Novel origin of lamin-derived cytoplasmic intermediate filaments in tardigrades
Figures
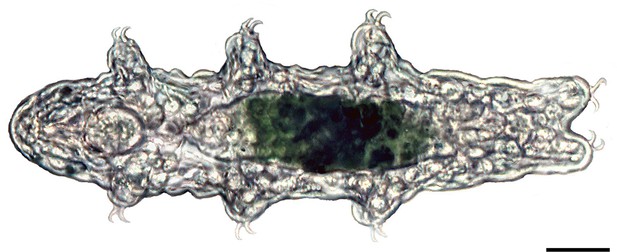
Light micrograph of a specimen of the tardigrade Hypsibius dujardini in dorsal view.
Anterior is left. Scale bar: 20 µm.

Structure and organization of the IF proteins of the tardigrade Hypsibius dujardini.
(A) Protein sequence alignment of H. dujardini IF proteins with human (Homo sapiens) lamins A and B1. The names of H. dujardini IF proteins were chosen according their structural similarities to known IF proteins (lamin-1 and lamin-2: lamin-like; cytotardin: cytoplasmic IF-like; see text for details). Note the sequence similarities of the rod domains (coil 1A, L1, coil 1B, linker L12 and coil 2) and the intermediate filament consensus motifs (highlighted in red with highly conserved parts in a box) among all three proteins. The positions of rod sub-domains are placed as described for human IF proteins (review Chernyatina et al., 2015). The nuclear localization signal in lamin-1, lamin-2, lamin A and lamin B1 is highlighted in green and the immunoglobulin fold (Ig fold) is marked in light orange. Note the absence of an Ig fold in cytotardin. Predictions (Jpred3, JNetPRED) of α-helices and β-sheets are indicated by red waved underlines and solid underlines, respectively. The C-terminal prenylation motif of lamins (CaaX) is marked in purple. The alignment (Clustal Omega) has been performed using Analysis Tool Web Services from the EMBL-EBI (McWilliam et al., 2013). (*) indicates positions which have a single, fully conserved residue. (:) Indicates conservation between groups of strongly similar properties — scoring > 0.5 in the Gonnet PAM 250 matrix. (.) Indicates conservation between groups of weakly similar properties — scoring ≤ 0.5 in the Gonnet PAM 250 matrix. (B) Organization of the three IF proteins of H. dujardini. Dark blue colour in the 1B coil of the rod domain indicates six heptads that have been lost in the chordate lineage of cytoplasmic intermediate filament proteins. The numbers denote the amino acid positions of the beginning and end of each rod sub-domain. 1A, 1B and 2, coiled-coil segments of the rod domain; CaaX, isoprenylation motif at the carboxyl terminus; LTD, lamin tail domain; NLS, nuclear localization signal.
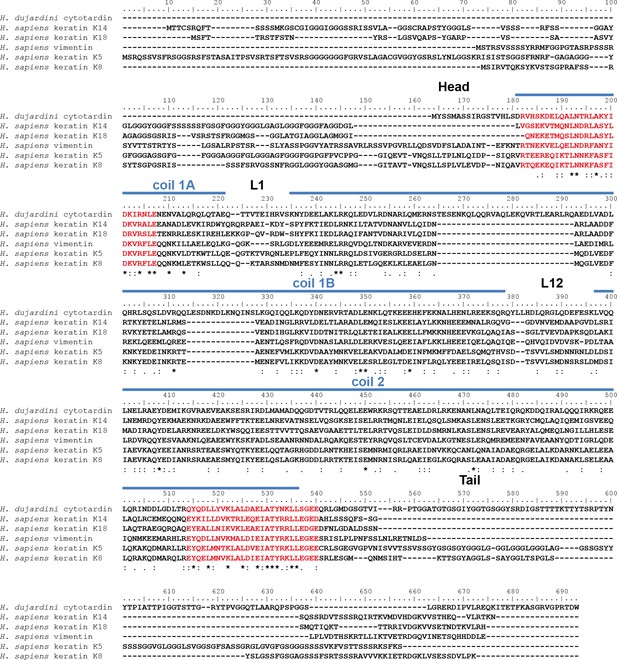
Protein sequence alignment of Hypsibius dujardini cytotardin with selected human (Homo sapiens) cytoplasmic IF proteins.
