Alleviation of neuronal energy deficiency by mTOR inhibition as a treatment for mitochondria-related neurodegeneration
Figures
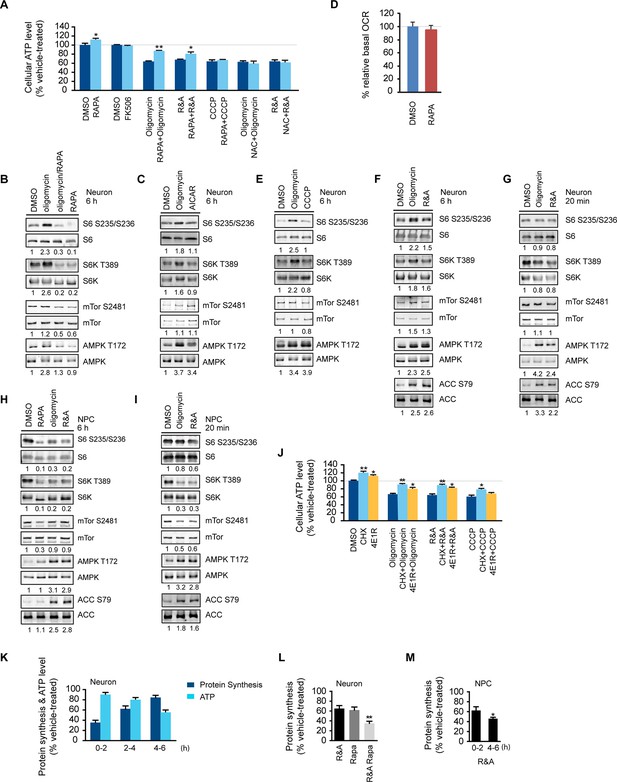
Rapamycin treatment increased neuronal ATP levels.
(A) The effect of rapamycin (RAPA) on cellular ATP level was examined in 5-week neurons differentiated from human neuroprogenitor cells (NPCs) derived from H9 ESCs. Rapamycin was used at 20 nM (final concentration). Mitochondrial dysfunction was mimicked by chemicals disrupting mitochondrial oxidative function: oligomycin (2 µM), blocking complex V (ATP synthase); rotenone and antimycin A (R&A; 1 µM each), complex I and III inhibitors; CCCP (20 µM), a mitochondrial uncoupler. All were prepared in DMSO as vehicle. N-acetylcysteine (NAC) was used at 750 µM (final concentration). The treatment was done for 6 hr with neurons grown in duplicate wells from the same batch of differentiation. The relative ATP level for each treatment was calculated as percentage after normalization to DMSO-treated neurons. Bars are mean ± SD, n=3. *p<0.05. **p<0.01, calculated by two-tailed t-test. (B) Immunoblot analysis of cell lysates prepared from neurons treated with oligomycin, rapamycin or both for 6 hr. The intensity of phosphorylated protein was quantified after normalization to non-phosphorylated signal, and was presented as fold change compared to control group treated with DMSO. (C) Immunoblot analysis of cell lysates prepared from neurons treated with oligomycin or AICAR for 6 hr. (D) Oxygen consumption rate (OCR) measurement by Seahorse extracellular flux analyzer. The basal OCRs of neurons treated with rapamycin for 6 hr were compared to neurons treated with DMSO as control. Bars are mean ± SD, n=3. (E) Immunoblot analysis of cell lysates prepared from neurons treated with CCCP for 6 hr. (F) Immunoblot analysis of cell lysates prepared from neurons treated with rotenone and antimycin-A for 6 hr. (G) Immunoblot analysis of cell lysates prepared from neurons treated with oligomycin or rotenone & antimycin-A for 20 min. (H) Immunoblot analysis of cell lysates prepared from NPCs treated with rapamycin, oligomycin, and rotenone/antimycin-A for 6 hr. (I) Immunoblot analysis of cell lysates prepared from NPCs treated with oligomycin or rotenone & antimycin-A for 20 min. (J) The effect of protein synthesis inhibition on cellular ATP level was examined in 5-week neurons differentiated from human neuroprogenitor cells (NPCs) derived from H9 ESCs. Cycloheximide (CHX) was used at 20 µg/ml, and 4E1RCat was used at 50 µM. The treatment was done for 2 hr with CHX and 4E1RCat alone, and for 6 hr when combined with mitochondrial inhibitors with neurons grown in duplicate wells from the same batch of differentiation. (K) Five-week neurons differentiated from human neuroprogenitor cells (NPCs) derived from H9 ESCs were treated with vehicle (DMSO) or rotenone & antimycin-A (R&A). Protein synthesis was measured by pulsing for 2 hr with 35S-Cys/Met every 2 hr from 0 to 6 hr, and 35S incorporation into protein and neuronal ATP levels were quantified and normalized to the DMSO-treated controls. Data are mean ± SD, n=3. (L) Five-week neurons differentiated from human neuroprogenitor cells (NPCs) derived from H9 ESCs were treated for vehicle (DMSO), rotenone & antimycin-A, rapamycin or both (R&A Rapa) for 4 hr. Protein synthesis was measured by labeling for 2 hr with 35S-Cys/Met from 2 to 4 hr. **p<0.01, calculated by two-tailed t-test. (M) Protein synthesis in NPCs derived from H9 ESC treated with rotenone & antimycin-A for 6 hr. Data are mean ± SD, n=3. All the experiments were repeated at least three times. (see associated Figure 1—source data 1).
