UPRER–immunity axis acts as physiological food evaluation system that promotes aversion behavior in sensing low-quality food
Figures
The stress response is induced in animals fed low-quality food, HK-E. coli.
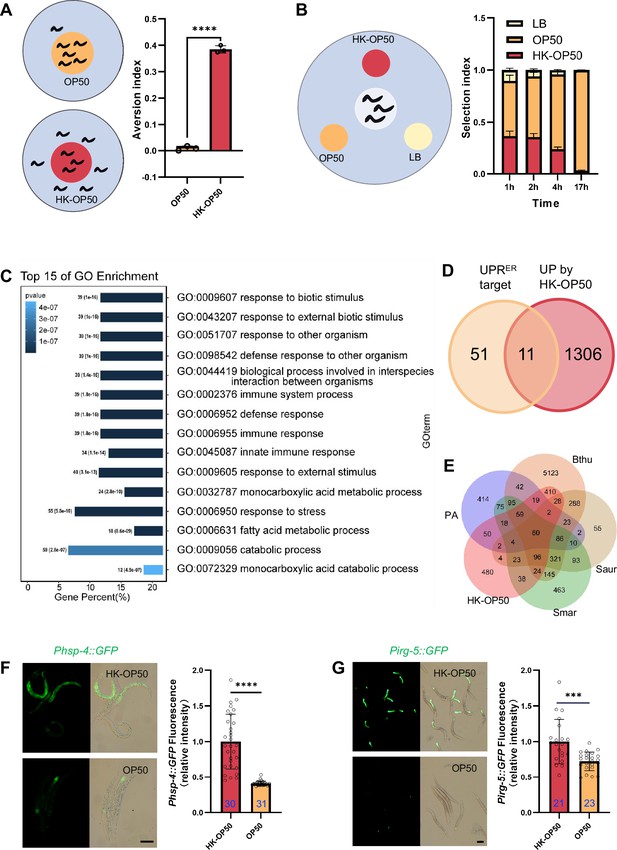
(A) Schematic drawing and quantitative data of the food aversion assay. Circles indicate the food spot for live (yellow) and HK-OP50 (red) bacteria, respectively. The animals were scored 16–17 hr after L1 worms were placed on the food spot. Data are represented as mean ± SD from three independent experiments, 79–129 animals/assay. (B) Schematic method and quantitative data of the food selection assay. Live (yellow), heat-killed (red) E. coli and LB as the buffer for E. coli were placed on indicated position. Synchronized L1 worms were place in the center spot. The selection index was calculated at the indicated time. Data are represented as mean ± SD from eight independent experiments, 123–792 animals/assay. (C) GO enrichment analysis of up-regulated genes in animals fed with HK-E. coli vs live E. coli. (D) Venn diagram showing numbers of UPRER target genes and up-regulated genes in animals fed HK-E. coli, and their overlap. (E) Venn diagram showing numbers of induction genes by four pathogenic bacteria and HK-E. coli induced genes, and their overlap. The gene expression data was extracted from published data of animals’ infection with Pseudomonas aeruginosa (PA) (Nakad et al., 2016), Bacillus thuringiensis (Bthu) (Sinha et al., 2012), Staphylococcus aureus (Saur) (Sinha et al., 2012), and Serratia marcescens (Smar) (Sinha et al., 2012). (F–G) GFP fluorescence images and bar graph showing that Phsp-4::GFP (F) and Pirg-5::GFP (G) were induced in animals fed with HK-E. coli. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. For all panels, Scale bar shows on indicated figures, 50 μm. * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001, ns: no significant difference. Precise p values are provided in Raw Data.
-
Figure 1—source data 1
Numerical data of Figure 1A–B and F–G and Figure 1—figure supplement 1G.
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig1-data1-v1.xlsx
Food selection assay of animals fed HK-E. coli or E. coli.
