Plasma membrane overgrowth causes fibrotic collagen accumulation and immune activation in Drosophila adipocytes
Figures
Endocytic defects cause Collagen accumulation in Drosophila adipocytes.
(A) Schematic depiction of Collagen IV production, secretion and incorporation into basement membranes. (B) shibire knock-down (BM-40-SPARC>shii) and dominant negative shibireK44A (BM-40-SPARC>shiDN) cause Vkg-GFP accumulation in third instar larva adipocytes (marked with RFP). (C) Confocal images of third instar larva adipocytes stained with anti-Dynamin antibody. Staining is absent upon shi knock-down and increased by shiK44A expression. Nuclei stained with DAPI. (D) Localization of Vkg-GFP and Cg25C (anti-Cg25C staining) in wild type and BM-40-SPARC>shii adipocytes. Collagen IV accumulates in the periphery of shii adipocytes. (E) Vkg accumulation in BM-40-SPARC>Rab5i and >Rab5DN adipocytes. (F) Vkg accumulation in BM-40-SPARC>Chci and >ChcDN adipocytes. (G) Presence of Vkg-GFP is reduced in discs from BM-40-SPARC>shii, >shiDN, >Rab5i and >Rab5DN larvae. Vkg-GFP decrease quantified in graph. n ≥ 5 for each genotype. Differences with wild type are in all cases significative (Mann–Whitney tests, **p < 0.01). (H) Elongation of the ventral nerve cord (VNC) in BM-40-SPARC>shiDN and >Rab5DN.
(A) Confocal images of adipocytes from shi1 and shi2 thermosensitive mutants.
Shifting larvae to restrictive temperature for 3 hr causes mild pericellular accumulation of Collagen IV (Vkg-GFP in green). myr-RFP membrane marker in red. (B) Western blots of hemolymph probed with an anti-Cg25C antibody (1:5000). Hemolymph was collected by turning 10 larvae inside-out inside 100 μl of PBS. 10 μl of 2-Mercaptoethanol-reduced sample (equivalent to the blood of 1 larva) were loaded per genotype. We bled wild type larvae (w1118) and larvae where vkg or Cg25C were knocked down in adipocytes (Cg>vkgi+tub-GAL80ts and Cg>Cg25Ci+tub-GAL80ts respectively). For vkg and Cg25C knock-down, and in order to circumvent embryonic/L1 lethality, temporary inhibition of GAL4-driven knock-down was achieved with thermosensitive GAL4 inhibitor tub-GAL80ts (larvae were grown at 18°C to prevent knock-down, transferred to 30°C to initiate knock-down in L1/L2 stage and bled 3 days later in L3 stage). Note that knock-down of Vkg increases Cg25C signal, expected as monomeric Cg25C cannot be incorporated into BMs in the absence of Viking (Pastor-Pareja and Xu, 2011). (C) Pericellular Vkg accumulation in adipocytes from BM-40-SPARC>Hrsi, >RN-trei, >AP-2αi and >AP-2μi larvae. (D) Western blots of hemolymph extracted from wild type (w1118), BM-40-SPARC>shii, >cacti and >Tl10B larvae probed with an anti-GFP antibody (1:5000). The amount of blood loaded in each well is equivalent to 1 larva.
UAS-Dcr2 expression (BM-40-SPARC-Gal4>UAS-Dcr2) does not affect Collagen IV localization (Vkg-GFP) in adipocytes compared to Vkg-GFP control larvae (+) and larvae expressing GAL4 but not Dcr2 (BM-40-SPARC-Gal4).
https://doi.org/10.7554/eLife.07187.005
Collagen accumulation in endocytosis-defective cells is pericellular and autonomous.