-
Figure 1—source data 1
- https://doi.org/10.7554/eLife.13378.004
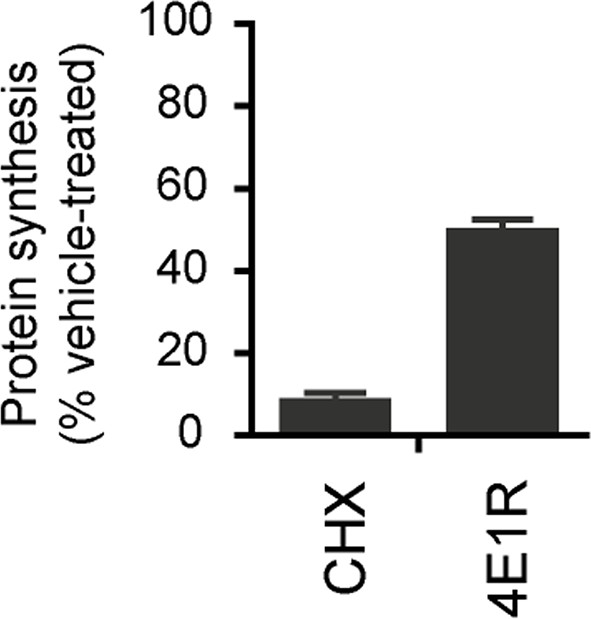
Five-week neurons differentiated from human neuroprogenitor cells (NPCs) derived from H9 ESCs were treated for vehicle (DMSO), cycloheximide and 4E1RCat.
Protein synthesis was measured by pulsing for 2 hr with 35S-Cys/Met, and 35S incorporation into protein was quantified and normalized to DMSO-treated control. Data are mean ± SD, n=3.
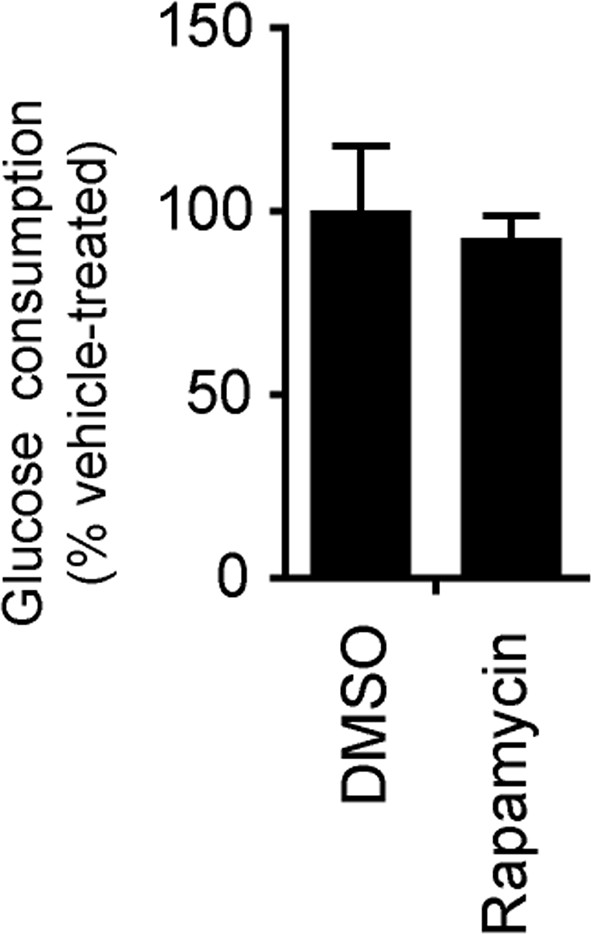
The glucose concentration in the medium growing 3-week neurons derived from H9 ESCs treated with DMSO and rapamycin for 8 hr were quantified by YSI 2950 metabolite analyzer.
