A synaptic F-actin network controls otoferlin-dependent exocytosis in auditory inner hair cells
Figures
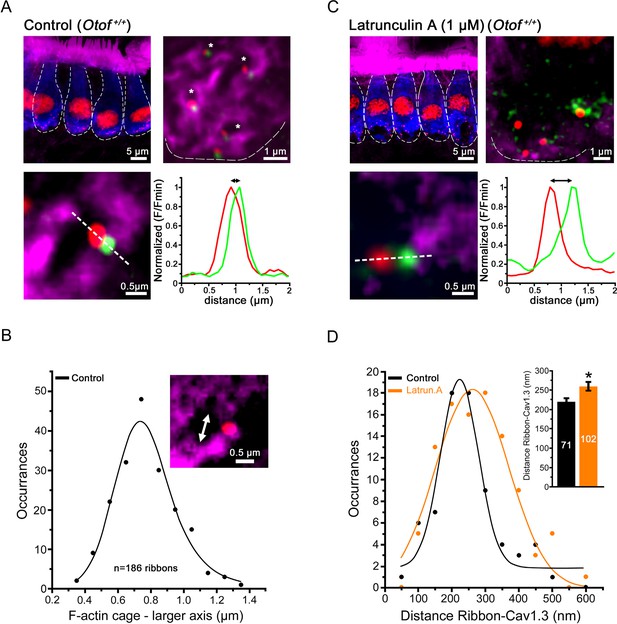
Confocal imaging of the synaptic F-actin cages in IHCs.
(A) Confocal images from averaged Z-stack projection (20 slices of 0.25 µm) of P13-IHCs labeled in blue with otoferlin-immuno-reactivity. Directly visualized with fluorescent-phalloidin (purple), F-actin intensively labelled the cuticular plate and the stereocilia but also in a punctated manner the synaptic basal pole of the IHCs. In this latter area, at higher magnification (averaged Z-stack projection of 8 slices of 0.25 µm), the synaptic F-actin forms a mesh of cages (see right panel where the blue channel of otoferlin is omitted; the cages are indicated by the white asterisks). At each border of the synaptic F-actin cages was generally attached one synaptic ribbon (red) and one associated Cav1.3 patch (green) as indicated in the lower left panel. The graph represents an example of fluorescent intensity profile through the white dashed line crossing the ribbon and the associated Cav1.3. (B) The graph indicates the Gaussian distribution of the larger axis (double white arrow head) of each F-actin cage. (C) A 45 min treatment with extracellular latrunculin-A disrupted the synaptic F-actin cages. The black holes at the base of the IHCs likely indicated swollen IHC active zones produced by the synaptic F-actin disorganization. At higher magnification (right panel), note also the disorganization of the Cav1.3 clusters (green) at the ribbons, as indicated by a larger distance in their respective fluorescent intensity profile distribution (bottom graph). (D) Comparative Gaussian distribution of the center mass distance between Cav1.3 and ribbon in controls (black, n = 71 active zones) and latrunculin-treated (orange, n = 102 active zones) IHCs. The inset histogram indicates the mean ± SEM distance in both conditions. *p<0.05.
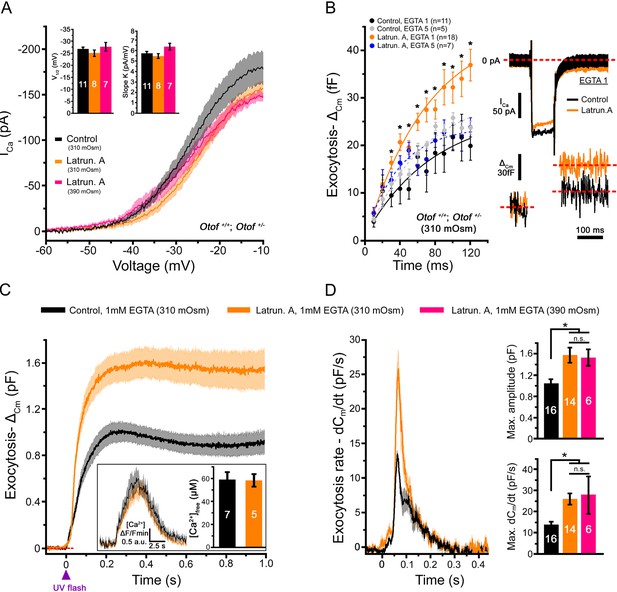
Latrunculin-A treatment facilitated exocytosis in IHCs.
