The selective estrogen receptor downregulator GDC-0810 is efficacious in diverse models of ER+ breast cancer
Figures
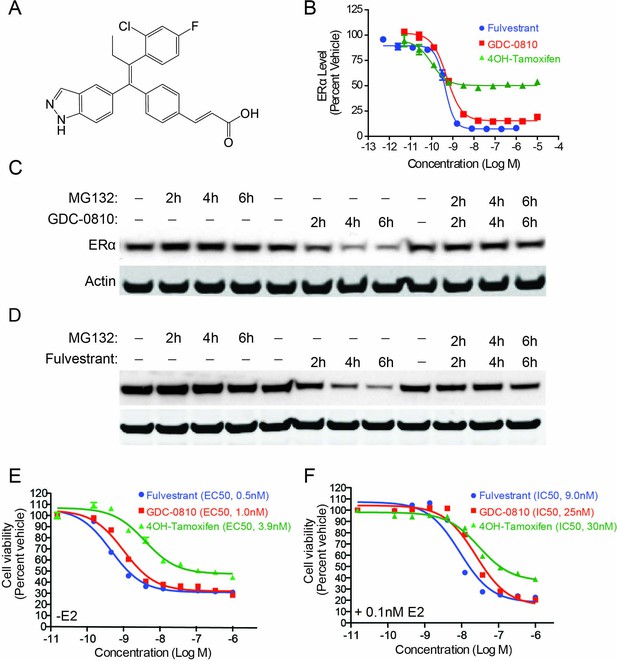
GDC-0810 induces proteasome-dependent degradation of ERα and suppresses proliferation of MCF7 cells.
(A) GDC-0810 structure. (B) MCF7 ERα In-Cell Western assay comparing GDC-0810 potency to fulvestrant and 4-hydroxytamoxifen. ERα levels are quantified by immunofluorescence assay, in triplicate, 4-hr post compound treatment. Error bars are SEM. (C) Western blot analysis assessing the effect of GDC-0810 (100 nM), and fulvestrant (100 nM) (D) on ERα levels at 2, 4 or 6 hr of treatment, in the presence or absence of the 26S proteasome inhibitor MG132 (10 μM). (E) MCF7 cell viability assay comparing GDC-0810 activity to fulvestrant and 4OH-tamoxifen, in the absence of exogenous estradiol. Viable cells are presented as percent CellTiter-Glo luciferase activity relative to the vehicle control after 5 day compound incubation. Error bars represent standard deviation from the mean, from biological quadruplicates. (F) MCF7 cell viability assay comparing GDC-0810 activity to fulvestrant and 4OH-tamoxifen, in the presence of 0.1 nM estradiol. Viable cells are presented as percent CellTiter-Glo luciferase activity relative to the vehicle control after 5 day compound incubation.
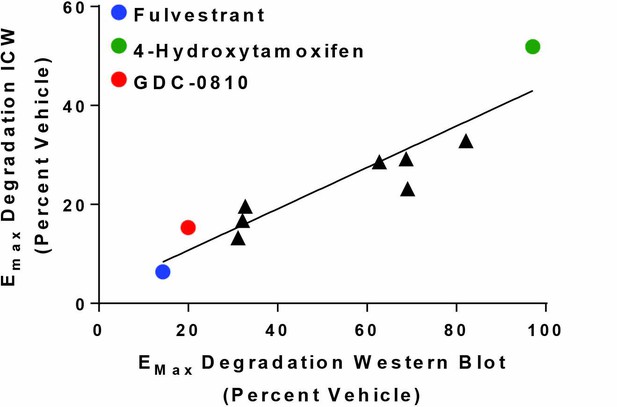
MCF7 ERα In-Cell western assay correlates with MCF7 western blot for most ER binders.
The average In-Cell Western Emax activity for GDC-0810 and ERα benchmark antagonists are plotted versus the average ligand induced ERα degradation induced by 20 hr treatment with 1 μM ligand performed in duplicate. For In-Cell Western assays n ≥ 2.
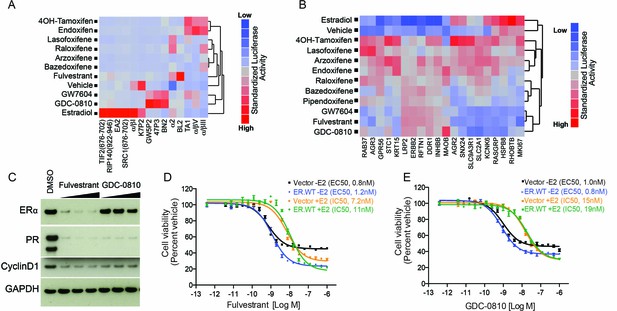
GDC-0810 promotes an ER confirmation distinct from 4OH-tamoxifen and fulvestrant.