Human type I keratins K14 (epidermis) and K18 (simple epithelia) are forming IFs from obligatory heterodimers with type II keratins K5 (epidermis) and K8, respectively. The type III IF protein vimentin is able to form homopolymeric IFs. The position of the cytotardin rod domain, containing the coiled-coil and linkers (coil 1A, L1, coil 1B, L12, coil 2; predicted from the protein sequence alignment in Figure 2), is indicated by blue lines. Note the sequence similarities of the rod domain, especially at the rod domain-flanking intermediate filament consensus motifs (highlighted in red). Note also 42 amino acids in the coil 1B of cytotardin, which have been deleted from the ancestral cytoplasmic IF protein gene in chordates. The alignment (Clustal Omega) has been performed using Analysis Tool Web Services from the EMBL-EBI (McWilliam et al., 2013). (*) indicates positions which have a single, fully conserved residue. (:) Indicates conservation between groups of strongly similar properties — scoring > 0.5 in the Gonnet PAM 250 matrix. (.) Indicates conservation between groups of weakly similar properties — scoring ≤ 0.5 in the Gonnet PAM 250 matrix.
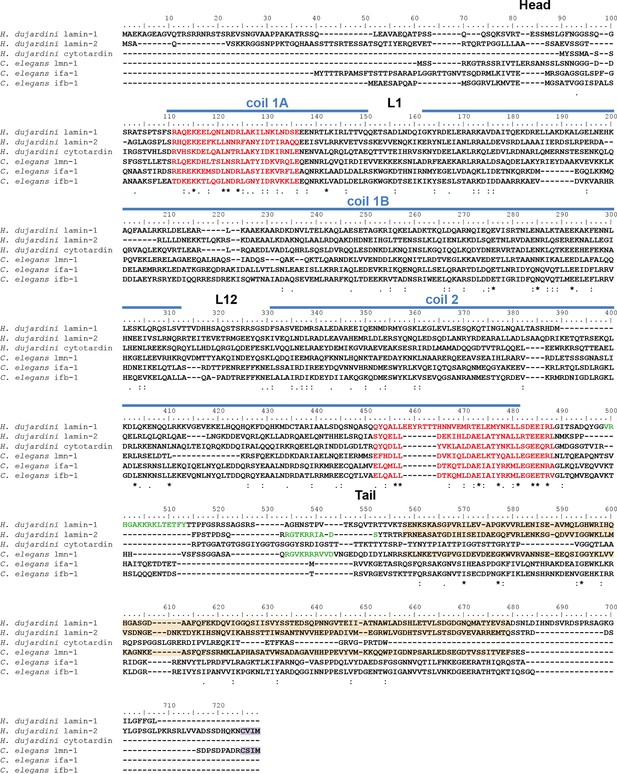
Protein sequence alignment of Hypsibius dujardini IF proteins with selected IF proteins from Caenorhabditis elegans.
C. elegans ifa-1 and ifb-1 are epithelial cytoplasmic intermediate filament proteins, whereas lmn-1 represents the single lamin of C. elegans. The position of the cytotardin rod domain, containing the coiled-coil and linkers (coil 1A, L1, coil 1B, L12, coil 2; predicted from the protein sequence alignment in Figure 2), is indicated by blue lines. The intermediate filament consensus motifs are highlighted in red, the immunoglobulin fold (Ig fold) is marked in light orange, and the C-terminal prenylation motif of lamins (CaaX) is marked in purple. Note the presence of a prenylation motif in H. dujardini lamin-2 and C. elegans lmn-1 and its absence in the other proteins. The alignment (Clustal Omega) has been performed using Analysis Tool Web Services from the EMBL-EBI (McWilliam et al., 2013). (*) indicates positions which have a single, fully conserved residue. (:) Indicates conservation between groups of strongly similar properties — scoring > 0.5 in the Gonnet PAM 250 matrix. (.) Indicates conservation between groups of weakly similar properties — scoring ≤ 0.5 in the Gonnet PAM 250 matrix.
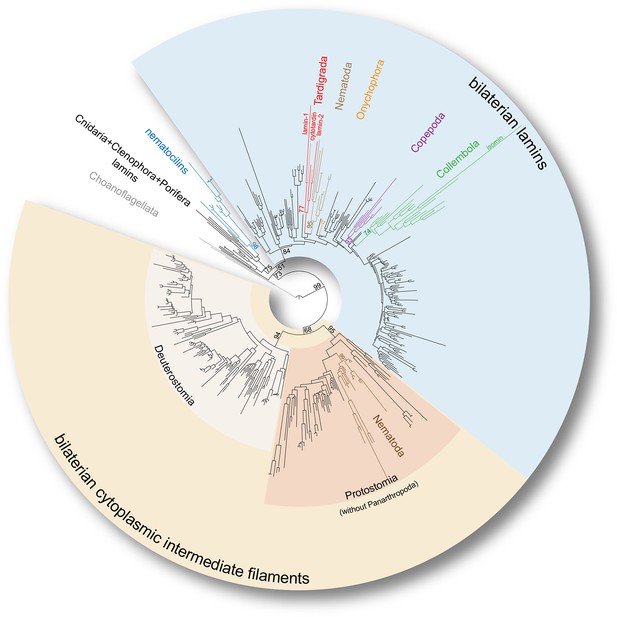
Phylogeny of the metazoan intermediate filament proteins illustrating the position of the three tardigrade IF proteins (highlighted in red).