(A) Ca2+ currents, evoked by a voltage-ramp protocol, were not significantly affected by latrunculin-A (orange). The parameters of the Boltzman fit are indicated by the histograms (mean ± SEM). Rising intracellular osmotic pressure from 310 mOsm to 390 mOsm did not affect the Ca2+ currents in latrunculin-treated IHCs (pink). For each curves, the darker line indicates the mean responses and the light filled area the standard error. (B) Exocytosis evoked by voltage-steps from -80 mV to -10 mV was largely facilitated after latrunculin-A treatment. At right, examples of Ca2+ currents and capacitance jumps (△Cm) are shown for a control (black traces) and latruncutin-treated IHC (orange traces). In 5 mM intracellular EGTA condition, the facilitation was greatly reduced (blue points). Data are represented as mean ± SEM. *p<0.05. (C) Exocytosis under Ca2+ uncaging was also facilitated after latrunculin-A treatment. The jump in the concentration of intracellular free Ca2+ was similar in control and latrunculin-treated IHCs (inset; p = 0.9). (D) Left, comparative exocytotic rates of control and latrunculin-treated IHCs obtained from the first derivative (dCm/dt) of the curves in C. Right, comparative maximum exocytotic amplitude and peak rate histograms (mean ± SEM). Increasing intracellular hydrostatic pressure from 310 to 390 mOsm (pink bars) did not affect exocytosis in latrunculin-treated IHCs. Numbers of cells are indicated in the histogram. *p<0.05 and n.s. as non significative.
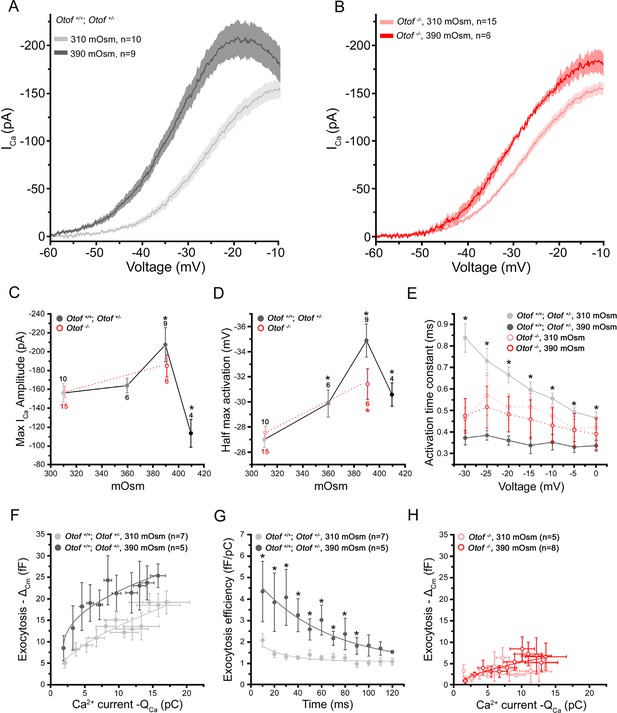
Intracellular hydrostatic pressure modulates Ca2+ currents and exocytosis.