(A) Vkg-GFP accumulation in a shii adipocyte (BM-40-SPARC>shii) expressing membrane marker myr-RFP. (B) shii adipocyte stained with phalloidin (F-actin). (C) shii adipocyte stained with cell-impermeable membrane dye FM4-64, labelling plasma membrane (PM) around accumulations. (D) shii adipocyte stained with fixable Texas-Red-coupled Dextran (70,000 MW), labelling PM around accumulations. (E) Antibody stainings of wild type and shii adipocytes performed without permeabilization (no detergent) in order to detect extracellular Collagen IV. In contrast to the accumulations in shii adipocytes, intracellular accumulations of Collagen IV in Tango1i adipocytes cannot be stained in the absence of permeabilization and are shown as a control. (F) Mosaic fat body (act-GAL4>shii flip-out clones, marked with RFP) showing Vkg-GFP accumulation in shii cells. Accumulation is suppressed by a GFP-targeting dsRNA (iGFPi). (G) Pericellular accumulation in mosaic shii fat body expressing Cg25C-RFP. Clones marked with GFP. (H) Localization of the endocytic marker Transferrin Receptor (Cg>TfR-GFP) in wild type adipocytes. (I) In shii adipocytes, TfR concentrates in PM pockets containing Collagen IV (anti-Cg25C). (J) In Rab11i adipocytes, TfR localizes to intracellular vesicles that completely fill the cytoplasm. No TfR is detected at the PM.
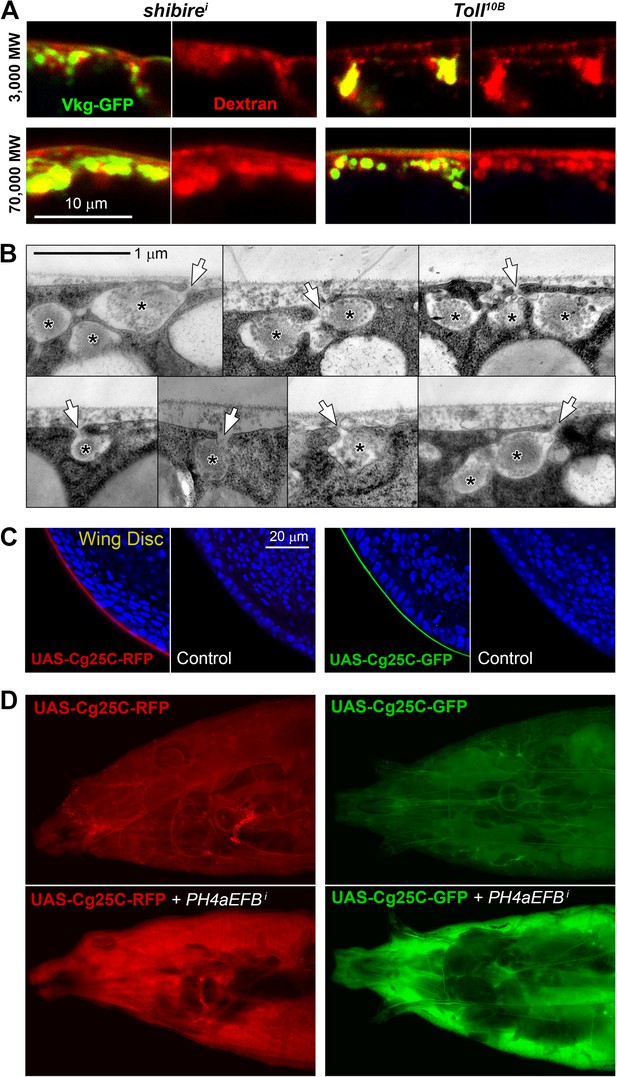
(A) Confocal images showing the PM of shii and Tl10B adipocytes stained with fixable Texas-Red-coupled Dextrans (3000 and 70,000 MW), labelling PM around Collagen IV (Vkg-GFP) accumulations.
(B) Electron micrographs of the PM in BM-40-SPARC>shii adipocytes showing instances of connection (arrows) between the pericellular accumulations (asterisks) and the extracellular space showing that these pockets are not isolated cisternae, but part of a very intricate PM. (C) Confocal images of wing discs (posterior ventral hinge) showing localization of Cg25C-GFP and Cg25C-RFP in the basement membrane after expression in the fat body controlled by Cg-GAL4 (Cg>Cg25C-GFP and Cg>Cg25C-RFP respectively). Images of wild type (w1118) discs are shown as controls to exclude auto-fluorescence. Nuclei stained with DAPI (blue). (D) Images of live larvae expressing Cg25C-RFP and Cg25C-GFP in the fat body (Cg>Cg25C-GFP and Cg>Cg25C-RFP). Knock-down of PH4αEFB, required for Collagen IV trimerization, causes tagged Cg25C to accumulate in the blood (note strong fluorescent signal filling the body cavity).