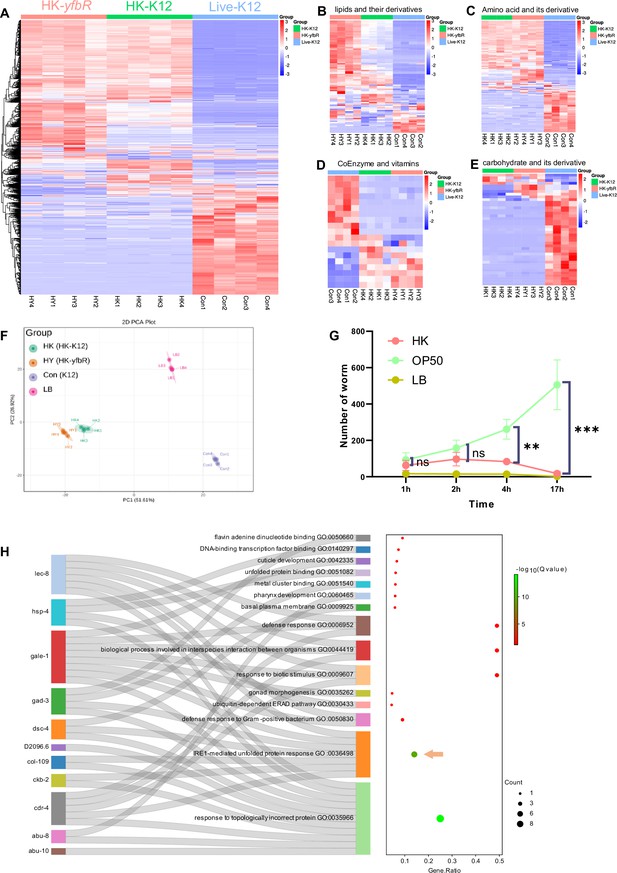
Relative to Figure 1. (A–E) Metabolomics analysis of different quality food (HK-K12, HK-yfbR and Live-K12). Cluster analysis of all metabolites (A), lipids and their derivatives (B), amino acids and their derivatives (C), coenzymes and vitamins (D), and carbohydrates and their derivatives (E) from Live-K12, HK-K12, and HK-yfbR. Color indicates the relative level of each metabolite. HK-K12: heat-killed E. coli wild-type K12; HK-yfbR: heat-killed E. coli mutant yfbR; K12: live E. coli wild-type K12. z-score for standardizing data, complete for bi-clustering algorithm, and Euclidean for distance method. (F) Principal component analysis to test the repeatability of the metabolic experiment. HK-K12: heat-killed E. coli wild-type K12; HK-yfbR: heat-killed E. coli mutant yfbR; K12: live E. coli wild-type K12. LB: LB buffer for culturing E. coli. (G) The number of worms in each position calculated at the indicated time, indicating that animals initially select both foods (1–2 hr), but eventually favor high-quality food (Live E. coli) until 17 hr. (H) GO enrichment analysis of UPRER dependent IRE-1 branch. For all panels, * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001, ns: no significant difference. Precise p values are provided in Raw Data.
Stress response of animals fed HK-E. coli or E. coli.
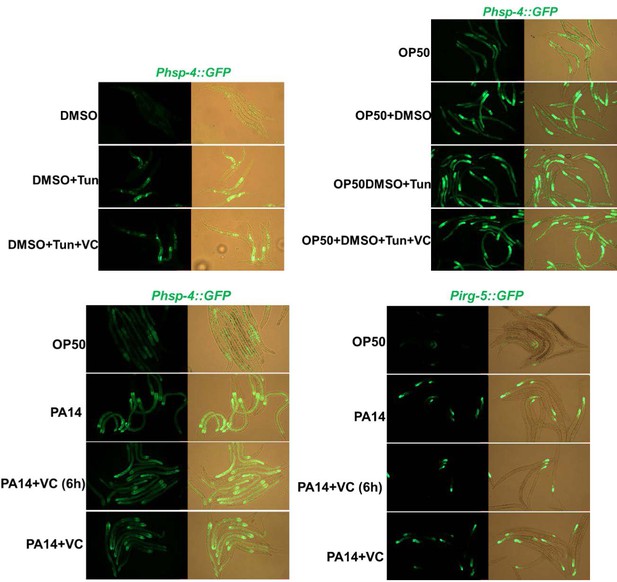
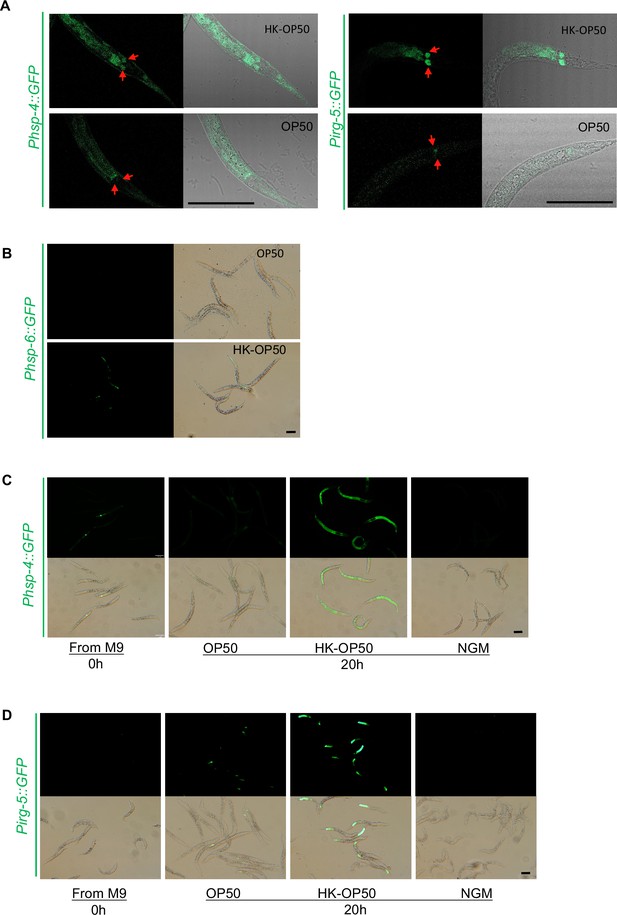
Relative to Figure 1. (A) UPRER reporter (Phsp-4::GFP) and immunity reporter (Pirg-5::GFP) were induced in the neuron and intestine of animals fed with HK-E. coli. Neurons are highlighted with a red arrow. (B) UPRmit reporter (Phsp-6::GFP) was weakly induced in animals fed with HK- E. coli. (C) UPRER reporter (Phsp-4::GFP) expression in animals under normal food (OP50), low-quality food (HK-OP50), and starved (M9: hatching L1 worm in M9, NGM) condition; L1 animals were cultured in OP50, HK-OP50 or starved NGM for 20 hr. (D) Immunity reporter (Pirg-5::GFP) expression in animals under normal food (OP50), low-quality food (HK-OP50), and starved (M9: hatching L1 worm in M9, NGM). L1 animals were cultured in OP50, HK-OP50 or starved NGM for 20 hr. For all panels, Scale bar shows on indicated figures, 50 μm.