(A) Comparative Ca2+ currents evoked by a voltage-ramp protocol in Otof+/- and Otof+/+IHCs (expressing otoferlin). Cells were recorded with intracellular osmotic pressure at 310 mOsm (n = 10) or at 390 mOsm (n = 9). The darker line indicates the mean responses and the light filled area the standard error. (B) Ca2+ currents in Otof-/- IHCs (lacking otoferlin) recorded in conditions similar to A. (C-D) Both maximum amplitude and half-maximum voltage-activation of Ca2+ currents were maximally affected at 390 mOsm. Note that the slopes of the Boltzman fit of the activation curve in A and B were found slightly affected in IHCs expressing otoferlin (310 mOsm: K = 5.90 ± 0.14 pA/mV, n = 10 and 390 mOsm: K = 5.36 ± 0,09 pA/mV, n = 9; p < 0.05) but not in Otof-/-IHCs (310 mOsm: K= 6.18 ± 0.20 pA/mV, n=6 and 390 mOsm: K= 5.89 ± 0.28, n = 6; p = 0.71) (data not shown). Data are represented as mean ± SEM, with the number of cells indicated above each point and (*) indicating p < 0.05. (E) Activation kinetics of Ca2+ currents, evoked by voltage-steps from -80 mV to different voltage levels, were significantly faster at 390 mOsm in hair cells expressing otoferlin but not in Otof-/- IHCs. For unknown reason the activation kinetics were faster in Otof-/-IHCs as compared to IHCs expressing otoferlin. Data are expressed as mean ± SEM. * p < 0.05. The number of cell is similar to D. (F) Ca2+ efficiency of exocytosis was plotted as the change in membrane capacitance (ΔCm) against the integral of the calcium current (QCa) when depolarizing IHCs at a constant voltage-step from -80 to -10 mV with increasing duration from 10 to 120 ms. Data points at 310 mOsm and 390 mOsm were fitted with a power function with a Ca2+ efficiency slope A = 2.47 ± 0.45 fF/pC and 9.30 ± 2.5 fF/pC (p < 0.05) and a (power cooperative index) = 0.33 and 0.51 (p = 0.52), respectively. The supralinear power cooperative index of 0.3 and 0.51 found here was somewhat lower to previous values reported by Cho et al. (2011) and Johnson et al (2005). (G) The exocytotic efficiency of each data point in F (ΔCm/QCa) was plotted for each depolarizing time at 310 mOsm and 390 mOsm. Data were fitted with an exponential function with 22.2 ± 10.4 ms and 50.1 ± 11.9 ms, respectively (p < 0.05). (H) Ca2+ efficiency of exocytosis (recorded in conditions similar to F) was unaffected when rising osmotic pressure from 310 to 390 mOsm in Otof-/-IHCs. Data were best fitted with a linear function with a similar slope of 0.12 ± 0.1 fF/pC and 0.48 ± 0.1 fF/pC (p = 0.2) at 310 and 390 mOsm, respectively. Data are represented as mean ± SEM. * p < 0.05.
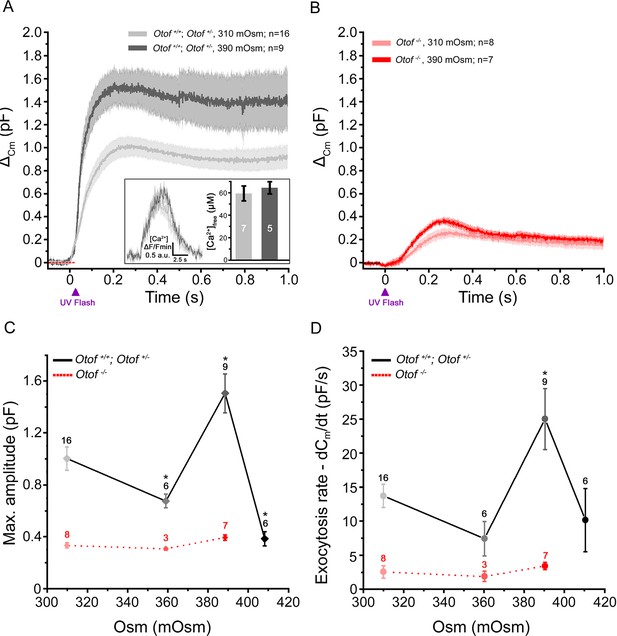
Exocytosis triggered by Ca2+ uncaging is sensitive to intracellular hydrostatic pressure.
(A) Exocytosis in IHCs expressing otoferlin was largely potentiated when rising osmotic pressure from 310 mOsm (light grey) to 390 mOsm (dark grey). The darker line in each condition indicates the mean responses and light filled area the standard error. The jump in the concentration of intracellular free Ca2+ was similar in 310 and 390 mOsm conditions (inset; p = 0.6). (B) Exocytosis evoked in Otof-/- IHCs in conditions similar to A. (C-D) Comparative maximum amplitude and peak exocytotic rate at various intracellular osmotic pressure. The results showed a maximum facilitation at 390 mOsm. These pressure effects were not seen on the residual slow exocytosis of Otof -/- IHCs. Data points are means ± SEM. The number of IHCs is indicated above each point. * p < 0.05.





