Microtubule networks in zebrafish hair cells facilitate presynapse transport and fusion during development
Figures
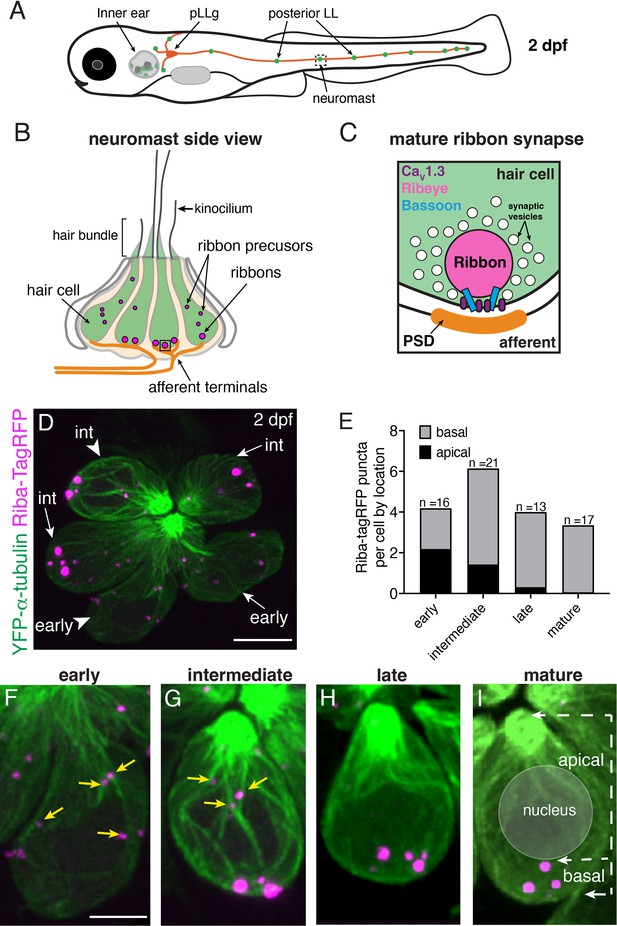
Ribbons associate with microtubules and change localization during development.
(A) Schematic of a larval zebrafish at 2 days post fertilization (dpf) with the location of the posterior-lateral line (pLL) indicated relative to the inner ear. Neuromasts (green) in the pLL contain sensory hair cells that are innervated by afferent projections from the posterior lateral-line ganglion (pLLg, orange). (B) Schematic of a neuromast at 2 dpf, viewed from the side. At 2 dpf, the majority of hair cells (green) are developing. At the top of the cells, the mechanosensory hair bundle is composed of actin-based stereocilia and a tubulin-based kinocilium. A shorter kinocilium and an abundance of small ribbon precursors are indicative of an immature stage. (C) Schematic of a ribbon synapse when mature. The dense presynapse or ribbon is made primarily of Ribeye. Ribbons tether synaptic vesicles near CaV1.3 channels at the plasma membrane, across from the postsynaptic density (PSD). Bassoon acts to anchor ribbons at the presynaptic AZ. (D) Example image of a neuromast at 2 dpf, viewed from top down. The microtubule network and ribbons are marked with YFP-Tubulin and Riba-TagRFP, respectively. In this example of 6 developing hair cells, two early and four intermediate cells are present. The cell bodies of an early and intermediate cell from this example (arrowheads) are expanded in (F and G). (E) Plot shows the average number of ribbons per hair cell at each developmental stage. Cell stage is determined by the height of the kinocilium. After an increase in ribbon number with development, there is a decrease upon maturation. The number of apically localized Riba-TagRFP puncta is high at early and intermediate stages and is lower in late and mature hair cells. In contrast, the number of basally-localized Riba-TagRFP puncta is low at early stages and becomes higher by intermediate stages (n=16, 21, 13, 17 hair cells for early, intermediate, late, and mature stages, respectively). (F–I) Example images of hair cells expressing YFP-Tubulin and Riba-TagRFP at early, intermediate, late, and mature stages. At the early stage, Riba-TagRFP puncta are spread throughout the cell body and are smaller in size. At the intermediate stage, the number of Riba-TagRFP puncta becomes more basally enriched and are larger in size. In late and mature hair cells, all Riba-TagRFP puncta are at the base of the cell and are fewer in number compared to the intermediate stage. The arrows in (I) highlight the apical and basal regions of the cell used for quantification of Riba-TagRFP puncta location in (E). Yellow arrows in (E) and (F) indicate precursors associated with microtubules. Scale bars in D=5 µm and in F=2 µm.
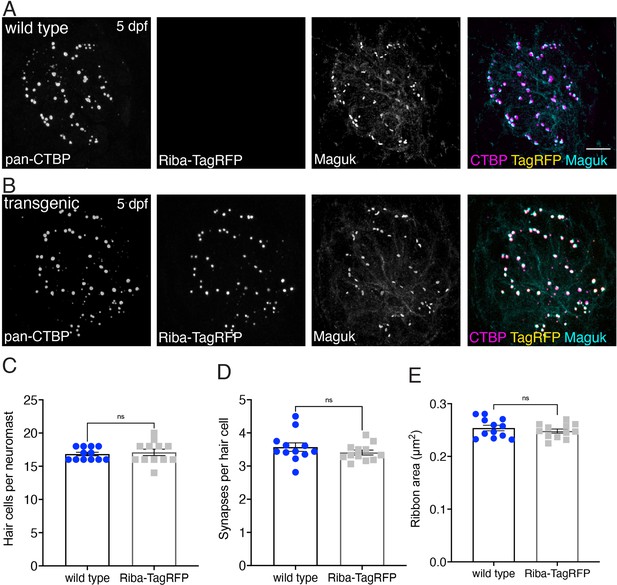
Riba-TagRFP transgenic fish have normal cell numbers, synapses per hair cell, and ribbon areas.
(A–B) Example immunostained images from pLL hair cells of wild type (A) and Riba-TagRFP transgenic fish (B) are shown at 5 dpf. The neuromasts are labeled with pan-CTBP (labels ribbons) and Maguk (labels postsynapses). (C–E) Quantification of these images shows no significant differences in the number of hair cells (C), p=0.728, the number of synapses per hair cell (D), p=0.283, or the ribbon areas >0.1 µm2 (E), p=0.392 between the wild type and transgenic fish (n=12 neuromasts for wild type and Riba-TagRFP). Error bars represent SEM. For comparisons, a Mann-Whitey test was used in C and an unpaired t-test was used in (D–E), scale bar in A=5 µm.
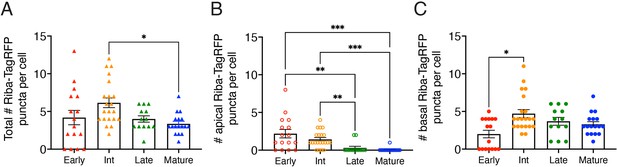
Ribbon number and apical-basal localization change during development.
(A) The total number of Riba-TagRFP puncta increases from early to intermediate stages. The total number of puncta becomes significantly reduced upon maturation. (B) The number of apically-localized Riba-TagRFP precursors is high at early and intermediate stages and is significantly reduced compared to late and mature hair cells. (C) The number of basally-localized Riba-TagRFP ribbons is low at early stages and becomes significantly higher by intermediate stages. Error bars represent SEM. For comparisons, a Kruskal-Wallis test was used in (A–C), *p<0.05, **p<0.01, ***p<0.001.
