Author response:
The following is the authors’ response to the original reviews
Public Reviews:
Reviewer 1:
Summary:
Identifying drugs that target specific disease phenotypes remains a persistent challenge. Many current methods are only applicable to well-characterized small molecules, such as those with known structures. In contrast, methods based on transcriptional responses offer broader applicability because they do not require prior information about small molecules. Additionally, they can be rapidly applied to new small molecules. One of the most promising strategies involves the use of “drug response signatures”-specific sets of genes whose differential expression can serve as markers for the response to a small molecule. By comparing drug response signatures with expression profiles characteristic of a disease, it is possible to identify drugs that modulate the disease profile, indicating a potential therapeutic connection.
This study aims to prioritize potential drug candidates and to forecast novel drug combinations that may be effective in treating triple-negative breast cancer (TNBC). Large consortia, such as the LINCS-L1000 project, offer transcriptional signatures across various time points after exposing numerous cell lines to hundreds of compounds at different concentrations. While this data is highly valuable, its direct applicability to pathophysiological contexts is constrained by the challenges in extracting consistent drug response profiles from these extensive datasets. The authors use their method to create drug response profiles for three different TNBC cell lines from LINCS.
To create a more precise, cancer-specific disease profile, the authors highlight the use of single-cell RNA sequencing (scRNA-seq) data. They focus on TNBC epithelial cells collected from 26 diseased individuals compared to epithelial cells collected from 10 healthy volunteers. The authors are further leveraging drug response data to develop inhibitor combinations.
Strengths:
The authors of this study contribute to an ongoing effort to develop automated, robust approaches that leverage gene expression similarities across various cell lines and different treatment regimens, aiming to predict drug response signatures more accurately. The authors are trying to address the gap that remains in computational methods for inferring drug responses at the cell subpopulation level.
Weaknesses:
One weakness is that the authors do not compare their method to previous studies. The authors develop a drug response profile by summarizing the time points, concentrations, and cell lines. The computational challenge of creating a single gene list that represents the transcriptional response to a drug across different cell lines and treatment protocols has been previously addressed. The Prototype Ranked List (PRL) procedure, developed by Iorio and co-authors (PNAS, 2010, doi:10.1073/pnas.1000138107), uses a hierarchical majority-voting scheme to rank genes. This method generates a list of genes that are consistently overexpressed or downregulated across individual conditions, which then hold top positions in the PRL. The PRL methodology was used by Aissa and co-authors (Nature Comm 2021, doi:10.1038/s41467-021-21884-z) to analyze drug effects on selective cell populations using scRNA-seq datasets. They combined PRL with Gene Set Enrichment Analysis (GSEA), a method that compares a ranked list of genes like PRL against a specific set of genes of interest. GSEA calculates a Normalized Enrichment Score (NES), which indicates how well the genes of interest are represented among the top genes in the PRL. Compared to the method described in the current manuscript, the PRL method allows for the identification of both upregulated and downregulated transcriptional signatures relevant to the drug’s effects. It also gives equal weight to each cell line’s contribution to the drug’s overall response signature.
The authors performed experimental validation of the top two identified drugs; however, the effect was modest. In addition, the effect on TNBC cell lines was cell-line specific as the identified drugs were effective against BT20, whose transcriptional signatures from LINCS were used for drug identification, but not against the other two cell lines analyzed. An incorrect choice of genes for the signature may result in capturing similarities tied to experimental conditions (e.g., the same cell line) rather than the drug’s actual effects. This reflects the challenges faced by drug response signature methods in both selecting the appropriate subset of genes that make up the signature and managing the multiple expression profiles generated by treating different cell lines with the same drug.
We appreciate the reviewer’s thoughtful feedback and their suggestion to refer to the Prototype Ranked List (PRL) manuscript. Unfortunately, since this methodology for the PRL isn’t implemented in an open-source package, direct comparison with our approach is challenging. Nonetheless, we investigated whether using ranks would yield similar results for the most likely active drug pairs identified by retriever. To do this, we calculated and compared the rankings of the average effect sizes provided by retriever. Although the Spearman (ρ \= 0.98) correlation coefficient was high, we observed that key genes are disadvantaged when using ranks compared to effect sizes. This difference is particularly evident in the gene set enrichment analysis, where using average ranks identified only one pathway as statistically significantly enriched. The code to replicate these analyses is available at https://github.com/dosorio/L1000-TNBC/blob/main/Code/.
