Social communication of predator-induced changes in Drosophila behavior and germ line physiology
Figures
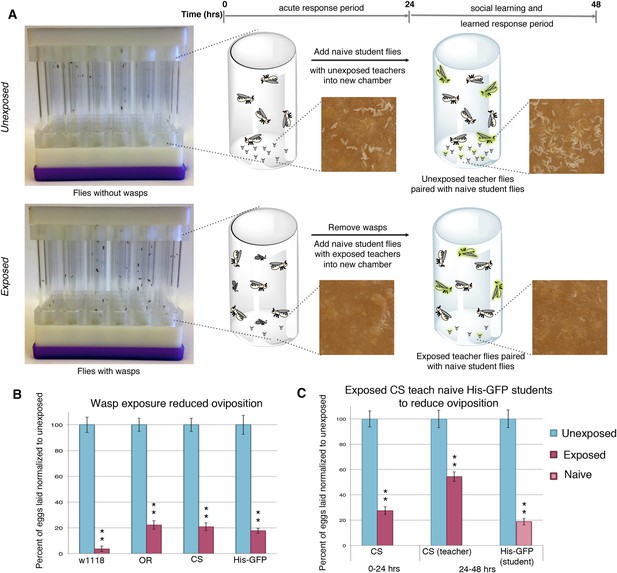
Flies respond to wasps by decreasing oviposition and are able to confer this information to naive flies.
(A) Standard exposure setup. (B and C) Percent of eggs laid normalized to unexposed. (B) Wild-type flies unexposed or exposed to wasps. (C) Canton-S teachers and His-GFP students. For (B) and (C), error bars represent standard error (n = 24 biological replicates) (**p < 1.0e-5).
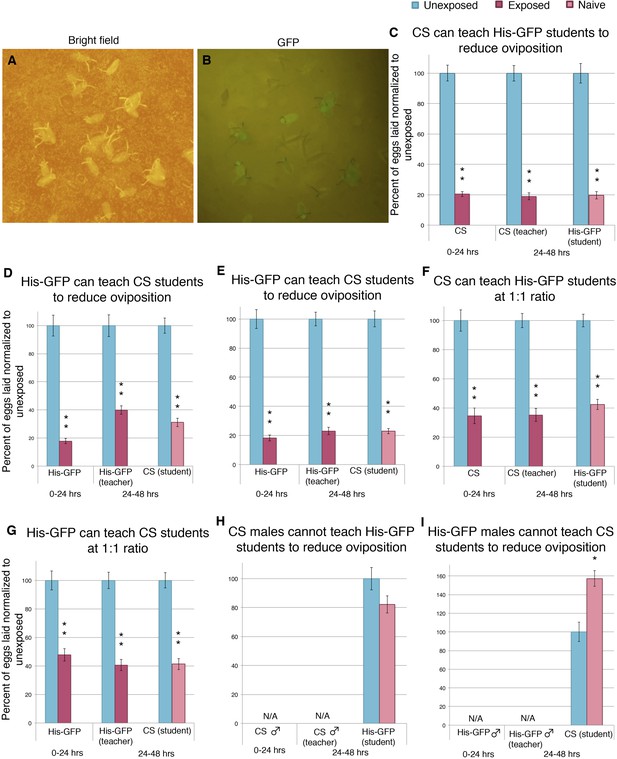
Social transmission of information from wasp-exposed female teacher fly to naive female student fly occurs.
(A) Bright field image of oviposition plate containing Canton-S and His-GFP. (B) Green fluorescent protein (GFP) channel image of oviposition plate containing Canton-S and His-GFP eggs. (C to I) Percent of eggs laid normalized to unexposed shown. (C) Second 24 replicates of Canton-S teachers and His-GFP students run in April 2014. (D) First 24 replicates of His-GFP teachers and Canton-S students run in August 2013. (E) Second 24 replicates of His-GFP teachers and Canton-S students run in April 2014. (F) 1:1 ratio of female teacher Canton-S flies to student His-GFP. (G) 1:1 ratio of female teacher His-GFP flies to student Canton-S flies. (H) 1:1 ratio of male teacher Canton-S flies to student His-GFP flies. (I) 1:1 ratio of male teacher His-GFP flies to student Canton-S flies. For (C) to (I), error bars represent standard error (n= 24 biological replicates) (*p < 0.05, **p < 1.0e-5).
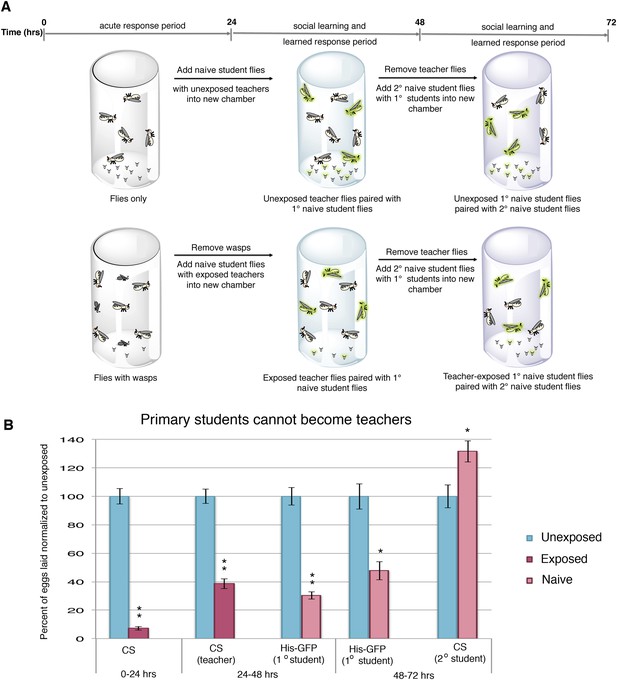
Student flies cannot become teachers.
(A) Standard exposure setup. (B) Teacher exposed primary student His-GFP flies paired with naive secondary student Canton-S flies. Error bars represent standard error (n = 24 biological replicates) (*p < 0.05, **p < 1.0e-5).
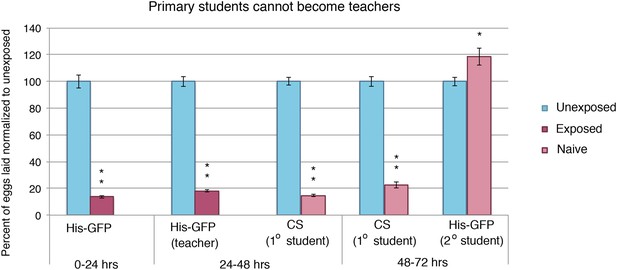
Student flies cannot become teachers.
Teacher exposed primary student Canton-S flies paired with naive secondary student His-GFP flies. Percent of eggs laid normalized to unexposed shown. Error bars represent standard error (n= 24 biological replicates) (*p < 0.05, **p < 1.0e-5).
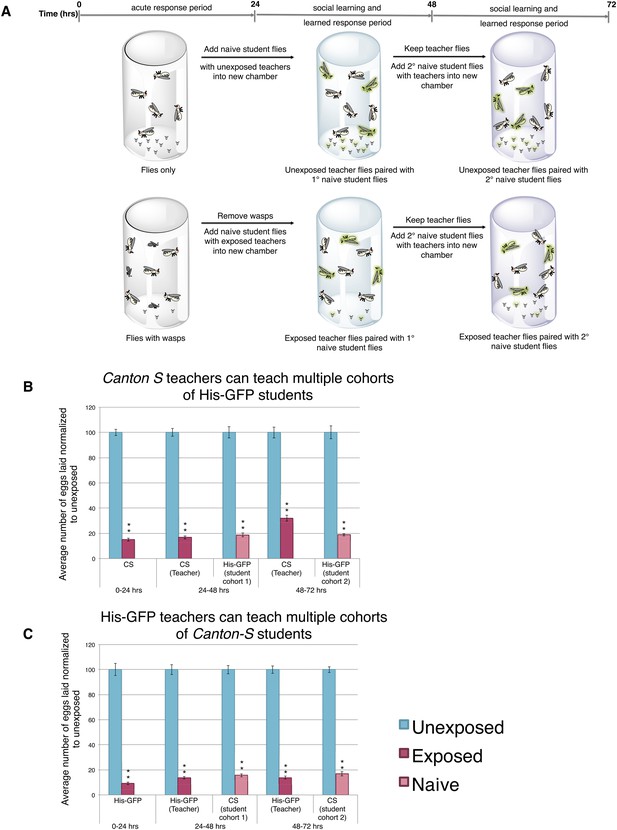
Teacher flies can teach multiple batches of students.
