Keratinocytes can modulate and directly initiate nociceptive responses
Figures
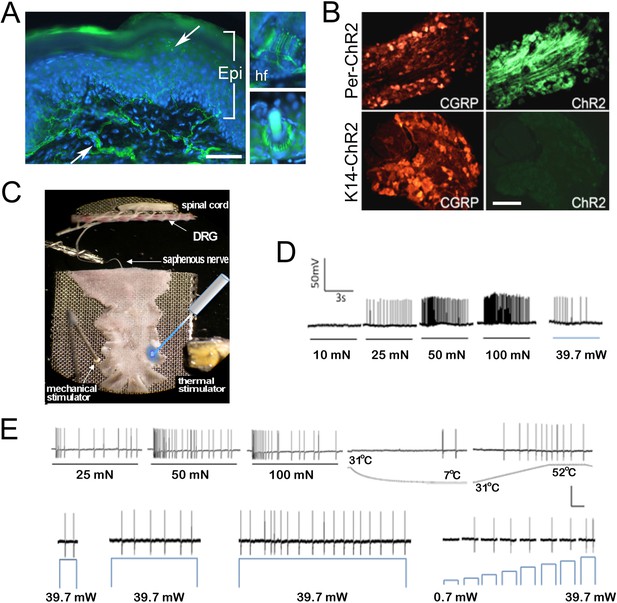
Light stimulates various types of cutaneous afferents in Prph-ChR2 transgenic mice.
(A). ChR2-YFP expression in unmyelinated and myelinated (lanceolate endings of hair shaft, panels on right) fibers of Prph-ChR2 mouse skin. Arrows indicate nerve fibers in dermis and epidermis (Epi); DAPI (blue) labeling demarcates keratinocytes. (B). ChR2 is expressed in DRG neurons of Prph-ChR2 but not KRT-ChR2 mice. CGRP labels peptidergic neurons. (C). Ex vivo preparation used for functional characterization of cutaneous afferents in response to mechanical, heat and laser stimulation. (D). Response of a Prph-ChR2 Aδ-HTMR to mechanical and blue laser stimulation. (E). Recordings from a CMHC nociceptor from a Prph-ChR2 mouse in response to mechanical, thermal and light stimulation. Calibration bars in (A) = 250 µm, (B) = 100 µm, (E) = 60 mV/1 s, top trace; 40 mV/1 s, bottom trace.
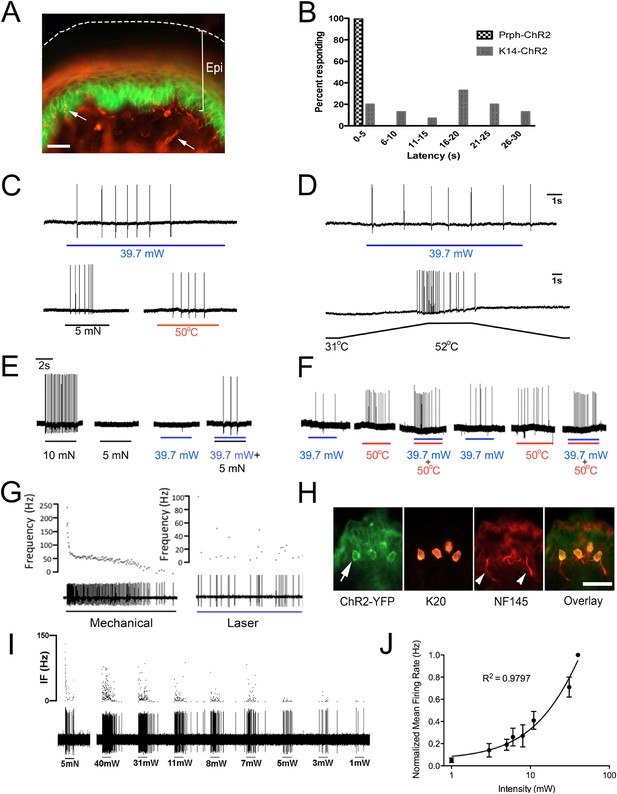
Blue light stimulates multiple subtypes of cutaneous afferents in KRT-ChR2 transgenic mice.
