Stable G protein-effector complexes in striatal neurons: mechanism of assembly and role in neurotransmitter signaling
Figures
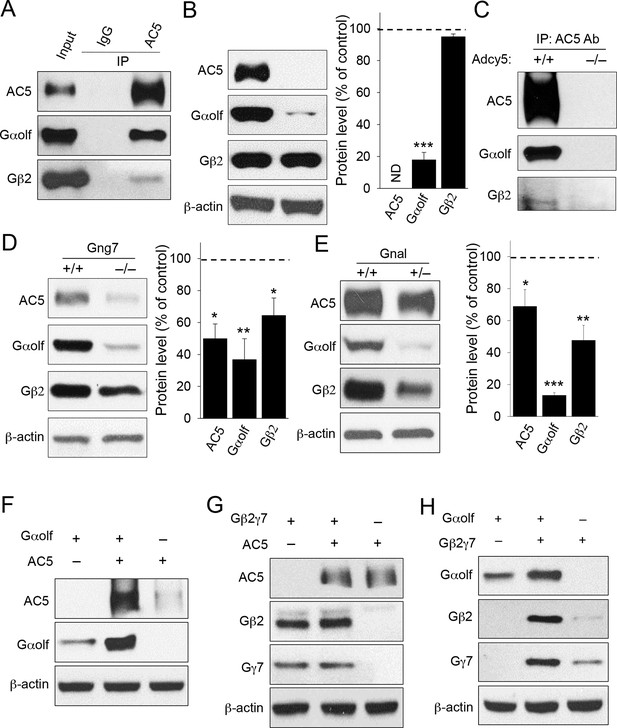
Co-dependence of heterotrimeric Golf subunits and AC5 in complex formation and expression.
(A) Immunoprecipitation of AC5 complexes from striatal lysates of wild type mice. Specific anti-AC5 antibodies but not non-immune IgGs pull down Gαolfβ2γ7 subunits from native striatal tissues. (B) Significant reduction of Gαolf expression in striatum tissue from mice lacking AC5. Total striatal lysates were analyzed by immunoblotting using indicated antibodies and quantified by densitometry. ***p<0.001, Student’s t-test, n = 3 mice. (C) Immunoprecipitation of AC5 from wild type (+/+) and Adcy5-/- tissues confirms specificity of AC5-Golf binding. Same anti-AC5 antibodies were used in both immunoprecipitaiton experiments and samples were processed in parallel. (D) Significant reduction of AC5 and Gαolf expression in striatal tissues from mice lacking Gγ7. Total striatal lysates from wild type and Gng7-/- mice were analyzed by immunoblotting with indicated antibodies and quantified by densitometry. *p<0.05, **p<0.01, Student’s t-test, n = 3 mice. (E) Significant reduction of AC5 and Gβ2 expression in striatal tissues from mice with reduced expression of Gαolf. Total striatal lysates from wild type and Gnal+/- mice were analyzed by immunoblotting with indicated antibodies and quantified by densitometry. *p<0.05, **p<0.01, ***p<0.001, Student’s t-test, n = 3 mice. (F) Mutual stabilization of AC5 and Gαolf upon co-transfection in HEK293 cells. Equal amounts of cDNAs were transfected into cells as described in the Methods section and protein expression was assessed by immunoblotting with specific antibodies (G) Lack of co-stabilization between AC5 and Gβ2γ7 subunits in co-transfected HEK293 cells. Indicated constructs were transfected into cells and changes in protein expression were monitored by immunoblotting with specific antibodies. (H) Mutual stabilization of Gαolf subunits with Gβ2γ7 subunits in co-transfected HEK293 cells. The experiment was conducted as described for panel G.