(A) Standard exposure setup for teachers teaching multiple batches of students. (B and C) Percent of eggs laid normalized to unexposed. (B) Canton-S flies unexposed or exposed to wasps and paired with primary and secondary His-GFP students. (C) His-GFP flies unexposed or exposed to wasps and paired with primary and secondary Canton-S students. For (B) and (C), error bars represent standard error (n = 24 biological replicates) (**p < 1.0e-5).
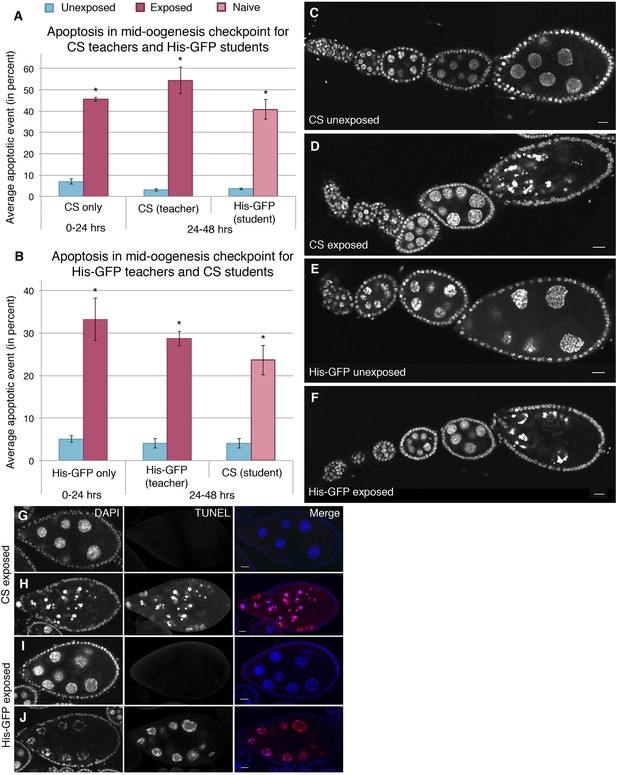
Stage-specific apoptosis observed in wasp-exposed teachers and teacher-exposed student flies.
(A and B) Average percent of apoptotic events for stage 7/8 egg chambers. (A) Canton-S exposed and unexposed ovary apoptosis. (B) His-GFP exposed and unexposed ovary apoptosis. (C to D) Canton-S unexposed/exposed ovariole. (E to F) His-GFP unexposed/exposed ovariole. (G to H) Canton-S transferase dUTP nick end labeling (TUNEL) staining performed on exposed fly ovaries. (I to J) His-GFPTUNEL staining. For (A) and (B), error bars represent standard error (n = 3 biological replicates from which 12 ovaries were scored for each group) (*p < 0.05). Scale bars, 20 μm.
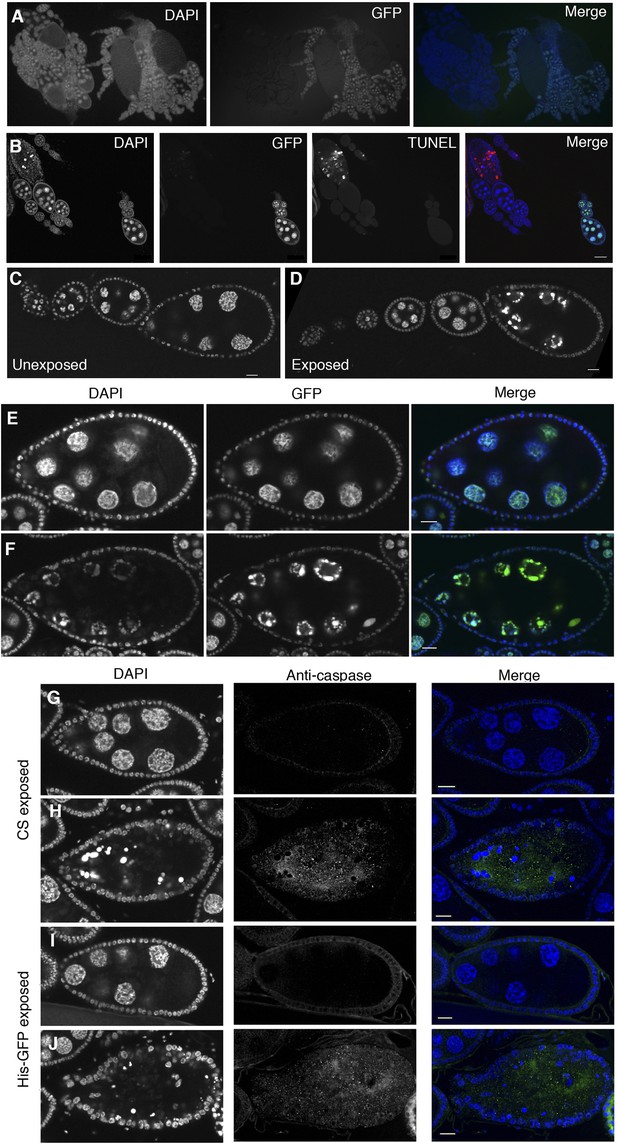
Stage-specific apoptosis is induced following wasp exposure.
(A) Canton-S and His-GFP ovaries intermingled are easily distinguishable. (B) Canton-S and His-GFP egg chambers that are undergoing apoptosis are easily distinguishable by using DAPI or TUNEL. (C to D) His-GFP unexposed/exposed ovary chain corresponding to Figure 3E,F, visualized using the GFP channel. (E to F) DAPI and His-GFP images of TUNEL-stained egg chambers corresponding to Figure 3I,J. (G to H) Canton-S exposed egg chambers stained with DAPI and anti-caspase. (I to J) His-GFP exposed egg chambers stained with DAPI and anti-caspase. Scale bars are 20 μm.
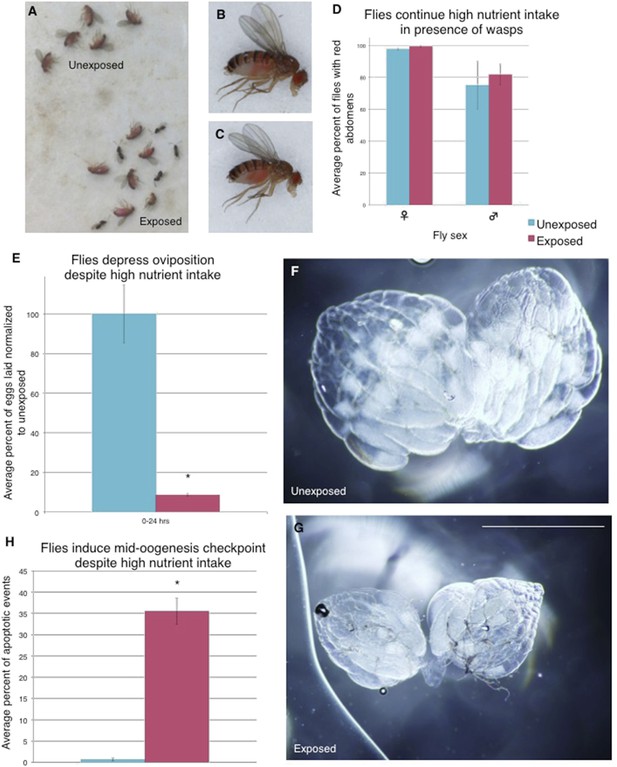
Flies continue to eat high-protein diet following wasp exposure but still depress oviposition.