The tree was obtained from a Maximum likelihood analysis under a dataset-specific GTR+G substitution model of 447 eukaryotic intermediate filament proteins (see Figure 3—figure supplement 1 for the full tree). Note that all tardigrade as well as collembolan (green) and copepod IF proteins (purple) belong to the bilaterian lamin clade (light blue). Hence, cytotardin and isomin are closer related to, for example, nematode lamins (light brown) than to nematode cytoplasmic IF proteins (dark brown), which are orthologs of the bilaterian cytoplasmic IF proteins (yellow). Selected bootstrap support values are given at particular nodes.
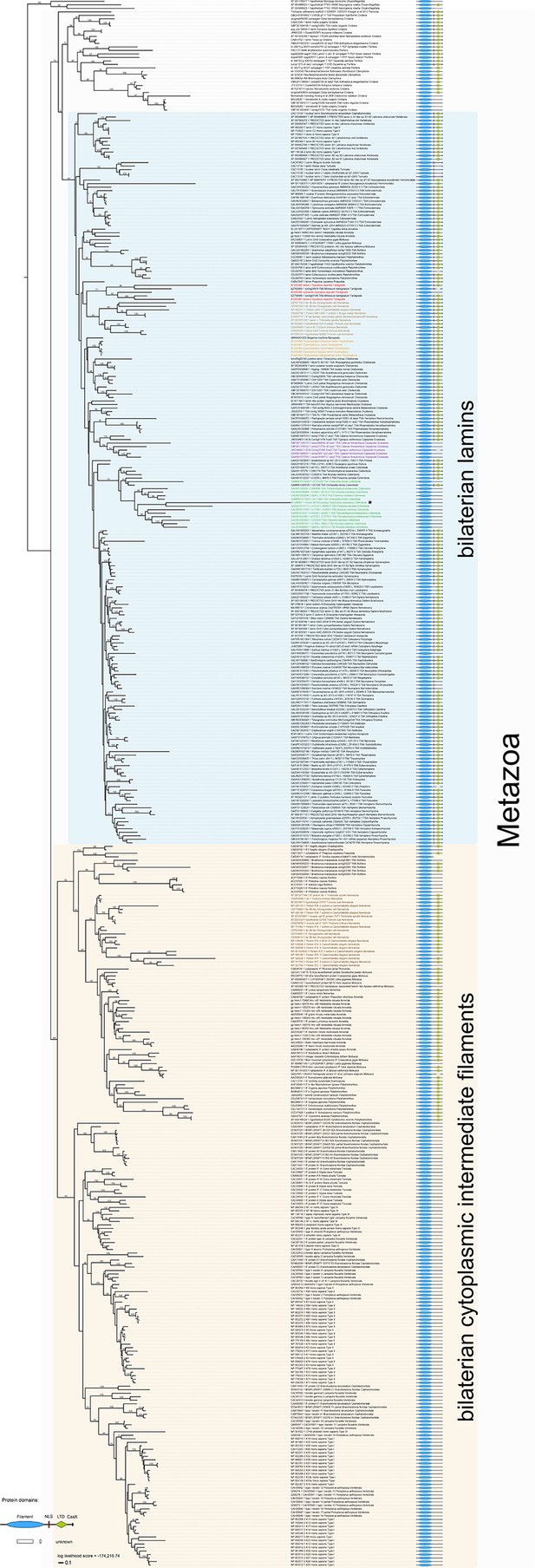
Maximum likelihood tree under a dataset-specific GTR+G substitution model and accession numbers of 447 eukaryotic intermediate filament proteins and the placement of the IF protein genes of the tardigrade Hypsibius dujardini (highlighted in red).
Lamins of onychophorans are highlighted in orange, copepods in purple, collembolans in green, nematodes in light brown and cytoplasmic IF proteins of nematodes in dark brown. Note the position of isomin of the collembolan Isotomurus maculatus, which has been interpreted as a putative cytoplasmic IF protein (Mencarelli et al., 2011), within a group of collembolan lamins (asterisk). The domain structure of each protein is depicted on the right. Note the absence of a nuclear localization signal (NLS) in cytotardin of H. dujardini as well as in the coloured sequences of copepods (purple) and collembolans (green). Bootstrap values from 1,000 pseudoreplicates ≥50% are given at the nodes. Scale bar indicates the number of substitutions per site. Abbreviations: CaaX, isoprenylation motif at the carboxyl terminus; LTD, lamin tail domain; NLS, nuclear localization signal.