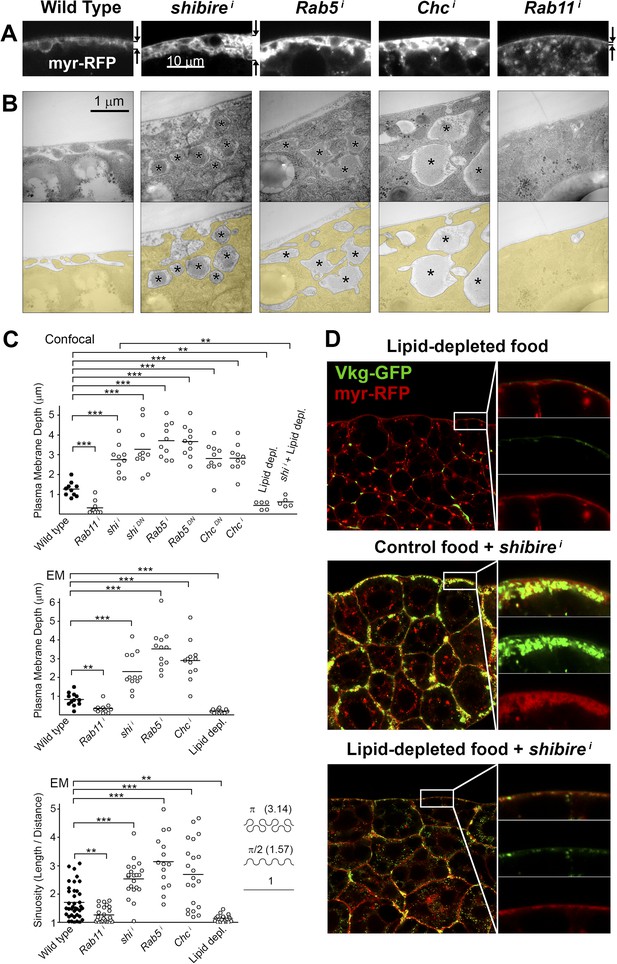
Pericellular Collagen trapping is due to PM overgrowth.
(A) Confocal sections of adipocyte PM (myr-RFP marker). PM expansion is observed in BM-40-SPARC>shii, >Rab5i and >Chci adipocytes, whereas PM flattening occurs in >Rab11i adipocytes along with accumulation of intracellular vesicles. (B) Electron micrographs of adipocyte PM from control, BM-40-SPARC>shii, >Rab5i, >Chci and >Rab11i larvae. Internal cell volume indicated through yellow transparency. Asterisks mark pericellular deposits. (C) Quantification of PM depth and sinuosity (see Figure 3—figure supplement 2). Depth measurements obtained from confocal (n = 12) and electron (n = 10) micrographs. PM sinuosity is the ratio between the length of PM between two points on that membrane and the linear distance separating them (n ≥ 15). Differences with controls were significative as indicated (Mann–Whitney tests, **p < 0.01, ***p < 0.001). (D) Adipocytes from wild type larvae grown on lipid-depleted food, BM-40-SPARC>shii larvae and BM-40-SPARC>shii larvae grown on lipid-depleted food. Vkg-GFP accumulation and PM excess are both suppressed by lipid-depletion.
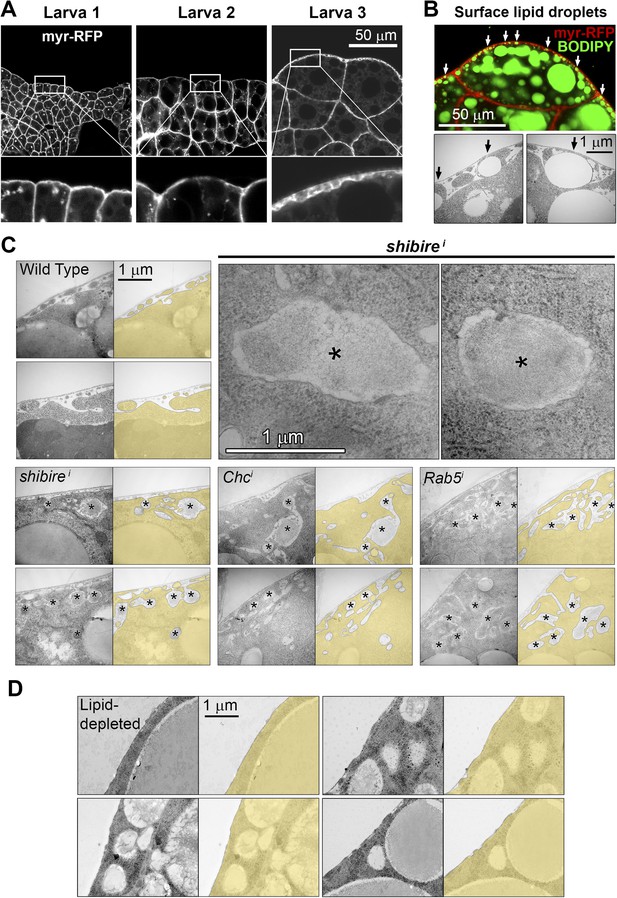
(A) Confocal images of fat body dissected from first, second and third instar larvae.