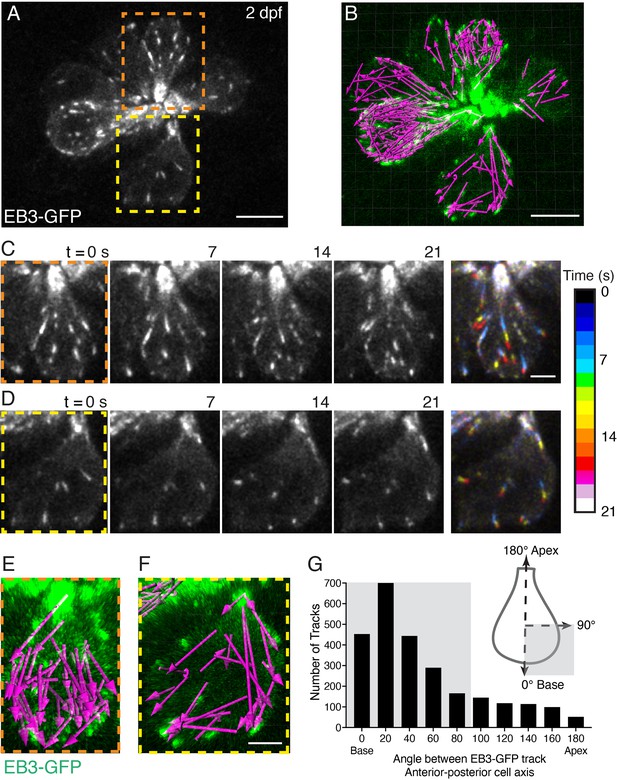
EB3-GFP tracks show plus ends of microtubules move to the cell base.
(A) Example image of a neuromast at 2 dpf. The growing or plus ends of microtubules are marked with EB3-GFP. In this example, the apex of 8 developing cells is at the center of the image, and the base of each cell is at the periphery. Two example cells are outlined (dashed lines) and expanded in more detail in (C–D), and (E–F). (B) A 22-min timelapse was taken of the example in (A). All EB3 tracks, indicated by magenta arrows (tracked in Imaris) detected during the timelapse are shown. (C–D) Example time courses of EB3-GFP in hair cells over 21 s; the cell apex is towards the top of each image. In the final image, the four images for each example (0–21 s) were projected over time as a pseudocolor image represented by the colormap. The pseudocolor images show that many EB3-GFP tracks move to the cell base. (E–F) The magenta arrows in (E) and (F) show all the EB3-GFP tracks acquired in the example cells in (C–D) over the entire 22-min duration. Arrows indicate the direction of travel. (G) The schematic in (G) shows how EB3-GFP tracks were aligned to each hair cell. Tracks moving toward the apex have a track angle of 180°, while those moving to the base have an angle of 0°. This analysis revealed that the majority of EB3-GFP tracks (shaded domains) move toward the base of the cell (n=7 neuromasts, 2598 tracks). Scale bars in A-B=5 µm and in C-F=2 µm.
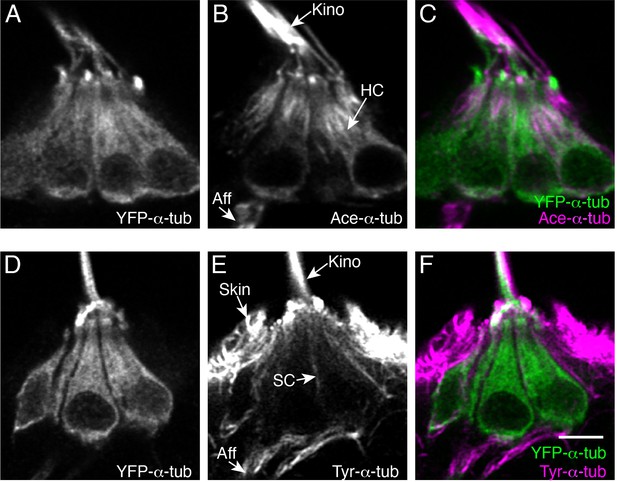
Microtubule modifications in lateral-line hair cells.
(A–F) Example immunostains of pLL hair cells expressing YFP-α-tubulin at 5 dpf, labeled either with acetylated-α-tubulin (A–C) or tyrosinated-α-tubulin (D–F). Panels (A) and (D) show YFP-α-tubulin, (B) and (E) show acetylated-α-tubulin or tyrosinated-α-tubulin labels, respectively. Panels (C) and (F) show the merged images. HC = hair cell; SC = supporting cell; Aff = afferent process; Kino = kinocilium; Scale bar in F=5 µm.
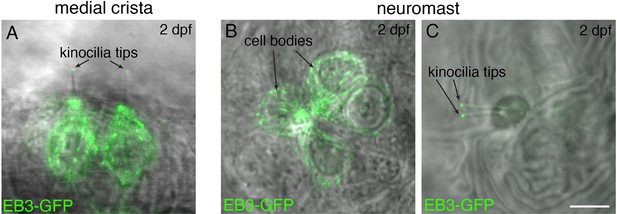
EB3-GFP label reveals microtubule plus ends at kinocilial tips in hair cells.
(A) Example side-view image of EB3-GFP localization in 2 hair cells of the medial cristae within the zebrafish inner ear at 2 dpf. (B–C) Example images of EB3-GFP localization in four hair cells within a pLL neuromast at 2 dpf. In (B), a maximum-intensity projection of EB3-GFP localization in the cell bodies is shown. In (C), a more apical projection shows the localization of EB3-GFP in the kinocilia of the same hair cells as (B). In both zebrafish inner ear and pLL hair cells, EB3-GFP is present in the cell bodies and at the tips of kinocilia. In all images, EB3-GFP is overlaid onto a transmitted light image. Scale bar in C=5 µm.
EB3-GFP dynamics in developing lateral-line hair cells.
Timelapse of the example of a pLL neuromast expressing EB3-GFP from Figure 2. The growing or plus ends of microtubules as visualized by capturing EB3-GFP dynamics. The timelapse was acquired on a LSM 780 confocal microscope every 7 s for 22 min. Maximum-intensity projection of the original z-stack is shown on the left side, played at five frames per second. The right side shows the same movie as the left side, except EB3-GFP tracks are color-coded and followed over time using the FIJI plugin TrackMate. Scale bar=5 µm.