Before drug treatment, 1 ml fresh neuronal growth medium was added to neurons grown in duplicated wells of 12-well plate. Glucose used was calculated by minus from glucose in fresh medium. Bars represent mean ± SD. n=3.
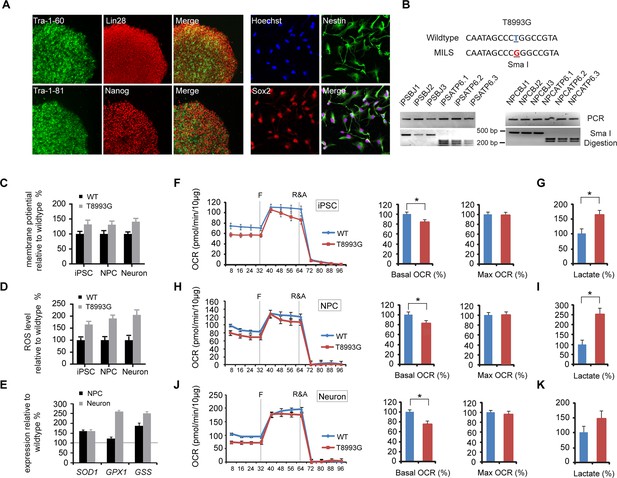
Established iPSCs and neuroprogenitor cells (NPC) from GM13411, a MILS fibroblast line.
(A) T8993G iPSC expressed pluripotency markers that included Tra-1–60, Lin28, Tra-1–81 and Nanog. NPCs derived from T8993G iPSCs were stained with anti-Sox2 and Nestin. (B) T8993G mutation generates a Sma I restriction enzyme site. T8993G iPSCs and NPC cells still retained the mutation as confirmed by PCR and Sma I digestion. DNA products were separated on agarose gel by electrophoresis. (C) Mitochondrial membrane potential analyzed by fluorescence-activated cell sorting (FACS) using TMRE staining. Two lines of iPSCs, NPCs and neurons derived from BJ fibroblasts and one from H9 hESCs were used as controls (WT). The relative mitochondrial membrane potential was presented as percentage compared to the mean of control. Bars are mean ± SD, n=3. The experiment was repeated three times. (D) Cellular reactive oxygen species (ROS) analyzed by FACS using CM-H2DCFDA staining. Two lines of iPSCs, NPCs and neurons derived from BJ fibroblasts and one from H9 hESCs were used as control (WT). The relative ROS level was presented as percentage compared to the mean of control. Bars are mean ± SD, n=3. The experiment was repeated three times. (E) T8993G NPCs and neurons had higher expression of oxidative stress response genes including SOD1, GPX1 and GSS. Two lines of iPSCs, NPCs and neurons derived from BJ fibroblasts and one from H9 hESCs were used as control (WT). The gene expression levels were quantified by real-time PCR after normalization to β-actin. The relative expression level was presented as percentage compared to the mean of control. Bars are mean ± SD, n=3. The experiment was repeated three times. (F, H, J) Oxygen consumption rate (OCR) measured by Seahorse extracellular flux analyzer. FCCP (F) is a mitochondrial uncoupler; rotenone and antimycin A (R&A) are complex I and III inhibitors. Error bars represent SD, n=6. Non-mitochondrial oxygen consumption has been subtracted. The relative percentage of basal and maximum OCR of T8993G iPSC, NPC and neurons at 3 weeks of differentiation were calculated by comparing to the mean of BJ and H9 cells (WT). The original data was in Figure 2—figure supplement 9. (G, I, K) Measurement of lactate secreted by iPSCs, NPCs and neurons at 3 weeks of differentiation. The relative percentage of secreted lactate from T8993G iPSC, NPC and neurons was calculated by comparing to the mean of BJ and H9 cells. Bars represent mean ± SD. n=3. *p<0.05. Calculated by two-tailed t-test. The experiments were repeated three times. (see associated Figure 2—source data 1).
-
Figure 2—source data 1
- https://doi.org/10.7554/eLife.13378.008
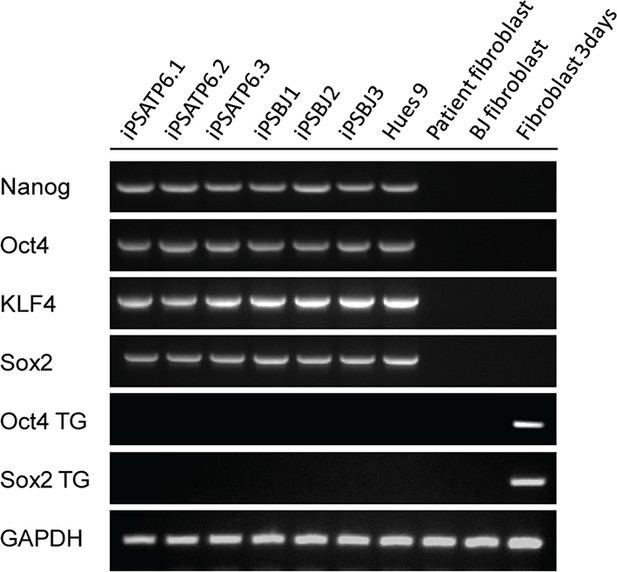
RT-PCR analysis of pluripotency genes, OCT4, NANOG, KLF4 and SOX2 in T8993G and BJ iPSCs.