Continued oviposition depression cannot be explained by a lack of nutrient intake that normally inactivates insulin signaling. The high-nutrient intake by exposed female flies suggests that an active insulin signaling pathway is inhibited or bypassed downstream of nutrient sensing. (A) Exposed and unexposed flies anesthetized immediately after 24-hr exposure period shows red food in abdomens. (B) Lateral view of unexposed fly. (C) Lateral view of exposed fly. (D) Percent of male and female flies with red food in abdomen, error bars are 95% confidence intervals. (E) Percent of eggs laid normalized to unexposed following 24-hr exposure period. All eggs on the food plate were counted, including eggs on the yeast paste. (F) Representative ovary dissected from unexposed fly. 36 total ovaries were dissected and examined across 3 replicates for each treatment. (G) Ovary dissected from exposed fly. Scale bar for (F) to (G) is 1.0 mm. (H) Average percent apoptosis in mid-oogenesis checkpoint for unexposed and exposed Canton S. For (D), (E), and (H), error bars represent standard error (n = 3 biological replicates. For (D), 100 female and 20 male flies were counted per replicate. For (E), 3 egg lay plates were counted per treatment. For (H), 3 biological replicates from which 12 ovaries were scored for each group) (*p < 0.05).
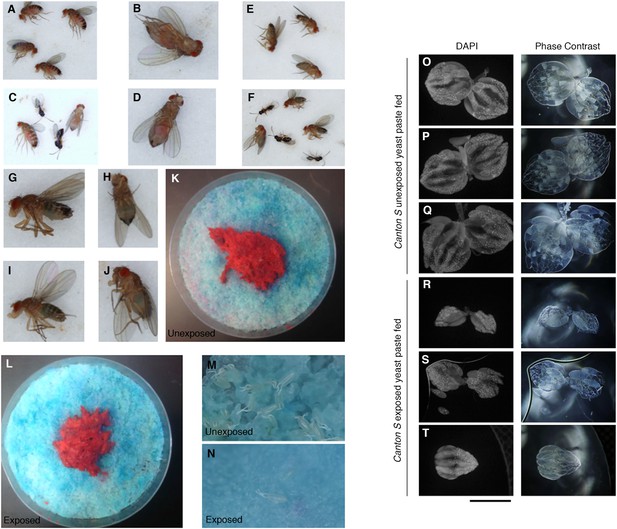
Flies continue to eat high protein diet following wasp exposure but still depress oviposition.
Additional evidence demonstrating that exposed flies eat rich nutrient food. (A) Unexposed flies after 24-hour period. (B) Ventral view of unexposed fly. (C) Exposed flies with wasps after 24-hour period. (D) Ventral view of exposed fly. (E) Unexposed flies after a 24-hour period fed only blue food have blue abdomens. (F) Exposed flies with wasps after a 24-hour period fed only blue food have blue abdomens. Percent of male and female flies with red food in abdomen. (G) Lateral view of unexposed fly. (H) Ventral view of unexposed fly. (I) Lateral view of exposed fly. (J) Ventral view of exposed fly. (K) Food plate from unexposed flies following 24-hour period. (L) Food plate from exposed flies following 24-hour period. (M) Zoom-in of food plate from unexposed flies showing many eggs. (N) Zoom-in of food plate from exposed flies showing few eggs. (O to Q) Unexposed Canton S ovaries from individuals fed yeast paste for 24 hours. (R to T) Exposed Canton S ovaries from individuals fed yeast paste for 24 hours.
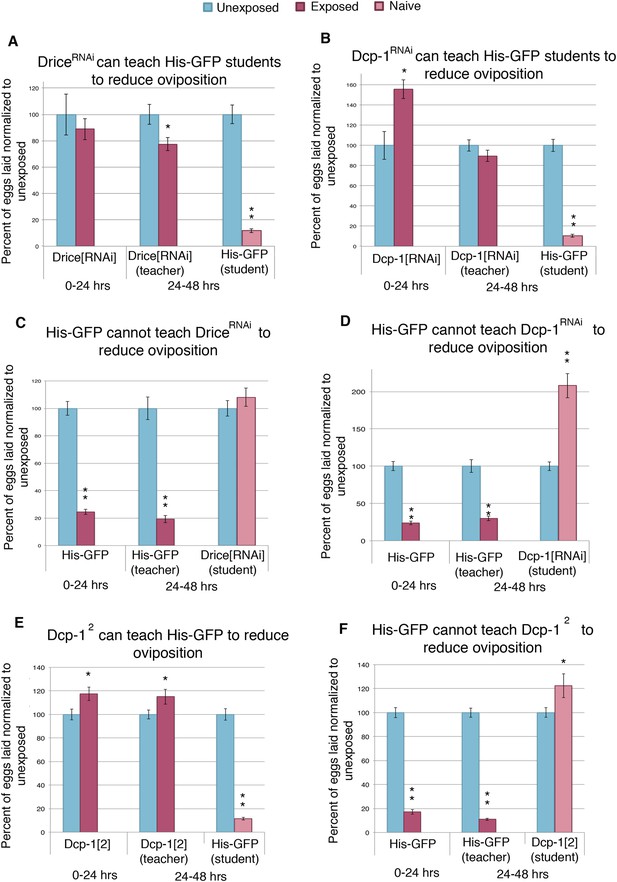
Socially transmitted oviposition depression in response to wasp exposure acts through the mid-oogenesis checkpoint.
(A to F) Percent of eggs laid normalized to unexposed. (A and C) Drice RNAi-knockdown as teachers and students. (B and D) Dcp-1 RNAi-knockdown as teachers and students. (E to F) Dcp-12 as teachers and students. For (A) to (F), error bars represent standard error (n = 24 biological replicates) (*p < 0.05, **p < 1.0e-5).
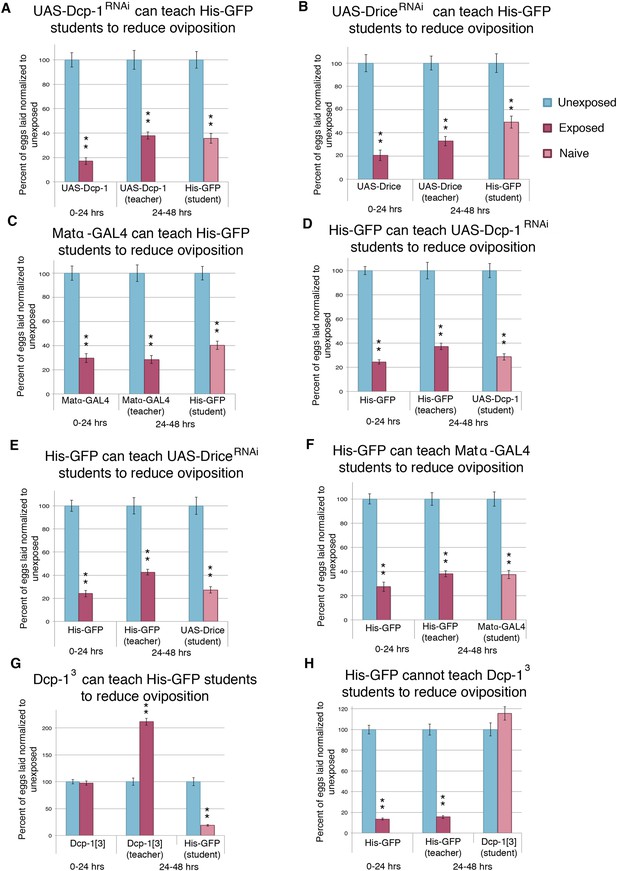
Socially transmitted oviposition depression acts through the mid-oogenesis checkpoint.
(A to H) Percent of eggs laid normalized to unexposed. (A to F) Teacher and student ability of GAL4 and UAS control parental strains from drice and Dcp-1 RNAi-knockdown crosses. (G) to (H) Teacher and student ability of Dcp-13. For (A) to (H), error bars represent standard error (n= 24 biological replicates). (**p < 1.0e-5).