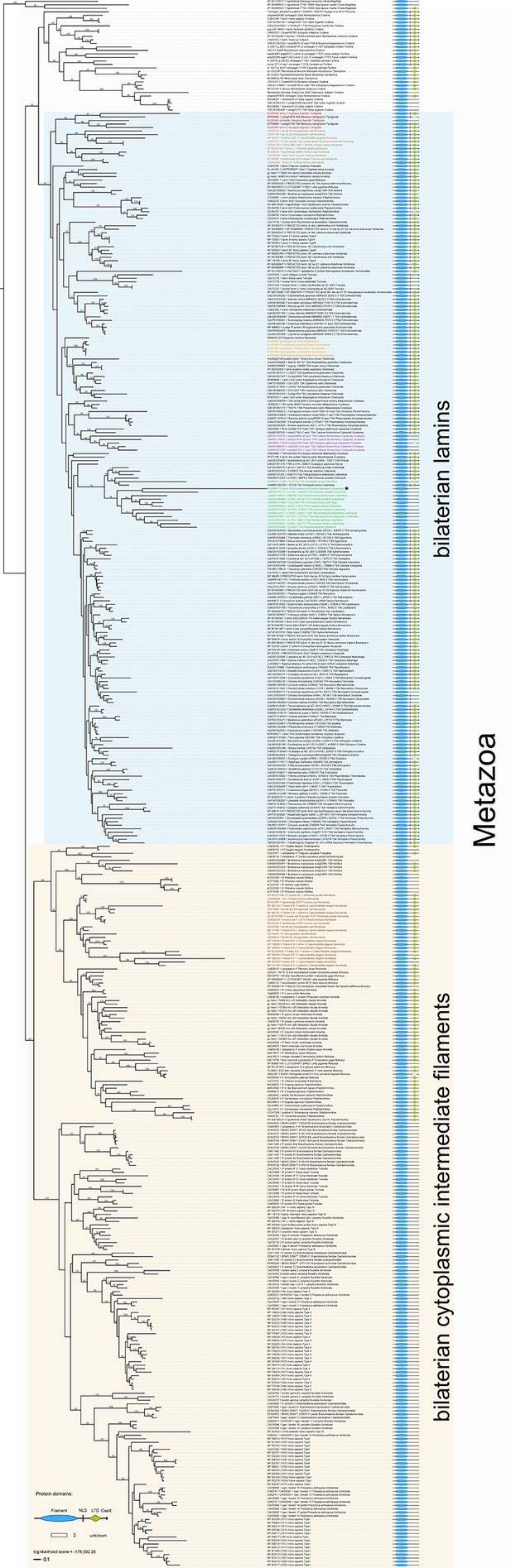
Maximum likelihood tree under the empirical LG+G substitution model and accession numbers of 447 eukaryotic intermediate filament proteins and the placement of the IF protein genes of the tardigrade Hypsibius dujardini (highlighted in red).
Lamins of onychophorans are highlighted in orange, copepods in purple, collembolans in green, nematodes in light brown and cytoplasmic IF proteins of nematodes in dark brown. Note the position of isomin of the collembolan Isotomurus maculatus, which has been interpreted as a putative cytoplasmic IF protein (Mencarelli et al., 2011), within a group of collembolan lamins (asterisk). The domain structure of each protein is depicted on the right. Note the absence of a nuclear localization signal (NLS) in cytotardin of H. dujardini as well as in the coloured sequences of copepods (purple) and collembolans (green). Bootstrap values from 1000 pseudoreplicates ≥50% are given at the nodes. Scale bar indicates the number of substitutions per site. Abbreviations: CaaX, isoprenylation motif at the carboxyl terminus; LTD, lamin tail domain; NLS, nuclear localization signal.
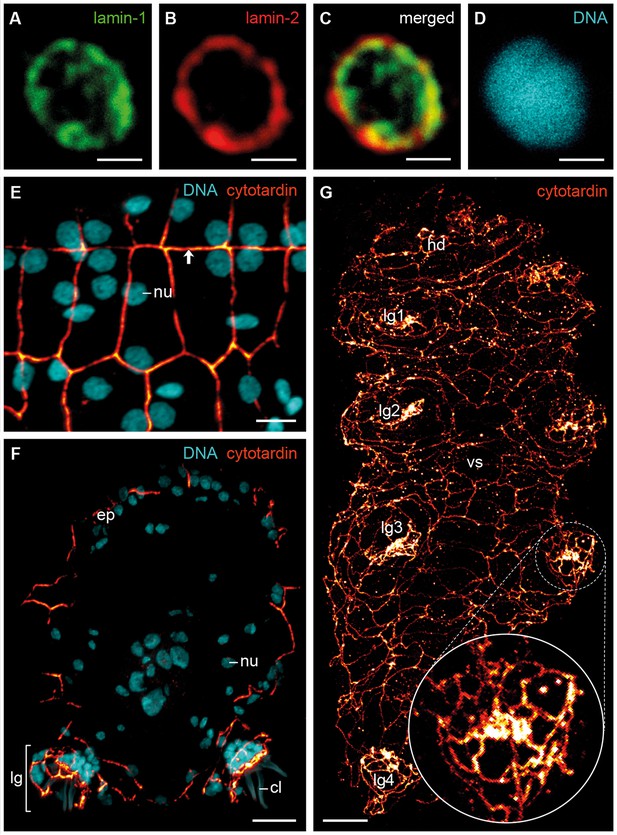
Immunofluorescence labelling of IF proteins in the tardigrade Hypsibius dujardini.