Animals evaluate food quality through UPRER (ire-1/xbp-1) - Innate immunity (pmk-1 MAPK) axis.
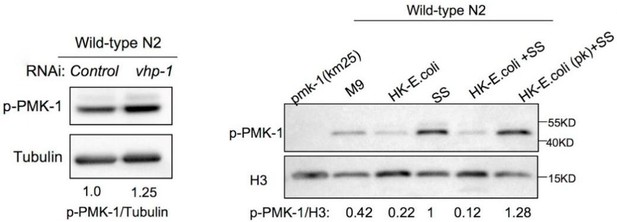
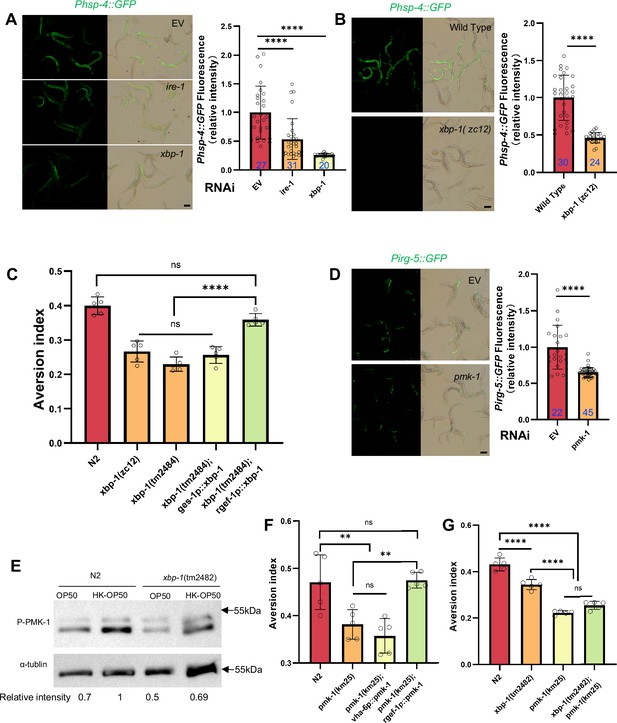
(A–B) GFP fluorescence images and bar graph showing that HK-E. coli induced Phsp-4::GFP was decreased in animals with ire-1 or xbp-1 RNAi treatment (A) or xbp-1 mutation (B). Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (C) Food aversion assay showing that xbp-1 mutation eliminated the discrimination against HK-E. coli. However, this effect is rescued by expressing xbp-1 in neurons rather than intestine. Data are represented as mean ± SD from five independent experiments, 156–763 animals/assay. (D) GFP fluorescence images and bar graph showing that HK-E. coli induced Pirg-5::GFP was decreased in animals with pmk-1 RNAi treatment. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (E) Western blot images showing the level of p-PMK-1 in L1 animals (Wild-type N2 and xbp-1 mutant) fed with OP50 or HK-OP50 for 4 hr. The level of p-PMK-1 is induced in animals fed HK-OP50. (F) Food aversion assay showing that pmk-1 mutation eliminated the discrimination against HK-E. coli. However, this effect is rescued by expressing pmk-1 in neurons rather than intestine. Data are represented as mean ± SD from five independent experiments, 168–492 animals/assay. (G) Food aversion assay in wild-type, xbp-1, pmk-1 and double mutant. Data are represented as mean ± SD from five independent experiments, 259–490 animals/assay. For all panels, Scale bar shows on indicated figures, 50 μm. * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001, ns: no significant difference. Precise p values are provided in Raw Data.
-
Figure 2—source data 1
Numerical data of Figure 2A–D and F–G; Figure 2—figure supplement 1A–C, F–H; and Figure 2—figure supplement 2A–C.
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig2-data1-v1.xlsx
-
Figure 2—source data 2
The raw western bolts for Figure 2E (labelled).
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig2-data2-v1.pdf
-
Figure 2—source data 3
The raw western bolts for Figure 2E (unlabelled, uncropped).
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig2-data3-v1.zip
UPRER and innate immunity pathway in animals are critical for evaluating HK-E. coli.
