day early as demonstrated by the presence of cuticular hairs (white triangles). All cuticle images were made with the same laser power and gain. (C) Lateral view of a 5.5 d embryo that failed to undergo katatrepsis after JHm treatment at 1.5 d AEL. The E1 cuticle covering its ventral surface shows enhanced chitin staining. The arrow shows the plane of the transverse image on the right; the ventromedial region of the embryo is still invaginated into the yolk. PI: propidium iodide staining. Dotted line is the midline. (D, E) Projected confocal Z-stacks of 10 d AEL embryos showing the E2 cuticle produced by controls (D) or by embryos treated with JHm at 4.5 d AEL. The cuticle of the control embryo (D) has a pebbly surface sculpturing, an egg tooth (blue triangle) but lacks cuticular eye lenses (red triangle). The JHm-treated embryo produced a cuticle typical of later stages. It is smooth, lacks an egg tooth, and has formed thickened, but abnormal, cuticular lenses (red triangle). md: mandible.
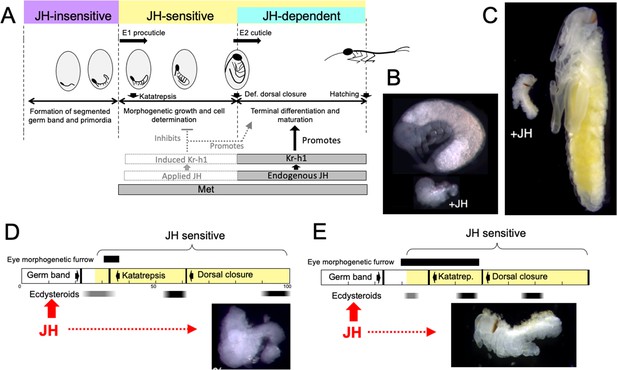
Comparison of responses of short germ band embryos to JH.
(A) Summary of the developmental responses of Thermobia domestica embryos to JH. Exogenous JH has little or no developmental effect up through germ band formation. Embryonic development is then altered by JH treatment starting just before deposition of the first embryonic (E1) cuticle, but endogenous JH does not appear until around definitive dorsal closure, when it is needed for terminal differentiation. (B, C) Photomicrographs of embryos of Thermobia (B) and of Schistocerca gregaria (C) comparing the size and morphology of a control embryo at definitive dorsal closure with a clutch mate that had been treated with JHm the day after oviposition (+JH). (D, E) Schematic summary of embryonic development of Thermobia (D) and Schistocerca (E) comparing the time of ecdysteroid surges and the onset of JH sensitivity with the time course of embryogenesis. The black bar shows the time of progressive patterning of the eye primordium. Eye patterning begins before Schistocerca embryos become sensitive to JH, but after Thermobia embryos become JH sensitive. Consequently, treatment with JHm (red) a day after oviposition results in Schistocerca embryos with tiny eyes but Thermobia embryos lack eyes all together. The times of ecdysteroids in the locust are based on data from Locusta migratoria (Lagueux et al., 1979).
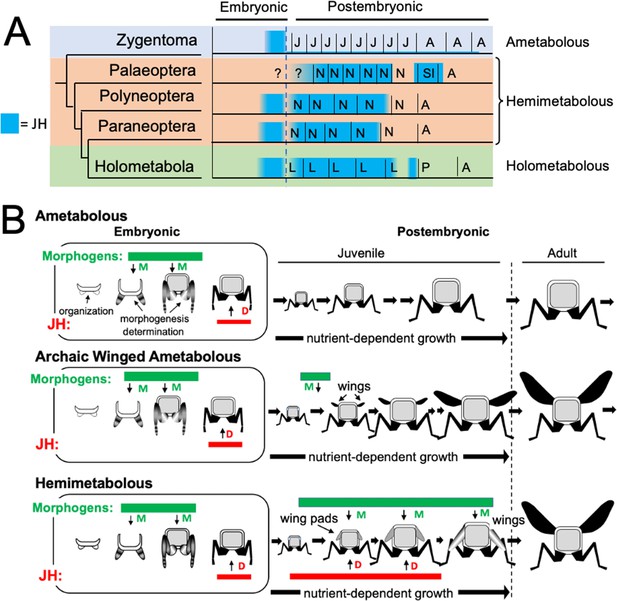
Phylogenetic shifts in JH production during embryonic and postembryonic development in insects.