Confocal laser-scanning micrographs. (A–D) Triple labelling of lamin-1 (green), lamin-2 (red) and DNA (cyan). Note the localization of lamin-1 within the nucleoplasm and that of lamin-2 at the nuclear periphery. (E, F) Double labelling of cytotardin (glow-mode) and DNA (cyan) on cryosections. (E) Tangential section of dorsolateral body wall. Arrow points to the dorsal midline. (F) Cross-section of a specimen. Dorsal is up. (G) Whole-mount preparation of a contracted specimen in ventral view. Anterior is up. Inset shows detail of the tip of a leg. cl, claw; ep, epidermis; hd, head; lg, leg; lg1–lg4, legs 1 to 4; nu, nucleus; vs, ventral body surface. Scale bars: (A–D) 1 µm, (E) 5 µm, (F, G) 10 µm.
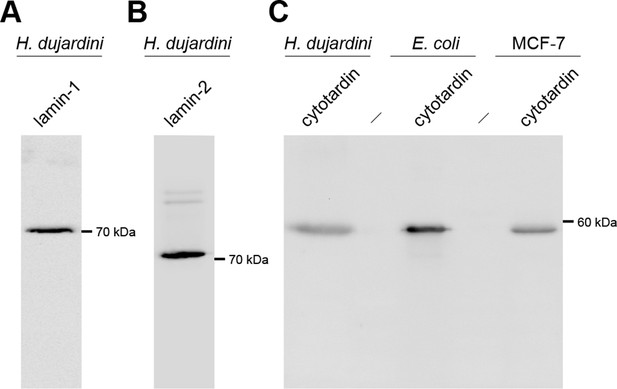
Western blots of lamin-1, lamin-2 and cytotardin antisera.
(A) Western blot analysis of lamin-1 protein expression in Hypsibius dujardini. Anti-lamin-1 antibody stains a band of ≈70 kDa, as expected. (B) Western blot analysis of lamin-2 protein expression in H. dujardini. Anti-lamin-2 antibody stains a band of ≈72 kDa, as expected. (C) Western blot analyses of cytotardin protein expression in H. dujardini, transformed Escherichia coli (E. coli) and transfected MCF-7 cells. Anti-cytotardin antibody stains a band of ≈57 kDa, as expected.
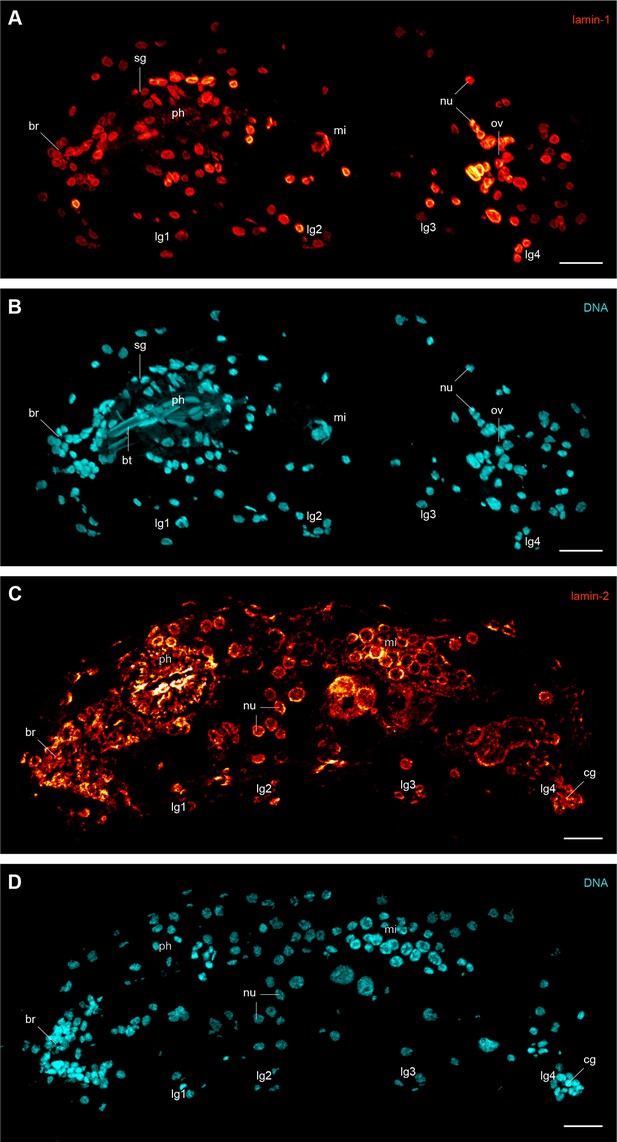
Immunofluorescence labelling of lamin-1 and lamin-2 in the tardigrade Hypsibius dujardini.