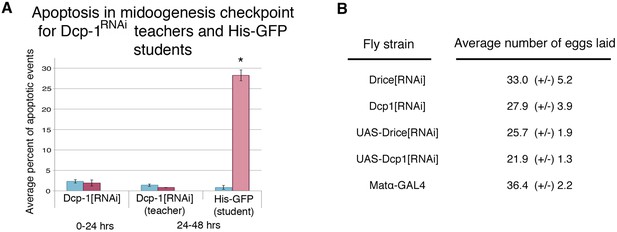
Further evidence indicating that oviposition depression acts through the mid-oogenesis checkpoint.
(A) Average percent of apoptotic events for stage 7/8 egg chambers in exposed and unexposed Dcp-1 RNAi-knockdown flies as teachers and His-GFP as students (n= 3 biological replicates from which 12 ovaries were scored for each group) (*p < 1.0e-5). (B) Average number of eggs laid for Drice and Dcp-1 RNAi and parental lines, shown with standard error. No significant difference in oviposition for unexposed flies was observed, P > 0.05 in all comparisons.
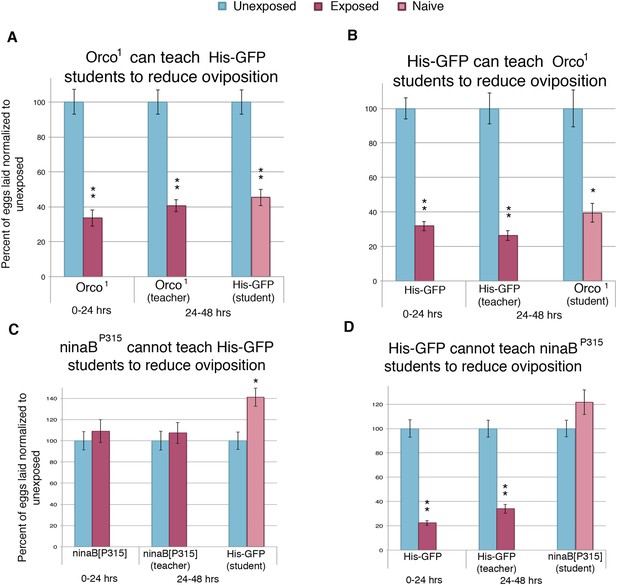
Flies respond to wasps and confer this information to naive flies through visual cues.
(A to D) Percent of eggs laid normalized to unexposed. (A to B) Smell mutants as teachers and students. (C to D) Sight mutants as teachers and students. For (A) to (D), error bars represent standard error (n = 24 biological replicates) (*p < 0.05, **p < 1.0e-5).
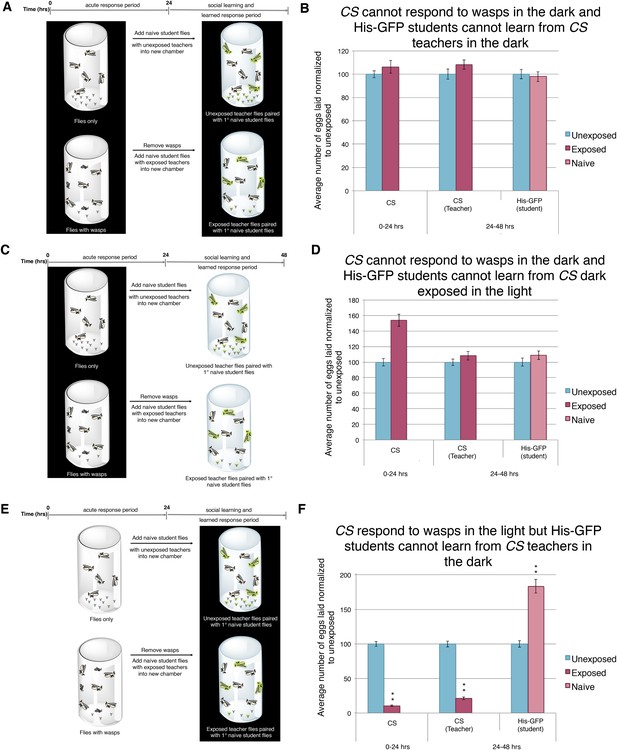
Acute and teaching response requires light.
(B, D, and F) Percent of eggs laid normalized to unexposed. (B, D, and F) Canton S as teachers and His-GFP as students. (A) Exposure setup when both acute and social response occurs in dark. (B) Results of experiment as described in (A). (C) Exposure setup when acute response occurs in the dark but social response occurs in the light. (D) Results of experiment as described in (C). (E) Exposure setup when acute response occurs in the light but social response occurs in the dark. (F) Results of experiment as described in (E). For (B), (D) and (F), error bars represent standard error (n = 24 biological replicates) (**p < 1.0e-5).
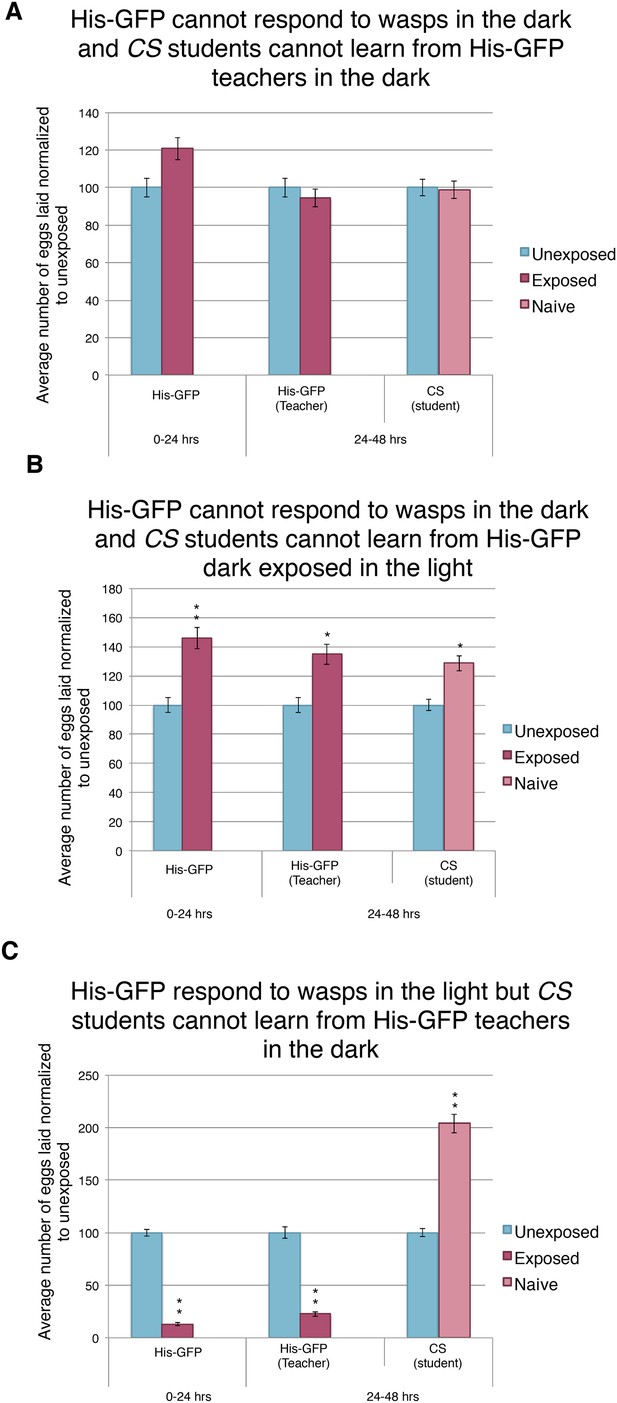
Further evidence indicating that learning requires light.
(A to C) Percent of eggs laid normalized to unexposed. (A to C) His-GFP as teachers and Canton-S as students. (A) His-GFP as teachers when both acute and social response occurs in dark. (B) His-GFP as teachers when acute response occurs in the dark but social response occurs in the light. (C) His-GFP as teachers when acute response occurs in the light but social response occurs in the dark. For (A to C) error bars represent standard error (n = 24 biological replicates) (*p < 0.05, **p < 1.0e-5).
