Author response:
The following is the authors’ response to the original reviews.
Public Reviews:
Reviewer #1 (Public review):
Summary:
Lumen formation is a fundamental morphogenetic event essential for the function of all tubular organs, notably the vertebrate vascular network, where continuous and patent conduits ensure blood flow and tissue perfusion. The mechanisms by which endothelial cells organize to create and maintain luminal space have historically been categorized into two broad strategies: cell shape changes, which involve alterations in apical-basal polarity and cytoskeletal architecture, and cell rearrangements, wherein intercellular junctions and positional relationships are remodeled to form uninterrupted conduits. The study presented here focuses on the latter process, highlighting a unique morphogenetic module, junction-based lamellipodia (JBL), as the driver for endothelial rearrangements.
Strengths:
The key mechanistic insight from this work is the requirement of the Arp2/3 complex, the classical nucleator of branched actin filament networks, for JBL protrusion. This implicates Arp2/3-mediated actin polymerization in pushing force generation, enabling plasma membrane advancement at junctional sites. The dependence on Arp2/3 positions JBL within the family of lamellipodia-like structures, but the junctional origin and function distinguish them from canonical, leading-edge lamellipodia seen in cell migration.
Weaknesses:
The study primarily presents descriptive observations and includes limited quantitative analyses or genetic modifications. Molecular mechanisms are typically interrogated through the use of pharmacological inhibitors rather than genetic approaches. Furthermore, the precise semantic distinction between JAIL and JBL requires additional clarification, as current evidence suggests their biological relevance may substantially overlap.
We have previously analyzed the effects of different ve-cadherin (cdh5) mutant alleles on EC rearrangements (Paatero et al., 2018; Sauteur et al., 2014).These mutants show complex defects (e.g. hypersprouting, reduced contact inhibition during anastomosis) in EC behavior early in vascular tube formation. We find that analysis of JBL dynamics and function is very difficult in such situations. The use of small molecule inhibitors allows acute interventions within a defined time-window and to avoid pleiotropic effects of genetic ablations. We have expanded our discussion on the distinction between JAIL and JBL and hope that this will clarify why – in our opinion – these terms should be used differentially in different cell biological contexts (see below and lines 348-374 in the manuscript).
Reviewer #2 (Public review):
Summary:
In Maggi et al., the authors investigated the mechanisms that regulate the dynamics of a specialized junctional structure called junction-based lamellipodia (JBL), which they have previously identified during multicellular vascular tube formation in the zebrafish. They identified the Arp2/3 complex to dynamically localize at expanding JBLs and showed that the chemical inhibition of Arp2/3 activity slowed junctional elongation. The authors therefore concluded that actin polymerization at JBLs pushes the distal junction forward to expand the JBL. They further revealed the accumulation of Myl9a/Myl9b (marker for MLC) at the junctional pole, at interjunctional regions, suggesting that contractile activity drives the merging of proximal and distal junctions. Indeed, chemical inhibition of ROCK activity decreased junctional mergence. With these new findings, the authors added new molecular and cellular details into the previously proposed clutch mechanism by proposing that Arp2/3-dependent actin polymerization provides pushing forces while actomyosin contractility drives the merging of proximal and distal junctions, explaining the oscillatory protrusive nature of JBLs.
Strengths:
The authors provide detailed analyses of endothelial cell-cell dynamics through time-lapse imaging of junctional and cytoskeletal components at subcellular resolution. The use of zebrafish as an animal model system is invaluable in identifying novel mechanisms that explain the organizing principles of how blood vessels are formed. The data is well presented, and the manuscript is easy to read.
Weaknesses:
While the data generally support the conclusions reached, some aspects can be strengthened. For the untrained eye, it is unclear where the proximal and distal junctions are in some images, and so it is difficult to follow their dynamics (especially in experiments where Cdh5 is used as the junctional marker). Images would benefit from clear annotation of the two junctions. All perturbation experiments were done using chemical inhibitors; this can be further supported by genetic perturbations.
We have added annotations to several figures and paid particular attention to the proximal and distal junctions.