myr-RFP membrane marker in white. PM convolution is apparent in third instar larvae. (B) Confocal (BODIPY staining) and electron micrographs showing surface lipid droplets (arrows) surrounded by PM in larva 3 adipocytes. (C) Electron micrographs of the PM of adipocytes from control w1118, BM-40-SPARC>shii, >Chci and >Rab5i third instar fat body. Intracellular volume indicated with a transparent yellow layer in right panels. Pericellular deposits of extracellular material marked by asterisks. (D) Electron micrographs of the PM of adipocytes from larvae grown in lipid-depleted medium.

Schematic explanation of PM sinuosity and depth measurements performed in electron micrographs.
See ‘Materials and methods’ section.
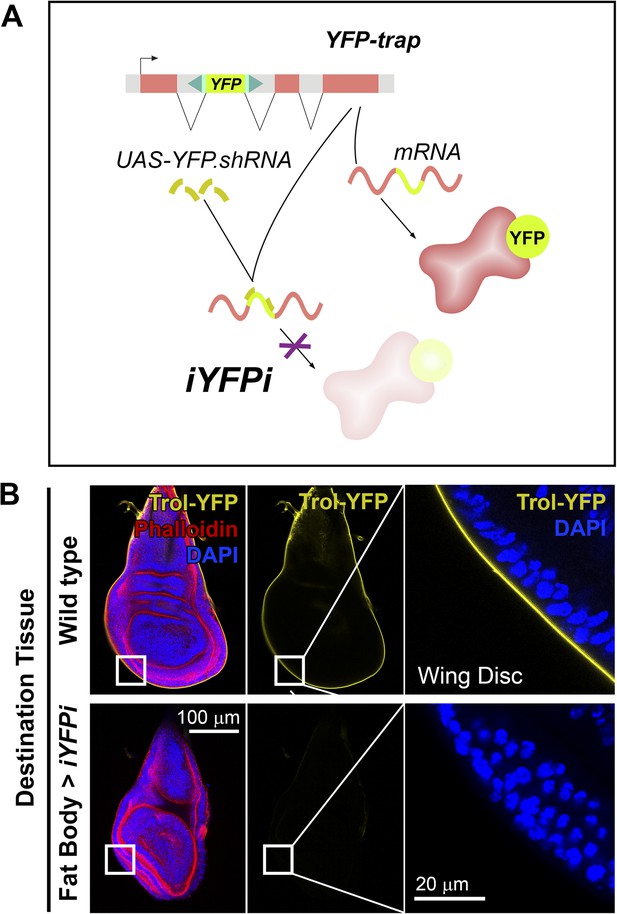
Perlecan, like Collagen IV, originates in the fat body.
(A) Schematic representation of the in vivo YFP interference strategy (iYFPi) to knock-down expression of YFP-trapped Perlecan (Trol-YFP) and ascertain its tissue of origin. Expression of a short hairpin RNA targets the YFP sequence in the YFP-trapped mRNA for degradation through RNAi. (B) Localization of Perlecan (Trol-YFP trap) in wing discs from trolCPTI-002049/Y flies. iYFPi in the fat body (BM-40-SPARC>iYFPi) eliminates expression of Trol-YFP in the wing disc and produces tissue hyperconstriction, a previously described trol loss-of-function phenotype (Pastor-Pareja and Xu, 2011). Phalloidin staining of F-actin in red to reveal disc deformation.
