Aven recognition of RNA G-quadruplexes regulates translation of the mixed lineage leukemia protooncogenes
Figures
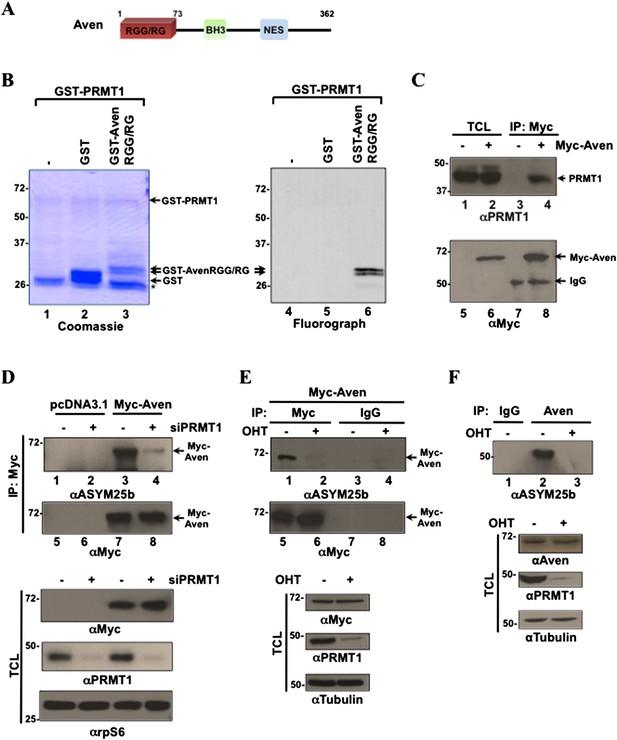
Aven is a substrate of PRMT1.
(A) Schematic diagram of Aven with its RGG/RG motif, putative BH3 domain, and nuclear export sequence (NES). (B) In vitro methylation assay with GST-PRMT1 and GST-AvenRGG/RG with (3H)-S-adenosyl-L-methionine as the methyl donor (n > 4). Proteins were resolved by SDS-PAGE, stained with Coomassie Blue (left), and analyzed by fluorography (right). The migration of the molecular mass markers is shown on the left in kDa and the migration of the GST-PRMT1, glutathione-S-transferase (GST), GST-AvenRGG/RG proteins is indicated with arrows. The asterisk (*) denotes degraded proteins from the GST-PRMT1 preparation. (C) HEK293T cells transfected with empty vector pcDNA3.1 (lanes 1, 3, 5, 7) or Myc-Aven-pcDNA3.1 (lanes 2, 4, 6, 8) were lysed and immunoprecipitated (IP) with anti-Myc antibodies (lanes 3, 4, and 7, 8). The total cell lysates (TCL) and the bound proteins were resolved by SDS-PAGE and immunoblotted with anti-PRMT1 (lanes 1–4) or −Myc antibodies (5–8). TCL denotes input TCL and IgG represents the heavy chain of immunoglobulin G. (D) HEK293T cells were cotransfected with siGFP (-siPRMT1) or siPRMT1 (+siPRMT1) and pcDNA3.1 or Myc-Aven plasmids. After 48 hr, the cells were lysed and IP with anti-Myc antibodies, the proteins were resolved by SDS-PAGE and immunoblotted with anti-ASYM25b (lanes 1–4), and anti-Myc antibodies (lanes 5–8). TCL were immunoblotted with anti-Myc, anti-PRMT1, and anti-rpS6 antibodies. The latter was to control for equal loading. (E) PRMT1FL/−;CreERT mouse embryo fibroblasts (MEFs) treated with 4-hydroxytamoxifen (OHT) for 6 days or left untreated were transfected with Myc-Aven followed by anti-Myc antibody immunoprecipitations and the methylation monitored by immunoblotting with ASYM25b (lanes 1–4) or anti-Myc antibodies (lanes 5–8). TCL were immunoblotted with anti-PRMT1, anti-Aven, and anti-tubulin antibodies as indicated. (F) PRMT1FL/−;CreERT MEFs treated with OHT for 6 days or left untreated were lysed and IP with anti-Aven (ab77014) or IgG antibodies. Immunoprecipitates were blotted with ASYM25b. TCL were immunoblotted with anti-PRMT1, anti-Aven, and anti-tubulin antibodies as indicated.
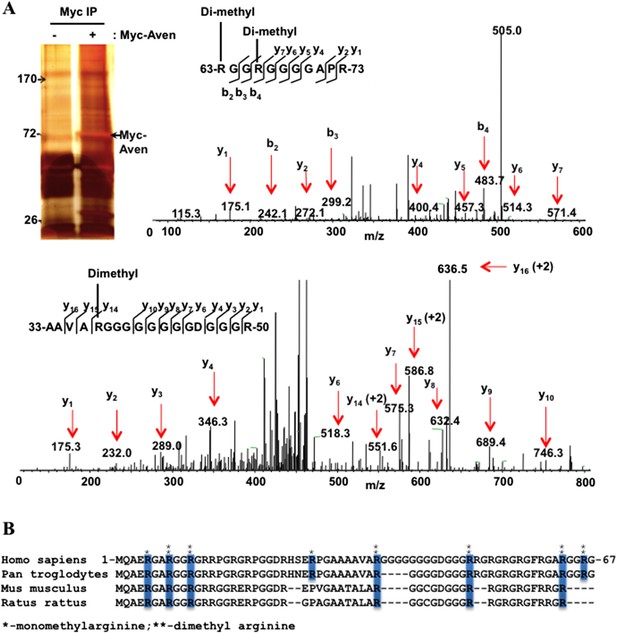
Aven harbors dimethylarginines within its RGG/RG motif.