The expression of ectopic OCT4 and SOX2 in the retrovirus vectors was also examined. Primary fibroblasts, H9 ESCs and day3-transduced fibroblasts were used as negative and positive controls respectively.

Karyotypes of three T8993G iPSC clones, 46, XY; and one BJ iPSC, 46, XY.
https://doi.org/10.7554/eLife.13378.010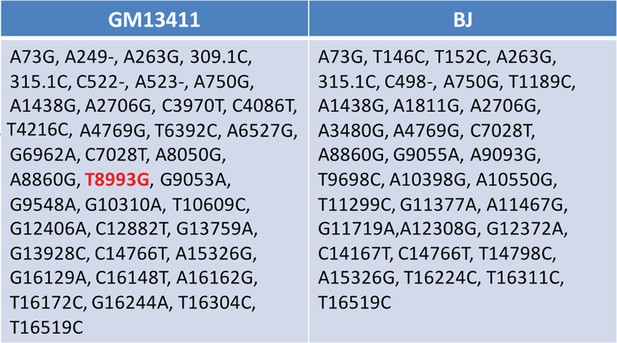
Sequencing of mitochondrial DNA extracted from T8993G (GM13411) and BJ iPSCs.
https://doi.org/10.7554/eLife.13378.011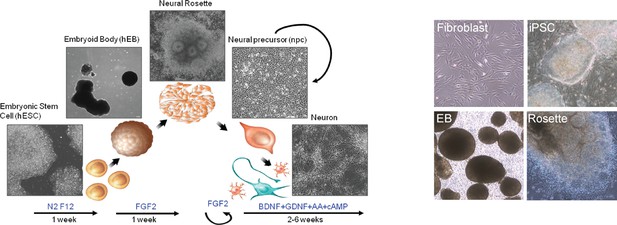
The outline of the protocol used to differentiate neurons from iPSCs; representative pictures of fibroblasts, iPSCs, embryoid bodies (EB) and neural rosettes.
https://doi.org/10.7554/eLife.13378.012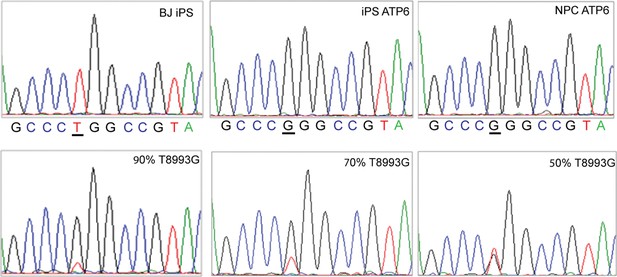
Sanger sequencing confirmed the T8993G mutation (upper panel, representative result).
The sensitivity of Sanger sequencing to detect T8993G mutation copy load was tested by mixing wild-type and T8993G PCR product at the ratios indicated.
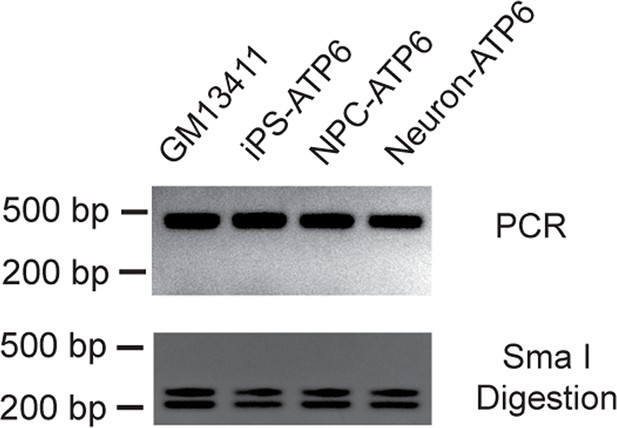
T8993G iPSCs, NPC cells and neurons all had an extremely high T8993G mtDNA mutation load as GM13411 fibroblast.
The mutations were confirmed by PCR and Sma I digestion. DNA products were separated on agarose gel by electrophoresis.