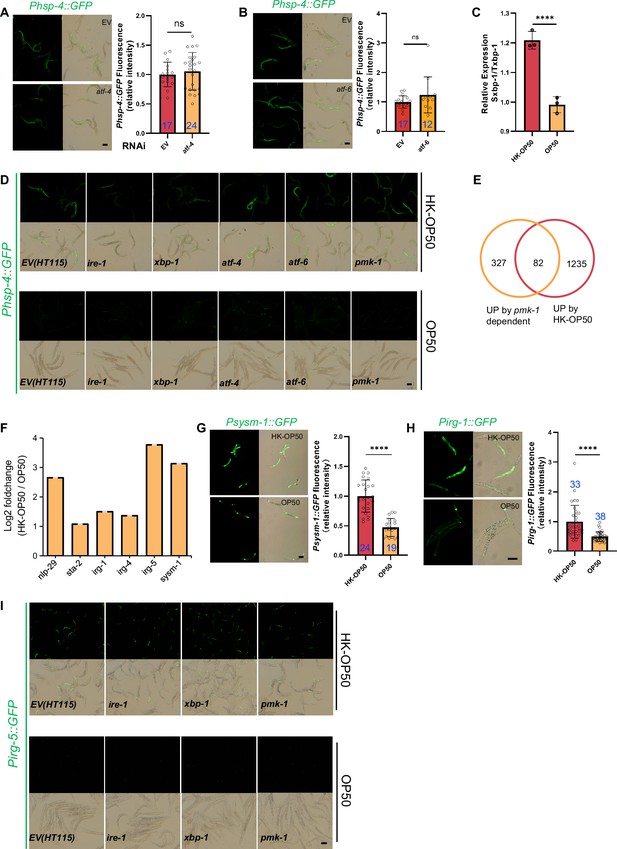
Relative to Figure 2. (A–B) GFP fluorescence images and bar graph showing that HK-E. coli induced Phsp-4::GFP was not affected in animals with atf-4 (A) or atf-6 (B) RNAi treatment. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (C) qPCR showing that IRE-1-mediated splicing of xbp-1 mRNA is induced in animals fed with HK-E. coli. (D) UPRER reporter (Phsp-4::GFP) expression in animals with candidate RNAi feeding OP50 or HK-OP50. (E) Venn diagram showing the numbers of PMK-1-dependent genes 30 and up-regulated genes in animals fed HK-E. coli, and their overlap. (F) The expression of PMK-1-dependent genes which was extracted from RNA-seq data from animals fed with HK-E. coli. The data from average of three independent experiments. (G–H) GFP fluorescence images and bar graph showing that Psysm-1::GFP (G) and Pirg-1::GFP (H) were induced in animals fed HK-E. coli. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (I) Immunity reporter (Pirg-5::GFP) expression in animals with candidate RNAi feeding OP50 or HK-OP50. For all panels, Scale bar shows on indicated figures, 50 μm. * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001, ns: no significant difference. Precise p values are provided in Raw Data.
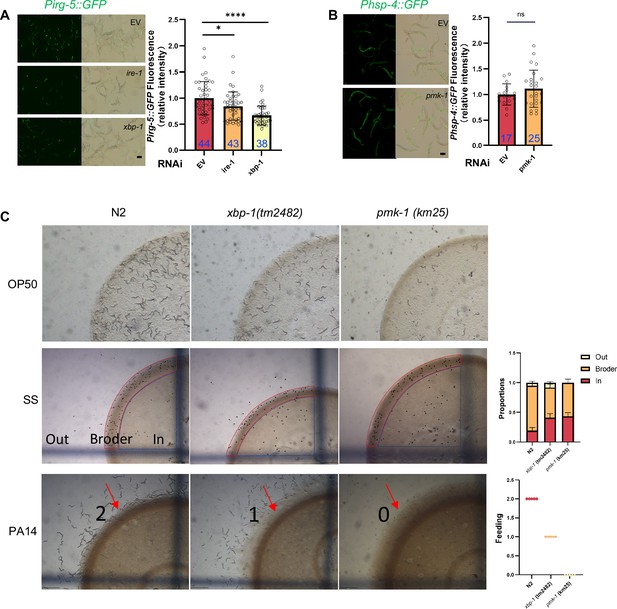
UPRER positively regulates innate immunity pathway in animals.
Relative to Figure 2. (A) GFP fluorescence images and bar graph showing that HK-E. coli induced Pirg-5::GFP was decreased in animals with ire-1 or xbp-1 RNAi treatment. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (B) GFP fluorescence images and bar graph showing that Phsp-4::GFP was not affected in animals with pmk-1 RNAi treatment. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (C) Images and bar graph showing that the avoidance behavior of N2, xbp-1 mutant, and pmk-1 mutant in response to different food source (Saprophytic staphylococci-SS, Pseudomonas aeruginosa-PA14, or OP50). ‘Broder’ indicates regions with thicker SS boundaries; ‘In’ denotes areas inside SS; ‘Out’ refers to areas without SS. The scale is as follows: 2 for animals capable of escaping from PA14 and feeding on PA14 located with edges; 1 for animals capable of escaping from PA14 but not feeding on PA14 located with edges; 0 for animals unable to escape from PA14 and being killed by PA14. Data are represented as mean ± SD from five independent experiments, 359–670 animals/assay. For all panels, Scale bar shows on indicated figures, 50 μm. * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001, ns: no significant difference. Precise p values are provided in Raw Data.