(A) Immunoprecipitates of Myc-Aven transfected HEK293T cells resolved by SDS-PAGE. The molecular mass markers are indicated in kDa. Mass spectrometry profile of Aven. LC-MS/MS analysis of the excised Myc-Aven band. The sequence of Aven from residues 63 to 73 is shown. LC-MS/MS analysis revealed the presence of a modified peptide RGGRGGGGAPR containing dimethylated R63 and R66. Analysis of the Aven peptide from residues 33 to 50 is shown as well as the dimethylation of R37. Similar analysis identified R8, R50, and R11 to be dimethylated (not shown). (B) The alignment of Aven N terminus from various eukaryotic species. The mono- (*) and di-methylated (**) arginine residues identified by LC-MS/MS analysis and conserved across various eukaryotic species are indicated with blue boxes.
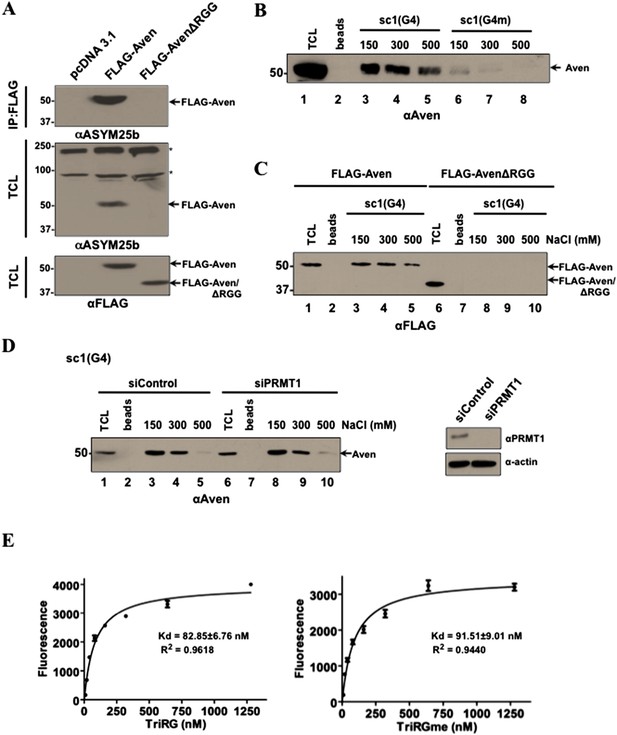
Aven binds G4 RNA sequences in an arginine methylation independent manner.
(A) U2OS cells transfected with pcDNA3.1, or expression vectors encoding FLAG-Aven or FLAG-AvenΔRGG were IP with anti-FLAG antibodies and immunoblotted with ASYM25b or anti-FLAG antibodies as indicated. The molecular mass markers are shown on the left in kDa and the migration of FLAG-Aven and FLAG-AvenΔRGG is shown. The asterisks (*) denote unknown arginine methylated proteins. (B) Biotinylated sc1 G4 bound to Streptavidin were incubated with HEK293T cell lysates. The bound proteins were washed with increasing NaCl (mM) as indicated and visualized by SDS-PAGE followed by immunoblotting with anti-Aven antibodies. (C) Biotinylated sc1 G4 or (G4m) bound to Streptavidin was incubated with cellular lysates expressing FLAG-Aven and FLAG-AvenΔRGG and detected as in panel B. (D) Biotinylated sc1 G4 RNA bound to Streptavidin beads was used to pull-down Aven from PRMT1-depleted HEK293T cells. Aven binding was performed as in panel B. PRMT1 depletion was confirmed by immunoblotting. (E) Biotinylated methylated and unmethylated Aven TriRG peptides were pre-bound on Streptavidin plates and were incubated with fluorescein-labeled sc1 G4 RNA. The bound RNA was quantified by measuring fluorescence at 521 nm. The experiment was performed twice in triplicates.
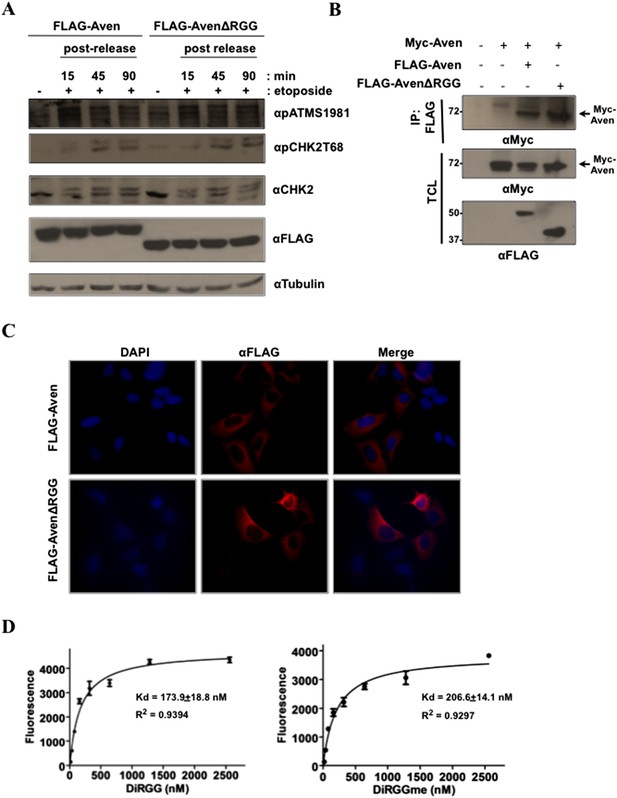
Aven RGG/RG motif binds RNA and does not regulate ATM activation, nor Aven cellular localization.