(A) ERα conformational profiling. A mammalian 2-hybrid assay was performed to monitor interaction of ERα with 14 conformation selective peptide probes. Luciferase signal was measured after 24 hr of compound treatment (1 μM for all compounds). Interaction profiles of ERα therapies, GDC-0810 and vehicle control from biological triplicates were analyzed by hierarchical clustering using the Ward algorithm and standardized data. (B) Transcriptional activity of benchmark ERα ligands in MCF7 cells, in the absence of exogenous estrogen. Transcriptional activity was monitored using a SERM discriminatory target gene set following 24 hr 1 μM ligand treatment. Data was log2 normalized followed by standardization and hierarchical clustering. (C) Doxycycline-inducible MCF7 cells were pre-cultured in estrogen-depleted medium with 100 ng/ml doxycycline for 2 weeks. Cells were then cultured in medium containing 10, 100 and 500 nM fulvestrant or GDC-0810 for 5 days, and the effect on levels of ER, PR and Cyclin D1 were assessed by Western Blot analysis. (D, E) Doxycycline-inducible MCF7 cells were pre-cultured in estrogen-depleted medium with 10 ng/ml doxycycline for 2 weeks before treatment. Cells were treated with a range of doses of fulvestrant or GDC-0810 for 7 days in estrogen-depleted medium with or without 0.1 nM E2. Cell viability was determined by CellTiter-Glo assay.
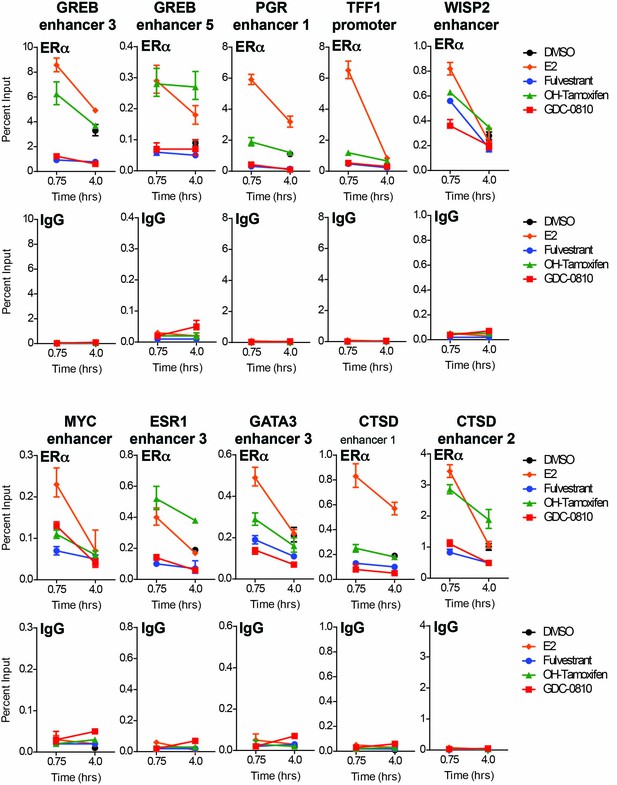
GDC-0810 does not promote the binding of ERα to known ER target sites.
ERα ChIP was performed on MCF7 cells following 45 min or 4 hr compound incubation. qPCR quantification was performed using oligonsucleotides directed to 10 distinct ERα binding sites. Data for ERα and non-specific IgG control is plotted as a percent of total input.
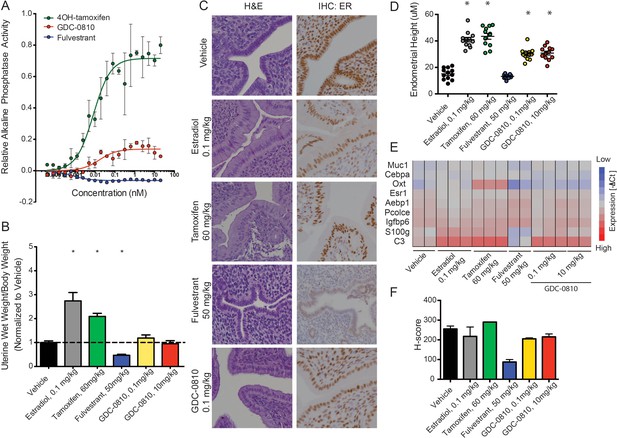
GDC-0810 displays mild estrogenic activity in vitro and in vivo.
(A) Alkaline phosphatase activity in Ishikawa endometrial cells stimulated with increasing concentrations of either 4OH-tamoxifen, GDC-0810 or fulvestrant, in the absence of estrogen. (B) Uterine wet weight (UWW) measurements from juvenile rats treated with individual specified ER ligands; 17α-estradiol was dosed at 0.1 mg/kg as a positive control. (C) Haematoxylin and eosin (H&E) staining, and anti-ER IHC were performed on tissues dissected as in B. (D) Endometrial cell height was digitally measured from the basement membrane to the apical (luminal) surface, using a digitally scanned image at 20X magnification. Three digital measurements were taken from each section, and 2 mice per condition were scored. Results are displayed as the mean endometrial cell height from two animals ± standard error (n = 2). (E) Gene expression in the uterus, after treatment with indicated compounds, was assessed using a rat Fluidigm panel. (F) H-scores were used to quantify the ER IHC from (C). H-scores were estimated manually, incorporating both intensity and percentage of positive nuclei, using the following formula: (0 x% negative)+(1 x% weak)+(2 x% moderate)+(3 x% strong) * Denotes significance (p<0.05) compared to Vehicle in 1-Way ANOVA and Dunnett's Multiple Comparison Test.