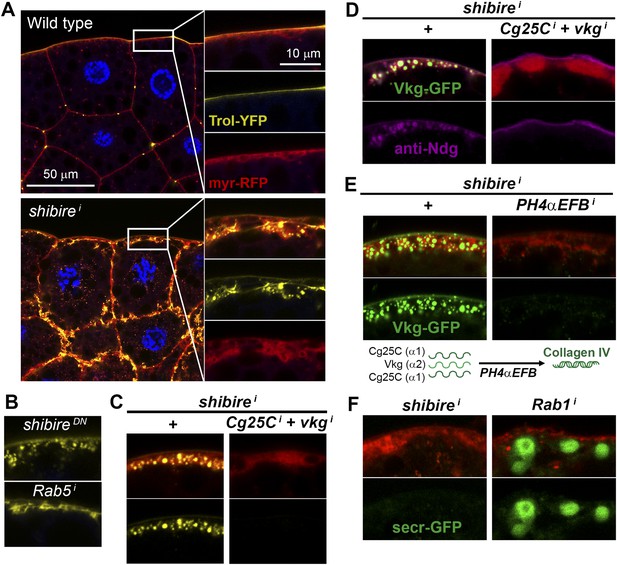
Pericellular deposits in adipocytes are fibrotic.
(A) Localization in wild type and BM-40-SPARC>shii adipocytes of Trol-YFP (trolCPTI-002049; see Figure 4). Perlecan accumulates in >shii adipocytes. (B) Perlecan accumulation in BM-40-SPARC>shiDN and >Rab5i adipocytes. (C) Perlecan accumulation in r4>shii adipocytes is suppressed by Collagen IV knock down. (D) Pericellular Nidogen accumulation (anti-Ndg staining) in r4>shii adipocytes is suppressed by Collagen IV knock down. (E) Pericellular Vkg accumulation in BM-40-SPARC>shii adipocytes is suppressed by knocking down prolyl-hydroxylase PH4αEFB. (F) BM-40-SPARC>shii adipocytes do not accumulate secretion marker secr-GFP. Intracellular secr-GFP retention in BM-40-SPARC>Rab1i is shown as a control.
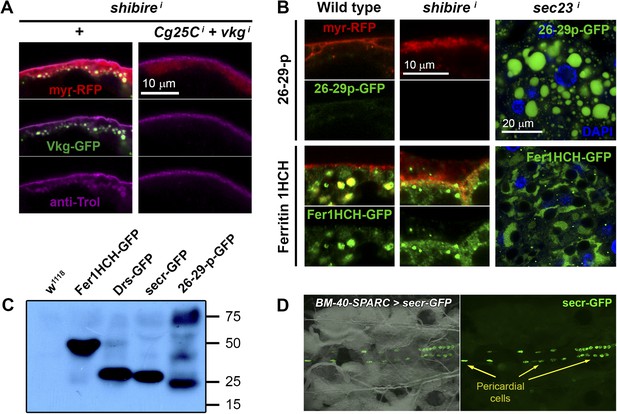
(A) PM of fat body adipocytes stained with anti-Trol antibody (magenta).
Pericellular accumulation of Perlecan (Trol) in r4>shii adipocytes is suppressed by additionally knocking down expression of vkg and Cg25C Collagen IV chains. (B) Localization of 26-29-protease (26-29-pCA06735 GFP-trap) and Ferritin 1HCH (Fer1HCHG188 GFP-trap) in adipocytes from wild type L3 larvae, Cg>shii L3 larvae and Cg>sec23i L2 larvae. Note intracellular accumulation upon sec23 knock-down, which confirms that 26-29-p and Fer1HCH are indeed adipocyte-secreted. (C) Western blots of hemolymph extracted from wild type (w1118), Fer1HCHG188, Drs-GFP, BM-40-SPARC>ecr-GFP and 26-29-pCA06735 larvae probed with anti-GFP antibody (1:5000). The amount of blood loaded in each well is equivalent to 2.5 larvae. Whereas Fer1HCH-GFP (expected molecular weight 50 kDa), Drs-GFP (34 kDa) and secr-GFP (27 kDa) are detected in the hemolymph as clear single bands, 26-29-p (87 kDa) seems to be processed. (D) Dorsal view of a live larva expressing secr-GFP in adipocytes (BM-40-SPARC>secr-GFP) showing accumulation of GFP in pericardial cells, a hemolymph filtering nephrocyte-like cell type.