We have previously analyzed the effects of different ve-cadherin (cdh5) mutant alleles on EC rearrangements (Paatero et al., 2018; Sauteur et al., 2014). These mutants show complex defects (e.g. hypersprouting, reduced contact inhibition during anastomosis) in EC behavior early in vascular tube formation. We find that analysis of JBL dynamics and function is very difficult in such situations. The use of small inhibitors allows acute interventions within a defined time-window and to avoid pleiotropic effects of genetic ablations.
Reviewer #3 (Public review):
The paper by Maggi et al. builds on earlier work by the team (Paatero et al., 2018) on oriented junction-based lamellipodia (JBL). They validate the role of JBLs in guiding endothelial cell rearrangements and utilise high-resolution time-lapse imaging of novel transgenic strains to visualise the formation of distal junctions and their subsequent fusion with proximal junctions. Through functional analyses of Arp2/3 and actomyosin contractility, the study identifies JBLs as localized mechanical hubs, where protrusive forces drive distal junction formation, and actomyosin contractility brings together the distal and proximal junctions. This forward movement provides a unique directionality which would contribute to proper lumen formation, EC orientation, and vessel stability during these early stages of vessel development.
Time-lapse live imaging of VEC, ZO-1, and actin reveals that VEC and ZO-1 are initially deposited at the distal junction, while actin primarily localizes to the region between the proximal and distal sites. Using a photoconvertible Cdh5-mClav2 transgenic line, the origin of the VEC aggregates was examined. This convincingly shows that VE-cadherin was derived from pools outside the proximal junctions. However, in addition to de novo VEC derived from within the photoconverted cell, could some VEC also be contributed by the neighbouring endothelial cell to which the JBL is connected?
Yes, the green (non-converted) VE-cadherin can indeed originate from either of the two cells. The main point we want to make, based on our observations, is that the red (converted) VE-cadherin from the proximal junction (as defined by the ROI) does not contribute to the distal junction.
As seen for JAILs in cultured ECs, the study reveals that Arp2/3 is enhanced when JBLs form by live imaging of Arpc1b-Venus in conjunction with ZO-1 and actin. Therefore Arp2/3 likely contributes to the initial formation of the distal junction in the lamellopodium.
Inhibiting Arp2/3 with CK666 prevents JBL formation, and filopodia form instead of lamellopodia. This loss of JBLs leads to impaired EC rearrangements.
Is the effect of CK666 treatment reversible? Since only a short (30 min) treatment is used, the overall effect on the embryo would be minimal, and thus washing out CK666 might lead to JBL formation and normalized rearrangements, which would further support the role of Arp2/3.
We have performed washout experiments and find that the ectopic filopodia disappear when the inhibitor is removed. This experiment is shown in supplementary Figure 3 and supplementary Movies 12 and 13.
From the images in Figure 4d it appears that ZO-1 levels are increased in the ring after CK666 treatment. Has this been investigated, and could this overall stabilization of adhesion proteins further prevent elongation of the ring?
This is an interesting thought and we haven take a closer look. There is quite a bit of sample-to-sample variation in the ZO1 signal. The quantification (Author response image 1) indicates that there is no increase in the CK666 treated embryos on average.
Author response image 1.
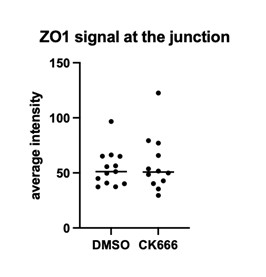
To explore how the distal and proximal junctions merge, imaging of spatiotemporal imaging of Myl9 and VEC is conducted. It indicates that Myl9 is localized at the interjunctional fusion site prior to fusion. This suggests pulling forces are at play to merge the junctions, and indeed Y 27632 treatment reduces or blocks the merging of these junctions.
For this experiment, a truncated version of VEC was use,d which lacks the cytoplasmic domain. Why have the authors chosen to image this line, since lacking the cytoplasmic domain could also impair the efficiency of tension on VEC at both junction sites? This is as described in the discussion (lines 328-332).
This line was used because it labels the entire JBL protrusion more clearly. We have also included an example using the VE-cad-Venus line (supplementary Figure 4b), which shows a Myl-Cherry pattern consistent with the other examples.
Since the time-lapse movies involve high-speed imaging of rather small structures, it is understandable that these are difficult to interpret. Adding labels to indicate certain structures or proteins at essential timepoints in the movies would help the readers understand these.