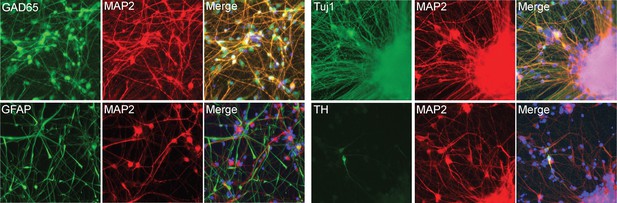
Neuronal marker staining.
https://doi.org/10.7554/eLife.13378.015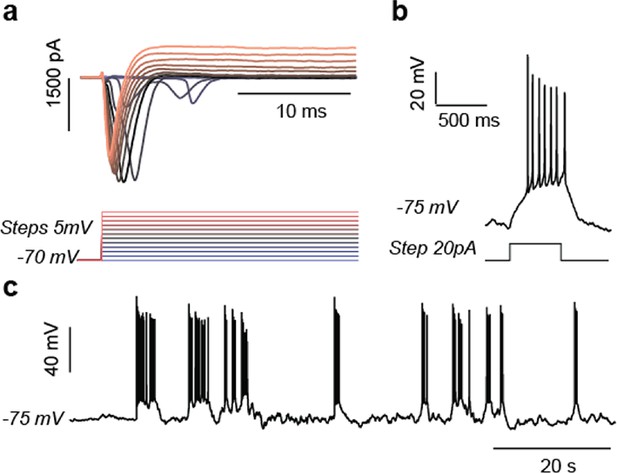
Electrophysiological study of T8993G and BJ 5-week neurons.
Representative results of T8993G neurons were shown. a. Evoked voltage dependent sodium and potassium currents recorded in voltage-clamp (-70mV). b. Evoked action potential. c. Spontaneous burst of action potentials..
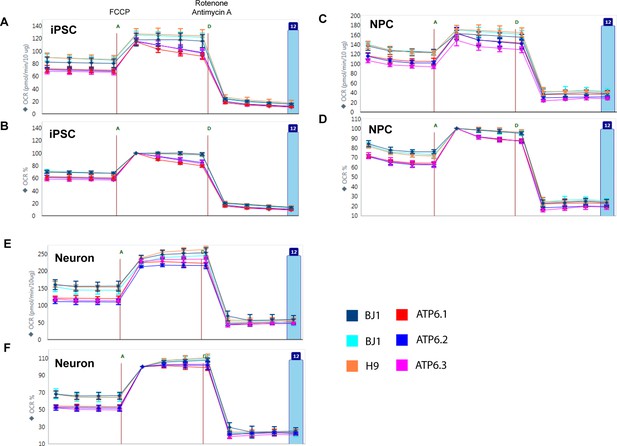
Oxygen consumption rate (OCR) analysis by Seahorse extracellular flux analyzer on BJ, H9 and ATP6 T8993G iPSCs, NPCs and 3-week neurons.
The data were analyzed by WAVE, software from Seahorse Bioscience. A, C and E present the OCR measurement normalized to protein content, while B, D, F show the OCR percentage change after normalization to measurement point 5. FCCP (F) is a chemical uncoupler of electron transport; rotenone and antimycin A (D) are complex I and III inhibitors. Error bars represent SD, n=6.
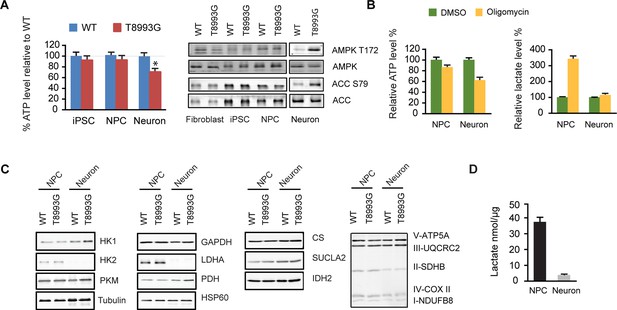
Shutoff of aerobic glycolysis during neuronal differentiation exposes mitochondrial ATP synthesis deficiency in T8993G MILS neurons.