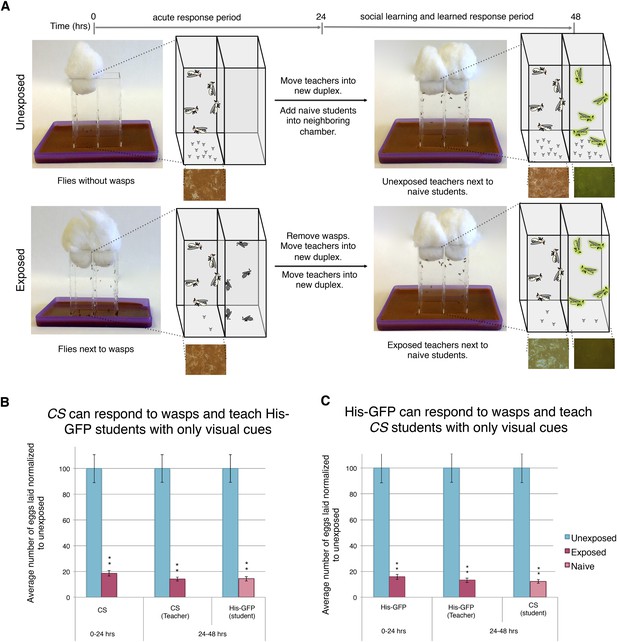
Visual cues are necessary and sufficient for learning.
(B and C) Percent of eggs laid normalized to unexposed. (A) Standard exposure setup using the Fly Duplex. The Fly Duplex ensures only visual cues are transferred between groups. (B) Canton S as teachers with His-GFP students. (C) His-GFP as teachers with Canton S as students. For (B and C) error bars represent standard error (n = 10 biological replicates) (**p < 1.0e-5).
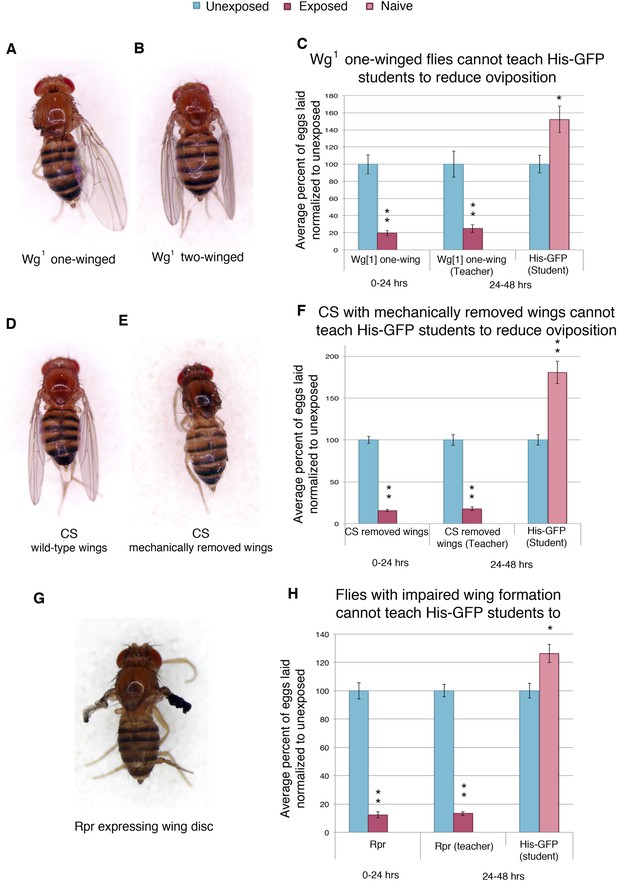
Teacher–student dynamics require wings to allow for communication to take place.
(For C, F, and H) Percent of eggs laid normalized to unexposed. (A) Dorsal view of wg1 with one wing. (B) Dorsal view of wg1 with two wings. (C) wg1 one-winged flies as teachers. (D) Dorsal view of Canton-S female. (E) Dorsal view of Canton-S female with clipped wings. (F) Canton-S flies with clipped wings as teachers. (G) Dorsal view of a female fly expressing reaper in the wing disc. (H) Flies expressing reaper in the wing disc as teachers. Error bars represent standard error (For (C) n = 18 biological replicates.) (For [F and H] n = 24 biological replicates) (*p < 0.05, **p < 1.0e-5).
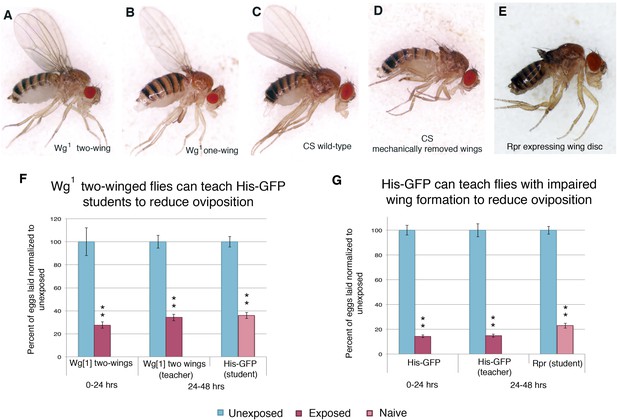
Teacher flies need wings in order to instruct student flies.
(A) Lateral view of one winged wg1 female. (B) Lateral view of two-winged wg1 female. (C) Lateral view of Canton-S female. (D) Lateral view of Canton-S female with clipped wings. (E) Lateral view of female fly expressing reaper in the wing disc. (F) wg1 two-winged flies as teachers. For (F) and (G), percent of eggs laid normalized to unexposed. (G) Flies expressing reaper (UAS-Rpr) in the wing disc as students. Error bars represent standard error (For [F and G] n= 24 biological replicates.) (**p < 1.0e-5).
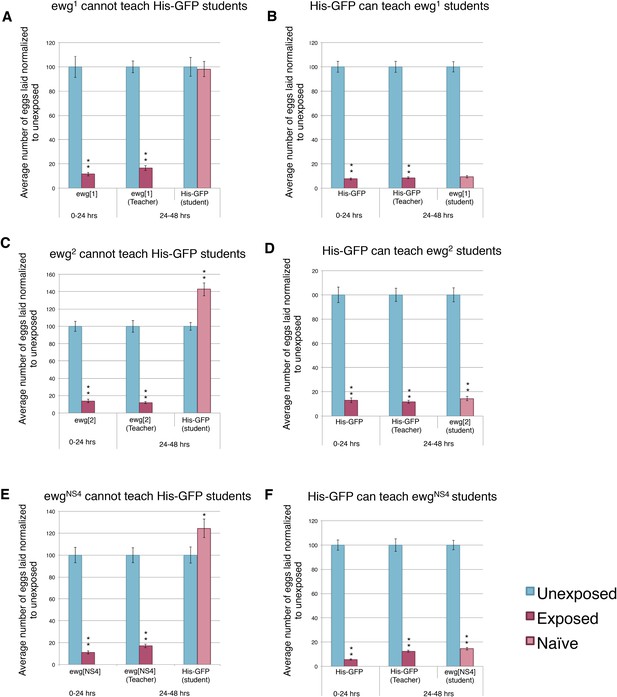
Teacher–student dynamics require functional wings to allow for communication to take place.
(For A to F) Percent of eggs laid normalized to unexposed. (A and B) ewg1 as teachers and students. (C and D) ewg2 as teachers and students. (E and F) ewgNS4 as teachers and students. Error bars represent standard error (For [A to F] n = 24 biological replicates.) (*p < 0.05, **p < 1.0e-5).