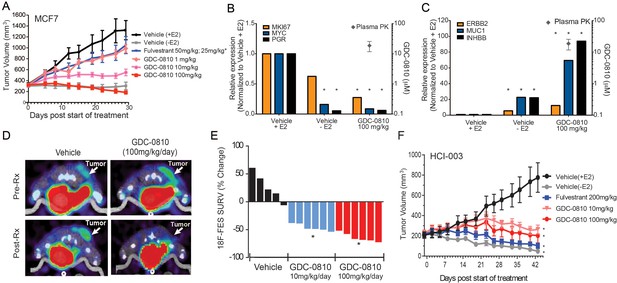
Antitumor activity and pharmacodynamic response of GDC-0810 in tamoxifen-sensitive breast cancer xenograft models.
(A) Tamoxifen-sensitive MCF7 tumor bearing animals were dosed with vehicle, fulvestrant (50 mg/kg on days 1, 3, 8; then 25 mg/kg 2x/week, s.c.) or GDC-0810 (1, 10, 100 mg/kg/day, p.o.) for 28 days in the presence of 60-day release 0.36 mg 17β-estradiol pellets. (B, C) Gene expression analysis of tumors treated with 100 mg/kg GDC-0810, compared to tumors in the presence or absence of estrogen pellets. Tumors were harvested on day 28 of the study; this was a separate study from that shown in (A). GDC-0810 plasma concentration is also shown. *p<0.05, n = 3. See Figure 4—figure supplement 1B for an extended panel of genes. (D) Representative FES-PET images of MCF7 tumors in the right dorsum (arrow) of mice treated with vehicle or GDC-0810 (100 mg/kg). Images were taken 1–2 hr after the dosing on the seventh day of treatment. (E) Percent change in FES SUVR after 6 days of treatment. Each bar represents the mean percent change in 18F-labeled estradiol SUVR. Vehicle-treated mice exhibited an average increase of SUVR of 26.1% whereas mice treated with 10 mg/kg and 100 mg/kg exhibited a 45.2% and 63.3% reduction in SUVR, respectively, compared to baseline (*p<0.0001 vs. vehicle). (F) HCI-003 patient derived xenograft tumors were implanted in mice containing a 1 mg 17β-estradiol beeswax pellet. Tumor bearing animals were dosed with vehicle, fulvestrant (200 mg/kg, 3x/week, s.c.), GDC-0810 (10 or 100 mg/kg/day, p.o.) for 43 days. One vehicle treated group had the 17β-estradiol pellets removed at treatment start to assure growth dependence on estradiol.
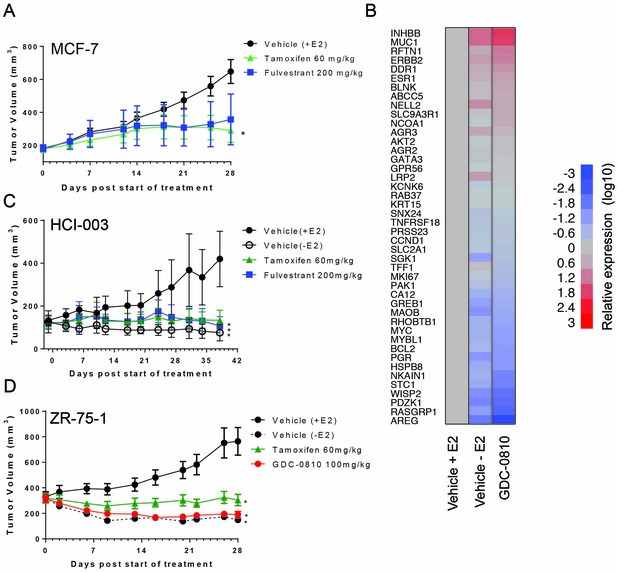
MCF7, HCI-003 and ZR-75-1 breast cancer xenograft models.