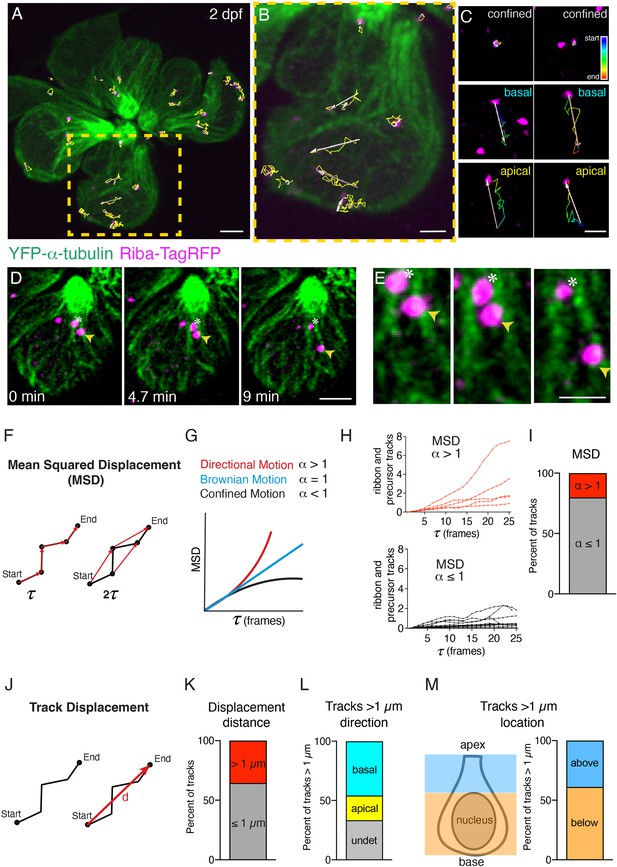
Ribbon precursors exhibit directional motion and confinement on microtubules.
(A –B) To quantify motion, ribbons and ribbon precursors were tracked in hair cells at early and intermediate stages at 2 dpf (example). Shown are tracks (in yellow) from an entire neuromast (A) and a single hair cell (B), obtained using Imaris, during a 30-min timelapse acquired every 50 s (also see Figure 3—video 1). (C) Magnified view shows individual tracks over time and examples of confinement and motion toward the cell base and apex. Pseudo-colored tracks indicate the timecourse of movement (blue start, red end). (D) Example of confined motion and directed motion on microtubules in a single hair cell. Images were obtained during a 9-min timelapse acquired every 20 s. The ribbon labeled by the asterisks remains confined, while the ribbon labeled with the yellow arrowheads moves along a microtubule, towards the cell base, over time (also see Figure 3—video 3). (E) A magnified image of the example shown in (D). (F) Mean squared displacement (MSD) vs time step was used to measure movement behaviors. Shown in red are the first- and second-time steps. The results are plotted in the form of MSD vs time step (τ) plots and the exponent (α) of the plots can be used to distinguish between the different types of motion observed: confined (α < 1), directional (α > 1), or Brownian motion (α = 1). (H) Example MSD plots of individual ribbon tracks from 2 control neuromasts (15 tracks MSD < 1 (black), five tracks MSD > 1 (red)). (I) The bar graph shows the percent of MSD tracks displaying confined (α < 1, 79.8%, gray), and directional motion (α > 1, 20.2%, red). (J) Track displacement vs time was used to measure movement behaviors with track > 1 µm indicative of directed motion. (K) The bar graph shows the percent of tracks with distances > 1 µm (35.6% red) and those with distances ≤ 1 µm (65.4%, gray). (L) The bar graph shows the percent of tracks with distances > 1 µm based on track direction (45.8% to the cell base, cyan; 20.8% to the cell apex, yellow; 33.3% undetermined direction, gray). (M) The bar graph shows the percent of tracks with distances > 1 µm based on location in the cell, above or below the nucleus (38.9% above the nucleus, blue; 61.1% below the nucleus, orange). In (K –M) n = 10 neuromasts, 40 hair cells, and 203 tracks. Scale bar in A, D=2 µm and B, C, E=1 µm.
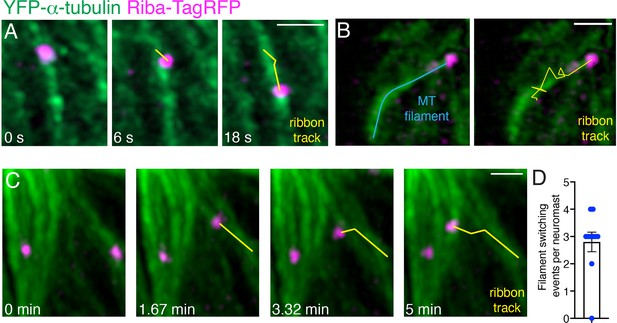
Ribbons move directionally on microtubules and can move between microtubule filaments.
(A) Example of ribbon movement on a microtubule directed towards the cell base. Ribbon precursor movement was imaged at 3 s intervals for ~5 min. Shown in (A) are frames 10, 12, and 16 (also see Figure 3—video 4). (B) Example of a ribbon movement along a microtubule towards the cell apex. Images were acquired every 50 s for ~40 min. The microtubule (MT) filament is indicated in blue. The track of the ribbon over frames 25–46 (21 frames, 17.5 min) is shown in yellow. The track is overlaid onto the last image of the timelapse (also see Figure 3—video 5). (C) Example of a ribbon moving along a microtubule towards the cell apex and then switching to another microtubule. Images were acquired every 98 s for 43 min. Shown in (C) are frames 10, 12, 14, and 16 (also see Figure 3—video 6). (D) Quantification shows an average of 2.8 filament switching events per neuromast during timelapses (timelapses acquired every 50–100 s for 30–70 min, n=10 neuromasts). Error bar represents SEM. Scale bars in A-C=1 µm.
Tracking precursors and ribbons in 3D using Imaris.
Timelapse movie of a pLL neuromast at 2 dpf. A partial cell volume was captured at 50 s intervals for ~30 min with a Zeiss LSM 780 with Airyscan. Microtubules are marked with YFP-tubulin (green), ribbons and precursors are marked with Riba-TagRFP (magenta). Tracks captured in Imaris are color-coded with time. This example is the same as Figure 3A–B. Maximum-intensity projections of the original z-stacks are shown, played at five frames per second. Scale bar=2 µm.
Tracking precursors and ribbons in 3D using Imaris.
Timelapse movie of a pLL neuromast at 2 dpf. A partial cell volume was captured at 60 s intervals for ~30 min with a Zeiss LSM 780 with Airyscan. Microtubules are marked with YFP-tubulin (green), ribbons and precursors are marked with Riba-TagRFP (magenta). Tracks captured in Imaris are shown in yellow. Maximum-intensity projections of the original z-stacks are shown, played at five frames per second. Scale bar=2 µm.
Directional motion to cell base and stationary precursors on microtubule.
Timelapse movie of a pLL neuromast at 2 dpf. A partial cell volume was captured at 20 s intervals for 9 min with a Zeiss LSM 980 with Airyscan 2. Microtubules are marked with YFP-tubulin (green), ribbons and precursors are marked with Riba-TagRFP (magenta). Maximum-intensity projections of the original z-stacks are shown played at five frames per second. This example is the same as Figure 3D–E. On the left side is a timelapse of the hair cell showing a ribbon precursor moving along a microtubule toward the cell base. In addition, a stationary ribbon precursor is also shown. On the right is a higher magnification view of these two ribbon precursors. The circles indicate spots identified using TrackMate, along with the track of the moving ribbon shown in yellow. Scale bar=2 µm.
Directional motion of precursor along a microtubule to the cell base.