Author response image 1.
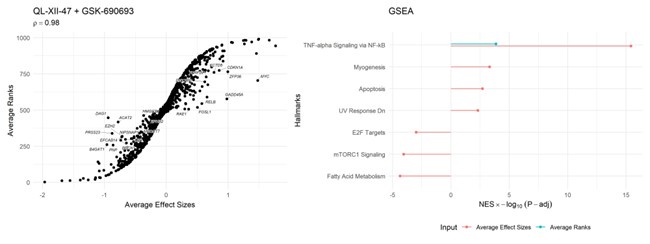
Given the similarity in purpose between retriever and the PRL approach, we have added the following statement to the introduction: “Previously, this goal was approached using a majority-voting scheme to rank genes across various cell types, concentrations, and time points. This approach generates a prototype ranked list (PRL) that represents the consistent ranks of genes across several cell lines in response to a specific drug.”
Regarding the experimental validation, we believe there is a misunderstanding about the evidence we provided. We would like to claridy that we used three different TNBC cell lines: CAL120, BT20, and DU4475. It’s important to note that CAL120 and DU4475 were not included in the signature generation process. Despite this, we observed effects that exceeded the additive effects expectations, particularly in the CAL120 cell line (Figure 5, Panel F).
Reviewer 2:
Summary:
In their study, Osorio and colleagues present ‘retriever,’ an innovative computational tool designed to extract disease-specific transcriptional drug response profiles from the LINCS-L1000 project. This tool has been effectively applied to TNBC, leveraging single-cell RNA sequencing data to predict drug combinations that may effectively target the disease. The public review highlights the significant integration of extensive pharmacological data with high-resolution transcriptomic information, which enhances the potential for personalized therapeutic applications.
Strengths:
A key finding of the study is the prediction and validation of the drug combination QL-XII-47 and GSK-690693 for the treatment of TNBC. The methodology employed is robust, with a clear pathway from data analysis to experimental confirmation.
Weaknesses:
However, several issues need to be addressed. The predictive accuracy of ’retriever’ is contingent upon the quality and comprehensiveness of the LINCS-L1000 and single-cell datasets utilized, which is an important caveat as these datasets may not fully capture the heterogeneity of patient responses to treatment. While the in vitro validation of the drug combinations is promising, further in vivo studies and clinical trials are necessary to establish their efficacy and safety. The applicability of these findings to other cancer types also warrants additional investigation. Expanding the application of ’retriever’ to a broader range of cancer types and integrating it with clinical data will be crucial for realizing its potential in personalized medicine. Furthermore, as the study primarily focuses on kinase inhibitors, it remains to be seen how well these findings translate to other drug classes.
We thank the reviewer for their thoughtful and constructive feedback. We appreciate your insights and agree that several important considerations need to be addressed.
We recognize that the predictive accuracy of retriever depends on the LINCS-L1000 and single-cell datasets. These resources may not fully represent the complete range of transcriptional responses to disease and treatment across different patients. As you mentioned, this is an important limitation. However, we believe that by extrapolating the evaluation of the most likely active compound to each individual patient, we can help address this issue. This approach will provide valuable insights into which patients in the study are most likely to respond positively to treatment.
On the in-vitro validation of drug combinations, we agree that while promising, these results are not sufficient on their own to establish clinical efficacy. Additional in-vivo studies will be essential in assessing the therapeutic potential and safety of these combinations, and clinical trials will be an important next step to validate the translational impact of our findings.
Lastly, we appreciate the reviewer’s comment about the focus of our study on kinase inhibitors. This result was unexpected, as we tested the full set of compounds from the LINCS-L1000 project. We agree that exploring other top candidates, including different drug classes, will be important for assessing how broadly retriever approach can be applied.
Reviewing Editor:
I appreciate the interesting and potentially impactful nature of your research; the reviewers have some concerns that I believe need to be addressed. While your research addresses an important and timely topic in cancer treatment, the current manuscript does not provide a substantial advance in its present form.
The significance of your findings is substantial, as you present a novel computational tool, ’retriever,’ which has the potential to revolutionize personalized cancer treatment strategies by predicting effective drug combinations for triple-negative breast cancer (TNBC). The integration of single-cell RNA-seq data with the LINCS-L1000 project’s transcriptional profiles is a powerful approach that could lead to more targeted and effective therapies. However, the manuscript would benefit from a more explicit discussion of how your work advances the field beyond current methodologies, particularly in the context of drug repurposing and combinatorial therapy.