(A) Tamoxifen and fulvestrant anti-tumor activity in MCF7 xenograft. Crl:NU-Foxn1nu mice implanted with 17-β Estradiol pellets (0.72 mg/pellet/60 days, Innovative Research of America) were injected with 1 × 107 MCF7 cells. 8 days later, animals bearing viable tumors were dosed orally with Vehicle (9% Peg-400:0.5% Tween–80:0.5% Povidone:90% 0.5% Carboxymethylcellulose) or tamoxifen (60 mg/kg/day), or subcutaneously with fulvestrant (200 mg/kg, 3x/week, 10% ETOH:Castor Oil) for 28 days. * Denotes significance (p<0.05) compared to Vehicle (+E2) in 1-Way ANOVA and Dunnett's Multiple Comparison Test. (B) Gene expression analysis, assessed using Fluidigm, of MCF7 tumors treated with GDC-0810, 100 mg/kg/day, p.o., on day 28, compared to tumors grown in the presence and absence of estrogen pellets at the same timepoint, n = 3. (C) Tamoxifen and fulvestrant display anti-tumor activity in the patient derived tumor, HCI-003. NOD.CB17-Prkdcscid/NcrCrl mice were implanted with HCI-003 tumor fragments and estradiol impregnated beeswax pellets. Animals were ovariectomized 19 days after fragment implant. Animals with viable tumors were dosed orally with Vehicle (9% Peg-400:0.5% Tween–80:0.5% Povidone:90% 0.5% Carboxymethylcellulose), tamoxifen (60 mg/kg/day), or fulvestrant (200 mg/kg 3x/week) for 38 days. Estradiol pellets were removed from one group of animals on the first day of dosing to serve as a control for estradiol dependence for cell growth (Vehicle (-E2)). For all graphs * denotes significance (p<0.05) compared to Vehicle (+E2) in 1-Way ANOVA and Dunnett's Multiple Comparison Test. (D) GDC-0810 displayed anti-tumor activity in ZR75-1 ER+ breast cancer tumors. Crl:NU-Foxn1nu mice implanted with 17-β Estradiol pellets (0.72 mg/pellet/60 days, Innovative Research of America) were injected with 5 × 106 ZR-75-1 cells. 21 days later, animals bearing viable tumors were dosed orally with Vehicle (9% Peg-400:0.5% Tween–80:0.5% Povidone:90% 0.5% Carboxymethylcellulose), tamoxifen (60 mg/kg/day), or GDC-0810 (100 mg/kg/day) for 28 days. Estradiol pellets were removed from one group of animals on the first day of dosing to serve as a control for estradiol dependence for cell growth [Vehicle (-E2)]. * Denotes significance (p<0.05) compared to Vehicle (+E2) in 1-Way ANOVA and Dunnett's Multiple Comparison Test.
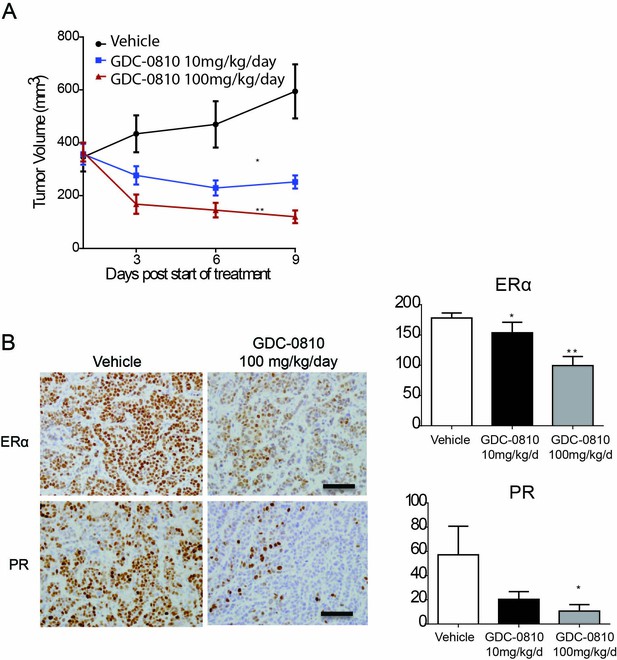
Activity of GDC-0810 in MCF7 xenograft tumors analyzed by FES-PET.
(A) MCF7 cells were injected s.c. into athymic ovariectomized mice supplemented with a 14-day release, 0.17-mg 17β-estradiol pellets. Four weeks later, mice bearing tumors ≥250 mm3 were randomized to treatment with vehicle, GDC-0810 10 mg/kg/day or 100 mg/kg/day (p.o.) for 9 days (*p<0.01 vs. vehicle, **p<0.005 vs. vehicle). (B) Left panel: Representative images of IHC for ERα and PR in mice treated with vehicle or GDC-0810. Scale bars: 200 µm. Right panel: Quantitative histoscores from tumors in left panel (H-score; *p<0.05, **p<0.005). Each bar: mean ± SEM.
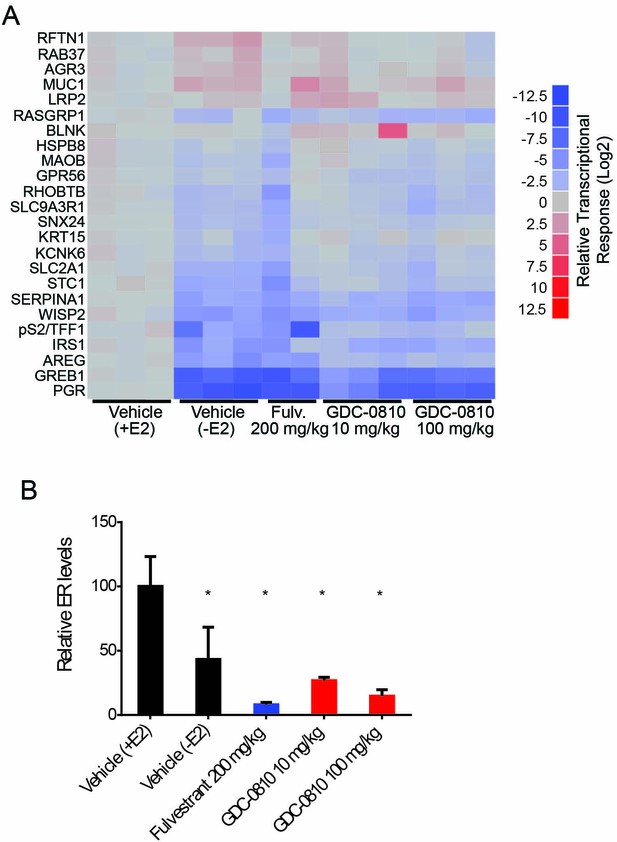
Pharmacodynamic activity of GDC-0810 and fulvestrant in HCI-003 xenograft tumors.