(A) Relative ATP level of T8993G compared to healthy control (BJ, H9 hESC) in iPSCs, NPCs and neurons. The relative percentage of ATP levels in T8993G was calculated by comparing to the mean of control cells respectively. Bars are mean ± SD, n=3. *p<0.05. Calculated by two-tailed t-test. Immunoblot analysis of AMPK Thr172 and ACC Ser79 phosphorylation in cell lysates prepared from primary fibroblasts, iPSCs, NPCs and neurons. (B) Cellular ATP level and secreted lactate from H9 NPCs and neurons treated with DMSO and oligomycin for 6 hr. The relative percentage of ATP levels was calculated by comparing to the mean of DMSO-treated cells respectively. Bars are mean ± SD, n=3. (C) Immunoblot analysis of representative enzymes in glycolysis, TCA and mitochondrial respiratory complexes in BJ and T8993G NPCs and neurons. 20 µg protein lysate from each sample were loaded for SDS-PAGE. (D) Measurement of lactate secreted by NPCs and neurons derived from human BJ iPSCs at 3 weeks. NPC and differentiated neurons at 3 weeks were incubated in fresh medium for 12 hr, and lactate in the medium is quantified. Bars represent mean ± SD of the absolute concentration of lactate after normalized to protein content. n=3. All the experiments were repeated at least three times. (see associated Figure 3—source data 1).
-
Figure 3—source data 1
- https://doi.org/10.7554/eLife.13378.019
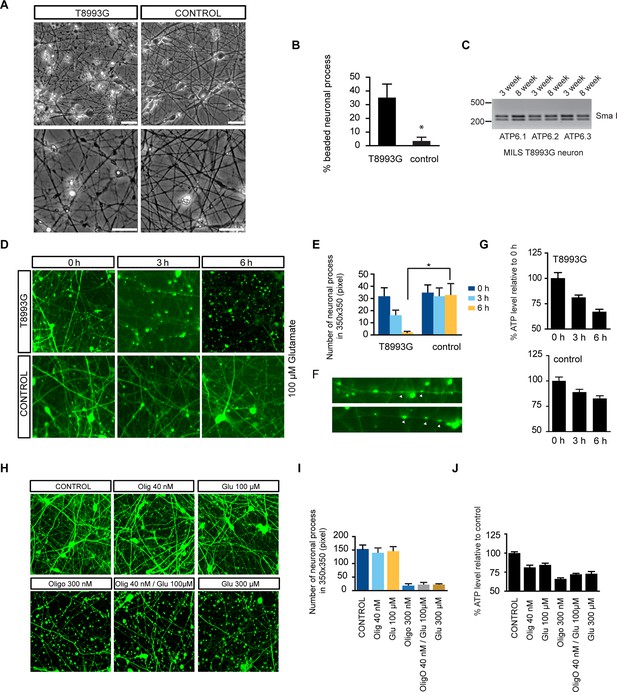
Degenerative phenotype of T8993G MILS neurons.
(A) Phase contrast photo of T8993G and BJ neurons at 8 weeks of differentiation. Scale bar, 20µm. (B) The percentage of neuronal processes containing neuritic beads was quantified by counting 20 neuronal processes for T8993G, BJ and H9 neurons as control. Bars are mean ± SD, n=3. *p<0.05 calculated by two-tailed t-test. (C) T8993G neurons differentiated at 3 and 8 weeks still retained the original high T8893G mutation load as confirmed by PCR and Sma I digestion. DNA products were separated on an agarose gel by electrophoresis. (D) Glutamate-induced toxicity test. Eight-week T8993G and BJ neurons containing DCX promoter-driven GFP were treated with 100 µM glutamate in neuron growth medium. (E) To quantify the extent of neuronal process collapse, the number of discernible neuronal processes in a fixed photo area (350x350 pixel) were counted at time points of 0, 3, and 6 hr. Bars are mean ± SD, n=3. *p<0.05 calculated by two-tailed t-test. (F) Neuritic beads, indicated by white arrows, formed along the axons. (G) Cellular ATP of 8-week T8993G and BJ neurons treated with 100 µM glutamate. The relative percentage of ATP level was calculated by comparing to the mean of 0 hr cells respectively. Bars are mean ± SD, n=3. (H) BJ neurons containing DCX promoter-driven GFP were treated with oligomycin and glutamate in neuron growth medium for 6 hr. (I) Quantification of the extent of neuronal process collapse. (J) Measurement of ATP level. The relative percentage of ATP level was calculated by comparing to the mean of untreated BJ neurons respectively. Bars are mean ± SD, n=3. All the experiments were repeated at least three times. (see associated Figure 4—source data 1).
-
Figure 4—source data 1
- https://doi.org/10.7554/eLife.13378.021
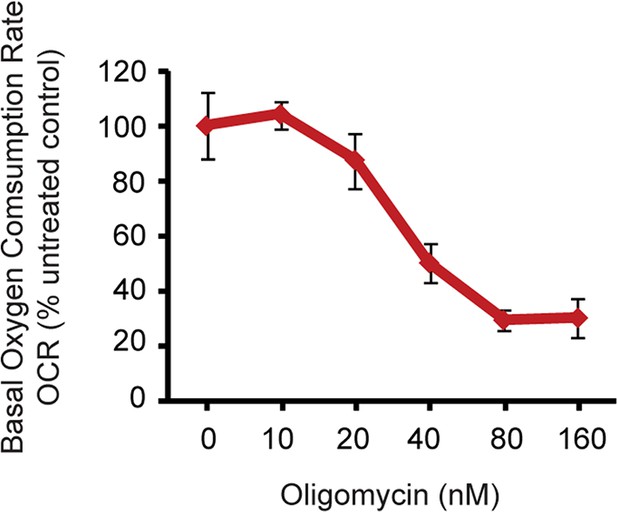
Oxygen consumption rate (OCR) analysis by Seahorse extracellular flux analyzer on neurons treated with Oligomycin.