(A) To investigate the role of RGG/RG motif in ataxia telangiectasia-mutated (ATM) activation, U2OS cells were transfected with FLAG-Aven and FLAG-AvenΔRGG. Transfected cells were treated with etoposide (50 ng/ml) for 30 min. Lysates collected at various time points post-treatment were separated by SDS-PAGE and immunoblotted with anti-pATM S1981, anti-FLAG, anti-pCHK2T68, anti-CHK2, and anti-tubulin antibodies. The experiment was performed three times and a representative experiment is shown. (B) U2OS cells were co-transfected with Myc-Aven and either FLAG-Aven or FLAG-AvenΔRGG. Immunoprecipitation was performed with anti-FLAG agarose beads and the membranes were immunoblotted with anti-FLAG and anti-Myc antibodies. 10% of the lysates were shown in the bottom panel to confirm the expression of the transfected constructs. The experiment was performed twice. (C) U2OS cells were transfected with FLAG-Aven and FLAG-AvenΔRGG. The cells were fixed and labeled for immunofluorescence with anti-FLAG antibodies. The experiment was performed three times. (D) Biotinylated methylated and unmethylated Aven DiRGG peptides were pre-bound on Streptavidin plates and were incubated with fluorescein-labeled sc1 G4 RNA. The bound RNA was quantified by measuring measuring fluorescence at 521 nm.
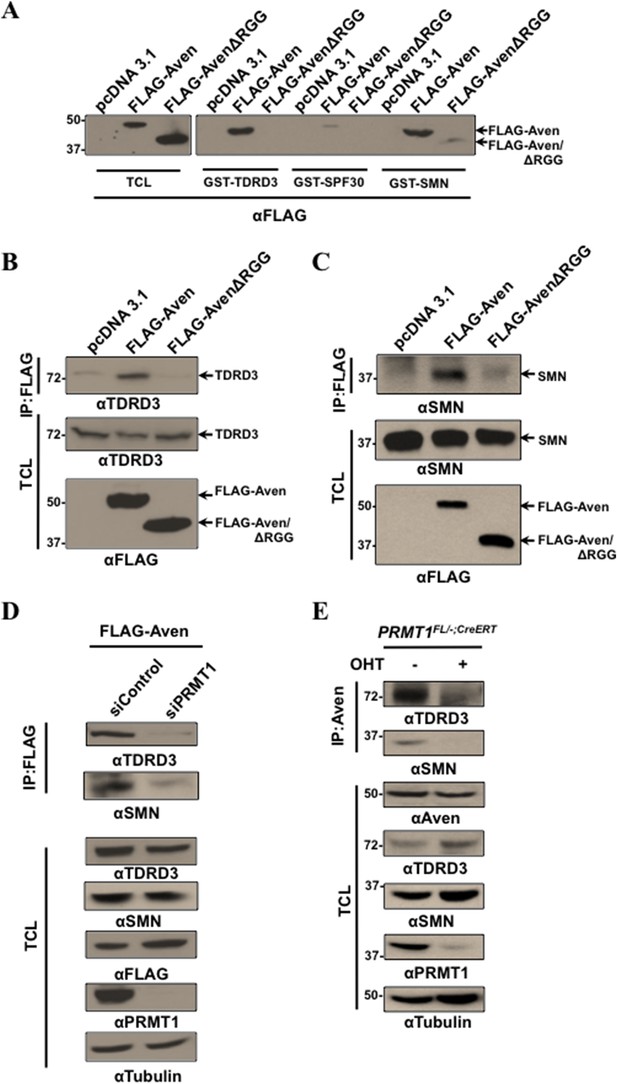
Tudor domains of TDRD3 and SMN recognize methylated Aven.
(A) Recombinant Tudor domains of TDRD3, SPF30, and SMN were fused GST and used in ‘pull-down’ assays with HEK293T lysates expressing pcDNA3.1 (control), FLAG-Aven, or FLAG-AvenΔRGG. The bound proteins were separated by SDS-PAGE and immunoblotted with anti-FLAG antibodies. (B, C) Lysates from HEK293T lysates expressing pcDNA3.1 (control), FLAG-Aven, or FLAG-AvenΔRGG were IP with anti-FLAG antibodies. Co-immunoprecipitation of endogenous TDRD3 and SMN was detected by immunoblotting. (D) Aven interaction with TDRD3 and SMN was reduced in cells deficient for PRMT1 using siRNAs. FLAG-Aven was co-expressed with either siControl or siPRMT1 in U2OS cells. Anti-FLAG antibody immunoprecipitations were performed and the presence of endogenous TDRD3 and SMN monitored by immunoblotting following separation by SDS-PAGE. (E) PRMT1FL/−;CreERT MEFs treated with OHT for 6 days or left untreated were lysed and IP with anti-Aven antibodies. Co-immunoprecipitation of endogenous TDRD3 and SMN was detected by immunoblotting (upper panels).
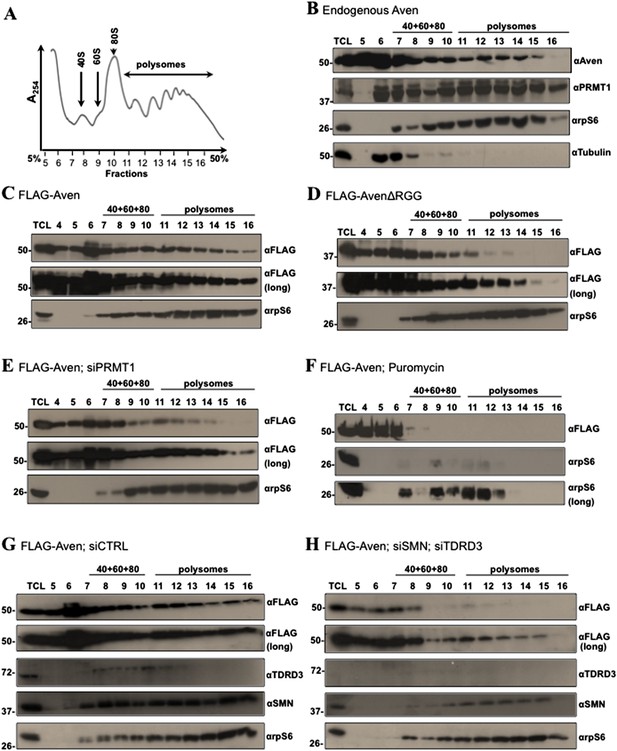
Methylation of Aven and its association with TDRD3 and SMN is required for polysomal localization.