(A) ER target gene transcription was analyzed in tumors 8 hr after the final treatment on day 43. qRT-PCR data from individual tumors [3 for Vehicle (+E2), Vehicle (-E2), and GDC-0810 10 and 100 mg/kg/day and 2 for Fulvestrant 200 mg/kg 3x/week] was normalized to the average of the Vehicle (+E2) treatment group and log2 transformed. (B) ERα protein levels were determined by western blot analysis of tumors 8 hr following the final compound treatment on day 43. ERα levels were normalized to a-tubulin and normalized to the average of the Vehicle (+E2) group. Data is presented as average of treatment group [3 for Vehicle (+E2), 5 for Vehicle (-E2), 4 for fulvestrant 200 mg/kg 3x/week and GDC-0810 100 mg/kg/day and 3 for GDC-0810 10 mg/kg/day]. Error is standard deviation. * Denotes significance (p<0.05) compared to Vehicle in 1-Way ANOVA and Dunnett's Multiple Comparison Test.
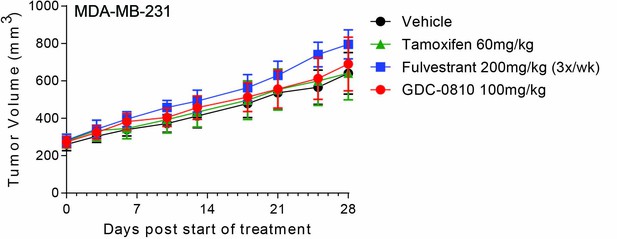
GDC-0810 is not efficacious in MDA-MB-231, an ER negative human breast cancer tumor model.
Crl:NU-Foxn1nu mice injected with 5 × 106 MDA-MB-231 cells were dosed orally with Vehicle (9% Peg-400:0.5% Tween–80:0.5% Povidone:90% 0.5% Carboxymethylcellulose), tamoxifen (60 mg/kg/day), GDC-0810 (100 mg/kg/day), or subcutaneously with fulvestrant (200 mg/kg, 3 times per week, 10% ETOH:Castor Oil) for 29 days.
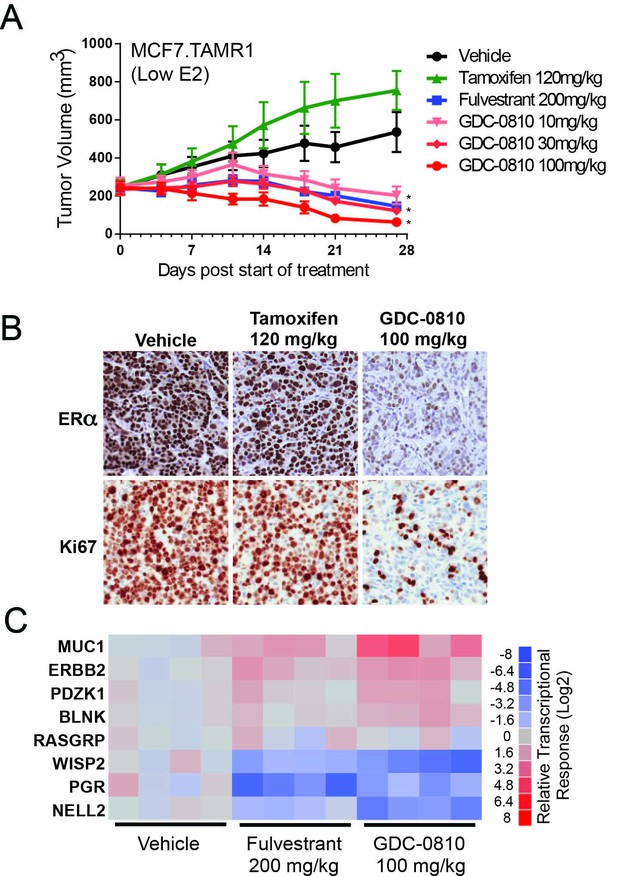
Antitumor activity of GDC-0810 in a tamoxifen-resistant breast cancer xenograft model.