(A). ChR2-YFP expression in keratinocytes of glabrous skin of KRT-ChR2 mouse. PGP9.5-positive nerve fibers (red) are in dermis and epidermis (arrows). (B). Plot of behavioral responses to blue laser across time intervals for Prph-ChR2 and KRT-ChR2 mice. All Prph-Cre mice showed an immediate response (within 5 s of stimulation). All KRT-ChR2 mice also responded at least once in 10 trials and with variable latencies (see Table 1). (C). Example showing activation of a CMH fiber type in response to blue laser applied to KRT-ChR2 skin in the ex vivo preparation. Responses of this fiber to mechanical and heat stimuli are shown below laser response. (D). Example of a train of action potentials elicited in a CH fiber type in response to laser activation of the KRT-ChR2 skin. Responses of this fiber to heat stimuli are shown below laser response. (E). In this KRT-ChR2 Aβ HTMR afferent laser stimulation does not produce firing when presented alone, but does in combination with subthreshold (5 mN) mechanical stimulation. (F). Light directly activates this KRT-ChR2 CMHC fiber and summates with noxious heat stimulation. (G). SA1 Aβ-low threshold mechanoreceptor responds to mechanical and laser stimulation. (H). SA1s terminate on ChR2-YFP (green) positive Merkel cells co-labeled with anti-K20 (orange). Anti-NFH (red) labels SA1 fiber. Calibration bars in (A) and (H) = 100 µm. (I). Light-evoked responses from a SA-1 fiber at varying intensities (1–40 mW) with instantaneous frequency depicted. Pulses were 5 s in duration with 30 s between pulses. (J). Normalized mean firing rate vs light intensity plotted on a log-intensity scale. Data from 8 afferents are averaged from ascending and descending steps of light intensity, and were fit with a Boltzman sigmoidal function (R2 = 0.98).
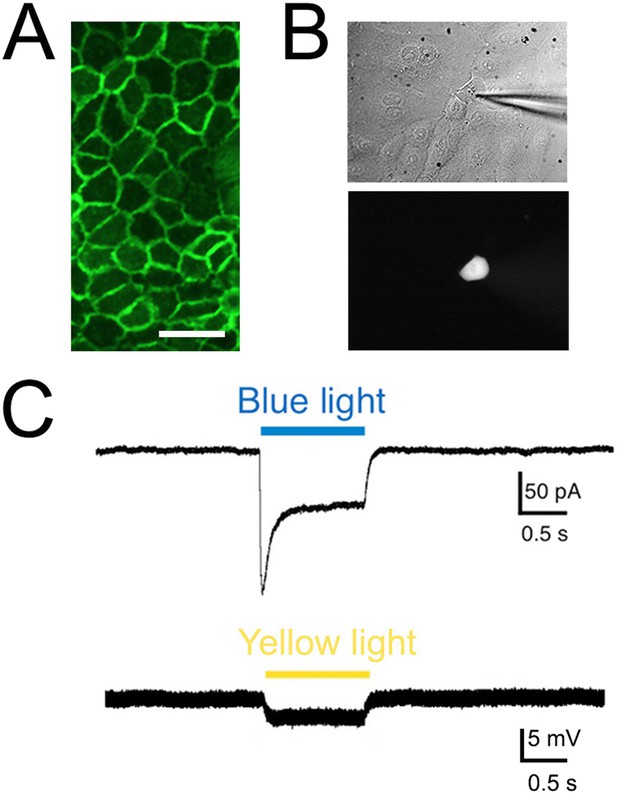
Light elicits current activation in cultured keratinocytes.
(A). Fluorescent ChR2-YFP protein in plasma membrane of keratinocytes cultured from skin of KRT-ChR2 mice. (B). IR-DIC images of patch pipette on single keratinocyte that was recorded from and then filled with Alexa 555 dye. (C). Representative trace illustrates typical current evoked by blue light stimulation of KRT-ChR2. Yellow light stimulation of KRT-NpHR keratinocytes also produced a change in voltage properties of the cell. Control KRT-Cre keratinocytes that were isolated in parallel showed no response to light (not shown). Bar in A is 40 µM.
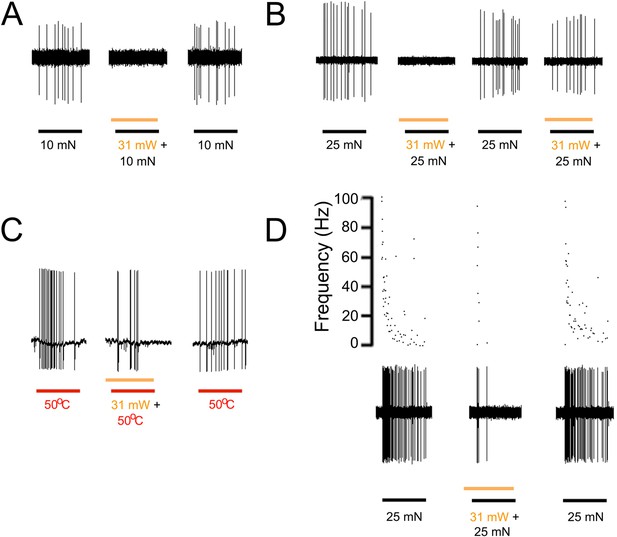
Yellow light inhibits AP firing in multiple subtypes of cutaneous afferents in KRT-NpHR mice.