(A) Cytoplasmic extracts from HEK293T cells were sedimented by centrifugation on a 5–50% sucrose gradient. Polysome profiles were obtained by continuous monitoring of UV absorbance at 254 nm. 40S, 60S, and 80S indicate the positions of the respective ribosomal subunits and the monomer on the gradient. (B) The distribution of endogenous Aven and PRMT1 across the gradient of panel A was monitored by immunoblotting. Ribosomal protein rpS6 was used as a loading control, whereas β-tubulin served as a cytoplasmic marker. (C–H) The distribution of FLAG-Aven or FLAG-AvenΔRGG across the gradient was monitored by immunoblotting as well as FLAG-Aven in siControl, siPRMT1, siSMNsiTDRD3, or with puromycin treatment. Both short (5 s, panels C–H) and long exposures (30 s, panels C–H) are shown. rpS6 was used as a loading control. The exposure time was determined using a standard curve with increasing amounts of lysates expressing FLAG-Aven immunoblotted with anti-FLAG antibodies for various times. Each polysomal profile experiment was performed independently three times.
-
Figure 4—source data 1
Quantitative mass spectrometry of Aven and AvenΔRGG interacting proteins.
- https://doi.org/10.7554/eLife.06234.009
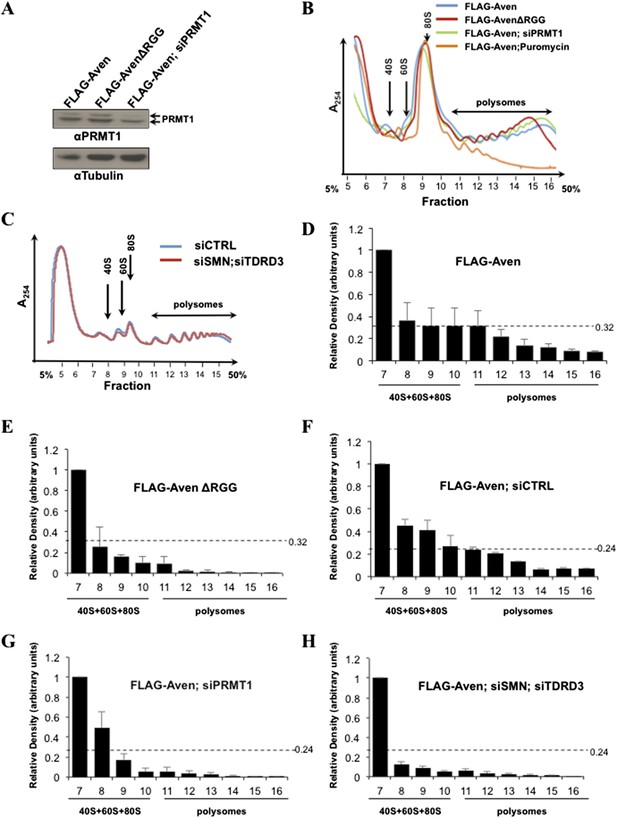
Polysomal profiles of siRNA-treated cells and quantification of FLAG-Aven and FLAG-AVENΔRGG in polysomal fractions.
(A) Immunoblots confirming the knockdown of PRMT1 and β-Tubulin was used as a loading control. The knockdown was ∼2.7-fold, as assessed by densitometric scanning. (B) Overlap of polysome profiles of cells overexpressing FLAG-Aven, FLAG-AvenΔRGG, FLAG-AVEN; siPRMT1, FLAG-AVEN; puromycin treated. Cytoplasmic extracts from the indicated cells were sedimented by centrifugation on a 5–50% sucrose gradient, shown as fraction numbers 5 to 16. Polysome profiles were obtained by continuous monitoring of UV absorbance at 254 nm. 40S, 60S, and 80S indicate the positions of the respective ribosomal subunits and the monomer on the gradient. (C) Overlap of polysome profiles of cells overexpressing FLAG-Aven; siGFP (siCTRL) or FLAG-AVEN; siSMNsiTDRD3. (D–H) Quantification of FLAG-Aven in each fraction of two representative polysome experiments using densitometry. Relative FLAG-Aven expression levels were normalized to rps6, and values expressed in terms of arbitrary densitometric units were shown as fold change of the most abundant fraction. Dashed lines indicate the densitometric units of the most abundant fraction in polysomes.