(A) Tamoxifen-resistant MCF7 tumors were implanted in animals supplemented with 60-day release 0.18 mg 17β-estradiol pellets. Tumor bearing animals were dosed with vehicle, tamoxifen (120 mg/kg/day p.o.), fulvestrant (200 mg/kg, 3x/week, s.c.) or GDC-0810 (10, 30 or 100 mg/kg/day, p.o.) for 27 days (B) Representative images of IHC for ERα and Ki67 in tamoxifen-resistant MCF7 xenograft tumors from (A) treated with vehicle, tamoxifen or GDC-0810. (C) Quantitative PCR analysis of ER-regulated genes in tamoxifen-resistant MCF7 xenograft tumors from (A), treated with vehicle, fulvestrant or GDC-0810. S.c. is sub-cutaneous dosing, and p.o. is per os (by mouth) oral gavage dosing. * Denotes significance (p<0.05) compared to Vehicle in 1-Way ANOVA and Dunnett's Multiple Comparison Test.
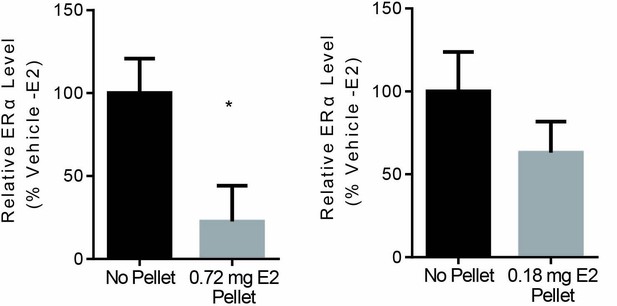
Effects of estradiol pellet on ERα levels in MCF7-TamR1 xenograft tumors.
TamR1 tumor fragments were implanted into Crl:NU-Foxn1nu mice implanted with either 0.72 or 0.18 mg/60 day 17-β estradiol pellets. Once tumors were established pellets were removed from 1 group. 28 days after pellet removal ERα protein levels were determined for 4 tumors from each group. Data is presented as average compared to Vehicle (-E2). Error is standard deviation. * Denotes significance (p<0.05) compared to Vehicle (-E2) by T-test.
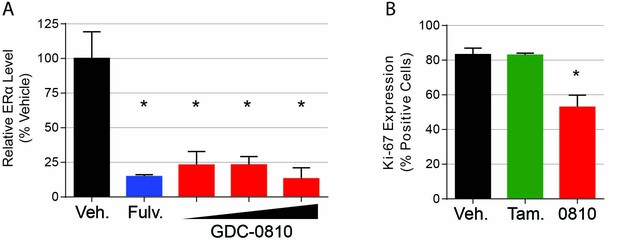
Pharmacodynamic activity of GDC-0810 in MCF7-TamR1 xenograft tumors.
(A) ERα protein levels were determined by western blot analysis of tumors 8 hr following the final compound treatment on day 27. ERα levels were normalized to α-tubulin and normalized to the average of the Vehicle (+E2) group. Data is presented as average of treatment group [4 tumors each for Vehicle (+E2), Vehicle (-E2), and fulvestrant 200 mg/kg 3x/week and 3 for GDC-0810 10, 30 and 100 mg/kg/day]. Error is standard deviation. * Denotes significance (p<0.05) compared to Vehicle in 1-Way ANOVA and Dunnett's Multiple Comparison Test. (B). KI-67 expression, quantified from IHC analysis.
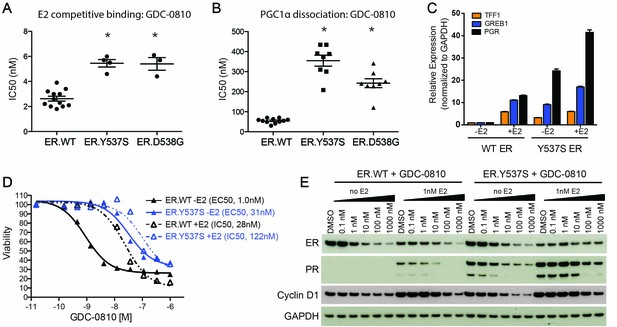
GDC-0810 antagonizes the estrogen-independent ER.Y537S mutant.
(A) A cell free, FRET-based E2 competitive binding assay (E2 present at EC80) was used to determine the binding of GDC-0810 to ER.WT, ER.Y537S and ER.D538G ligand binding domains. Shown are the IC50 values calculated from multiple independent experiments, with mean and standard deviation indicated (B) A cell free, FRET-based PGC1α recruitment assay was used to evaluate the effect of GDC-0810 on ER:PGC1α interaction in the presence of agonist (EC80), using either purified wild-type or mutant ERα ligand binding domains (LBD). Shown are the IC50 values calculated from multiple independent experiments, with mean and standard deviation indicated. (C) Quantitative RT-PCR analysis of ER-regulated genes from CRISPR-Cas9 engineered ER.Y537S cells, in the absence and presence of estrogen, highlighting the E2 independent pathway activity of MCF7 cells expressing ER.Y537S. (D) Cell viability assays, measuring the effect of GDC-0810, were performed on MCF7 ER.WT (black lines) and ER.Y537S (blue lines) cells, in the absence (solid lines) and presence (dotted lines) of estrogen. (E) Western blot analysis evaluating levels of ERα, as well as PR and cyclin D1 as ER targets, in ER.WT and ER.Y537S cells. Cells were treated with GDC-0810 for 24 hr.