The strength of the evidence presented is robust, as evidenced by the systematic testing of 152 drug response profiles and 11,476 drug combinations. The identification of QL-XII-47 and GSK-690693 as promising treatment candidates for TNBC is a significant outcome that warrants further exploration. To enhance the manuscript, it would be beneficial to include a more detailed analysis of the biological pathways and mechanisms of action associated with these drugs, as well as a broader experimental validation beyond the cell lines tested.
Taken together, I encourage you to address the issues raised and consider resubmitting a revised version of your work.
Following the suggestions of the reviewing editor, we have included a more detailed discussion on how retriever advances the field, especially in the context of drug repurposing and combinatorial therapy in precision medicine, going beyond current methodologies.
We agree with the suggestion of the editor to offer a more detailed analysis of the biological pathways and mechanisms of action related to these drugs. Consequently, we have expanded our evaluation of these mechanisms. We utilized the Biological Process Gene Ontology to identify changes associated with the mechanisms of each compound individually, as well as the proposed drug combination. Our findings reveal that the statistically significant processes are closely related to cancer deregulation, cross-validating our previous report using the Cancer Hallmarks.
Author response image 2.
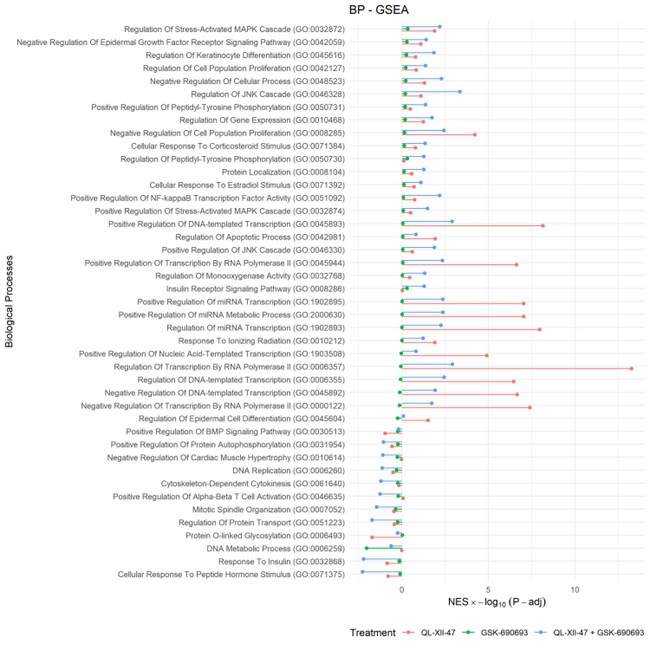
Recommendations for the authors:
Reviewer 1:
(1) The LINCS-L1000 project is introduced in the manuscript as a resource for published transcriptional profiles of several cell lines. Since the original citation, it has been expanded into a vast resource, and the description probably needs to reflect the recent version of LINCS.
We agree with the reviewer that the LINCS-L1000 project is introduced in the manuscript as a resource for transcriptional profiles of several cell lines. Since the original citation, the project has grown into a much larger resource.
To reflect this, we have added a 2022 citation that summarizes efforts to link omics signatures with biological mechanisms using iLINCS: Pilarczyk, Marcin, et al. ”Connecting omics signatures and revealing biological mechanisms with iLINCS.” Nature communications 13.1 (2022): 4678.
Reviewer 2:
(1) It would be beneficial for the manuscript if the authors could expand on the potential limitations inherentto the ’retriever’ tool. This discussion could insightfully address how the foundational assumptions of the analysis may influence the predictive accuracy and the extent to which dataset quality could affect the reliability of the outcomes.
We agree with the reviewer that expanding on the limitations of retriever would help raise awareness of the underlying assumptions in the analysis and how they affect the predictive accuracy and reliability of the results.