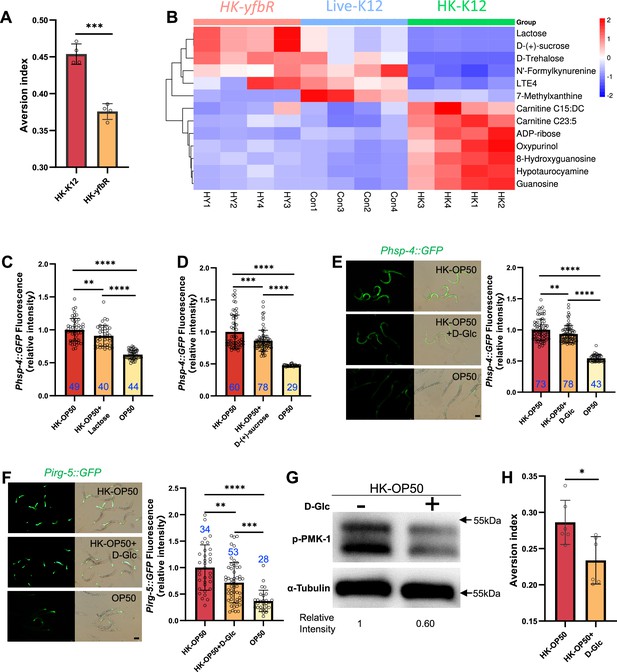
HK-E. coli is low sugar food, which induce stress response and avoidance behavior in animals.
(A) Food aversion assay showing that wild-type animals eliminated the discrimination against HK-E. coli when yfbR is mutated in E. coli. Data are represented as mean ± SD four independent experiments, 251–490 animals/assay. (B) Heat map showing the 13 differential metabolites from HK-K12, HK-yfbR, and K12 in four independent experiments. Color indicates the relative level of each metabolite. (C–D) The bar graph showing that HK-E. coli induced Phsp-4::GFP was decreased in animals with lactose (C) or D-(+)-sucrose (D) supplementation. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (E–F) GFP fluorescence images and bar graph showing that HK-E. coli induced Phsp-4::GFP (E) and Pirg-5::GFP (F) were decreased in animals with D-(+)-glucose (D-Glc) supplementation. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (G) Western blot images showing the level of p-PMK-1 in L1 animals fed HK-E. coli with or without D-(+)-glucose (D-Glc) supplementation for 4 hr. The level of p-PMK-1 is decreased in animals fed HK-OP50 +D Glc. (H) Food aversion assay showing that wild-type animals eliminated the discrimination against HK-E. coli with D-Glc supplementation. Data are represented as mean ± SD six independent experiments, 190–492 animals/assay. For all panels, Scale bar shows on indicated figures, 50 μm. * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001, ns: no significant difference. Precise p values are provided in Raw Data.
-
Figure 3—source data 1
Numerical data of Figure 3A, C–F and H; Figure 3—figure supplement 1B–C; and Figure 3—figure supplement 2C–D, G.
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig3-data1-v1.xlsx
-
Figure 3—source data 2
The raw western bolts for Figure 3G (labelled).
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig3-data2-v1.pdf
-
Figure 3—source data 3
The raw western bolts for Figure 3G (unlabelled, uncropped).
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig3-data3-v1.zip
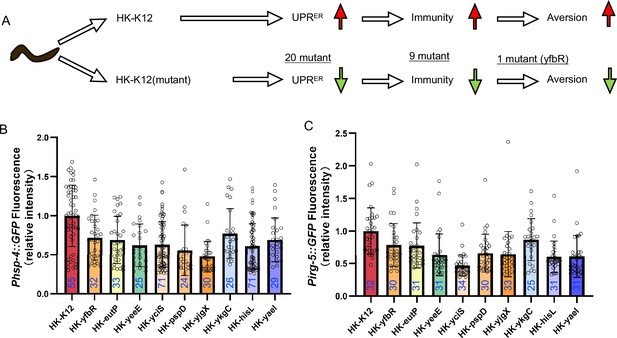
E. coli Keio mutant screening.
Relative to Figure 3. (A) Flow chart of strategy for E. coli Keio mutant screening. We identified 20 E. coli mutants that did not induce hsp-4::GFP through the UPRER reporter (Pirg-5::GFP) after three rounds of screening (Supplementary file 3a). From these 20 E. coli mutants, we identified 9 E. coli mutants that did not induce Pirg-5::GFP through the immunity reporter (Pirg-5::GFP) screening (Supplementary file 3a). (B–C) The bar graph showing that HK-E. coli induced Phsp-4::GFP (B) and Pirg-5::GFP (C) was decreased in animals fed mutant E. coli (Heat-killed). Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. For all panels, * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001, ns: no significant difference. Precise p values are provided in Raw Data.
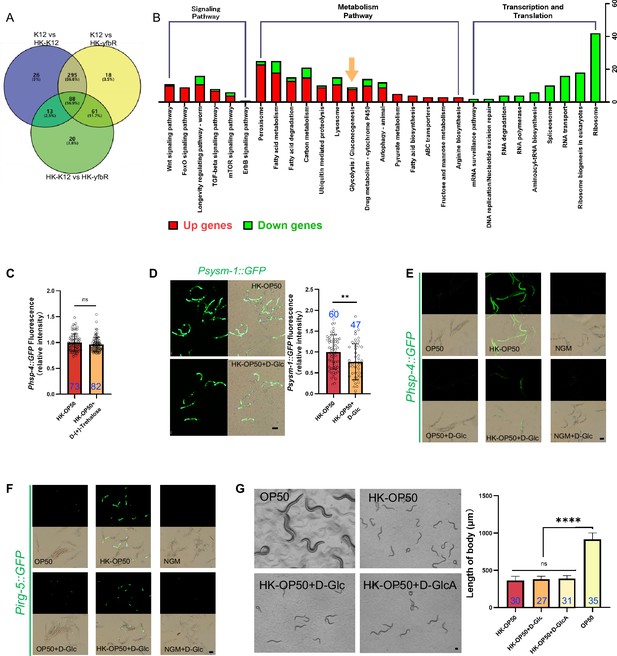
Low sugar food, HK-E. coli, induce stress response and avoidance behavior in animals.