We have added annotations and labels to all movies. We have also improved annotations in several figures (i.e. Figs. 1, 2, 5, 6 and 7)
Recommendations for the authors:
Reviewing Editor Comments:
Overall, the reviewers are supportive of the manuscript but identify a number of areas where the clarity of the presented data could be improved, and further quantification could be provided to strengthen your conclusions. We would encourage you to address these minor concerns as best you can and to consider the recommendations of all three reviewers when deciding how to revise your manuscript.
Reviewer #1 (Recommendations for the authors):
Lumen formation is a fundamental morphogenetic event essential for the function of all tubular organs, notably the vertebrate vascular network, where continuous and patent conduits ensure blood flow and tissue perfusion. The mechanisms by which endothelial cells organize to create and maintain luminal space have historically been categorized into two broad strategies: cell shape changes, which involve alterations in apical-basal polarity and cytoskeletal architecture, and cell rearrangements, wherein intercellular junctions and positional relationships are remodeled to form uninterrupted conduits. The study presented here focuses on the latter process, highlighting a unique morphogenetic module, junction-based lamellipodia (JBL), as the driver for endothelial rearrangements.
JBL are described as oscillating membrane protrusions emerging at endothelial junctions, operating in a ratchet-like manner to mediate convergent cell movements. This ratchet mechanism allows endothelial cells to approach each other, thereby aligning and joining local luminal segments into a continuous vascular structure. The study employs in vivo high-resolution time-lapse imaging, a technically demanding method that captures spatiotemporal dynamics of cytoskeletal and adhesion complexes during JBL activity with unprecedented detail.
The key mechanistic insight from this work is the requirement of the Arp2/3 complex, the classical nucleator of branched actin filament networks, for JBL protrusion. This implicates Arp2/3-mediated actin polymerization in pushing force generation, enabling plasma membrane advancement at junctional sites. The dependence on Arp2/3 positions JBL within the family of lamellipodia-like structures, but the junctional origin and function distinguish them from canonical, leading-edge lamellipodia seen in cell migration.
An intriguing observation is that a novel junction arises at the distal pole of a JBL. This distal junction is formed from a pool of VE-cadherin that is spatially redistributed from regions outside the initial JBL domain. The distal junction then merges with the proximal junction through a process dependent on actomyosin contractility, as was judged by Myl9 recruitment.
The alternation between pushing forces (Arp2/3-dependent JBL protrusion) and pulling forces (actomyosin-driven junction fusion) defines JBL as a bidirectional mechanical module. Inhibition of actomyosin prevents merging of proximal and distal junctions, thereby stalling lumen continuity. This two-phase system, actin-based extension followed by actomyosin-mediated constriction, ensures both elongation and maturation of endothelial arrangements, ultimately securing vascular patency.
This manuscript represents a robust and thoughtfully executed study that advances our understanding of lumen formation during vascular development. The overarching conclusions are well substantiated, and the results section provides a clear and detailed exposition of the key findings. I appreciate the explanatory movie at the end. Nevertheless, I offer several remarks for further improvement:
(1) The fluorescent images presented are visually compelling, yet lack quantitative analysis in the initial figure. Although quantification is included in Figure 3, it is advisable to incorporate this analysis into Figure 1 as well. Early presentation of quantification will help the reader to appreciate the impact and significance of the findings from the outset.
We appreciate the reviewer’s suggestion and have now added line graphs to measure the spatiotemporal intensities of the Utrophin and ZO-1 reporters in Figure 1b. These measurements demonstrate the sequence of F-actin protrusion and subsequent junctional movement. In Figure 1a, we have added a double-headed arrow which shows the overall movement of the junction towards the dorsal side of the forming DLAV.
(2) For the fluorescence images, further quantitative analysis of membrane overlap, either in terms of width or pixel overlap, would enhance the rigor of the study. Temporal quantification of overlap may provide valuable insights into the stability and reproducibility of the process across experimental replicates.
JBL are quite heterogenous with respect to size, shape and dynamics, which makes quantifications of membrane overlap (JBL size) across experimental replicates difficult. We have published some quantifications on JBL orientation and oscillation in our previous paper (Paatero et al., 2018, Nat. comm. Figures 1 and 2), which are in agreement with our current study.