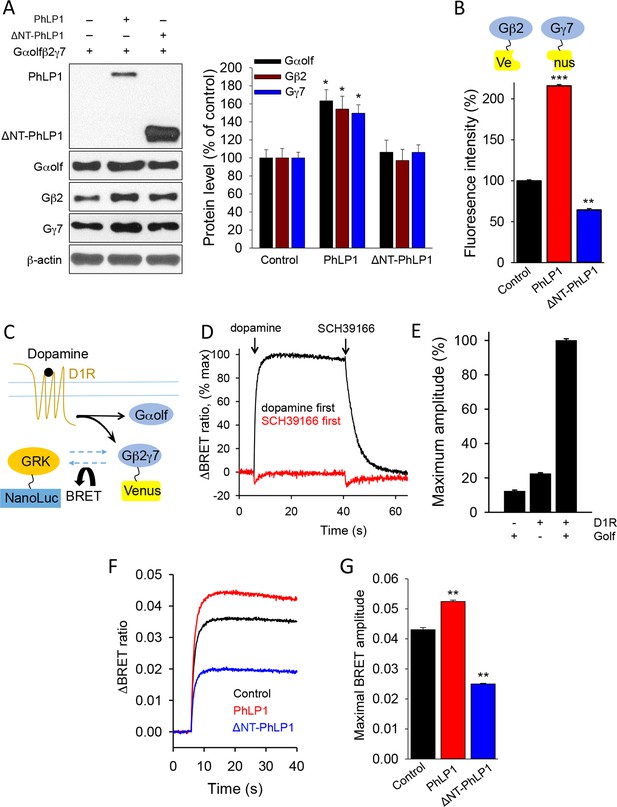
PhLP1 facilitates functional assembly of Gαolfβ2γ7 complex.
(A) Left, Full length of PhLP1 but not its N terminally truncated mutant ΔNT-PhLP1 increases the expression level of Gαolfβ2γ7 subunits upon overexpression in HEK293 cells. Right, quantification of immunoblot data from 3 independent experiments. Data were normalized to the individual protein expression in the control group without PhLP1 transfection. Data were analyzed by One-Way ANOVA (Gαolf F[2, 9] = 8.731, p = 0.008; Gβ2 F[2, 9] = 6.688, p = 0.017; Gγ7 F[2, 9] = 11.107, p = 0.004). *p<0.01 compared to the control group post hoc Tukey’s test. (B) Full length PhLP1 facilitates, while ΔNT-PhLP1 inhibits Gβ2γ7 assembly. Venus fluorescence intensity was used as a readout of Gβ2γ7 complex assembly in a complementation experiment in transfected HEK293 cells. Data were analyzed by One-Way ANOVA (F[2, 15] = 2719.521, p<0.001). **p<0.01, ***p<0.001 compared with the control group, post hoc Tukey’s test. (C) Schematic diagram of BRET sensor strategy for examining the dissociation and reassociation of Gαolf and Gβ2γ7 subunits upon D1Rs activation and inactivation. (D) Representative BRET response traces. Cells transfected with D1R and Golf were stimulated by 100 μM dopamine followed by 100 μM SCH39166 (black) or by 100 μM SCH39166 followed by 100 μM dopamine (red). First and second ligands were applied at 5 and 40 s, respectively. (E) Control experiments examining the requirement of both Golf and D1R to transduce the signal. Cells were transfected with the three different conditions, Golf only, D1R only, or D1R plus Golf. Each bar represents the mean of 6 replicates. (F) Representative BRET signal traces in response to D1 receptor activation with dopamine (100 μM). (G) Comparison of maximal BRET ratios. Data were analyzed by One-Way ANOVA (F[2, 9] = 706.655, p<0.001). **p<0.01 compared with the control group, post hoc Tukey’s test.
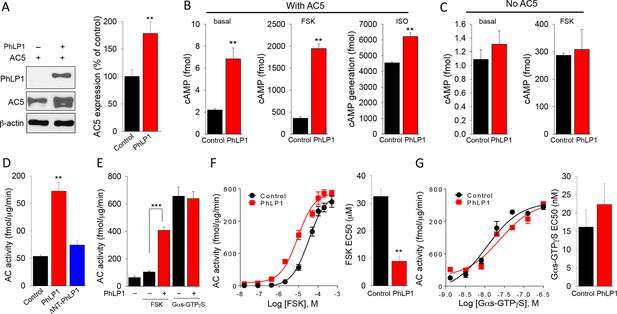
PhLP1 augments expression and activity of AC5.