Confocal laser-scanning micrographs. Sagittal sections of specimens. Anterior is left, dorsal is up. Note the presence of lamin-1 and lamin-2 inside all nuclei. Cuticular structures of the foregut are autofluorescent. (A, B) Double labelling of lamin-1 (glow-mode) and DNA (cyan) on a cryosection. (C, D) Double labelling of lamin-2 (glow-mode) and DNA (cyan) on a cryosection. br, position of the brain; bt, buccal tube; cg, claw gland; lg1–lg4, legs 1 to 4; mi, position of the midgut; nu, nuclei; ov, ovary; ph, pharynx; sg, salivary gland. Scale bars: (A–D) 10 µm.
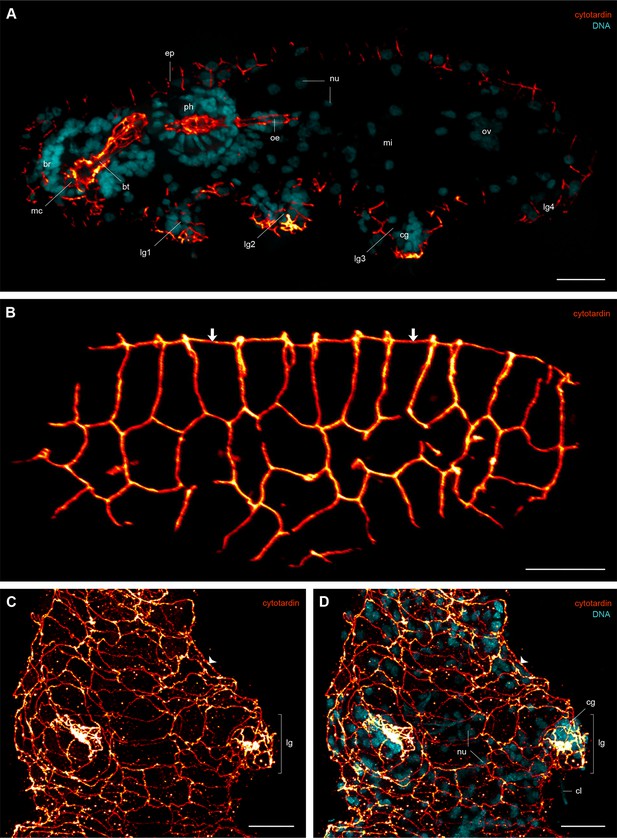
Immunofluorescence labelling of cytotardin in the tardigrade Hypsibius dujardini with focus on the epidermis.
Confocal laser-scanning micrographs. (A) Double labelling of cytotardin (glow-mode) and DNA (cyan) on a cryosection. Sagittal section of a specimen. Anterior is left, dorsal is up. Note the exclusive presence of cytotardin filaments in the epidermis and tissues of the foregut. (B) Anti-cytotardin immunolabelling on a cryosection. Tangential section of dorsolateral body wall. Arrows point to the dorsal midline. (C, D) Double labelling of cytotardin (glow-mode) and DNA (cyan) on a whole-mount preparation of a contracted specimen. Detail of the third pair of legs in ventral view. Anterior is up. Note the apical position of cytotardin-filament belts in the epidermal cells (arrowheads) and their dense arrangement in the tips of each leg. br, brain; bt, buccal tube; cg, claw gland; cl, claw; ep, epidermis; lg, leg; lg1–lg4, legs 1 to 4; mc, mouth cone; mi, position of the midgut; nu, nuclei; oe, oesophagus; ov, ovary; ph, pharynx. Scale bars: (A–D) 10 µm.