Timelapse movie of a pLL neuromast at 2 dpf. A partial cell volume was captured at 3 s intervals for 5 min with a Zeiss LSM 980 with Airyscan 2. Microtubules are marked with YFP-tubulin (green), ribbons and precursors are marked with Riba-TagRFP (magenta). Maximum-intensity projections of the original z-stacks are shown, played at five frames per second. On the left side is a timelapse of the hair cell showing a ribbon precursor moving along a microtubule toward the cell base. On the right is a higher magnification view of this ribbon precursor. TrackMate was used to generate the track of the precursor along the microtubule in yellow. This example is the same as Figure 3—figure supplement 1A. Scale bar=2 µm.
Directional motion of precursor along microtubule to cell apex.
Timelapse movie of a pLL neuromast at 2 dpf. A partial cell volume was captured at 50 s intervals for 18 min with a Zeiss LSM 780 with Airyscan. Microtubules are marked with YFP-tubulin (green), ribbons and precursors are marked with Riba-TagRFP (magenta). Maximum-intensity projections of the original z-stacks are shown, played at five frames per second. On the left side is a timelapse of the hair cell showing a ribbon precursor moving along a microtubule toward the cell apex. On the right is a higher magnification view of the precursor. The circle indicates a spot identified using TrackMate, along with the track of the moving ribbon shown in yellow. This example is the same as Figure 3—figure supplement 1B. Scale bar=2 µm.
Precursor switching between microtubules.
Timelapse movie of a pLL neuromast at 2 dpf. A partial cell volume was captured at ~100 s intervals for 40 min with a Zeiss LSM 980 with Airyscan. Microtubules are marked with YFP-tubulin (green), ribbons and precursors are marked with Riba-TagRFP (magenta). Maximum-intensity projections of the original z-stacks are shown, played at five frames per second. On the left side is a timelapse of the hair cell showing a ribbon precursor moving along a microtubule toward the hair cell apex; the precursor then moves to another microtubule. On the right is a higher magnification view of the ribbon precursor during the timelapse. The precursor movement is quite fast and at this interval can be observed twice in a single z-stack during movement between microtubules at timepoints 2, 4, and 15. This example is the same as Figure 3—figure supplement 1C. Scale bar=2 µm.
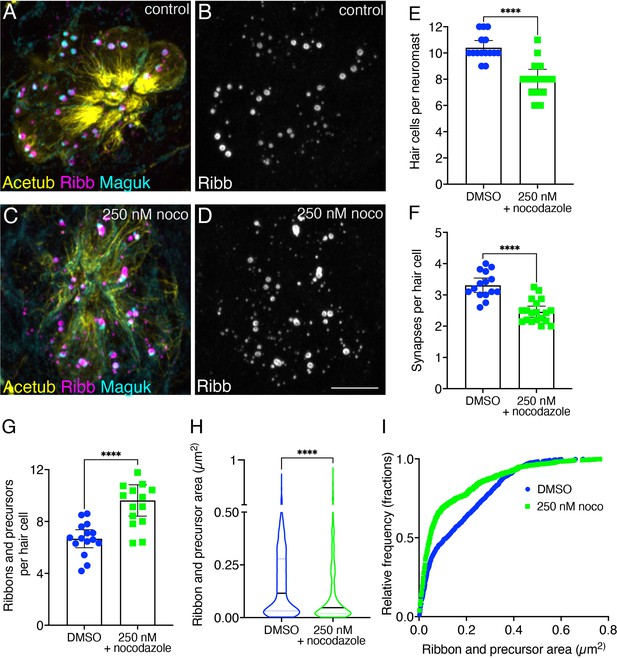
Overnight microtubule destabilization increases ribbon numbers, and decreases synapse counts and ribbon areas.
(A–D) Example immunostain of a neuromast at 3 dpf after an overnight treatment with 250 nM nocodazole (C–D) or DMSO (A–B). Acetylated-α-tubulin (Acetub) labels microtubules, Ribeyeb (Ribb) labels precursors and ribbons, and Maguk labels postsynapses. (E–G) After an overnight treatment with 250 nM nocodazole, there are significantly fewer hair cells per neuromast (E), p<0.0001, fewer complete synapses per cell (F), p<0.0001, and more ribbons and precursors per cell (G), p<0.0001 compared to controls (n=15 neuromasts for control and 250 nM nocodazole treatments). (H–I) After an overnight treatment with 250 nM nocodazole the average area of Ribb puncta was significantly lower compared to controls (H), p<0.0001 (n=1008 and 1135 Ribb puncta for control and 250 nM nocodazole treatments). In (I), the relative frequency of all the areas of Ribb puncta are plotted in nocodazole treatment and controls. For comparisons, an unpaired t-test was used in (E–G), and a Mann-Whitney test was used in (H). Error bars represent SEM in E-G. In H the median and first and third quartiles are shown. Scale bar in D=5 µm.
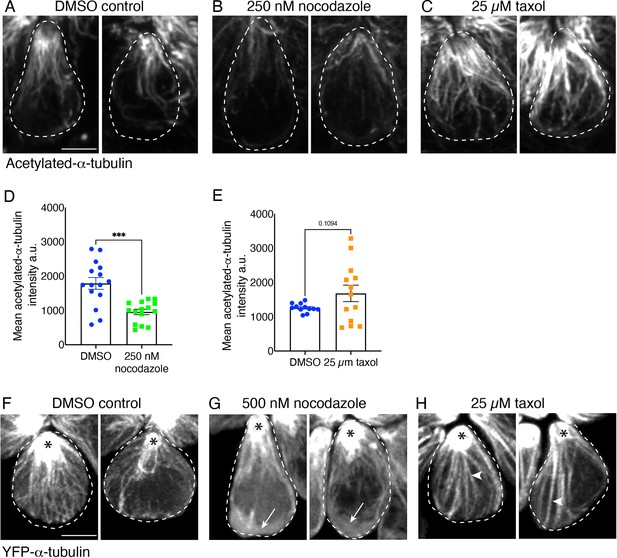
Nocodazole and taxol treatment impact microtubules in lateral-line hair cells.
(A–C) Example side-view images of individual hair cells treated overnight with 250 nM nocodazole (B) or 25 µM taxol (C) overnight compared to DMSO control (A). Hair cells were fixed and stained with acetylated-α-tubulin at 3 dpf. In (B), fewer microtubule networks are observed in nocodazole-treated hair cells. In contrast, in (C), more intense, stable microtubules are observed in taxol-treated hair cells. (D) Quantification reveals a significant reduction in the mean acetylated-α-tubulin intensity in hair cells treated overnight with nocodazole, indicating microtubule disruption (n=15 neuromasts for each condition, p<0001). (E) Although the mean acetylated-α-tubulin intensity levels were elevated after treatment with 25 µM taxol overnight, the elevation was not significant (n=12 neuromasts for DMSO control and 13 for taxol treatment, p=0.109). (F–H) Example side-view in vivo images of individual hair cells at 2 dpf expressing YFP-α-tubulin treated at 2 dpf with 500 nM nocodazole (G) or 25 µM taxol (H) for 3–4 hrs, compared to DMSO controls (F). In (G) fewer microtubule networks along with more diffuse YFP-α-tubulin label (white arrows) is observed in nocodazole-treated hair cells. In contrast, in (H), more intense and long, stable microtubules (white arrowheads) are observed in taxol-treated hair cells. All hair cells in (F–H) show persistent and stable microtubules at the cell apex (black asterisks). Images in (A–C) and (F–H) were maximum-intensity projected and displayed using the same settings. For comparisons, an unpaired t-test was used in (D) and (E). Error bars represent SEM. Scale bar in A and F=2.5 µm.