(3) When referencing the role of Arp2/3, the authors employ an ArpC1b transgenic fish. The results section should thus specifically address the involvement of ArpC1b, rather than generalizing to Arp2/3. In the discussion, it would be appropriate to speculate on the potential involvement of the complete Arp2/3 complex. Notably, the use of CK is acknowledged as a broadly accepted inhibitor of actin polymerization.
As ArpC1b is a subunit of an active Arp2/3 complex (Padrick et al., 2011), we have used an ArpC1b-Venus as a readout for Arp2/3 localization. The construct has been validated before in cell culture (Law et al., 2021) as well as in zebrafish (Malchow et al., 2024) and the spatiotemporal distribution of the reporter shown to be consistent with Arp2/3 complex. We are stating this in the results section (lines 173-178) and subsequently use the term Arp2/3 to facilitate reading of the text. In the corresponding figure legends, we are maintaining the term ArpC1b. CK666 interferes with the dimerization of Arp2 and Arp3 subunits and thus prevents activity of the Arp2/3 complex.
(4) The discussion regarding JAIL versus JBL involvement remains challenging to interpret. If JAIL structures arise from the loss of cell-cell contacts, both JAIL and JBL resemble membrane protrusions and are likely governed by similar molecular mechanisms, predominantly actin polymerization and Arp2/3 activity, with probable contribution from Rac1 signaling. The precise semantic distinction between JAIL and JBL warrants further clarification, as their biological relevance may be overlapping.
We agree with the reviewer. Below we outline the reasons why lamellipodial protrusions that emanate from cell-cell junctions should not be indiscriminately called JAIL, but that JAIL and JBL constitute different cellular activities acting in different tissue contexts. We have modified the text in the Discussion (lines 348-374).
(1) JAIL have originally been described in cell culture experiments (Abu-Taha et al., 2014). According to this and subsequent papers by the same group, local dissolution of endothelial adherens junctions (i.e. downregulation of VE-cadherin) triggers the formation of lamellipodia-like structures. These protrusions eventually retract, followed by the reestablishment of EC junctions.
(2) In our in vivo studies, we observed lamellipodial protrusions during endothelial cell rearrangements, and we call these structures JBL (Paatero et al., 2018). While JBL appear very similar to JAIL in general (i.e. regulation by Arp2/3 and its localization), we also observe two critical differences. For one, JBL form while maintaining the original (proximal) junction. Moreover, a distal junction is formed at the front edge of the JBL, leading to a “double junction” configuration. In our current manuscript, we have examined the role of actomyosin contractility and find that it correlates with and is required for the merging of proximal and distal junctions during JBL cycles. These observations indicates that the proximal and distal junctions are essential components of JBL function during endothelial cell elongation and rearrangements. These salient and distinct features prompted us to adopt the term junction-based-lamellipodia (JBL), in order to differentiate them from JAIL.
(3) We like to argue that JAIL and JBL represent similar but different lamellipodia-like protrusions. JAILs are associated with the maintenance of endothelial integrity, and control permeability and trans-endothelial cell migration, as has been suggested by several publications (Cao et al., 2017; Kipcke et al., 2025; Seebach et al., 2021; Taha et al., 2014). In contrast, JBL drive cell rearrangements, by step-wise elongation of cell junctions leading to convergent cell movements.
(4) Although JAIL have also been implicated in endothelial cell migration (Cao and Schnittler, 2019; Cao et al., 2017; Seebach et al., 2021), neither junctional patterns nor junctional dynamics have been analyzed in this context. We therefore propose that JAIL and JBL are actin-based protrusions forming at endothelial cell-cell junctions, but act in different contexts to provide cell motility (JBL) or endothelial integrity (JAIL).
(5) Some of the quantification plots, specifically in figures 5d and 6c, do not display significant differences or distribution patterns. It would be beneficial to revise these graphs to clearly represent statistical significance and underlying data distributions.