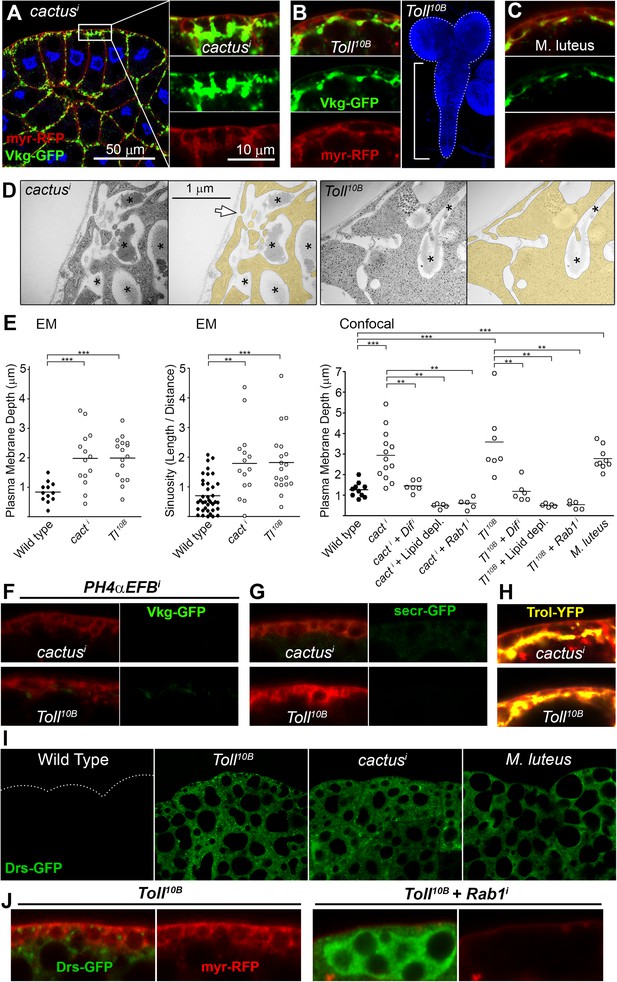
Fibrotic deposits and PM overgrowth upon Toll activation.
(A) Pericellular Vkg deposits (Vkg-GFP) and PM overgrowth in BM-40-SPARC>cacti adipocytes. (B) Vkg deposits and VNC elongation in BM-40-SPARC>Tl10B larvae. (C) Vkg deposits and PM overgrowth in adipocytes 1 day after infection with Micrococcus luteus. (D) Electron micrographs of BM-40-SPARC>cacti and >Tl10B adipocytes. The arrow marks connection of the deposits to the extracellular space. (E) Measurements of PM depth and sinuosity in adipocytes of indicated genotypes. Depth measurements obtained from confocal (n ≥ 7) and electron micrographs (n ≥ 10). Sinuosity measured in electron micrographs (n ≥ 15). Differences with wild type or appropriate control as indicated were significative in all cases (Mann–Whitney tests, **p < 0.01, **p < 0.001). (F) Pericellular Vkg accumulation in BM-40-SPARC>cacti and >Tl10B adipocytes is suppressed by knocking down prolyl-hydroxylase PH4αEFB. (G) Secretion marker secr-GFP does not accumulate in BM-40-SPARC>cacti or >Tl10B adipocytes. (H) Pericellular Perlecan deposits (Trol-YFP) in BM-40-SPARC>cacti and >Tl10B adipocytes. (I) Induction of antimicrobial peptide Drosomycin (Drs-GFP) fills adipocyte cytoplasm in BM-40-SPARC>cacti, >Tl10B and Micrococcus luteus-infected larvae. (J) Rab1 knock-down causes intracellular Drosomycin retention and suppresses PM overgrowth in BM-40-SPARC>Tl10B adipocytes.