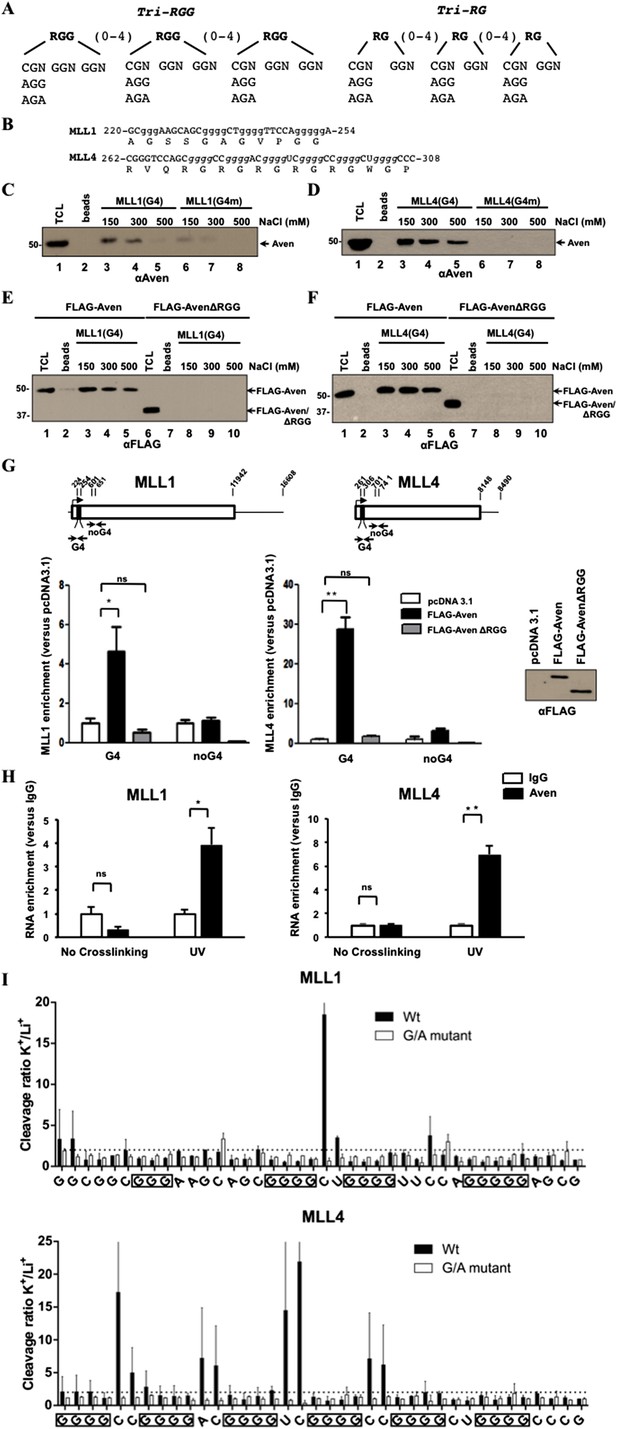
Aven RGG/RG motif binds G4 RNA structures of MLL1 and MLL4.
(A, B) RNA sequences of the RGG/RG motifs and the PG4 motifs of MLL1 and MLL4. (C, D) Biotinylated MLL1 G4 or a mutant sequence (G4m), biotinylated MLL4 G4 or a mutant sequence (G4m) bound to Streptavidin beads were incubated with HEK293T cell lysates. The bound proteins were washed with increasing concentrations of NaCl and visualized by SDS-PAGE followed by immunoblotting with anti-Aven antibodies. (E, F) HEK293T cells expressing FLAG-Aven and FLAG-AvenΔRGG were processed as in panel C, D except the bound proteins were visualized by immunoblotting with anti-FLAG antibodies. (G) Photocrosslinking IP experiments were performed using anti-FLAG antibodies. The bound RNA was analyzed in triplicate from two biological replicates by RT-qPCR with the primers spanning the PG4 sequence or a sequence ∼300 nucleotides downstream. The levels of bound RNA in immunoprecipitates were normalized to the levels of the total RNA in the input. Mean values are expressed as fold enrichment over pcDNA3.1. Error bars represent ±SEM. *p < 0.05, **p < 0.001, n.s. non-significant. The experiment was performed twice. (H) Photocrosslinking IP experiments were performed on HEK293T cells using anti-Aven antibodies. The bound RNA was analyzed in triplicates by real-time RT-PCR with the primers spanning the PG4 sequence, as indicated in panel G. The level of bound RNA in immunoprecipitates was normalized to the levels of the total RNA in the input. Mean values are expressed as fold enrichment over IgG. Error bars represent ±SEM. *p < 0.05, **p < 0.001, n.s. non-significant. The experiment was performed twice. (I) In-line probing of MLL1 and MLL4 PG4. The nucleotide sequence of the mixed lineage leukemias (MLLs) PG4 is shown below, the boxed guanines represent the predicted G-tracks. K+/Li+ ratios of the band intensities of the MLLs G4 (black) and G/A-mutant (white) for each nucleotide are shown. Error bars represent ± standard deviation. The experiment was performed twice. The dashed line represents a twofold change, an arbitrary set threshold that indicates G4 formation when exceeded.
-
Figure 5—source data 1
G4 sequences in coding regions of mRNAs.
- https://doi.org/10.7554/eLife.06234.012
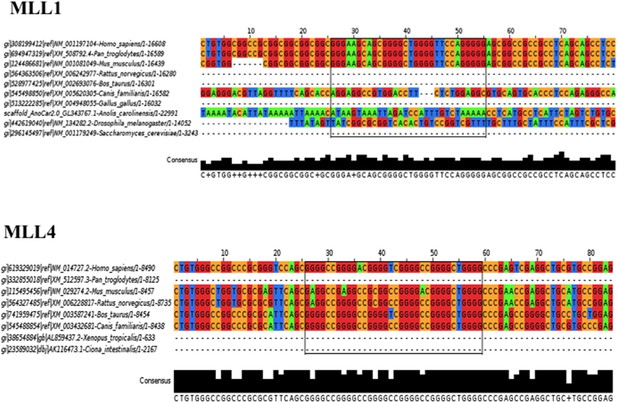
Sequence conservation of the MLL1 and MLL4 PG4 sequences.