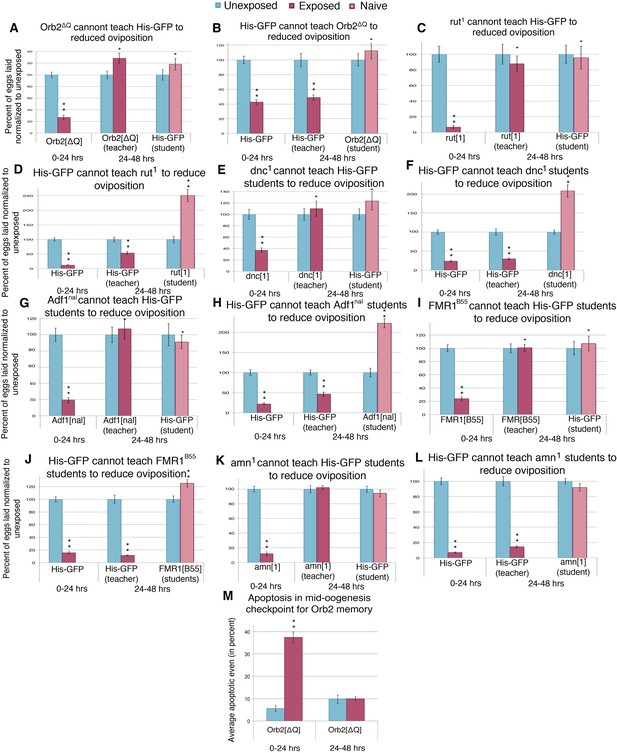
Learning mutants are unable to teach or be students.
(A to L) Percent of eggs laid normalized to unexposed. (A to B) Orb2ΔQ as teacher and student. (C to D) rut1 as teacher and student. (E and F) dnc1 as teacher and student. (G and H) Adf1nal as teacher and student. (I and J) FMR1B55 as teacher and student. (K and L) amn1 as teacher and student. For (M), average percent of apoptotic events for stage 7/8 egg chambers. (M) Orb2ΔQ exposed and unexposed ovary apoptosis. Error bars represent standard error. (For [A] to [L] n = 24 biological replicates.) (For [M] n = 3 biological replicates from which 12 ovaries were scored for each group) (*p < 0.05, **p < 1.0e-5).
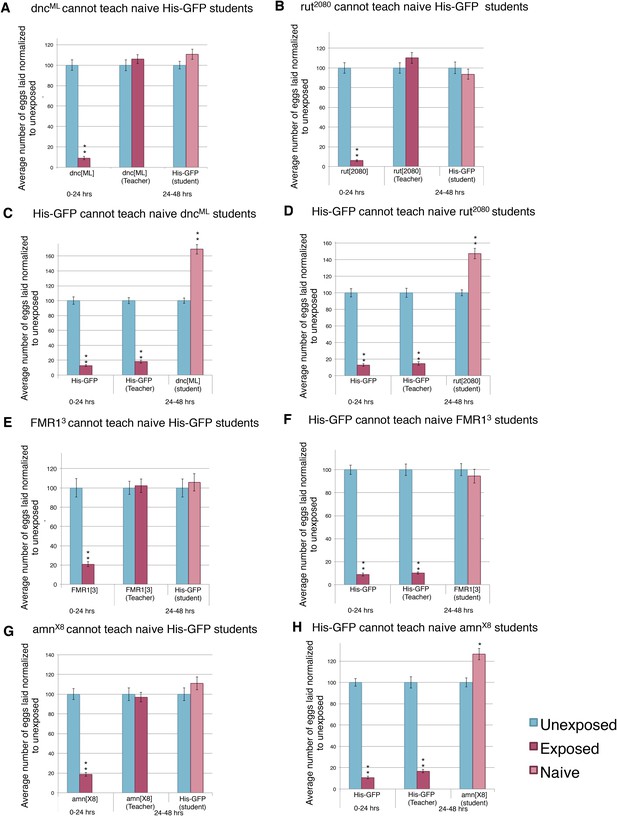
Learning mutants are unable to teach or be students.
(A to H) Percent of eggs laid normalized to unexposed. (A and C) dncML as teacher and student. (B and D) rut2080 as teacher and student. (E and F) FMR13 as teacher and student. (G and H) amnX8 as teacher and student. Error bars represent standard error (n= 24 biological replicates) (**p < 1.0e-5).

Learning and teaching require a continuous neural input from the brain.
(A and D) Percent of eggs laid normalized to unexposed. (A) Canton-S teachers with heads removed after acute exposure. (B) Dorsal view of representative Canton-S female. (C) Dorsal view of representative Canton-S female with no head. (D) Flies expressing tetanus toxin (UAS-TeTx) in mushroom body (MB) as teacher. Error bars represent standard error (For [A] and [D] n = 24 biological replicates.) (**p < 1.0e-5).
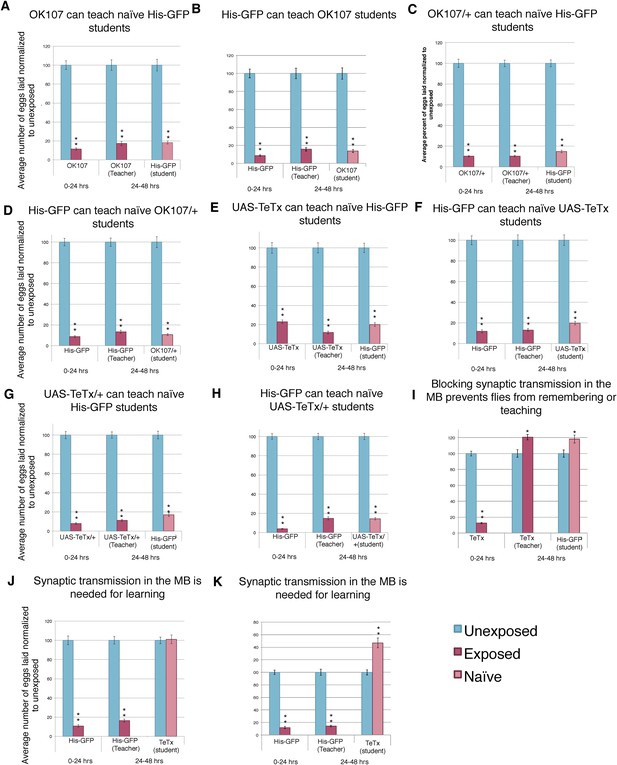
Blocking synaptic transmission in the MB prevents teacher behavior and student learning.
(A to E) Percent of eggs laid normalized to unexposed. (A and B) OK107 GAL4 as teachers and students. (C and D) OK107/+ GAL4 as teachers and students. (E and F) UAS-TeTx as teachers and students. (G and H) UAS-TeTx/+ as teachers and students. (I) Flies expressing tetanus toxin in MB as teachers using MB 247 GAL4. (J) Flies expressing tetanus toxin in MB as students using MB 247 GAL4 (K) Flies expressing tetanus toxin in MB as students using OK107 GAL4. Error bars represent standard error (For [A] to [K] n = 24 biological replicates.) (*P < 0.05, **P < 1.0e-5).
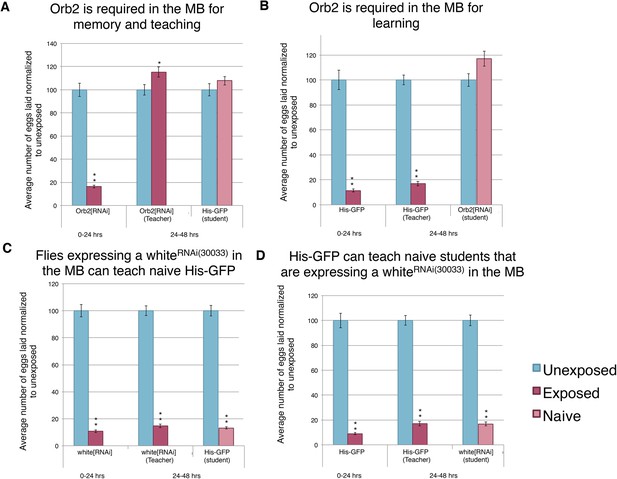
Knockdown of Orb2 in the MB results in defective learning.
(A to D) Percent of eggs laid normalized to unexposed. (A to B) Orb2 RNAi-knockdown as teachers and students. (C to D) white RNAi-knockdown as teachers and students. (For [A] to [D] n = 24 biological replicates.) (*p < 0.05, **p < 1.0e-5).
Expression of an RNAi hairpin in the MB does not induce defective learning and memory.
(A to D) Percent of eggs laid normalized to unexposed. (A to B) Teacher and student ability of UAS control parental strains from Orb2 RNAi-knockdown cross. (C to D) White RNAi-knockdown as teachers and students. (For [A] to [D] n= 24 biological replicates.) (**p< 1.0e-5).