Relative to Figure 3. (A) Venn diagram showing the number of differentially metabolites in HK-E. coli-K12, HK-E. coli-yfbR and E. coli. (B) KEGG enrichment analysis of differentially expressed genes in animals fed HK-E. coli vs live E. coli. We noticed that most of glycolysis/gluconeogenesis genes are up-regulated in animals fed HK-E. coli. (C) The bar graph showing that HK-E. coli induced Phsp-4::GFP was not affected in animals with D-(+)-trehalose supplementation. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (D) GFP fluorescence images and bar graph showing that HK-E. coli induced Psysm-1::GFP was decreased in animals with D-(+)-glucose (D-Glc) supplementation. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (E) UPRER reporter (Phsp-4::GFP) expression animals with D-Glc supplementation under OP50, HK-OP50, or NGM condition. (F) immunity reporter (Pirg-5::GFP) expression animals with D-Glc supplementation under OP50, HK-OP50, or NGM condition. (G) Development of animals after 48 h fed with OP50, HK-OP50, HK-OP50 +D Glc, and HK-OP50 +D GlcA. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. For all panels, Scale bar shows on indicated figures, 50 μm. * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001, ns: no significant difference. Precise p values are provided in Raw Data.
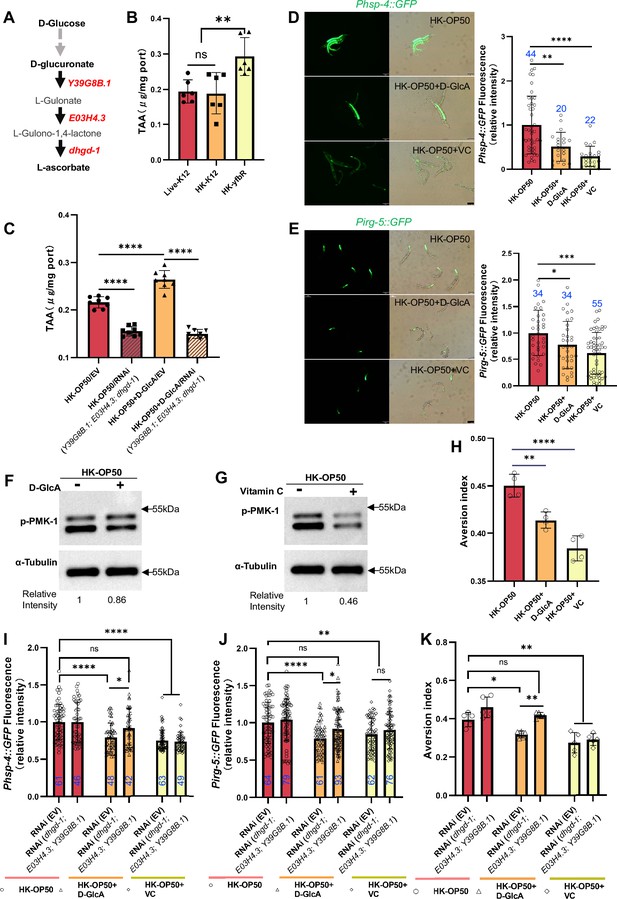
Vitamin C biosynthesis pathway is critically involved in evaluating sugar in the food.
(A) Cartoon illustration of a simplified, Vitamin C biosynthesis pathway in C. elegans. The relevant coding genes of enzymes was labeled with red. (B) The level of total L-ascorbic acid (TAA), also known as vitamin C, in animals fed with Live-K12, HK-K12, or HK-yfbR. Data are represented as mean ± SD from six independent experiments. (C) The level of total L-ascorbic acid (TAA) in animals (control or knockdown of Vitamin C biosynthesis genes) fed with HK-E. coli with or without D-glucuronate (D-GlcA) supplementation. Data are represented as mean ± SD from eight independent experiments. (D–E) GFP fluorescence images and bar graph showing that HK-E. coli induced Phsp-4::GFP (D) and Pirg-5::GFP (E) were decreased in animals with D-GlcA or Vitamin C supplementation. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (F–G) Western blot images showing the level of p-PMK-1 in L1 animals fed with HK-E. coli with D-GlcA or Vitamin C supplementation for 4 hr. The level of p-PMK-1 is decreased in animals with D-GlcA (F) or Vitamin C (G) supplementation. (H) Food aversion assay showing that wild-type animals eliminated the discrimination against HK-E. coli with D-GlcA or Vitamin C supplementation. Data are represented as mean ± SD from four independent experiments, 153–292 animals/assay. (I–K) The bar graph showing that suppression of HK-E. coli induced Phsp-4::GFP (I), Pirg-5::GFP (J) and food avoidance (K) by D-GlcA supplementation was abolished in animals with RNAi of VC biosynthesis genes, which was not affect by Vitamin C supplementation. Blue numbers are the number of worms scored from at least three independent experiments and Data are represented as mean ± SD. (I-J) Data are represented as mean ± SD from five independent experiments, 252–537 animals/assay. (K) For all panels, Scale bar shows on indicated figures, 50 μm. * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001, ns: no significant difference. Precise p values are provided in Raw Data.