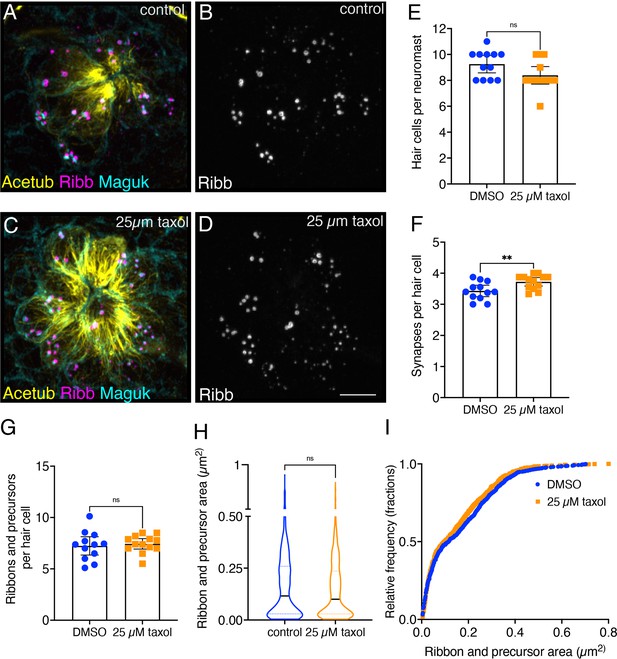
Overnight microtubule stabilization slightly increases synapse counts.
(A–D) Example immunostain of a neuromast at 3 dpf after an overnight treatment with 25 µM taxol (C–D) or DMSO (A–B). Acetylated-α-tubulin (Acetub) labels microtubules, Ribeyeb (Ribb) labels precursors and ribbons, and Maguk labels postsynapses. (E–G) After an overnight treatment with 25 µM taxol, there are similar numbers of hair cells per neuromast (E), p=0.059, more complete synapses per cell (F), p=0.009, and no change in the number of ribbons and precursors per cell (G), p=0.672 compared to controls (n=12 and 13 neuromasts for control and 25 µM taxol treatments). (H–I) After an overnight treatment with 25 µM taxol, the average area of Ribb puncta was not changed compared to controls (H), p=0.153 (n=800 and 817 Ribb puncta for control and 25 µM taxol treatments). In (I), the relative frequency of all the areas of Ribb puncta are plotted in taxol treatment and controls. For comparisons, an unpaired t-test was used in (E–G), and a Mann-Whitney test was used in (H). Error bars represent SEM in E-G. In H the median and first and third quartiles are shown. Scale bar in D=5 µm.
Microtubule dynamics in hair cells change upon treatment with nocodazole and taxol.
Timelapse movies of pLL neuromasts at 2 dpf captured after treatment with 0.1% DMSO (control, left side), 25 µM taxol (Taxol, middle), or 250 nM nocodazole (Noc, right side) for 30 min. Microtubules are marked with YFP-tubulin (gray) and ribbons are marked with Riba-TagRFP (magenta). The timelapses of partial cell volumes were acquired with a Zeiss LSM 780 with Airyscan every 50–80 s for 30 min. Maximum-intensity projections of the original z-stacks are shown, played at five frames per second. The taxol-treated neuromast has more stabilized microtubules and very little cytoplasmic tubulin (depolymerized tubulin), indicating that the microtubules are more stable than the control. In the nocodazole-treated neuromast, there are fewer microtubules and more diffuse cytoplasmic tubulin compared to the control. Scale bar=2 µm.
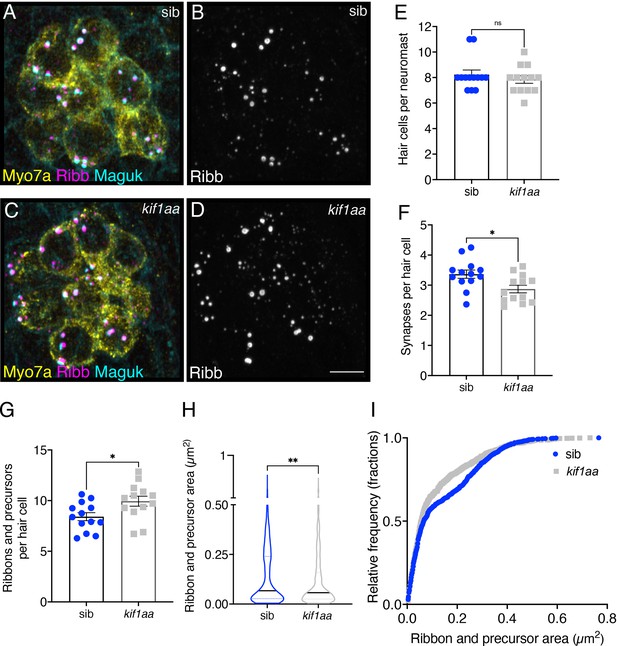
Loss of Kif1aa increases precursor numbers and decreases synapse counts.
(A–D) Example immunostain of neuromasts at 3 dpf in kif1aa germline mutants (C–D) or sibling control (A–B). Myosin7a labels hair cells, Ribeyeb (Ribb) labels precursors and ribbons, and Maguk labels postsynapses. (E–G) In kif1aa mutants, there is no change in the number of hair cells per neuromast (E), p=0.418, but there are fewer complete synapses per cell (F), p=0.014, and more ribbons and precursors per cell (G), p=0.024 compared to sibling controls (n=13 neuromasts for control and kif1aa mutants). (H–I) In kif1aa germline mutants, the average area of Ribb puncta was significantly lower compared to sibling controls (H), p=0.005 (n=896 and 1008 Ribb puncta for control and kif1aa mutants). In (I), the relative frequency of all the areas of all Ribb puncta are plotted in kif1aa mutants and sibling controls. For comparisons, an unpaired t-test was used in (E–G), and a Mann-Whitney test was used in (H). Error bars represent SEM in E-G. In H the median and first and third quartiles are shown. Scale bar in D=5 µm.
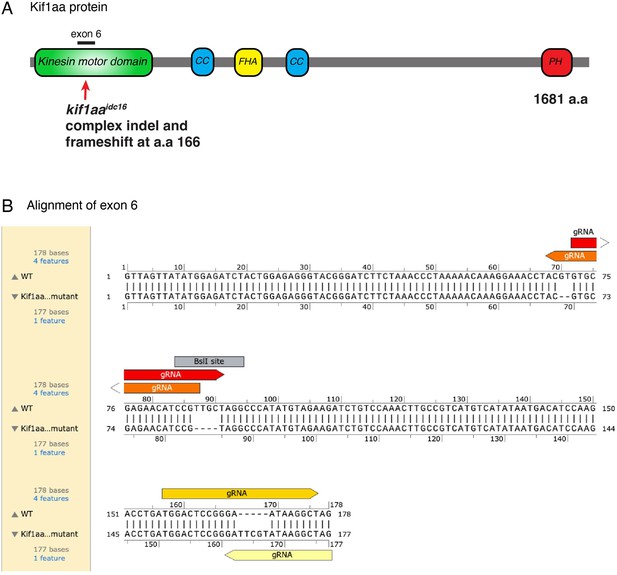
Kif1aa protein and exon 6 lesions.