Because of the spatiotemporal heterogeneity, it is difficult to perform statistical quantifications across samples. In Figure 5c/d, we have imaged/analyzed myl9-EGFP in a mosaic situation, in which only one of interacting cells expresses high levels of myl9-EGFP. This is a rare situation and we managed to image only this example. Nevertheless, it is consistent with our other expression data of myl9-reporters and also with our previous photoconversion experiments using photoconvertible UCHD (Paatero et al., 2018, Figure 4), which shows that actin-rich JBL form at the front end of the endothelial cell in the direction of junction elongation. In Figure 5d, we have quantified the average intensity of GFP signal within the region of interest. The newly added error bars indicate the standard deviation between pixel intensities within the ROI.
In Figure 6c, we have analyzed the Myl9b-mCherry intensities and find that it is redistributed during a JBL cycle. The spatial distribution is evident from the heat-map and we have not included a standard deviation. Myl9b-mCherry levels are very heterogenous and is not possible to quantify intensities across samples. We have, however, included four more examples of Myl9b-mCherry distribution in Supplementary Figure 4. The patterns observed in these samples are consistent with those in Figure 6.
(6) The observation of myosin recruitment does not inherently imply a concomitant increase in actomyosin contractile activity. The inclusion of phospho-MLC staining would considerably strengthen the evidence for enhanced actomyosin activity.
This is a good suggestion and we have extensively tried different anti-P-Myl antibodies (and protocols), but did not get them to work reliably on zebrafish embryos. We therefore rely on published work that has established the correlation between the recruitment of myosin light chain and increased actomyosin tension (Fernandez-Gonzalez et al., 2009; Munjal et al., 2015).
Reviewer #2 (Recommendations for the authors):
(1) Figure 1a is not described/mentioned in the Results.
The have corrected this (lines 102-108). We have also added measurements to better present the different dynamics of F-actin (UCHD) and ZO1 within the JBL and the relative endothelial cell movements (see Figure 1b), as suggested by reviewer#1.
(2) In Figure 3a, the authors claim that Arp2/3 is deposited at the distal side of the junction ring. While it is clear where the proximal junction is (ZO1-rich), the distal junction is less so (hardly any ZO1). It is therefore difficult to agree based on this time-lapse imaging that Arpc1b-Venus is at the distal junction. Can the authors please include panels showing merged channels and annotate where the proximal and distal junctions are?
The activation of the Arp2/3 complex and the formation of the distal junction are sequential events. We see that ArpC1b oscillates with an accumulation at the onset and during JBL protrusion. In contrast, the distal junction is formed when the protrusive activity has been stopped. One caveat of the analysis shown in Figure 3a is that our ZO1 reporters label the distal junction only very weakly – this is in particular the case for the ZO1-tdTomato knock-in. The distal junction is better visible in VE-cadherin and UCHD reporters, as shown in Figures 5 to 7.
(3) In Figures 3b and c, it is also difficult to distinguish proximal and distal junctions in these images. Please mark the boundaries in the image panels (Figure 3b) and indicate on the x-axis where the proximal and distal junctions are (Figure 3c).
In Figure 3b, we show ArpC1b-Venus and mRuby-UCHD side-by-side. This Figure demonstrates that the Arp2/3 complex maintains its position at the front of the JBL during the protrusive phase (always distal to the UCHD signal). The imaging is done at very short intervals (1/30sec), which makes it difficult to follow entire oscillations due to photo-bleaching of the ArpC1b reporter.
(4) The treatment of CK666 resulted in perturbed localization of Arpc1b-Venus. Therefore, the inhibition of junctional elongation can also be explained by the mislocalization of Arp2/3, rather than the inhibition of Arp2/3 activity at the junctions. Can the authors discuss this or perform another experiment that is more specific to manipulating Arp2/3 activity?
CK666 is a well-established inhibitor of Arp2/3. Structural and functional analyses have shown that CK666 interferes with the interaction between Arp2 and Arp3, thereby preventing the activation of the complex (Hetrick et al., 2013; Padrick et al., 2011). We therefore conclude that the phenotypes we observe in CK666 treatment are due to Arp2/3 inhibition.
It is possible that CK666 prevents ArpC1b binding to the Arp2/3 complex. However, published work suggests that ArpC1b can bind to Arp2/3 also in its inactive state (Chou et al., 2022). Thus, we can only speculate why we lose localization ArpC1b under CK666. We prefer not to do so.