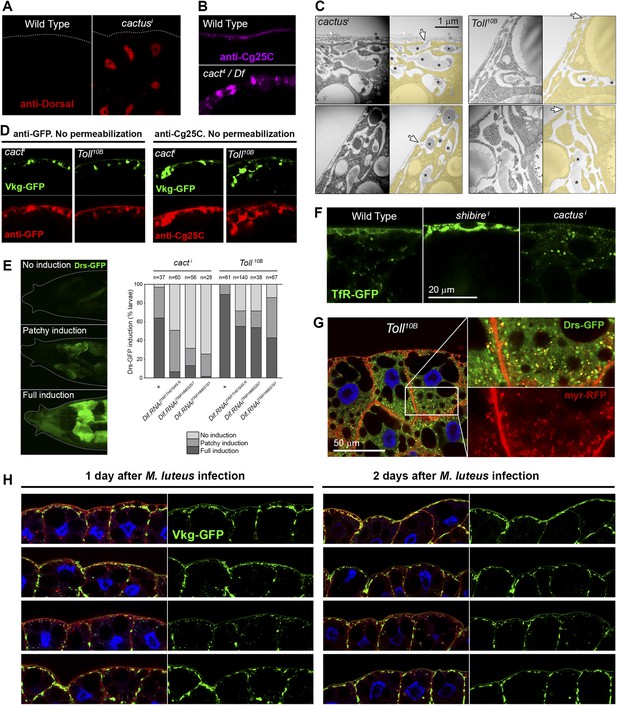
(A) Nuclear accumulation of the Toll downstream transcription factor Dorsal (anti-Dorsal staining) in BM-40-SPARC>cacti adipocytes.
(B) Pericellular Cg25C accumulation in adipocytes of mutant cact4 over cact-uncovering deficiency Df(2L)r10. (C) Electron micrographs of the PM of BM-40-SPARC>cacti and >Tl10B adipocytes. Asterisks mark pericellular deposits. Arrows mark visible connections of the deposits to the extracellular space. (D) Antibody stainings of cacti and Tl10B adipocytes performed without permeabilization (no detergent) in order to detect extracellular Collagen IV. (E) Knock-down of Dif decreases the expression of Toll target gene Drosomycin (Drs-GFP) activated by cacti and Tl10B (BM-40-SPARC>cacti and >Tl10B adipocytes). (F) Localization of endocytic marker TfR-GFP in wild type, Cg>shii and Cg>cacti adipocytes. Intracellular TfR vesicles are seen in cacti adipocytes, same as wild type. (G) Drosomycin-containing vesicles (Drs-GFP) in the cytoplasm of BM-40-SPARC>Tl10B adipocytes. (H) Pericellular retention of Collagen IV (Vkg-GFP) at the PM of adipocytes from larvae dissected 1 or 2 days after Micrococcus luteus infection.
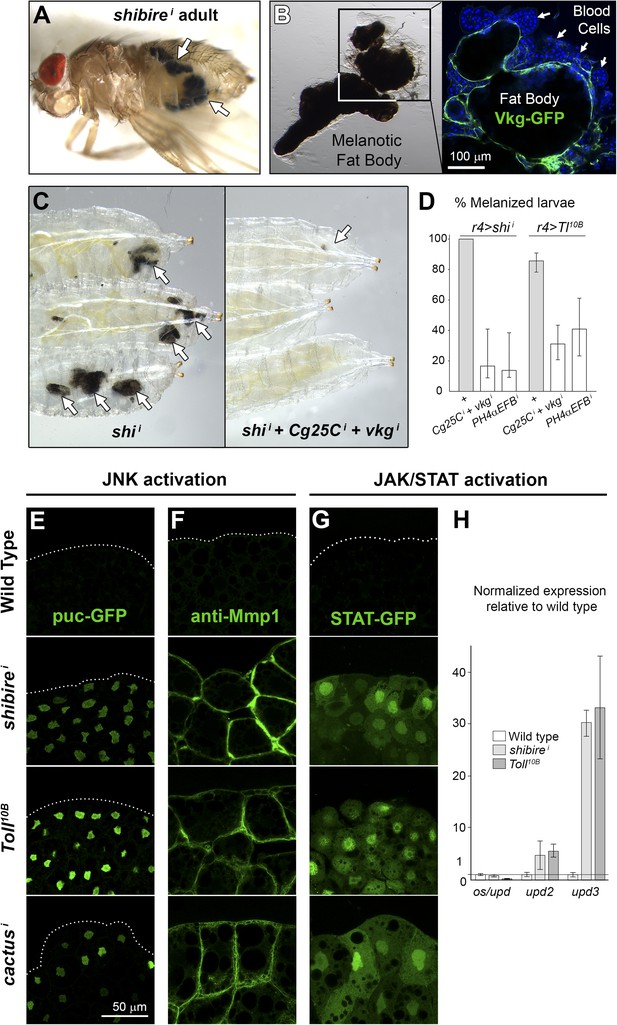
Immune response to fibrotic deposits.