(A) Overview of the Kif1aa protein and major domains (coiled-coil (CC), fork-head associated (FHA), pleckstrin homology (PH)). The location of the germline kif1aa lesion in the kinesin motor domain within exon 6 is indicated. (B) The DNA sequence of exon 6 (178 bp) in wild type and kif1aa germline mutants. The four gRNAs used to make the kif1aa germline mutant are shown. Two deletions and one insertion are present in kif1aa germline mutants. The BslI restriction site used for genotyping is shown. This DNA alignment was done in SnapGene.
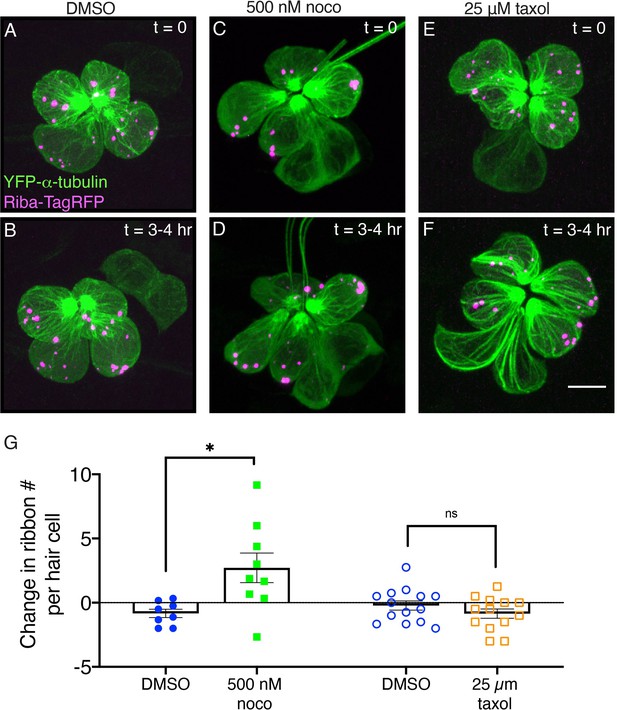
Microtubule destabilization increases ribbon numbers in vivo.
(A, C, E) Example images of neuromasts at 2 dpf. The microtubule network and ribbons are marked with YFP-tubulin and Riba-TagRFP, respectively. Neuromasts were imaged immediately after a 30-min treatment with DMSO (control) (A), 500 nM nocodazole (C) or 25 µM taxol (E), (t=0). (B, D, F) The same neuromasts in A, C, E were reimaged after an additional 3–4 hr of treatment. (G) Quantification reveals that after 3–4 hr nocodazole treatment, there are more Riba-TagRFP puncta per hair cell compared to controls (n=9 and 8 neuromasts for nocodazole and DMSO, p=0.013). In contrast, after a 3–4 hr taxol treatment, there was no significant change in the number of Riba-TagRFP puncta per hair cell compared to controls (n=13 and 14 neuromasts for taxol and DMSO, p=0.256). An unpaired t-test was used for comparisons in (G). Error bars represent SEM. Scale bar in F=5 µm.
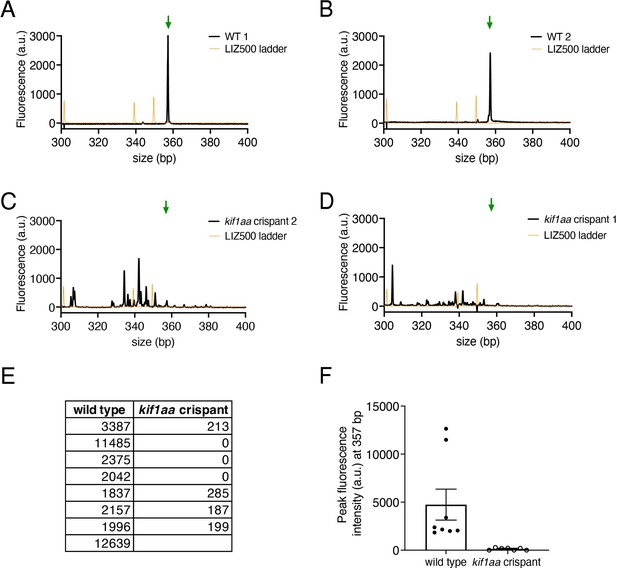
kif1aa crispant verification via genotyping via fluorescent fragment analysis.
Each fish that was imaged in our kif1aa experiments was genotyped using fragment analysis of fluorescent PCR products primers: kif1aa_FWD_fPCR and kif1aa_REV_fPCR (see Key Resources Table) (Carrington et al., 2015). (A–D) Shown are example graphs from wild type (A, B) and kif1aa F0 crispants (C, D). The wild-type graphs have a distinct peak at 357 bp (green arrow), while the kif1aa crispants graphs show that this peak is dramatically reduced. The multiple secondary peaks in (C–D) are indicative of indels up to ~50 bp in kif1aa F0 crispants. (E–F) Quantification of fluorescence intensity values (a.u.) at 357 bp for wild type and kif1aa F0 crispants are shown in a table (E) and a graph (F), demonstrating successful genomic cutting in the kif1aaF0 crispants. kif1aa F0 crispants without robust genomic cutting were excluded from our analyses.

Loss of Kif1aa does not impact ribbon numbers over 3–4 hr.
(A-D) Example images of neuromasts at 2 dpf. The microtubule network and ribbons are marked with YFP-tubulin and Riba-TagRFP, respectively. Neuromasts were imaged immediately in sibling control (A) and kif1aa F0 crispants (C), and after 3–4 hr, (B,D). (E) Quantification reveals that after 3–4 hr the number of Riba-TagRFP puncta per hair cell is the same in control and kif1aa F0 crispants (n=12 and 11 neuromasts for control and kif1aa F0 crispants, p=0.427). An unpaired t-test was used for the comparison in (E). Error bars represent SEM. Scale bar in A=5 µm.
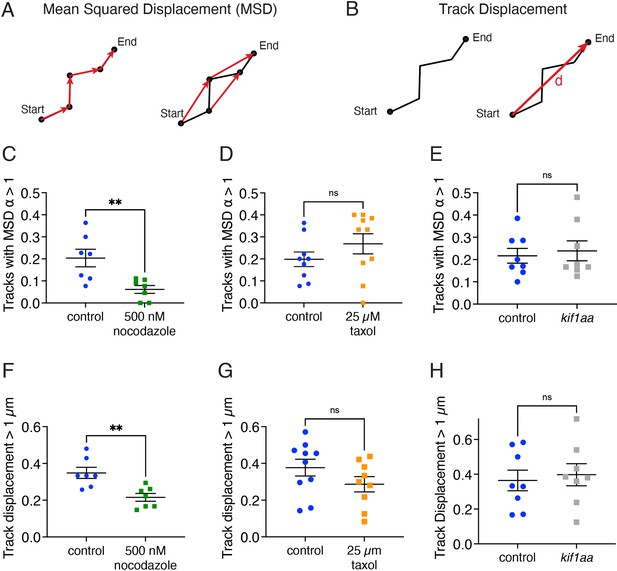
Ribbon precursors require intact microtubules, but not Kif1aa for directional motion.