https://doi.org/10.7554/eLife.06234.013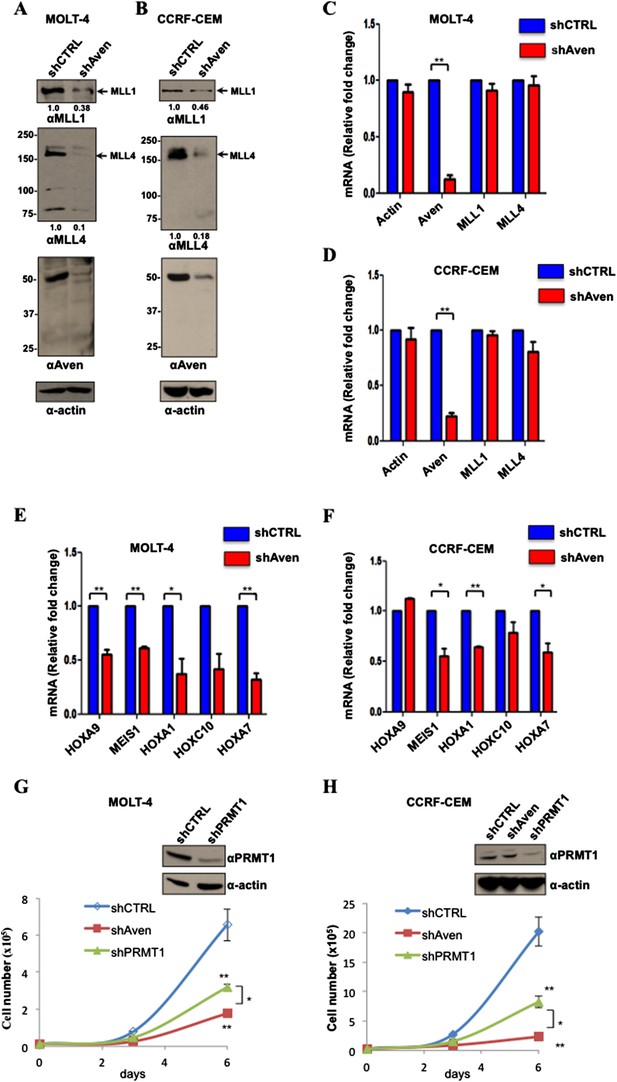
Aven regulates MLL1 and MLL4 protein expression required for leukemic cell survival.
(A, B) Cellular lysates from MOLT-4 and CCRF-CEM cells stabling expressing shCTRL or shAven were separated by SDS-PAGE and immunoblotted with anti-MLL1, anti-MLL4, anti-Aven, and β-actin antibodies. The experiment was performed three times. (C–F) RT-qPCR of the indicated mRNAs was performed from RNA isolated from shCTRL and shAven MOLT-4 and CCRF-CEM cells and expressed as a relative fold change normalized to rpS6 levels. Error bars ±SEM is shown. The significance was measured by the Student's t-test and defined as *p < 0.05, **p < 0.001, n = 3. (G, H) Proliferation curves for shControl (CTRL), shPRMT1, and shAven MOLT-4 and CCRF-CEM cells are shown. Immunoblots confirm the depletion of PRMT1 in MOLT-4 and CCRF-CEM cells. Error bars ± standard deviation is shown. The data were analyzed using ANOVA (Analysis of Variance) followed by post hoc comparison using Tukey test. *p < 0.05, **p < 0.001, n = 3.
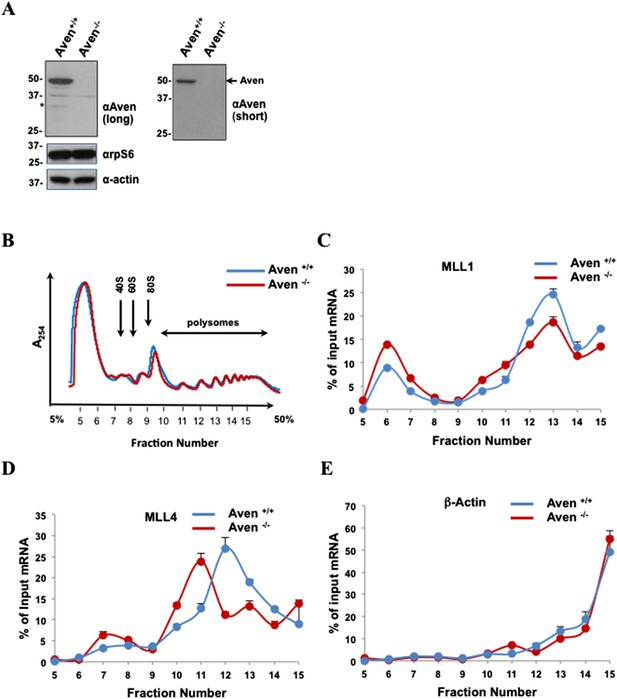
Aven regulates polysomal association of MLL1 and MLL4, but not β-actin mRNA.
(A) Aven-deficient HEK293T cells were generated by CRISPR/Cas9. Stable clones were obtained Aven+/+ (clone #7) and Aven−/− (clone #2). Anti-Aven, anti-rpS6, and anti-β-actin immunoblots of TCL are shown. The asterisks denote a minor Aven species of lower molecular mass. The band at ∼37 kDa is a non-specific band recognized by the anti-Aven antibody. n = 3. (B) Polysome profiles of Aven+/+ and Aven−/− HEK293T cells are shown. Cytoplasmic extracts from HEK293T cells were sedimented by centrifugation on a 5–50% sucrose gradient, shown as fraction numbers 5–15. Polysome profiles were obtained by continuous monitoring of UV absorbance at 254 nm. 40S, 60S, and 80S indicate the positions of the respective ribosomal subunits and the monomer on the gradient. (C–E) The indicated polysomal fractions were isolated, the RNA purified, and the presence of MLL1, MLL4, or β-actin was quantified by qRT-PCR. mRNAs in each fraction are represented as the percentage of input. Error bars represent ±SEM, n = 5.