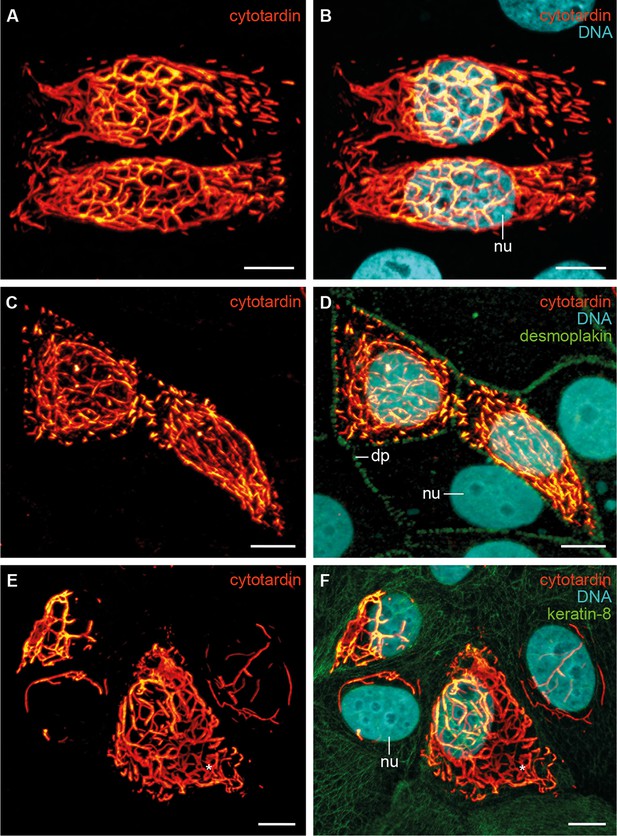
Immunolocalization of exogenous cytotardin in human MCF-7 epithelial cells.
(A, B) Double labelling of cytotardin (glow-mode) and DNA (cyan). Note the cytoplasmic cytotardin filamentous network surrounding the nucleus and extensions close to cell borders. Short cytotardin filaments are aligned along the plasma membrane, different from the arrangement seen in tardigrade epithelial cells. (C, D) Triple labelling of cytotardin (glow-mode), desmoplakin (green) and DNA (cyan). Note that cytotardin forms a cytoplasmic filamentous network extending from the perinuclear area to the cell membrane. Note also that it is not co-localised with desmoplakin. (E, F) Triple labelling of cytotardin (glow-mode), keratin-8 (green) and DNA (cyan). Endogenous keratin networks have been displaced by cytotardin filaments from the perinuclear region without being disrupted (asterisk). dp, desmosomal plaque; nu, nucleus. Scale bars: (A–F) 10 µm.
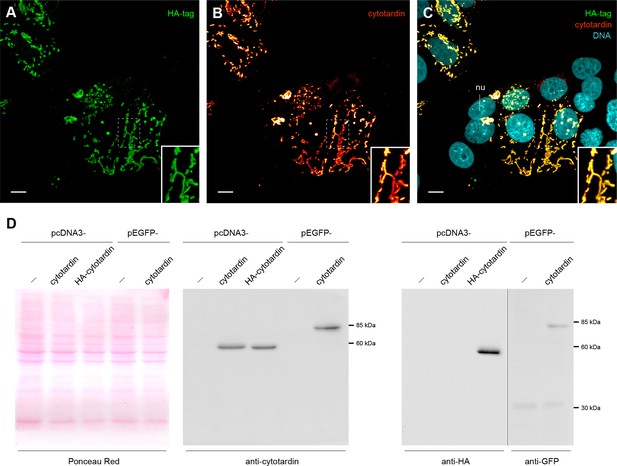
Characterization of cytotardin antiserum.
(A–C) Triple labelling of cytotardin (glow-mode), HA-tag (green) and DNA (cyan) in MCF-7 cells transfected with pcDNA3-HA-cytotardin plasmids. Anti-cytotardin and anti-HA-tag staining show similar signals confirming the specificity of anti-cytotardin antibodies. HA-cytotardin is cytoplasmic and forms short filaments able to localize close to cell borders. (D) Western blot analysis of cytotardin in MCF-7 cells transfected with pcDNA3-cytotardin, pcDNA3-HA-cytotardin, pEGFP-cytotardin and the corresponding empty vectors. Left panel shows a Ponceau red-staining of the blotted cell extract as loading control. Anti-tag (right panel) compared to anti-cytotardin (middle panel) staining shows the expression of the exogenous protein and specificity of the antibodies for immunoblotting. nu, nucleus. Scale bars: (A–C) 10 µm.