(A) Melanotic fat body in a ppl>shii fly. (B) Melanized fat body from an r4>shii larva. Hemocytes (blood cells) encapsulate the tissue. (C) Knock-down of Collagen IV reduces fat body melanization in r4>shii larvae. Cultures maintained at 30°C. (D) Percentage of larvae displaying signs of melanization in indicated genotypes. n ≥ 30 per genotype. Differences with r4>shii and >Tl10B controls were significative (χ2 tests, ***p < 0.001). Cultures maintained at 30°C. (E) Induction of c-Jun N-terminal kinase (JNK) downstream puckered (puc-GFP enhancer trap) in BM-40-SPARC>shii, >Tl10B and >cacti adipocytes. (F) Induction of Matrix Metallo-Protease 1 (anti-Mmp1 staining) in BM-40-SPARC>shii, >Tl10B and >cacti adipocytes. (G) Expression of JAK/STAT activity reporter 10XSTAT-GFP in BM-40-SPARC>shii, >Tl10B and >cacti adipocytes. (H) Expression of JAK/STAT-activating ligands in wild type, BM-40-SPARC>shii and >Tl10B adipocytes assessed by real time RT-PCR. Error bars represent 95% confidence intervals. rp49 expression was used for normalization.
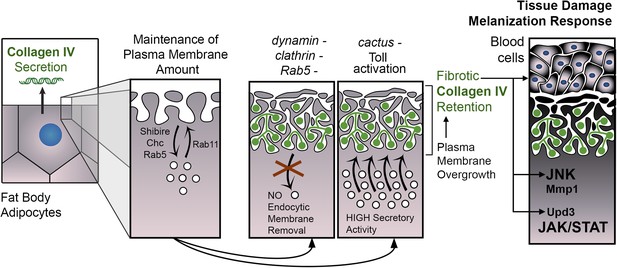
PM overgrowth leads to adipocyte fibrosis.
Schematic representation summarizing the genesis of fibrotic deposits caused by PM excess and the ensuing reaction by the immune system. Defective endocytosis or excess secretion induced by Toll activity cause PM overgrowth in fat body adipocytes, which leads to hyperconvoluted PM morphology and pericellular trapping of Collagen IV and other extracellular matrix (ECM) proteins in the cell cortex. Fibrotic Collagen IV deposits trigger an immune response, as evidenced by tissue melanization and activation of the JAK/STAT and JNK pathways.
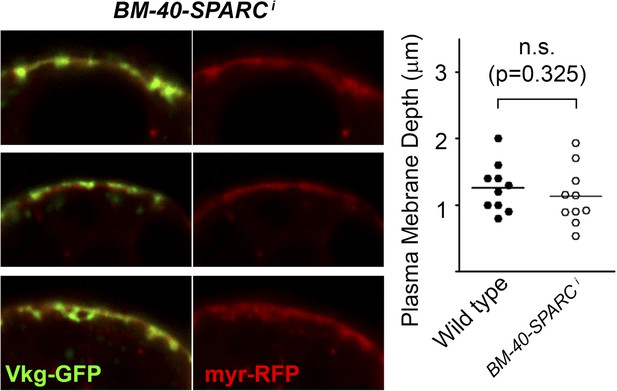
Knock-down of BM-40-SPARC (BM-40-SPARC>BM-40-SPARCi) causes PM accumulation of Collagen IV without PM overgrowth.
https://doi.org/10.7554/eLife.07187.018Additional files
-
Supplementary file 1
Hits in RNAi screening causing plasma membrane accumulation of Collagen IV in adipocytes.
- https://doi.org/10.7554/eLife.07187.019
-
Supplementary file 2
Detailed genotypes.
- https://doi.org/10.7554/eLife.07187.020
-
Supplementary file 3
Origin of antibodies, mutants and transgenes used in this study.
- https://doi.org/10.7554/eLife.07187.021