(A). Yellow light decreases AP firing in response to mechanical stimulation in this Aδ-HTMR afferent. (B). In this CMH-fiber the response to mechanical stimulation is decreased with the initial yellow laser stimulation; a smaller decrease in AP firing occurred with a second laser presentation. (C). This CMH-fiber showed decreased firing in response to heat in the presence of yellow laser stimulation. (D). Responses of a SA1 fiber to mechanical stimulation are significantly reduced by activation of NpHR in epidermal keratinocytes (which are likely Merkel cells). Laser stimuli (orange bars) occurred 1 s prior to mechanical (black bar) or heat (red bar) stimuli. Duration of each stimulus was either 5 s (mechanical and heat) or 6 s (laser).
Videos
KRT-ChR2 mice exhibit nocifensive behaviors in response to blue light.
Blue light stimulation of channelrhodopsin expressing keratinocytes in the skin of KRT-ChR2 mice induces behavioral withdrawal responses. This mouse exhibits foot lifting at ∼9 s after light exposure on the glabrous skin of the hind foot.
Tables
KRT-ChR2 mice respond to blue light stimulation of paw skin
| Mouse strain | Sex | Responses/10 |
|---|---|---|
| KRT-ChR2 1 | Female | 4 |
| KRT-ChR2 2 | Female | 3 |
| KRT-ChR2 3 | Female | 1 |
| KRT-ChR2 4 | Male | 3 |
| KRT-ChR2 5 | Male | 1 |
| KRT-ChR2 6 | Male | 3 |
| Mean | 2.5 |
| KRT-Cre | Male | 0 |
| KRT-Cre | Male | 0 |
| WT | Female | 0 |
| KRT-Cre | Female | 0 |
| WT | Female | 0 |
| Mean | 0.0 |
-
All KRT-ChR2 mice respond to light applied to foot plantar skin whereas control littermates (n = 5) showed no response. The number of nocifensive responses (paw lifting, biting, licking) out of 10 stimulations was recorded. In total, light evoked responses in KRT-ChR2 mice in 17 of 60 total trials (28%). Control KRT-Cre mice lack the ChR2 gene whereas WT controls lack both transgenes.
Number of primary afferents recorded from Prph-ChR2, KRT-ChR2 and KRT-NpHR mice that showed responses to light stimulation
| Prph-ChR2 | KRT-ChR2 | KRT-NpHR | ||||
|---|---|---|---|---|---|---|
| Cell type | Responsive | Unresponsive | Responsive (direct) | Unresponsive | Responsive | Unresponsive |
| SA1 | 0 | 3 | 21 (21) | 0 | 16 | 0 |
| RA (Aβ) LTMR | 0 | 4 | 0 | 15 | 0 | 9 |
| RA (Aδ) LTMR | 0 | 1 | 0 | 6 | 0 | 2 |
| A-HTMR (Aβ) | 1 | 1 | 3 (2) | 1 | 2 | 5 |
| A-HTMR (Aδ) | 0 | 2 | 4 (2) | 6 | 5 | 7 |
| CM | 7 | 0 | 1 (0) | 1 | 2 | 4 |
| CC | 0 | 2 | 0 | 1 | 0 | 1 |
| CH | 4 | 3 | 4 (2) | 3 | 0 | 1 |
| CMC | 0 | 1 | 1 (0) | 1 | 1 | 1 |
| CMH | 11 | 3 | 6 (2) | 0 | 7 | 5 |
| CMHC | 3 | 3 | 6 (2) | 0 | 2 | 1 |
-
Fibers that were activated directly by light stimulation of KRT-ChR2 keratinocytes are in parentheses.
-
Cell types recorded from are: SA1, slowly adapting type 1; RA (Aβ), rapidly adapting A beta low-threshold mechanoreceptor; RA (Aδ), rapidly adapting A delta low-threshold mechanoreceptor, A-HTMR, high-threshold mechanoreceptor(Aβ); A-HTMR, high-threshold mechanoreceptor (Aδ); CM, C mechanoreceptor; CC, C cold receptor; CH, C heat receptor; CMC, C mechano-cold receptor; CMH, C mechano-heat receptor; CMHC, C mechano-heat and cold receptor.