(A) Mean squared displacement (MSD) vs time was used to quantify the proportion of ribbon and precursor tracks with a velocity and α>1, a behavior indicative of directionally moving tracks. Shown are the first- and second-time steps. (B) To further quantify directional motion, the proportion of tracks with large displacements >1 µm was quantified. Track displacement was measured as the distance between the start and end point of the track. (C) Hair cells treated with 500 mM nocodazole have fewer directional tracks (α>1) compared to controls (p=0.007). (D) In hair cells treated with 25 µM taxol, there are not significantly fewer directional tracks (α>1) compared to controls (p=0.24). (E) In hair cells lacking Kif1aa, there are not significantly fewer directional tracks (α>1) compared to controls (p=0.70). (F) Hair cells treated with 500 nM nocodazole have fewer ribbons with track displacements >1 µm compared to control (p=0.004). (G) There is no change in track displacement >1 µm in hair cells treated with 25 µM taxol (p=0.17). (H) There is no change in track displacement >1 µm in hair cells lacking Kif1aa (p=0.71). N=7 neuromasts for DMSO control and nocodazole for (C) and (F); n=9 and 10 neuromasts for DMSO control and taxol for (D) and (G); n=8 neuromasts for control and kif1aa for (E) and (H). An unpaired t-test was used in (C–H). Error bars represent SEM.
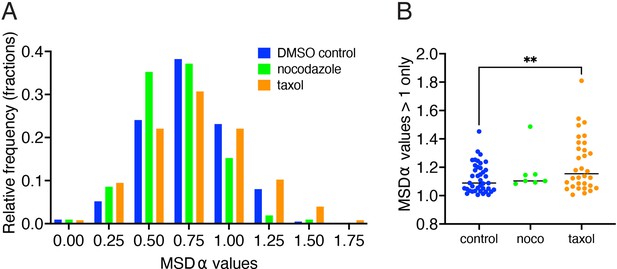
A more stable microtubule network results in directional ribbon tracks with higher mean squared displacement (MSD) α values.
(A) Distributions of individual ribbon track MSD α values are shown for control, nocodazole- (500 nM) and taxol- (25 µM) treated hair cells. Compared to control, taxol treatment shifts the distribution towards higher α values, while nocodazole shifts the distribution towards lower α values (total number of tracks analyzed: control (239), nocodazole (113), and taxol (140)). (B) When examining only α>1 values (indicative of directional motion), there are significantly higher α values in taxol-treated samples compared to control (p=0.013). A one-way ANOVA was used for the comparison in (B). Error bars represent SEM.
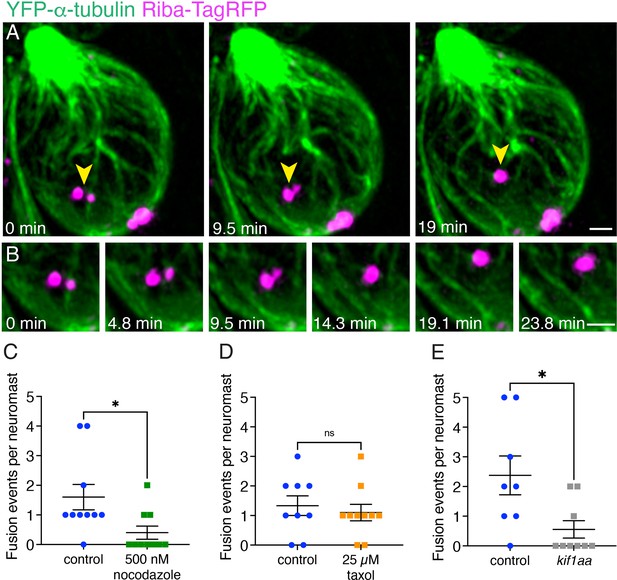
Microtubules and Kif1aa are required for fusion events.
(A) An example of two ribbon precursors undergoing fusion on microtubules (yellow arrowheads). (B) A zoomed-in montage of the example from (A) is shown where the association of each precursor with microtubules can be seen during the process of fusion (also see Figure 8—video 1). (C–D) Destabilizing the microtubules with nocodazole treatment reduces the number of fusion events observed in timelapses (p=0.023). Taxol treatment has no effect (p=0.60). (E) Loss of Kif1aa significantly reduces the number of fusion events observed in timelapses compared to control (p=0.018). N=10 neuromasts for DMSO control and nocodazole for (C); n=9 and 10 neuromasts for DMSO control and taxol for (D); n=8 and 9 neuromasts for control and kif1aa for (E). An unpaired t-test was used for comparisons in (C–E). Error bars represent SEM. Scale bars in (A) and B=1 µm.
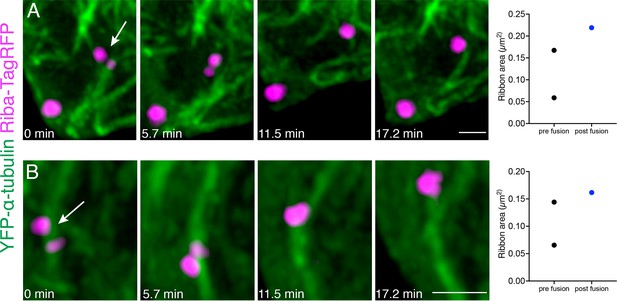
Ribbon precursor fuse on or near microtubules.
(A–B) Two examples of ribbon precursor fusion on or near microtubules. Ribbons were tracked over 13 frames that were acquired every 5.7 min. Shown in (A) are frames 3–6. Shown in (B) are frames 4–7. The arrows indicate ribbons of interest. Plotted on the right of each example is the change in area before (pre fusion) the two precursors fuse into one larger precursor (post fusion). These examples correspond to Figure 8—videos 2 and 3. Scale bars in A-B=1 µm.
Ribbon precursors attached to microtubules undergo fusion.
Timelapse movie of a pLL neuromast at 2 dpf. A partial cell volume was captured at 4.8 min intervals for 100 min with a Zeiss LSM 780 with Airyscan. Microtubules are marked with YFP-tubulin (green) and ribbons are marked with Riba-TagRFP (magenta). Maximum-intensity projections of the original z-stacks are shown, played at five frames per second. Two ribbon precursors associate with microtubules and fuse near the hair cell base. This example is the same as Figure 8A–B. Scale bar=2 µm.
Ribbon precursors attached to microtubules undergo fusion.
Timelapse movie of a pLL neuromast at 2 dpf. A partial cell volume was captured at 5.7 min intervals for 75 min with a Zeiss LSM 780 with Airyscan. Microtubules are marked with YFP-tubulin (green) and ribbons are marked with Riba-TagRFP (magenta). Maximum-intensity projections of the original z-stacks are shown, played at two frames per second. This example is the same as Figure 8—figure supplement 1A. Scale bar=2 µm.