-
Figure 4—source data 1
Numerical data of Figure 4B–E and H–K and Figure 4—figure supplement 1A–E.
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig4-data1-v1.xlsx
-
Figure 4—source data 2
The raw western bolts for Figure 4F–G (labelled).
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig4-data2-v1.pdf
-
Figure 4—source data 3
The raw western bolts for Figure 4F–G (unlabelled, uncropped).
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig4-data3-v1.zip
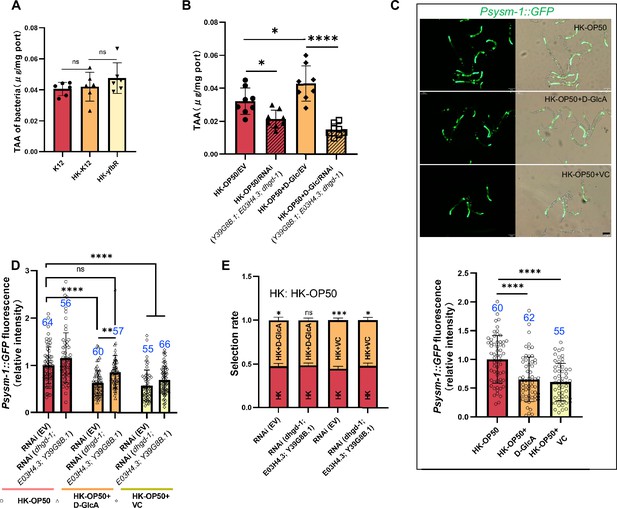
Vitamin C biosynthesis pathway is critical for evaluating low sugar.
Relative to Figure 4. (A) The level of total L-ascorbic acid (TAA) in Live-K12, HK-K12, or HK-yfbR. Data are represented as mean ± SD from six independent experiments. (B) The level of total L-ascorbic acid (TAA) in animals fed HK-E. coli with or without D-Glc supplementation. Data are represented as mean ± SD from eight independent experiments. (C) GFP fluorescence images and bar graph showing that HK-E. coli induced Psysm-1::GFP was decreased in animals with D-GlcA or vitamin C supplementation. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (D) The bar graph showing that suppression of HK-E. coli induced Psysm-1::GFP by D-GlcA supplementation was abolished in animals with RNAi of VC biosynthesis genes, which was not affect by vitamin C supplementation. Blue numbers are the number of worms scored from at least three independent experiments. Data are represented as mean ± SD. (E) Food selection assay showing that the preference of HK-E. coli with D-GlcA supplementation was abolished in animals by RNAi of vitamin C biosynthesis genes. Data are represented as mean ± SD from six independent experiments, 427–775 animals/assay. For all panels, Scale bar shows on indicated figures, 50 μm. * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001, ns: no significant difference. Precise p values are provided in Raw Data.
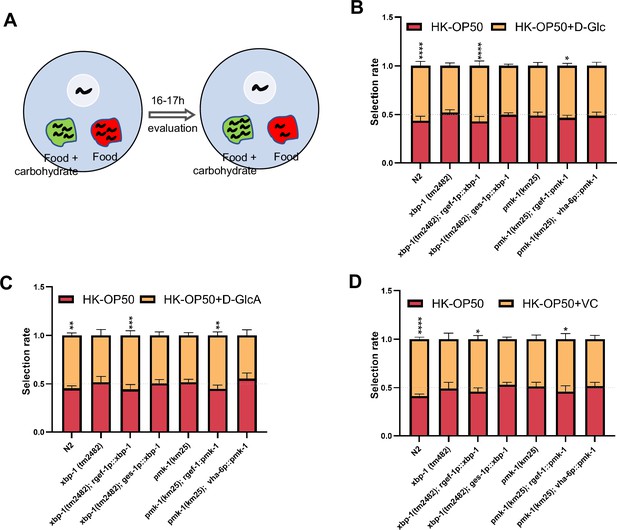
Animals evaluate sugar and vitamin C through neuronal XBP-1 and PMK-1.
(A) Schematic method of the food selection assay. Food (red) and food with carbohydrate (D-Glc, D-GlcA, or VC) supplementation (green) was placed on indicated position. Synchronized L1 worms were then place in plate. After 16-17h, the selection index was calculated. (B–D) Food selection assay showing that xbp-1 or pmk-1 mutation eliminated the preference of HK-E. coli with D-Glc (B), D-GlcA (C) or Vitamin C (D) supplementation, which was rescued in xbp-1 or pmk-1 mutant animals by expressing XBP-1 or PMK-1 in neurons rather than intestine. Data are represented as mean ± SD from five independent experiments, 68–647 animals/assay (B). Data are represented as mean ± SD from six independent experiments, 83–701 animals/assay (C). Data are represented as mean ± SD from six independent experiments, 67–1035 animals/assay (D). For all panels, Scale bar shows on indicated figures, 50 μm. * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001, ns: no significant difference. Precise p values are provided in Raw Data.
-
Figure 5—source data 1
Numerical data of Figure 5B–D and Figure 5—figure supplement 1A–C.