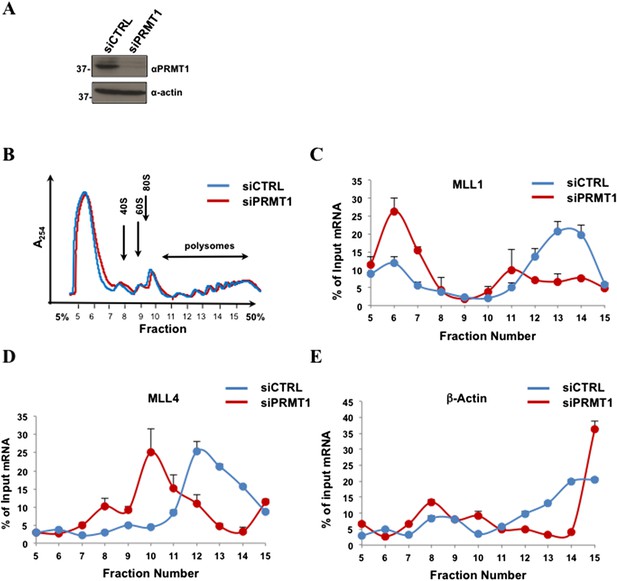
PRMT1 is required for the polysomal association of MLL1 and MLL4, but not β-actin mRNA.
(A) PRMT1 was depleted by siRNA and cell extracts were immunoblotted with anti-PRMT1 or anti-β-actin antibodies. (B) Polysome profiles of siGFP (siCTRL) or siPRMT1 HEK293T cells are shown. Cytoplasmic extracts from HEK293T cells were sedimented by centrifugation on a 5–50% sucrose gradient, shown as fraction numbers 5–15. Polysome profiles were obtained by continuous monitoring of UV absorbance at 254 nm. 40S, 60S, and 80S indicate the positions of the respective ribosomal subunits and the monomer on the gradient. (C–E) The indicated polysomal fractions were isolated, the RNA purified, and the presence of MLL1, MLL4, or β-actin was quantified by qRT-PCR. mRNAs in each fraction are represented as the percentage of input. Error bars represent ±SEM, n = 2.
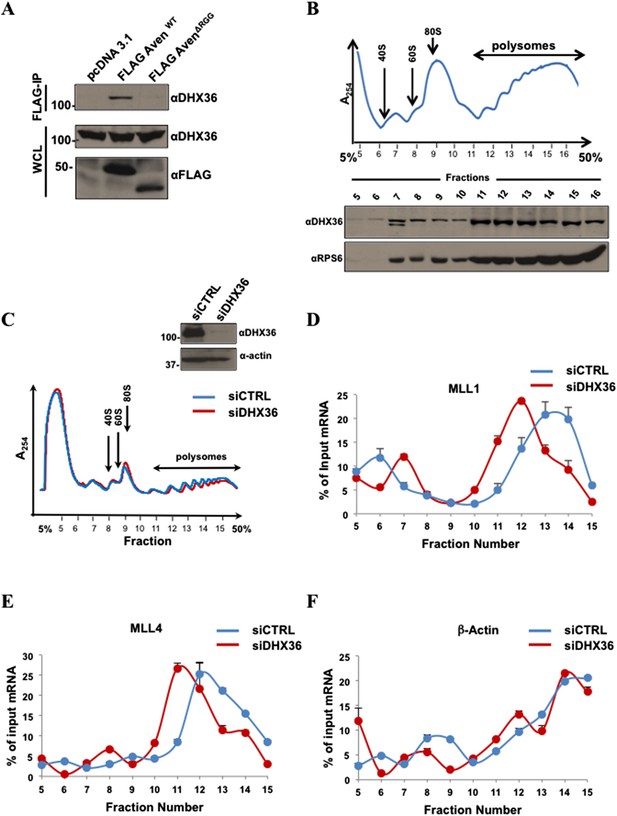
DHX36 is required for MLL1 and MLL4 mRNA polysomal association.
(A) HEK293T cells expressing FLAG-Aven and the FLAG-AvenΔRGG were IP with anti-FLAG agarose beads and the bound proteins were immunoblotted with anti-DHX36 antibodies. TCL were immunoblotted with anti-DHX36 and anti-FLAG antibodies as indicated. (B) Proteins from the polysomal fractions isolated from HEK293T cells were TCA precipitated, separated by SDS-PAGE, and immunoblotted with anti-DHX36 and anti-rpS6 antibodies. The experiment was performed n = 4 times and a typical polysomal profile is shown. (C) Immunoblots of TCL from siGFP (CTRL) and siDHX36-transfected HEK293T cells are shown. Polysome profiles siCTRL and siDHX36-transfected HEK293T cells. Cytoplasmic extracts from HEK293T cells were sedimented by centrifugation on a 5–50% sucrose gradient, shown as fraction numbers 5 to 15. Polysome profiles were obtained by continuous monitoring of UV absorbance at 254 nm. 40S, 60S, and 80S indicate the positions of the respective ribosomal subunits and the monomer on the gradient. (D–F) The indicated polysomal fractions were isolated, total RNA isolated, and the presence of MLL1, MLL4, or β-actin was quantified by RT-qPCR. mRNAs in each fraction are represented as the percentage of input. Error bars represent ±SEM, n = 2.