(A) Co-expression of PhLP1 significantly increases AC5 expression in HEK293 cells. **p<0.01 Students’ t-test, n = 3. (B) PhLP1 enhances cAMP levels under basal condition, or in response to stimulation with forskolin (FSK, 1 μM, 5 min) or isoproterenol (ISO, 1 μM, 5 min) in AC5 expressing HEK293 cells. **p<0.01 Student’s t-test, n = 3. (C) PhLP1 has no effect on cAMP generation in HEK293 cells without AC5 overexpression. n = 3 (D) Effects of purified recombinant full length PhLP1 and ΔNT-PhLP1 on adenylyl cyclase activity in striatal membranes. Assays were performed in the absence of GTP. Striatal membranes were pre-incubated with 0.5 μM of purified proteins at 4°C for 20 min and then subjected to adenylyl cyclase activity assay. Data were analyzed by One-Way ANOVA (F[2, 9] = 35.477, p<0.001). **p<0.01, post hoc Tukey’s test. n = 3 (E) Purified recombinant PhLP1 enhances forskolin-stimulated adenylyl cyclase activity in striatal membranes. Membranes were pre-incubated with 0.5 μM of purified proteins at 4°C for 20 min. Membranes were then stimulated with 1 μM forskolin or 80 nM Gαs-GTPγS. Data were analyzed by One-Way ANOVA (F(4, 15) = 52.291, p<0.001).***p<0.001, post hoc Tukey’s test. n = 3 (F) Effect of purified recombinant PhLP1 on the dose response of forskolin-mediated activation of adenylyl cyclase in striatal membranes. The EC50 for forskolin was 32.5 ± 2.7 μM in the control reaction and 8.9 ± 1.6 μM in PhLP1-treated reaction. **p<0.01 Student’s t-test, n = 3. (G) Effect of purified recombinant PhLP1 on the dose response of Gαs-GTPγS-mediated activation of adenylyl cyclase in striatal membranes. The EC50 for Gαs-GTPγS was 16.2 ± 4.7 nM in the control reaction and 22.3 ± 5.7 nM in PhLP1-treated reaction (n = 3 reactions).
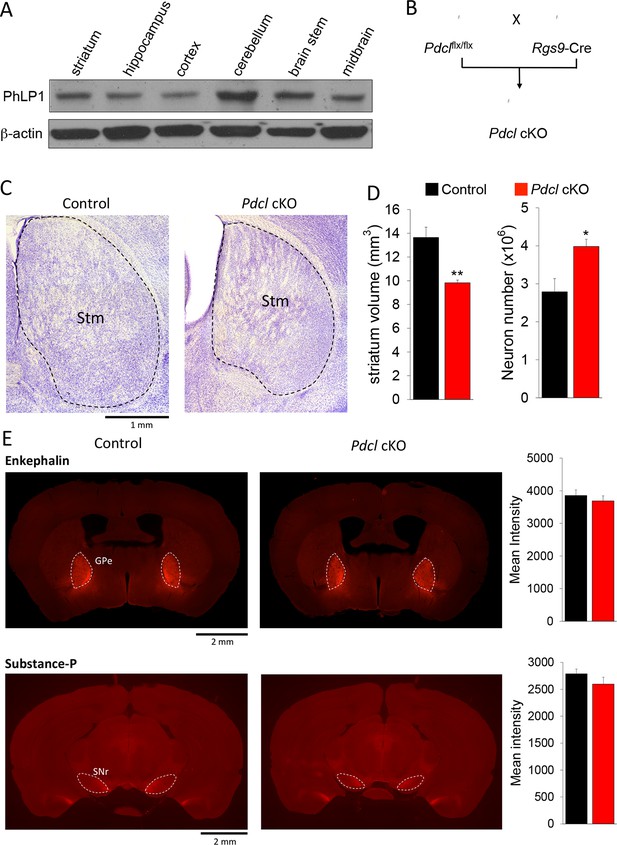
Elimination of PhLP1 does not impact survival and connectivity of striatal neurons.