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig5-data1-v1.xlsx
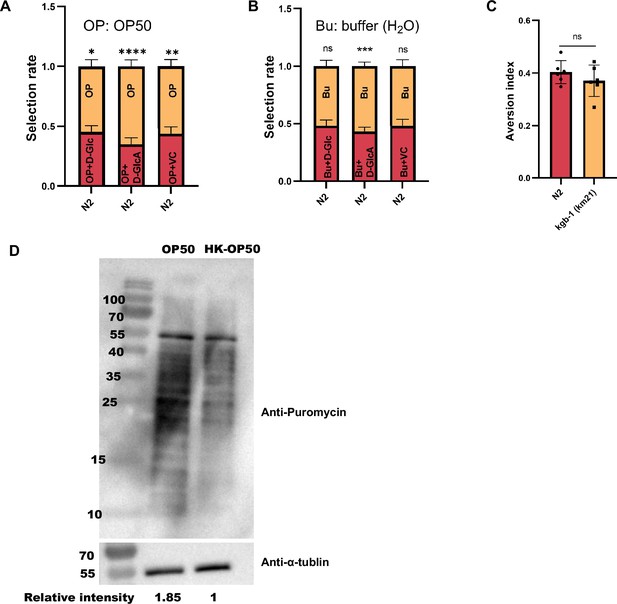
Food behavior of animals.
Relative to Figure 5. (A) Food selection assay for OP50 and OP50 +D Glc, D-GlcA, or VC, respectively. Data are represented as mean ± SD from five independent experiments, 241–1182 animals/assay. (B) Food selection assay for buffer (H2O) & buffer (H2O)+D Glc, D-GlcA, or VC, respectively. Data are represented as mean ± SD from five independent experiments, 8–153 animals/assay. (C) Food avoidance assay for N2 and kgb-1 mutant animals fed with HK-E. coli. Data are represented as mean ± SD from six independent experiments, 348–660 animals/assay. (D) Translation ability of animals fed with OP50 or HK-OP50 are presented by western blot of puromycin-labeled peptides. For all panels, * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001, ns: no significant difference. Precise p values are provided in Raw Data.
-
Figure 5—figure supplement 1—source data 1
The raw western bolts for Figure 5—figure supplement 1D (labelled).
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig5-figsupp1-data1-v1.pdf
-
Figure 5—figure supplement 1—source data 2
The raw western bolts for Figure 5—figure supplement 1D (unlabelled, uncropped).
- https://cdn.elifesciences.org/articles/94181/elife-94181-fig5-figsupp1-data2-v1.zip
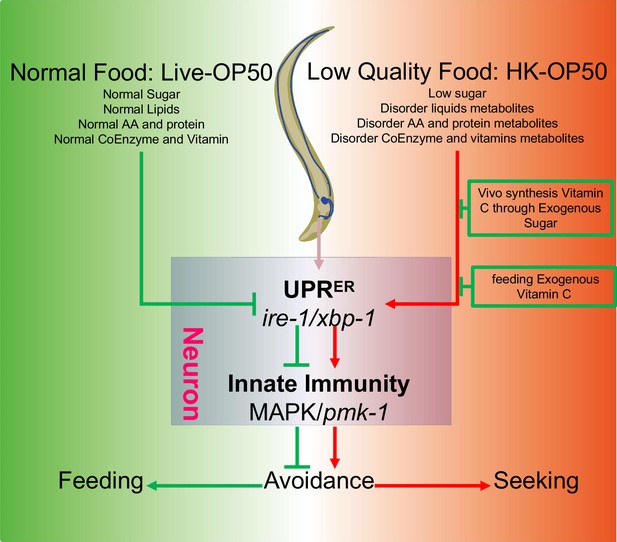
Schematic model of physiological food evaluation system in evaluating/sensing sugar and vitamin C through UPRER (IRE-1/XBP-1) - Innate immunity (PMK-1/p38 MAPK) axis.
Vitamin C level is low in animals fed low sugar food, HK-E. coli. Sugar and Vitamin C deficiency activate cellular UPRER and immune response, which promote animals to leave low-quality food and seek better food for survival. This cellular stress regulated physiological food evaluation system depends UPRER (IRE-1/XBP-1) - Innate immunity (PMK-1/p38 MAPK) axis in neuron.

L1 animals obtained from mothers fed E. coli strains OP50, HT115, or K12 exhibited similar UPR induction under HK-E. coli OP50 feeding conditions.
Additional files
-
Supplementary file 1
Metabolism-seq analysis.
- https://cdn.elifesciences.org/articles/94181/elife-94181-supp1-v1.xlsx
-
Supplementary file 2
RNA-seq analysis.
- https://cdn.elifesciences.org/articles/94181/elife-94181-supp2-v1.xlsx
-
Supplementary file 3
Screening data for E. coli mutant keio library.
- https://cdn.elifesciences.org/articles/94181/elife-94181-supp3-v1.xlsx
-
Supplementary file 4
Metabolism-seq data of HK-K12, HK-yfbR and K12.
- https://cdn.elifesciences.org/articles/94181/elife-94181-supp4-v1.xlsx
-
Supplementary file 5
RNA-seq data of animals fed with HK-E. coli OP50 and E. coli OP50.
- https://cdn.elifesciences.org/articles/94181/elife-94181-supp5-v1.xlsx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/94181/elife-94181-mdarchecklist1-v1.docx