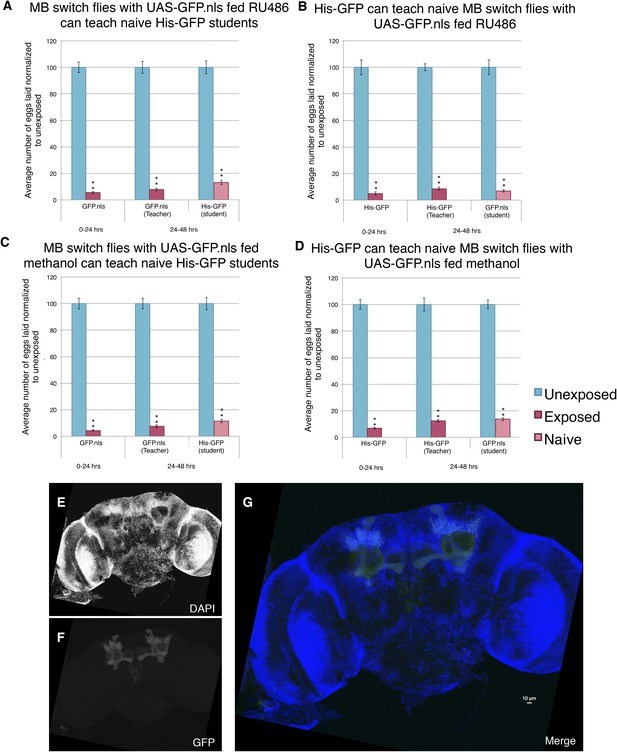
Induction of GFP in the MB using the Gene-Switch System does not perturb learning and memory.
(A to D) Percent of eggs laid normalized to unexposed. (A to B) GFP induction with RU486 feeding in the MB as teachers and students. (C to D) Lack GFP induction with methanol feeding in the MB as teachers and students. Brains from flies expressing the GeneSwitch construct (RU486+) in the MB along with a GFP nuclear localization signal (nls) showing (E) DAPI, (F) GFP expression, and (G) the merged image. Scale bar = 10 μm. (For [A] to [D] n = 24 biological replicates.) (**p < 1.0e-5).
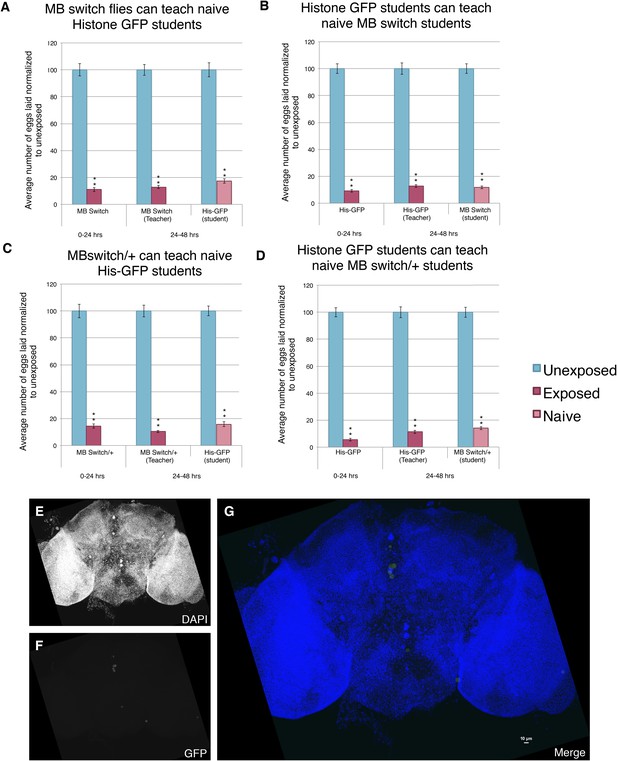
Further evidence demonstrating that induction of GFP in the MB using the GeneSwitch System does not perturb learning and memory.
(A to D) Percent of eggs laid normalized to unexposed. (A and B) MB GeneSwitch GAL4 as teachers and students. (C and D) MB GeneSwitch GAL4 /+ as teachers and students. Brains from flies not expressing the GeneSwitch construct (methanol) in the MB along with a GFP nls showing (E) DAPI, (F) GFP expression, and (G) the merged image. Scale bar = 10 μm. (For [A] to [D] n= 24 biological replicates.) (**p< 1.0e-5).
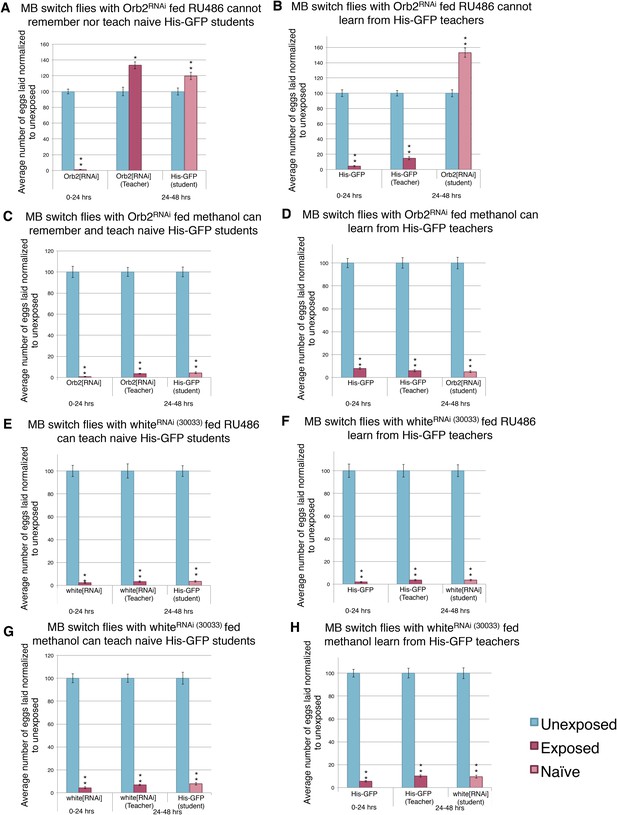
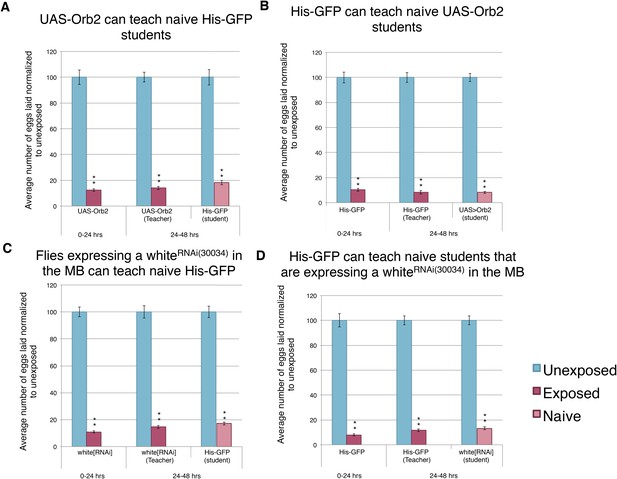
Knockdown of Orb2 in the MB using the GeneSwitch System results in defective learning.
(A to H) Percent of eggs laid normalized to unexposed. (A to B) Orb2 RNAi-knockdown in the MB (GeneSwitch) fed RU486 as teachers and students. (C to D) Orb2 RNAi-knockdown in the MB (GeneSwitch) not fed RU486 (methanol fed). (E to F) White RNAi-knockdown in the MB (GeneSwitch) fed RU486 as teachers and students. (G to H) White RNAi-knockdown in the MB (GeneSwitch) not fed RU486 (methanol fed). (For [A] to [H] n = 24 biological replicates.) (*p < 0.05, **p < 1.0e-5).