Ribbon precursors attached to microtubules undergo fusion.
Timelapse movie of a pLL neuromast at 2 dpf. A partial cell volume was captured at 5.7 min intervals for 75 min with a Zeiss LSM 780 with Airyscan. Microtubules are marked with YFP-tubulin (green) and ribbons are marked with Riba-TagRFP (magenta). Maximum-intensity projections of the original z-stacks are shown, played at two frames per second. This example is the same as Figure 8—figure supplement 1B. Scale bar=2 µm.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (D. rerio) | Tübingen zebrafish; TU | Other | Background stain | ZFIN:ZDB-GENO-990623–3 |
| Genetic reagent (D. rerio) | Tg(myo6b:ctbp2a-TagRFP)idc11Tg | Wong et al., 2019 | Transgenic line | ZFIN: ZDB-ALT-190102–4 |
| Genetic reagent (D. rerio) | Tg(myo6b:YFP-Hsa.TUBA)idc16Tg | Ohta et al., 2020 | Transgenic line | ZFIN: ZDB-ALT-210824–2 |
| Genetic reagent (D. rerio) | myo6b:EB3-GFPidc23Tg | This paper | Transgenic line | ZFIN: ZDB-ALT-230928–1 |
| Genetic reagent (D. rerio) | kif1aaidc24 | This paper and (David et al., 2024) | Mutant strain | ZFIN: ZDB-ALT-240416–3 |
| Transfected construct (plasmid, injected) | myo6b:EB3-GFP | This paper | Tol2 transgenesis construct | ZFIN: ZDB-TGCONSTRCT-230928–1 |
| Recombinant DNA reagent | p5E-pmyo6b | Trapani et al., 2009 | Gateway entry clone | |
| Recombinant DNA reagent | pME-EB3-GFP | Kawano et al., 2022 | Gateway entry clone | |
| Recombinant DNA reagent | pDestTol2pACryGFP | Berger and Currie, 2013 | Addgene plasmid # 64022, RRID:Addgene_64022 | |
| Recombinant DNA reagent | p3E-polyA | Kwan et al., 2007 | Gateway entry clone | |
| Sequence-based reagent | kif1aa gRNA | This paper | gRNA | 5’-ACGGATGTTCTCGCACACGT(AGG)–3’ |
| Sequence-based reagent | kif1aa gRNA | This paper | gRNA | 5’-GTGCGAGAACATCCGTTGCT(AGG)–3’ |
| Sequence-based reagent | kif1aa gRNA | This paper | gRNA | 5’-TGGACTCCGGGAATAAGGCT(AGG)–3’ |
| Sequence-based reagent | kif1aa gRNA | This paper | gRNA | 5’-AGAATACCTAGCCTTATTCC(CGG)–3’. |
| Sequence-based reagent | kif1aa_FWD | This paper | PCR primers | 5’-AACACCAAGCTGACCAGTGC-3’ |
| Sequence-based reagent | kif1aa_REV | This paper | PCR primers | 5’-TGCGGTCCTAGGCTTACAAT-3’ |
| Sequence-based reagent | kif1aa_FWD_fPCR | This paper | PCR primers | 5’-TGTAAAACGACGGCCAGT-AAATAGAGATTCACTTTTAATC-3’ |
| Sequence-based reagent | kif1aa_REV_fPCR | This paper | PCR primers | 5’- GTGTCTT-CCTAGGCTTACAATGCTTTTGG-3’ |
| Antibody | anti-Myosin-VIIa (rabbit polyclonal) | Proteus Biosciences | Cat# 25–6790, RRID:AB_10015251 | IF(1:1000) |
| Antibody | anti-pan-Maguk (mouse monoclonal IgG1) | Millipore | Cat# MABN72, RRID:AB_10807829 | IF(1:500) |
| Antibody | anti-GFP (chicken polyclonal) | Aves labs | Cat# GFP-1010, RRID:AB_10000240 | IF(1:1000) |
| Antibody | anti-tyrosinated-α-tubulin (mouse monoclonal IgG2a) | Sigma Aldrich | Cat# MAB1864-I, RRID:AB_2890657 | IF(1:1000) |
| Antibody | anti-acetylated-α-tubulin (mouse monoclonal IgG2b) | Sigma Aldrich | Cat# T7451, RRID:AB_609894 | IF(1:5000) |
| Antibody | anti-Ribeyeb (mouse monoclonal IgG2a) | Sheets et al., 2011 | IF(1:10,000) | |
| Antibody | anti-CTPB (mouse monoclonal IgG2a) | Santa Cruz | Cat# sc-55502, RRID:AB_629339 | IF(1:1000) |
| Antibody | Anti-mouse secondary antibodies (goat polyclonal) | Thermo Fisher Scientific | Cat# A-2114; RRID:AB_2535779 | IF(1:1000) |
| Antibody | Anti-mouse secondary antibodies (goat polyclonal) | Thermo Fisher Scientific | Cat# A-21143 RRID;AB_2535779 | IF(1:1000) |
| Antibody | Anti-mouse secondary antibodies (goat polyclonal) | Thermo Fisher Scientific | Cat# A-2113; RRID;AB_2535771 | IF(1:1000) |
| Antibody | Anti-mouse secondary antibodies (goat polyclonal) | Thermo Fisher Scientific | Cat# A-21240; RRID:AB_2535809 | IF(1:1000) |
| Antibody | Anti-mouse secondary antibodies (goat polyclonal) | Thermo Fisher Scientific | Cat# A-21242; RRID:AB_253581 | IF(1:1000) |
| Antibody | Anti-mouse secondary antibodies (goat polyclonal) | Thermo Fisher Scientific | Cat# A-2124; RRID:AB_2535810 | IF(1:1000) |
| Antibody | Anti-rabbit secondary (goat polyclonal) | Thermo Fisher Scientific | Cat# A-11008; RRID;AB_143165, | IF(1:1000) |
| Antibody | Anti-chicken secondary (goat polyclonal) | Thermo Fisher Scientific | Cat# A-11039, RRID:AB_2534096 | IF(1:1000) |
| Commercial assay, kit | LIZ500, fPCR dye standard | Applied Biosystems | Cat# 4322682 | |
| Peptide, recombinant protein | Bs1I | New England Biolabs | Cat# R0555S | |
| Peptide, recombinant protein | Cas9 protein | Integrated DNA technologies | Cat# 1081059 | |
| Chemical compound, drug | ethyl 3-aminobenzoate methanesulfonate salt | Sigma Aldrich | Cat# A5040 | |
| Chemical compound, drug | nocodazole | Sigma Aldrich | Cat# SML1665 | 250–500 nM |
| Chemical compound, drug | Paclitaxel | Sigma Aldrich | Cat# 5082270001 | 25 µM |
| Software, algorithm | Imaris 9.9 | Oxford Instruments | RRID:SCR_007370 | |
| Software, algorithm | Matlab R2020b | Mathworks | RRID:SCR_001622 | |
| Software, algorithm | MSDanalyzer | Tarantino et al., 2014 | ||
| Software, algorithm | Prism 10 | Graphpad | RRID:SCR_002798 | |
| Software, algorithm | FIJI | Open source | RRID:SCR_002285 |