The following paragraph was added to the Discussion section: “Although retriever represents a significant advancement in extracting disease-specific drug response profiles from the LINCS-L1000 dataset. Several limitations must be considered when interpreting its results. One key limitation is the restricted scope of gene expression data in the LINCS-L1000 project, which includes expression profiles for only 1,000 genes. While this gene set provides valuable insights into broad transcriptional changes, it may not fully capture the complexity of cellular responses to drug treatments. A possible solution to this limitation relies on imputation techniques to address the missing quantification in the gene expression matrix. The accuracy of the imputed values is dependent on the quality of the imputation model and the completeness of the available data. Consequently, there is an inherent risk that the imputed values may not accurately represent the true and complete underlying biological response.”
(2) Enhancing the manuscript with a more detailed exploration of the clinical ramifications of the study’s findings would be valuable. The authors might consider including how these predictions could be strategically incorporated into the design of clinical trials, thereby bridging the gap between computational predictions and clinical application.
We appreciate the opportunity provided by the reviewer to expand on the potential of retriever for the design of clinical trials and clinical application.
The following paragraph was added to the discussion section: “Finally, we have shown that the approach implemented in retriever method can predict effective drug combinations for patients with triplenegative breast cancer (TNBC), but its potential goes beyond that. It can also be applied to single-cell RNA sequencing data from individual tumors and other diseases for which a the single-cell transcriptomic profile of a normal control population is available. In line with this, the LINCS project has released datasets for iPSC-derived cardiomyocytes and motor neurons, opening up new possibilities for precision medicine not only in cancer but also in a variety of other diseases. By predicting the most effective drug and combination treatments for each patient, clinical trials can be designed to target the right populations with the responsive transcriptional phenotype, leading to more successful outcomes.”
(3) It would be insightful if the authors could discuss the potential for drug resistance in the context of thedrug combinations identified by ’retriever’. An analysis of this phenomenon could provide critical insights into the longevity and effectiveness of the proposed treatment strategies.
We agree with the reviewer that the potential for drug resistance is a critical consideration when evaluating any therapeutic strategy in cancer, especially when using drug combinations. While the current study focuses on identifying effective drug pairings using ‘retriever’, we recognize that the emergence of resistance could limit their long-term utility. We have addressed the topic within the introduction: “Nonetheless, monotherapy in cancer is highly susceptible to the development of resistance following an initial response to treatment. Combination therapy, or the simultaneous administration of multiple drugs to treat a disease, has evolved into the standard pharmacological regimen for treating complex diseases such as cancer. Combination therapy prevent tumor evolution and help inhibit the development of drug resistance in cancer, thereby improving patient survival.”
(4) Providing details regarding the computational resources necessary for the implementation of ’retriever’,along with any limitations associated with these requirements, could greatly enhance the transparency and reproducibility of the methodology. Such information would be instrumental for other researchers seeking to apply this tool in their own work.
The following paragraph was added to the data availability section of the manuscript: “The retriever package is available from the Kuijjer Lab repository https://github.com/kuijjerlab/retriever or from the CRAN repositories at https://cran.r-project.org/package=retriever, and it is implemented as an R multiplatform package that can run on standard laptops or desktops with around 16 GB of RAM, making it accessible for most users. It is designed to work on Windows, macOS, and Linux. While the package can function with modest hardware, performance may vary based on dataset size and complexity. For larger datasets, systems with more RAM or cloud-based resources may improve efficiency.”
(5) A thoughtful discussion on the ethical considerations surrounding the use of patient-derived data in thedevelopment and validation of ’retriever’ would round out the manuscript. Addressing issues of data privacy and the ethical use of such data could set a precedent for responsible research practices in the field of computational biology and personalized medicine.
We agree with the reviewer on the need of discussing the ethical considerations surrounding the use of patient-derived data in the validation, development and re-purposing of drugs for disease treatment.
The following paragraph was added to the discussion section: “We want to highlight the important ethical considerations involved in using patient-derived data for drug development and repurposing, particularly around data privacy, informed consent, and the reliability of predictive models. To protect patient privacy, it is crucial to adhere to data protection laws, such as HIPAA and GDPR, and to rigorously de-identify data to minimize the risk of re-identification. Additionally, datasets must be diverse and representative to prevent bias, ensuring that predictive models are applicable to a broad population. Computational models should undergo extensive validation before being used in clinical settings to ensure their accuracy and transparency. Ethical protocols for data sharing must also be established to respect patient autonomy and control over their data. Furthermore, continuous monitoring and validation of drug predictions are necessary to ensure treatment safety, effectiveness, and fairness.”