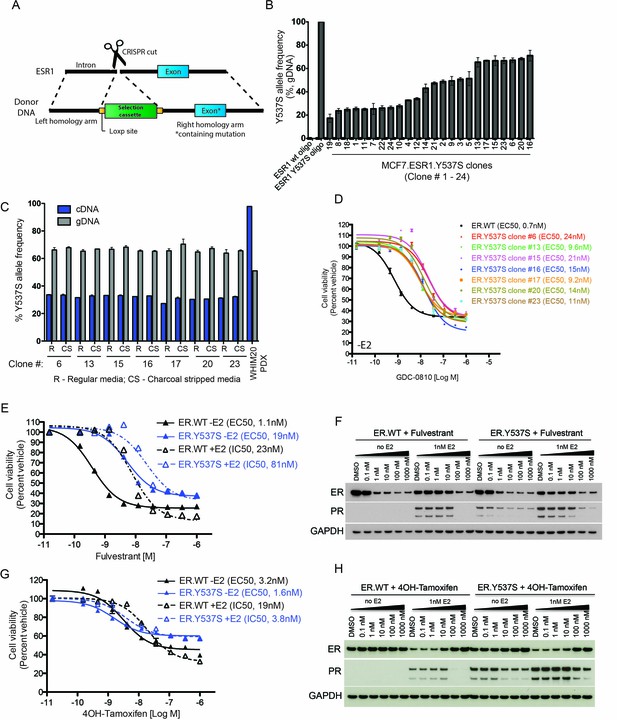
Characterization of MCF7 ER.Y573S knock-in cells.
(A) Schematic detailing the strategy to engineer the ER.Y537S mutation into the endogenous ESR1 locus in MCF7 cells. (B) Y537S-specific ddPCR on genomic DNA was used to evaluate the mutant allele frequency in 24 independent knock-in clones. (C) Expression of the mutant allele was evaluated in 7 clones, as well as in the WHIM20 PDX model by performing Y537S-specific ddPCR on cDNA. (D) The effect of GDC-0810 on the viability of MCF7 ER.WT and multiple MCF7 ER.Y537S clones in the absence of estrogen; all mutant clones exhibit a similar right shift relative to ER.WT cells. (E) The effect of fulvestrant on cell viability was compared across MCF7 ER.WT (black lines) and ER.Y537S (blue lines) cells in the absence (solid lines) and presence (dotted lines) of estrogen. (F) Western blot analyses were performed on ER.WT and ER.Y537S cells in the presence and absence of fulvestrant and estrogen. Cells were harvested 24 hr after drug administration. (G) The effect of 4OH-tamoxifen on cell viability was compared across MCF7 ER.WT (black lines) and ER.Y537S (blue lines) cells in the absence (solid lines) and presence (dotted lines) of estrogen. (H) Western blot analyses were performed on ER.WT and ER.Y537S cells in the presence and absence of 4OH-tamoxifen and estrogen. Cells were harvested 24 hr after drug administration.
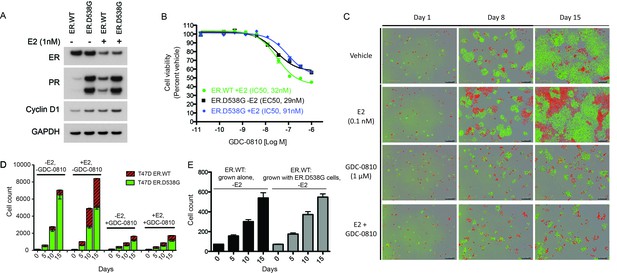
GDC-0810 exhibits activity in T47D ER.D538G knock-in cells.
(A) Western blot analysis of T47D ER.WT and ER.D538G cells in the presence and absence of estrogen. (B) Cell viability assays of GDC-0810 activity in ER.WT and ER.D538G cells. (C) Representative images of T47D ER.WT (red) and ER.D538G (green) cells, grown under specified conditions. Wells, in which cells had been plates at a 1:1 ratio on day 1, were imaged on a regular basis to enable quantification of ER.WT/red vs. ER.D538G/green cells over time. (D) Quantification of cell competition experiments in which labeled T47D.WT cells (red), were mixed at a 1:1 ratio with labeled T47D.D538G cells (green), and grown in the presence and absence of estrogen and GDC-0810. 4 images per condition were quantified. (E) Cell counts for ER.WT cells grown alone or in the presence of ER.D538G cells.