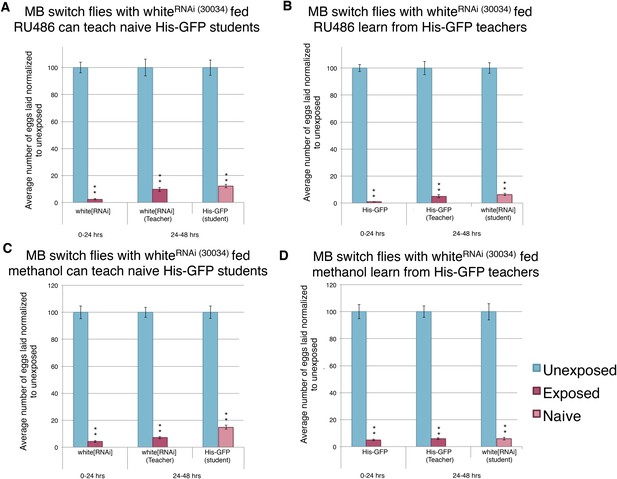
Expression of an RNAi hairpin in the MB using the GeneSwitch System does not perturb learning and memory.
(A to D) Percent of eggs laid normalized to unexposed. (A to B) White RNAi-knockdown in the MB (GeneSwitch) fed RU486 as teachers and students. (C to D) White RNAi-knockdown in the MB (GeneSwitch) not fed RU486 (methanol fed). (For [A] to [D] n= 24 biological replicates.) (**p < 1.0e-5).
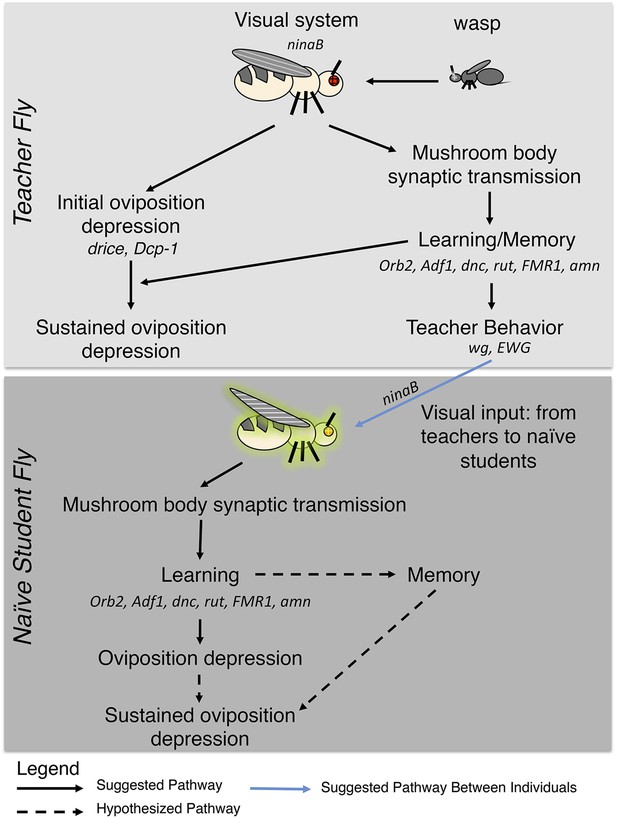
Pathway model for fly-wasp mediated social learning.
Initial oviposition depression during the 0- to 24-hr acute response period and information transmission during social learning 24- to 48-hr periods are not coupled. Sustained oviposition depression requires learning and memory genes in both teachers and students. Alleles tested for indicated genes were ninaBP315, Orb2ΔQ, Adf1nal, dnc1, dncML, rut1, rut2080, FMR1B55, FMR13, amn1, amnX8, wg1, ewg1, ewg2, ewgNs4, and drice-RNAi, Dcp-1-RNAi, Dcp-11, Dcp-13.
Additional files
-
Supplementary file 1
Absolute number of apoptotic egg chambers. (A) Absolute number of apoptotic egg chambers in Canton-S flies immediately following wasp exposure or mock exposure (0- to 24-hr period in oviposition experiments). Each replicate, replicate sum, and replicate average along with standard error is shown (*p < 0.05, **p < 0.001). (B) Absolute number of apoptotic egg chambers in GFP-Histone flies immediately following wasp exposure or mock exposure (0- to 24-hr period in oviposition experiments). Each replicate, replicate sum, and replicate average along with standard error is shown (*p < 0.05, **p < 0.001). (C) Absolute number of apoptotic egg chambers in teacher Canton-S flies 24 hr following wasp exposure or mock exposure (24- to 48-hr period in oviposition experiments). Each replicate, replicate sum, and replicate average along with standard error is shown (*p < 0.05, **p < 0.001). (D) Absolute number of apoptotic egg chambers in teacher GFP-Histone flies 24 hr following wasp exposure or mock exposure (24- to 48-hr period in oviposition experiments). Each replicate, replicate sum, and replicate average along with standard error is shown (*p < 0.05, **p < 0.001). (E) Absolute number of apoptotic egg chambers in Canton-S flies fed yeast paste immediately following wasp exposure or mock exposure (0- to 24-hr period in oviposition experiments). Each replicate, replicate sum, and replicate average along with standard error is shown (*p < 0.05, **p < 0.001). (F) Absolute number of apoptotic egg chambers in student GFP-Histone flies 24 hr following wasp exposure or mock exposure (24- to 48-hr period in oviposition experiments). Each replicate, replicate sum, and replicate average along with standard error is shown (*p < 0.05, **p < 0.001). (G) Absolute number of apoptotic egg chambers in student Canton-S flies 24 hr following wasp exposure or mock exposure (24- to 48-hr period in oviposition experiments). Each replicate, replicate sum, and replicate average along with standard error is shown (*p < 0.05, **p < 0.001). (H) Absolute number of apoptotic egg chambers in Dcp-1[RNAi] flies immediately following wasp exposure or mock exposure (0- to 24-hr period in oviposition experiments). Each replicate, replicate sum, and replicate average along with standard error is shown (*p < 0.05, **p < 0.001). (I) Absolute number of apoptotic egg chambers in teacher Dcp-1[RNAi] flies 24 hr following wasp exposure or mock exposure (24- to 48-hr period in oviposition experiments). Each replicate, replicate sum, and replicate average along with standard error is shown (*p < 0.05, **p < 0.001). (J) Absolute number of apoptotic egg chambers in student GFP-Histone flies, paired with Dcp-1[RNAi] teachers, 24 hr following wasp exposure or mock exposure (24- to 48-hr period in oviposition experiments). Each replicate, replicate sum, and replicate average along with standard error is shown (*p < 0.05, **p < 0.001). (K) Absolute number of apoptotic egg chambers in Orb2ΔQ flies immediately following wasp exposure or mock exposure (0- to 24-hr period in oviposition experiments). Each replicate, replicate sum, and replicate average along with standard error is shown (*p < 0.05, **p < 0.001). (L) Absolute number of apoptotic egg chambers in Orb2ΔQ flies 24 hr following wasp exposure or mock exposure (24- to 48-hr period in oviposition experiments). Each replicate, replicate sum, and replicate average along with standard error is shown (*p < 0.05, **p < 0.001).
- https://doi.org/10.7554/eLife.07423.033
-
Supplementary file 2
Genotypes of each fly strain used in study. Names used in study, followed by full genotype and location acquired from shown.
- https://doi.org/10.7554/eLife.07423.034
-
Supplementary file 3
Statistical analyses and corresponding P-values shown for main figures. Comparison groups, statistical test performed, sample size, and P-values are shown for a corresponding figure.
- https://doi.org/10.7554/eLife.07423.035
-
Supplementary file 4
Corresponding P-values generated from t-tests are shown for supplementary figures. Comparison groups, sample size, and P-values are shown for a corresponding figure.
- https://doi.org/10.7554/eLife.07423.036
-
Supplementary file 5
Corresponding P-values generated from t-tests are shown for supplementary file 1A–L. Comparison groups, sample size, and P-values are shown for a corresponding file.
- https://doi.org/10.7554/eLife.07423.037
-
Supplementary file 6
Corresponding raw average egg counts corresponding to main figures are shown.
- https://doi.org/10.7554/eLife.07423.038
-
Supplementary file 7
Corresponding raw average egg counts corresponding to supplementary figures are shown.
- https://doi.org/10.7554/eLife.07423.039