Plasmids, primers, and restriction enzymes used for the cloning of tardigrade lamin and cytotardin genes.
https://doi.org/10.7554/eLife.11117.017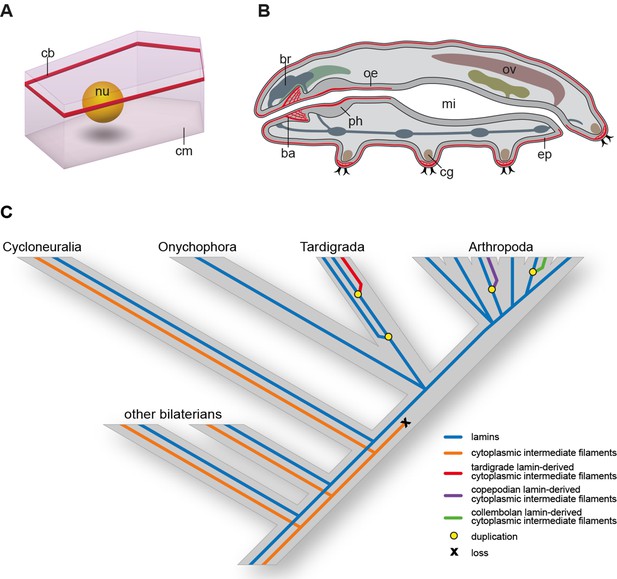
Distribution of cytotardin within the cell and across the tissues in Hypsibius dujardini and the evolutionary history of cytoplasmic intermediate filament proteins.
(A) Diagram of an epidermal cell of H. dujardini with a belt-like arrangement of cytotardin. (B) Diagram of H. dujardini showing the distribution of cytotardin (red), which is confined to the ectodermal tissues. (C) Scenario of the independent origin of lamin-derived cytoplasmic intermediate filaments in tardigrades, collembolans, and copepods. Note that the cytoplasmic IFs in these three lineages (indicated in red, purple, and green, respectively) evolved independently from the cytoplasmic IFs of other bilaterians (highlighted in orange). ba, buccal apparatus; br, brain; cb, cytotardin filament belt; cg, claw gland; cm, cell membrane; ep, epidermis; mi, midgut; nu, nucleus; oe, oesophagus; ov, ovary; ph, pharynx.
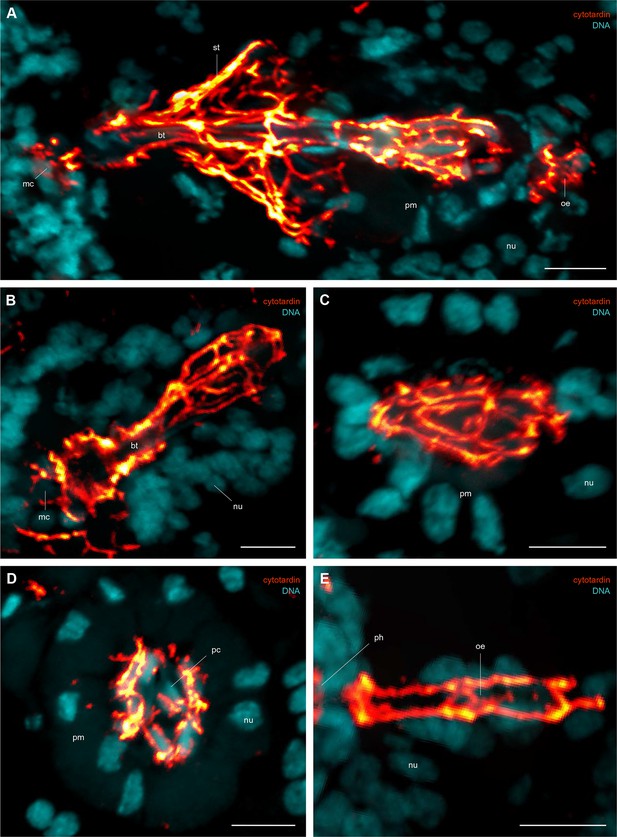
Immunofluorescence labelling of cytotardin in the tardigrade Hypsibius dujardini with focus on the foregut.
Confocal laser-scanning micrographs. Double labelling of cytotardin (glow-mode) and DNA (cyan) on sagittal (A–C, E) and cross sections (D) of cryosectioned specimens. Cuticular structures of the foregut are autofluorescent. Anterior is left (A–C, E); dorsal is up (in A–E). (A) Overview of the foregut. Note the dense arrangement of cytotardin arrays in the epithelia of the foregut, especially those surrounding the stylets. (B) Detail of the buccal tube. (C) Detail of the pharynx. (D) Cross-section of the pharynx. (E) Detail of the oesophagus. bt, buccal tube; mc, mouth cone; nu, nucleus; oe, oesophagus; pc, placoid; ph, pharynx; pm, pharyngeal myoepithelium; st, epithelium surrounding the stylets. Scale bars: (A–E) 5 µm.
Videos
The tardigrade Hypsibius dujardini survives freezing.
This time-lapse video shows thawing specimens of the tardigrade H. dujardini after 4 days frozen in ice. One specimen starts with minuscule movements of one leg after 20 min of thawing and fully recovers locomotion within 120 min.