By measuring the basal OCRs with different concentration of oligomycin, 40 nM was found to partially inhibit ATP synthase
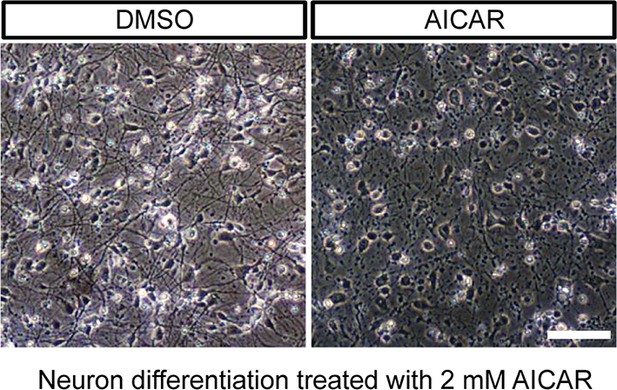
Effect of AICAR on neuron differentiation.
AICAR (2 mM) treatment started from day 2 of BJ neuron differentiation, and DMSO as control. The photo shows neurons after 6 days of treatment. (B) AICAR (2 mM) was added to 6-week BJ neurons. Two days of AICAR treatment led to extensive cell death.
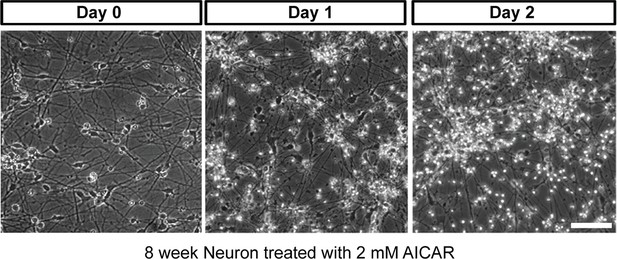
Effect of AICAR on 6-week differentiated neurons.
AICAR (2 mM) was added to 6-week BJ neurons. Two days of AICAR treatment led to extensive cell death.
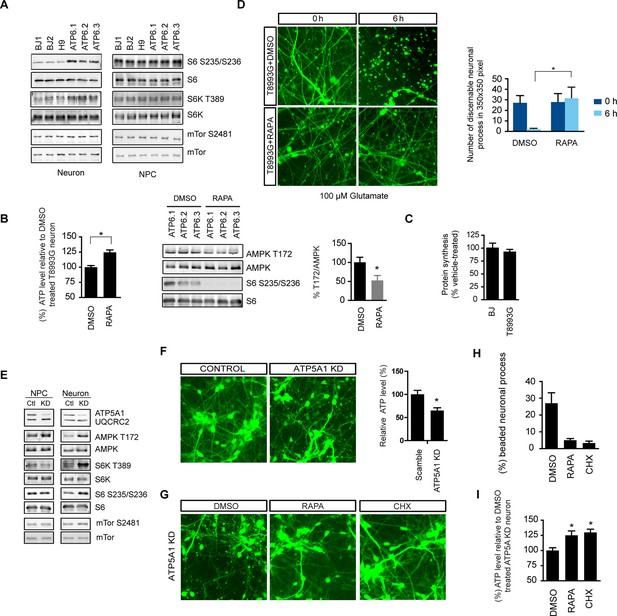
Rapamycin treatment alleviates ATP deficiency and aberrant AMPK activation in T8993G MILS neurons.