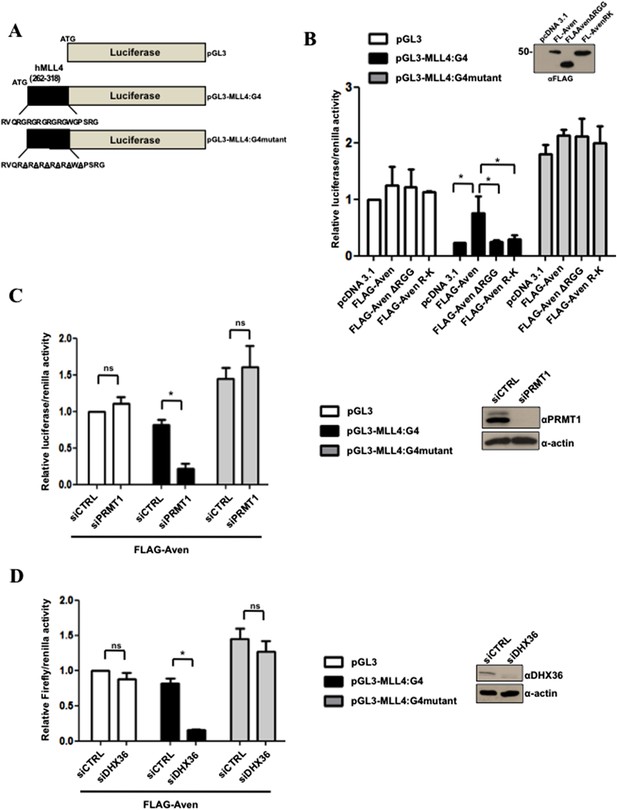
PRMT1 and Aven RGG/RG motif required for optimal translation of MLL4 G4 sequence.
(A) Schematic of the luciferase reporter plasmid pGL3, as well as the chimeric pGL3-MLL4-G4 and pGL3-MLL4-G4mutant. pGL3-MLL4-G4 harbors the human MLL4 G4 sequence nucleotide 262 to 318 inserted in-frame at the N-terminus of luciferase, while pGL3-MLL4-G4mutant contains glycine to alanine mutations that disrupts the G4 structure. (B) Aven−/− HEK293T cells were transfected with the following reporter genes pGL3, pGL3-MLL4-G4, or pGL3-MLL4-G4mutant and pRenilla as well as pcDNA3.1, FLAG-Aven, FLAG-AvenΔRGG, or FLAG-AvenR-K. The cells were harvested 24 hr post-transfection and dual luciferase assays were performed. The relative luciferase/Renilla ratio was normalized to 1.0 in pGL3 pcDNA3.1 transfected cells. Extracts were immunoblotted with anti-FLAG antibodies to confirm Aven, AvenΔRGG, or AvenR-K expression. Error bars represent standard deviation values. The experiments were performed three independent times (n = 3) and each independent experiment was performed in technical triplicates. The significance was measured by ANOVA followed by post hoc comparison using Tukey test. *p < 0.05. (C) HEK293T cells were co-transfected with FLAG-Aven and either siGFP (siCTRL) or siPRMT1 along with the following reporter genes pGL3, pGL3-MLL4-G4, or pGL3-MLL4-G4mutant and pRenilla. The cells were harvested 24 hr post-transfection and dual luciferase assays were performed. The relative luciferase/Renilla ratio was normalized to 1.0 in pGL3 siCTRL transfected cells. Extracts were immunoblotted with anti-PRMT1 or anti-β-actin antibodies, as indicated. Error bars represent standard deviation values. The experiments were performed three independent times (n = 3) and each independent experiment was performed in technical triplicates. The significance was measured by ANOVA followed by post hoc comparison using Tukey test. *p < 0.05, n.s. non-significant. (D) HEK293T cells were co-transfected with FLAG-Aven and either siGFP (siCTRL) or siDHX36 along with the following reporter genes pGL3, pGL3-MLL4-G4, or pGL3-MLL4-G4mutant and pRenilla. The cells were harvested 24 hr post-transfection and dual luciferase assays were performed. The relative luciferase/Renilla ratio was normalized to 1.0 in pGL3 siCTRL transfected cells. Extracts were immunoblotted with anti-DHX36 or anti-β-actin antibodies, as indicated. The error bars represent ± the standard deviation. Experiments were performed three times (n = 3) and each experiment was analyzed in triplicates. The significance was measured by ANOVA followed by post hoc comparison using Tukey test. *p < 0.05, n.s. non-significant.
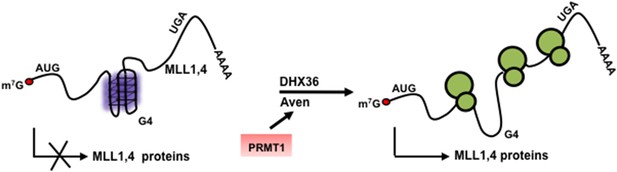
Model denoting the role of arginine methylated Aven by PRMT1 and DHX36 in the translation of G4 harboring MLL1 and MLL4 proteins.
https://doi.org/10.7554/eLife.06234.019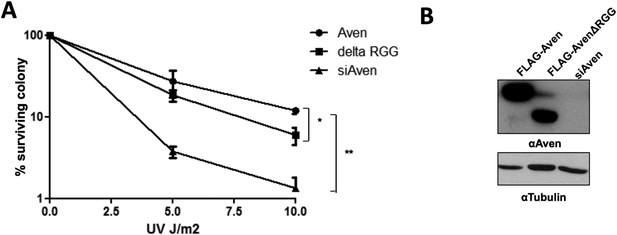
The Aven RGG/RG motif is required for the cell survival. (A)
U2OS cells stably expressing either FLAG-Aven, FLAG-AvenΔRGG or siAven were seeded in 10-cm dishes. 24 h following plating, the cells were treated with the indicated doses of UV in duplicates. The visible colonies were counted at 14 days after treatment, respectively, and normalized to 100% without the treatment. Error bars represent SEM (n = 4). Significance was measured by ANOVA followed by post hoc comparison using Tukey test. *p< 0.05, **p< 0.01. (B) Western blots to confirm the expression of FLAG-Aven, FLAG-AvenΔRGG and the knock down of endogenous Aven. Tubulin was used as a loading control.