(5) In Figures 5d and 6c, is the quantification of Myl9 intensity of one cell only? If so, can the authors show the dynamics of average Myl9 intensity i) between forwarding and non-forwarding JBL poles and ii) as the proximal and distal junctions merge several endothelial cells?
Figure 5c/d depicts two interacting cells, expressing different levels of Myl9a-EGFP. This is a rare experimental situation and we managed to image only this example. We quantified the average signal at both poles of the junctional ring within a region of interest. The newly added error bars indicate the standard deviation between pixel intensities within the ROI. The analysis has been done on immunofluorescent images, therefore a dynamic analysis over time is not possible.
In Figure 6c, we have analyzed the Myl9b-mCherry intensities and find that it is redistributed during a JBL cycle. The spatial distribution is evident from the heat-map and we have not included a standard deviation. Myl9b-mCherry levels are very heterogenous and is not possible to quantify intensities across samples. We have, however, included four more examples of Myl9b-mCherry distribution in Supplementary Figure 4. The patterns observed in these samples are consistent with those in Figure 6.
(6) Figure 5. The 'f' in the figure legend should be 'e' since there is no panel 'f'.
We have corrected this.
(7) Figure 7. As the boundaries for proximal and distal junctions are not always clear, especially when Cdh5 appears as clusters, how do you determine where the two junctions are in order to measure the interjunctional space? Please offer a clearer explanation in the Methods.
We have added the following in the M&M.
“Junctional merging tracking
Speed of junctional merge was evaluated by monitoring isolated junctional rings during DLAV formation. Inhibitor treatment Y-27632 (75 μM) or DMSO (1%) were applied 30 min before mounting. The same concentrations of chemicals were applied to the low-melting-point agarose mounting medium and the E3 medium on top of it before imaging and imaging the junctions for 10-15 min on an Olympus SpinSR spinning disc microscope. Distances were measured using Fiji software. In each frame, the interjunctional distance was defined as the maximum distance between the proximal and distal junctions. A line was manually drawn between the proximal and distal junctions in Fiji, and its length was recorded. The same proximal and distal junction landmarks were used consistently across all time points.”
(8) One would think that upon the inhibition of junctional mergence (by ROCK inhibition), actin polymerization would persist to push the distal junction forward to elongate the JBL. However, there is instead a decrease in junctional elongation (Figure 7b). Can the authors speculate why? Additionally, junction elongation can probably be achieved by continuous "pushing" of the distal junction alone (through actin polymerization). Can the authors speculate why there is a need/what is the benefit of merging proximal and distal junctions for junction elongation?
These are all very interesting questions, but they are quite complex and would require extensive and speculative answers, which is outside the scope of this study. Nevertheless, here are a few quick thoughts on these issues.
(1) When endothelial cells elongate, they have to overcome tensile forces at the junctions (generated by the subjunctional actomyosin belt). JBL are providing a tractive and deforming force, which overcomes the tensile force and thus promotes junctional elongation.
(2) The distal junction is then providing an anchor to which the actin cytoskeleton can attach. The space between proximal and distal junction becomes a compartment of local actomyosin contraction, which provides the force for the ratchet to move the proximal junction forward junctional mergence.
(3) Thus, it is not the protrusion (pushing) itself that elongates the cell but the elongation of the junction (driven by actomyosin contraction)!
(4) The maintenance of the proximal junction is most likely needed to ensure endothelial integrity during the JBL cycles.
(5) How the frequency and the size of JBLs is regulated is not known. One possible player that might be involved is an internal clock mechanism (e.g. a feedback loop via small GTPases (such as Rac) Arp2/3 regulation). Another possibility is that JBL size is limited by it sweeping up basally localized VE-cadherin (in cis-configuration). Increasing cell-cell adhesion (by VE-cad trans-interactions between the JBL and the underlying cell) eventually stop the protrusion. It is also possible that an cell-autonomously controlled mechanism of F-actin polymerization (actin pulses) are involved in the regulation of the JBC cycle length.
(9) The animation showing the molecular mechanism of JBL function during endothelial junction elongation (Video 25) is very helpful in understanding the dynamic coupling between junctional proteins, actomyosin cytoskeleton, and junction remodelling. However, I wonder why there are no Myosin II proteins binding to the actin bundles during the merging of proximal and distal junctions (between 0:25 and 0:28), since this is one of the main findings by the authors in this study.