(A) PhLP1 expression in different brain regions from adult mice as determined by immunoblot analysis. (B) Generation of PhLP1 conditional knockout out. Pdclflx/flx mice were crossed with Rgs9-Cre mice to generate striatal specific PhLP1 conditional knockout (Pdcl cKO) mice (C) Representative images of Nissl-stained coronal brain sections from adult control and Pdcl cKO mice. Stm, striatum. (Scale bar, 1 mm). (D) Left: Striatal volume of adult Pdcl cKO mice was reduced by 30% as compared with control mice (n = 4 mice each). Error bars represent SEM. Student’s t test: **p<0.01. Right: Counts of striatal neurons in Pdcl cKO and control mice obtained from Nissl-stained sections. The striatal neuron counts were increased by 43% compared with control mice (n = 4 mice each). Error bars represent SEM. Student’s t test: *p<0.05. (E) Left: Representative images of anti-enkephalin and anti-substance-P stained brain sections. GPe, external globus pallidus. SNr, substantia nigra pars reticulate. (Scale bar, 2 mm.) Right: Quantification of immunofluorescent signal for enkephalin and substance-P. Mean intensity of signals from both antibodies showed no significant difference between control and Pdcl cKO mice. Error bars represent SEM.
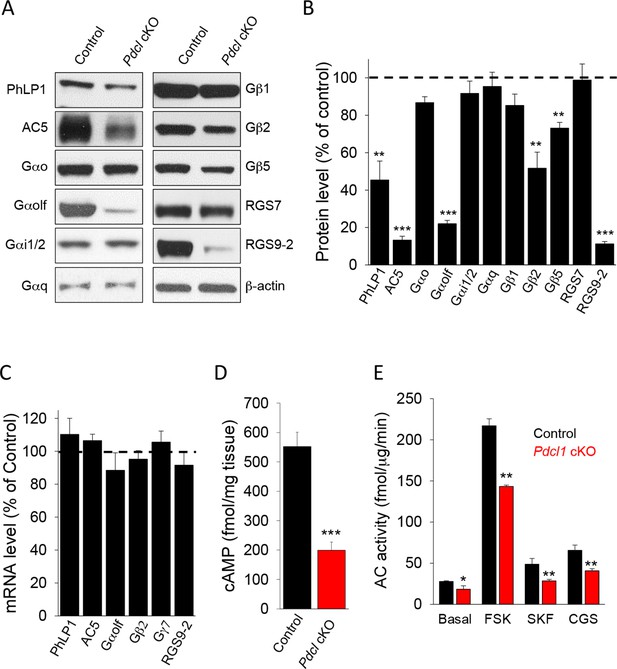
Elimination of PhLP1 in striatal neurons significantly impairs expression and function of AC5-Golf complex.
(A) Representative immunoblot data of AC5, different G protein subunits and RGS proteins in stratal tissues from adult control or Pdcl cKO mice. (B) Quantification of protein levels. Data were normalized to the percentage of protein level in control mice. **p<0.01, ***p<0.001, Student’s t-test, n = 3 mice. (C) mRNA quantification in striatum tissue from control and Pdcl cKO mice. Data were normalized to the percentage of mRNA level in control mice. (D), Basal cAMP level was reduced in the striatum tissue of Pdcl cKO mice. ***p<0.001, Students’ t-test, n = 3 mice. (E) Striatal membrane adenylyl cyclase activity is reduced in Pdcl cKO mice. Adenylyl cyclase activity was measured either under basal conditions (without stimulation), or in the presence of forskolin (FSK, 1 μM), D1R specific agonist SKF38391 (SKF, 10 μM) or A2AR agonist CGS21680 (CGS, 10 μM). *p<0.05, **p<0.01, Student’s t-test, n = 3 reactions.