(A) Depiction of JH titers in ametabolous, hemimetabolous, and holometabolous insects. Titers are based on direct measurements or are estimated from Kr-h1 expression. The ametabolous order, Zygentoma, provides the only data for an ametabolous insect, Thermobia (Figure 2A; Fernandez-Nicolas et al., 2023). Palaeopteran postembryonic data from the mayfly, Cloeon dipterum (Kamsoi et al., 2021). Polyneopteran titers based on locusts (Temin et al., 1986; Truman and Riddiford, 1999) and Blattella (Maestro et al., 2010). Paraneopteran from Pyrrhocoris (Konopova et al., 2011) and Holometabolous for Manduca (Bergot et al., 1981a; Borst et al., 1987; Fain and Riddiford, 1975). (B) A scenario for the role of postembryonic JH in the evolution of the wing pad. In the ametabolous condition, as typified by Thermobia, the major phase of body morphogenesis (M) is confined to mid-embryogenesis, followed by the appearance of JH which supports differentiation (D). In archaic winged insects a new, postembryonic phase of morphogenesis supports wing formation in the young juvenile. The small wings then undergo positive allometric growth until they are large enough to support flight of the older juveniles and the adults. The postembryonic, reappearance of JH during wing morphogenesis redirected development to make the wing pad. This compromise developmental program is then maintained until the end of juvenile growth when the disappearance of JH allows wing differentiation.
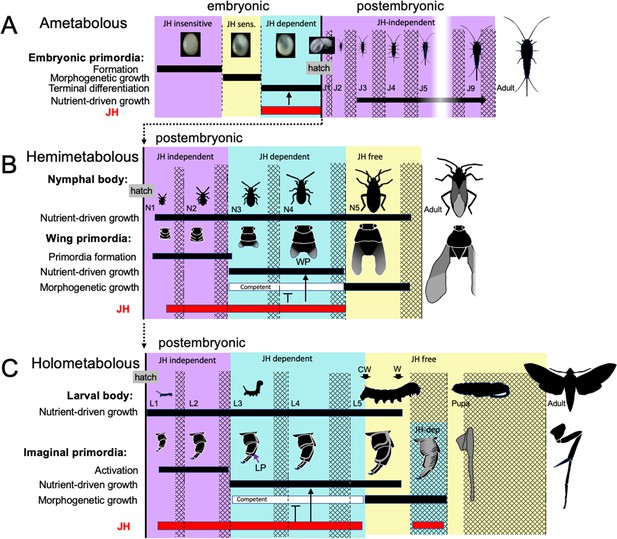
Comparison of the developmental effects of JH in Thermobia with its postembryonic actions in hemimetabolous and holometabolous insects.
(A) As summarized in Figure 10, early Thermobia embryogenesis is JH insensitive, but JH sensitivity is acquired just prior to katatrepsis and the production of the first embryonic cuticle. JH appears after definitive dorsal closure and is required for the terminal differentiation of the juvenile. Once the juvenile form is established, JH is not then needed for its maintenance. (B) The early nymphal instars of hemimetabolous insects are also JH independent. Morphogenesis, associated with wing development, begins around the third nymphal stage and is correlated with the appearance of JH dependence. JH allows wing pad (WP) growth but suppresses its morphogenesis until the disappearance of JH ushers in the JH-free period which allows wing morphogenesis and differentiation. (C). The function of JH in holometabolous larvae is like that seen in nymphs except that imaginal primordia (like the leg primordia [LP]) are widely spread through the larval body and involve almost all of the adult organs. The imaginal primordia and the larval cells differ in their JH requirements during the prepupal period (see text). The pupal-adult transition then requires the absence of JH. CW: critical weight checkpoint; W: wandering stage; double crosshatch: period of new cuticle production preceding each ecdysis.
Tables
Specialized animals and reagents.
| Reagent / animal | Source | Catalog number |
|---|---|---|
| Thermobia domestica | 30+year laboratory colony | est. from wild caught |
| pyriproxyfen | Sumitomo Co | #95737-68-1 |
| 7 ethoxyprecocene | Sigma Aldrich | #65383-73-5 |
| DAPI | Sigma Aldrich | #28718-90-3 |
| Calcofluor White | Sigma Aldrich | #4193-55-9 |
| Propidium Iodide | Thermo Fisher Scientific | #440301000 |
| Oregon Green phalloidin | Thermo Fisher Scientific | #O7466 |
| rabbit anti-distal-less | gift from Grace Panganiban | |
| rabbit anti P-HistoneH3 (ser 10) | EDM Millipore | #06–570 |
| Normal Donkey Serum | Jackson ImmunoResearch | #017-000-121 |
| AF488 Donkey anti-rabbit IgG | Jackson ImmunoResearch | #711-545-152 |
| AF594 Donkey anti-rabbit IgG | Jackson ImmunoResearch | #711-585-152 |
| DPX mountant | Electron Microscopy Sciences | # 13512 |
Primers used for real-time PCR.
| gene | sequence accession number (NCBI) | forward primer (5'–3') | reverse primer (5'–3') |
|---|---|---|---|
| Met | JN416986.1 | TACTCCATCCACACAGTCAAGG | TTCCGTGATTCGACGATCTCTC |
| Kr-h1 | JN416989.1 | ACTCCGTCGAATGGTACTAGTG | GTTCTTGCATGGGAGGAAACTG |
| myo | GASN02042720.1 | TTCAACAAGCAAGCCCACAAG | ACACCGATCCACTACCATTCC |
| rp49 | AB689035.1 | CTAAAGAGGAACTGGCGCAAAC | GTTTAGTCTTCTTGGCGCTTCC |