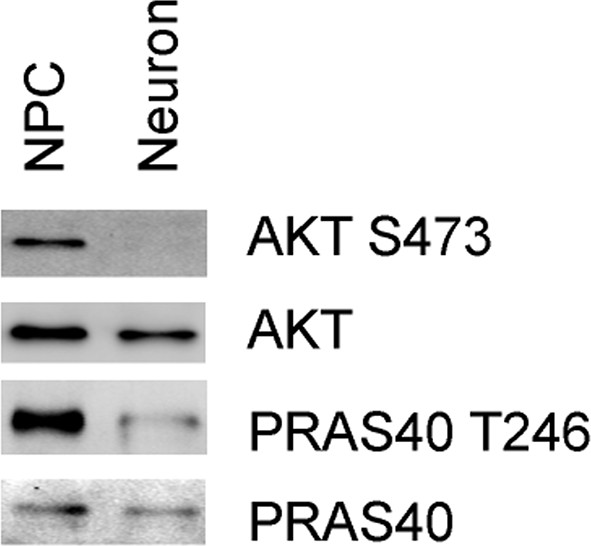
(A) Immunoblot analysis of phosphorylation of ribosomal S6, S6K and mTOR in 3-week neurons and NPCs. (B) Effect of rapamycin on ATP level was examined in T8993G neurons. Five-week T8993G neurons were treated with rapamycin (20 nM) and DMSO for 6 hr. The relative ATP levels of rapamycin-treated neurons were calculated as a percentage compared to the mean of DMSO-treated T8993G neurons. Bars are mean ± SD, n=3. *p<0.05 calculated by two-tailed t-test. AMPK Thr172 phosphorylation was examined by immunoblot analysis and quantified. (C) Five-week neurons differentiated from BJ and T8993G NPCs were used to measure protein synthesis rate. Protein synthesis are pulsed for 2 hr with 35S-Cys/Met. 35S incorporation into protein were quantified and normalized to the total protein. Data are mean ± SD, n=3. (D) The effect of rapamycin on glutamate-induced toxicity test. Eight-week T8993G neurons containing DCX promoter-driven GFP were treated with 100 µM glutamate in neuron growth medium with rapamycin (20 nM) or DMSO. To quantify the extent of neuronal process collapse, the number of discernible neuronal processes in a fixed photo area (350x350 pixel) were counted at time points of 0 hr and 6 hr. Bar are mean ± SD, n=3. *p<0.05 calculated by two-tailed t-test. (E) Immunoblot analysis of phosphorylation of ribosomal AMPK, S6, S6K and mTOR in 3-week neurons and NPCs depleting of ATP5A1. Control (Ctl) lysate were from cells infected with scramble shRNA. (F) Three-weeks ATP5A1-depleting BJ neurons containing DCX promoter-driven GFP. Neurons were infected with lenti-shRNA ATP5A1 and scramble shRNA from day 2 of differentiation. Relative ATP levels were quantified after normalized to protein content. Data are mean ± SD, n=3. (G) Three-weeks ATP5A1-depleted BJ neurons containing DCX promoter-driven GFP treated with DMSO, rapamycin and cycloheximide (200 ng/ml) for 12 hr. (H) Quantification of beaded neuronal process. (I) Measurement of ATP level. All the experiments were repeated at least three times. (see associated Figure 5—source data 1).
-
Figure 5—source data 1
- https://doi.org/10.7554/eLife.13378.026
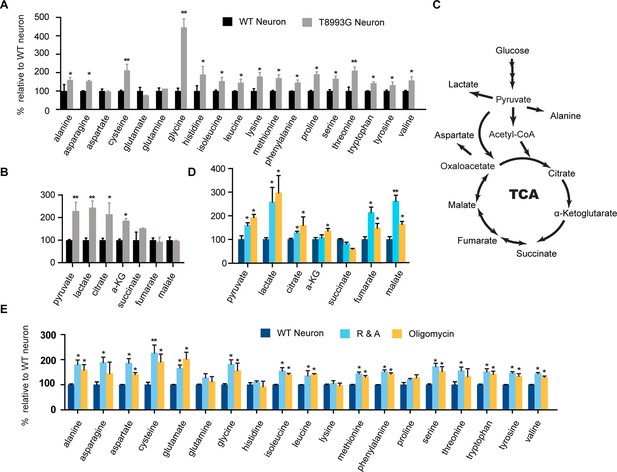
Metabolite profiling of amino acids, TCA and glycolysis intermediates.
(A) Metabolites measured by gas chromatography mass spectrometry (GC-MS). The metabolites were extracted from 3-week T8993G and control including two BJ and one H9 neurons. Relative cellular amino acids were shown. Bar are mean ± SD, n=3. (B) Metabolites of glycolysis and TCA reactions. The relative amount of metabolites in T8993G neurons was presented as percentage compared to the mean of control. Bar are mean ± SD, n=3. (C) A simplified metabolic flow diagram of glycolysis and the TCA cycle. (D, E) 3-week BJ neurons were treated with oligomycin (40 nM) and rotenone and antimycin A (1 µM each) for 6 hr. Bar are mean ± SD, n=3. *p<0.05. **p<0.01, calculated by two-tailed t-test. (see associated Figure 6—source data 1).
-
Figure 6—source data 1
- https://doi.org/10.7554/eLife.13378.028