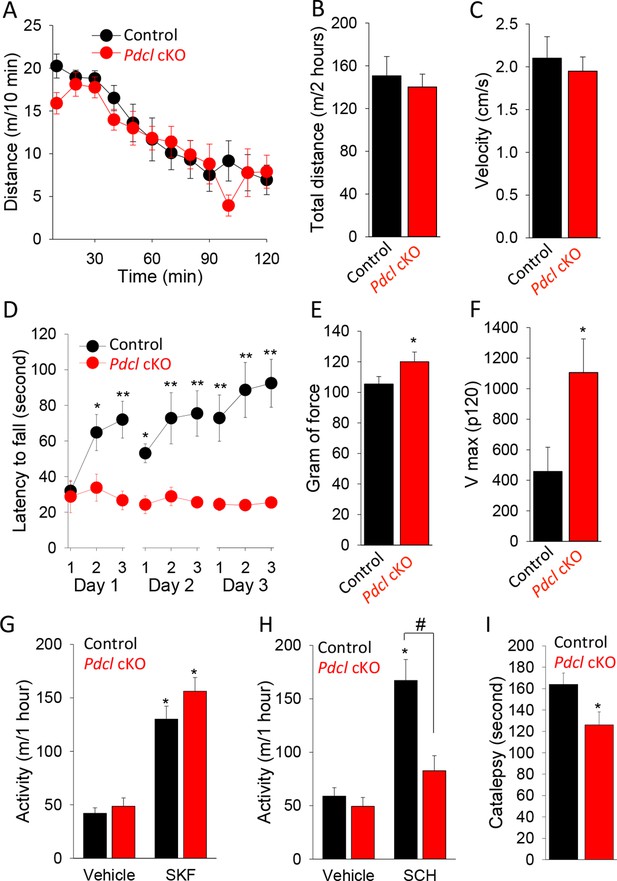
Behavioral consequences of PhLP1 elimination in striatal neurons.
(A) Pdcl cKO mice display normal basal locomotion and habituation to a novel environment. (B) Total distance traveled in 2 hr in the open field chamber. (C) Average velocity in the open field chamber. (D) Pdcl cKO mice exhibit severe deficits in motor learning behavior in rotarod test. There were statistically significant differences between genotype as determined by Two-way ANOVA (F[1, 153] = 63.518, p<0.001). *p<0.05, **p<0.01, ***p<0.001, post hoc Tukey’s test, n = 9 and 10 for control mice and Pdcl cKO mice, respectively. (E) Increased grip strength of the forelimbs in Pdcl cKO mice. *p<0.05, Student’s t-test, n = 8 mice for each genotype. (F) Increased acoustic startle response of Pdcl cKO as measured by Vmax in response to 120 dB white noise bursts. (G) Normal locomotor response to D1R agonist SKF 38,393 (SKF, 50 mg/kg, i.p.) in Pdcl cKOcompared to control mice. Mice were injected with vehicle or SKF 38,393 (SKF, 50 mg/kg, i.p.) and immediately put in open field chambers. The locomotion was recorded for 1 hr. Data were analyzed by Two-way ANOVA (treatment F[1, 28]=96.068, p<0.001, genotype F[1, 28] = 2.679, p = 0.113). *p<0.01 post hoc Tukey’s test compared to the vehicle control of the same genotype. n=8 mice per each genotype. (H) Blunted response to A2AR antagonist SCH58261 (SCH, 3 mg/kg, i.p.) treatment. Data were analyzed by Two-way ANOVA (treatment F(1, 28) = 42.819, p<0.001, genotype F(1, 28) = 20.181, p<0.001). *p<0.01 post hoc Tukey’s test compared to the vehicle control of the same genotype, #p<0.05 post hoc Tukey’s test in comparison between genotypes in response to SCH 58261. n = 8 mice per each genotype. (I) Reduced catalepsy in response to D2R antagonist haloperidol (2 mg/kg, i.p.) in Pdcl cKO mice. Catalepsy was measured in the bar test 1 hr after haloperidol (2 mg/kg, i.p.) administration. *p<0.05, Student’s t-test. n = 8 mice per each genotype.
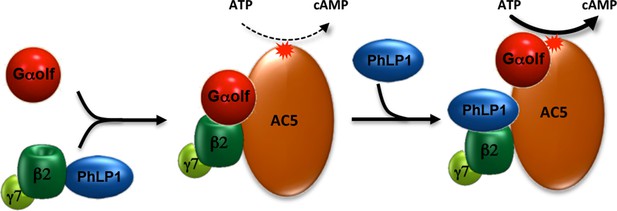
Schematic illustration of PhLP1 involvement in regulating Golf-AC5 complex assembly and signaling.
PhLP1 promotes biogenesis of Gβ2γ7 and assembly of its complex with Golf. The trimeric Gαolfβ2γ7 forms stable complexes with AC5 contributing to its proteolytic stability. In addition, PhLP1 regulates cAMP production by influencing Golf arrangement on AC5.