Since we show two JBL cycles, we want to spread the information over both of them.
References:
Cao, J. and Schnittler, H. (2019). Putting VE-cadherin into JAIL for junction remodeling. J. Cell Sci. 132.
Cao, J., Ehling, M., März, S., Seebach, J., Tarbashevich, K., Sixta, T., Pitulescu, M. E., Werner, A. C., Flach, B., Montanez, E., et al. (2017). Polarized actin and VE-cadherin dynamics regulate junctional remodelling and cell migration during sprouting angiogenesis. Nat. Commun. 8, 1–20.
Chou, S. Z., Chatterjee, M. and Pollard, T. D. (2022). Mechanism of actin filament branch formation by Arp2/3 complex revealed by a high-resolution cryo-EM structure of the branch junction. Proc. Natl. Acad. Sci. U. S. A. 119, e2206722119.
Fernandez-Gonzalez, R., Simoes, S. de M., Röper, J. C., Eaton, S. and Zallen, J. A. (2009). Myosin II Dynamics Are Regulated by Tension in Intercalating Cells. Dev. Cell 17, 736–743.
Hetrick, B., Han, M. S., Helgeson, L. A. and Nolen, B. J. (2013). Small molecules CK-666 and CK-869 inhibit actin-related protein 2/3 complex by blocking an activating conformational change. Chem. Biol. 20, 701–712.
Kipcke, J. P., Odenthal-Schnittler, M., Aldirawi, M., Franz, J., Bojovic, V., Seebach, J. and Schnittler, H. (2025). TNF-α induces VE-cadherin-dependent gap/JAIL cycling through an intermediate state essential for neutrophil transmigration. Front. Immunol. 16,.
Law, A. L., Jalal, S., Pallett, T., Mosis, F., Guni, A., Brayford, S., Yolland, L., Marcotti, S., Levitt, J. A., Poland, S. P., et al. (2021). Nance-Horan Syndrome-like 1 protein negatively regulates Scar/WAVE-Arp2/3 activity and inhibits lamellipodia stability and cell migration. Nature Communications 2021 12:1 12, 5687-.
Malchow, J., Eberlein, J., Li, W., Hogan, B. M., Okuda, K. S. and Helker, C. S. M. (2024). Neural progenitor-derived Apelin controls tip cell behavior and vascular patterning. Sci. Adv. 10, 1174.
Munjal, A., Philippe, J. M., Munro, E. and Lecuit, T. (2015). A self-organized biomechanical network drives shape changes during tissue morphogenesis. Nature 524, 351–355.
Paatero, I., Sauteur, L., Lee, M., Lagendijk, A. K., Heutschi, D., Wiesner, C., Guzmán, C., Bieli, D., Hogan, B. M., Affolter, M., et al. (2018). Junction-based lamellipodia drive endothelial cell rearrangements in vivo via a VE-cadherin-F-actin based oscillatory cell-cell interaction. Nat. Commun. 9,.
Padrick, S. B., Doolittle, L. K., Brautigam, C. A., King, D. S. and Rosen, M. K. (2011). Arp2/3 complex is bound and activated by two WASP proteins. Proc. Natl. Acad. Sci. U. S. A. 108, E472–E479.
Sauteur, L., Krudewig, A., Herwig, L., Ehrenfeuchter, N., Lenard, A., Affolter, M. and Belting, H. G. (2014). Cdh5/VE-cadherin promotes endothelial cell interface elongation via cortical actin polymerization during angiogenic sprouting. Cell Rep. 9, 504–513.
Seebach, J., Klusmeier, N. and Schnittler, H. (2021). Autoregulatory “Multitasking” at Endothelial Cell Junctions by Junction-Associated Intermittent Lamellipodia Controls Barrier Properties. Front. Physiol. 11,.
Taha, A. A., Taha, M., Seebach, J. and Schnittler, H. J. (2014). ARP2/3-mediated junction-associated lamellipodia control VE-cadherin-based cell junction dynamics and maintain monolayer integrity. Mol. Biol. Cell 25, 245–256.