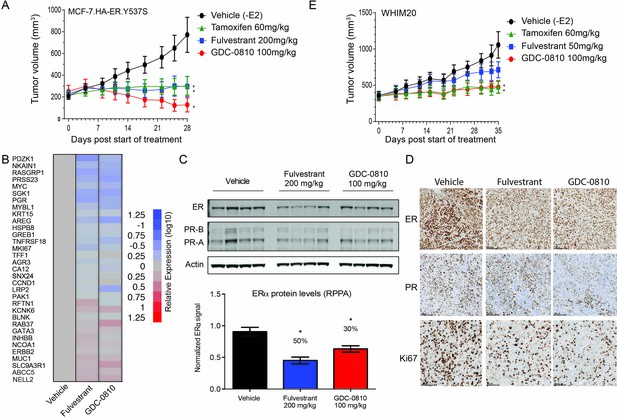
GDC-0810 antagonizes estrogen-independent ER.Y537S-expressing tumors in vivo.
(A) MCF7 HA-ER.Y537S overexpressing tumors were implanted in animals without supplemental 17β-estradiol pellets. Tumor bearing animals were dosed with vehicle, tamoxifen (60 mg/kg/day p.o.), fulvestrant (200 mg/kg, 3x/week, s.c.) or GDC-0810 (100 mg/kg/day, p.o.) for 28 days. (B) Gene expression analysis of WHIM20 tumors after 4 days of dosing, harvested 8 hr after the final dose. (C) Western blot and quantification of ER levels by RPPA, assessing the effect of GDC-0810 and fulvestrant on ER and PR levels in the WHIM20 PDX model. (D) Representative IHC images of WHIM20 tumors treated with either vehicle, fulvestrant or GDC-0810 and probed using anti-ER, anti-PR, or anti-Ki67 antibodies. (E) WHIM20 tumors were implanted in mice without supplemental 17β-estradiol pellets. Upon reaching ~200 mm3,tumor bearing animals were dosed with vehicle, tamoxifen (60 mg/kg/day p.o.), fulvestrant (50 mg/kg, days 1, 3 and 8, 25 mg/kg 1x/week thereafter, s.c.) or GDC-0810 (100 mg/kg/day, p.o.) for 35 days. * Denotes significance (p<0.05) compared to Vehicle in 1-Way ANOVA and Dunnett's Multiple Comparison Test.
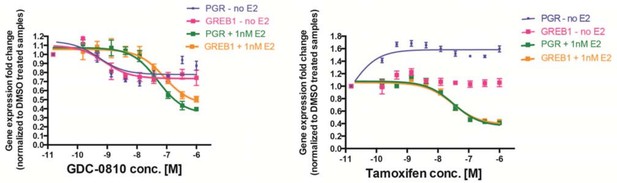
MCF7 cells were treated with increasing concentrations of either GDC-0810 (left) or 4OH-tamoxifen (right), for 6 hours, prior to analysis of PGR and GREB1 as ER target genes, by Taqman assays.
Gene expression was normalized to DMSO control (set to 1) either in the presence or absence of 1nM E2.
Tables
In vitro properties of GCD-0810.
https://doi.org/10.7554/eLife.15828.004| Compound | ER binding∗ | Transcription† | Cell viability‡ | ERα degradation§ | ||||
|---|---|---|---|---|---|---|---|---|
| ERα | ERβ | 3X ERE~LUC | CellTiter-Glo | In-Cell Western | ||||
| Ki [nM] | IC50 [nM] | Emax [% E2] | IC50 [nM] | Emax [% E2] | EC50 [nM] | Emax [% Veh.] | ||
| GDC-0810 | 3.8 ± 1.6 | 3.7 ± 4.0 | 1.3 ± 0.8 | 6.1 ± 2.8 | 2.5 ± 2.1 | 24.6 ± 3.3 | 0.65 ± 0.50 | 15.3 ± 3.4 |
| 4-OH Tam | 2.2 ± 1.3 | 3.6 ± 1.7 | 6.7 ± 3.6 | 4.7 ± 2.9 | 0.53 ± 0.25 | 48.0 ± 4.7 | 0.14 ± 0.04 | 51.9 ± 2.7# |
| Fulvestrant | 13.1 ± 10.8 | 13.2 ± 7.6 | 0.3 ± 0.2 | 4.1 ± 2.6 | 0.56 ± 0.70 | 25.4 ± 3.7 | 0.39 ± 0.18 | 6.4 ± 2.0 |
-
∗ Binding affinities (Ki) of GDC-0810, 4-hydroxytamoxifen (4-OHT), and fulvestrant for ERα and ERβ. Shown are the mean and standard deviation of 3–4 experiments run in duplicate.
† ERα antagonist reporter assay. Results are the mean and standard deviation of 3 experiments.
-
‡ Relative cell viability after 5 d incubation with compound. Shown are the mean and standard deviation of more than 50 assays run in triplicate.
§ Relative ERα immunofluorescence activity in MCF7 In-Cell Western.
-
# The apparent reduction in ERα immunoreactivity is not reproduced in western blots.
Additional files
-
Supplementary file 1
Supplementary data tables related to the specificity for GDC-0810 in binding and activation of ER relative to other nuclear hormone receptors.
(A) Radioligand binding assay (B) GDC-0810 nuclear hormone receptor reporter activity; agonist mode (C) GDC-0810 nuclear hormone receptor reporter activity; antagonist mode.
- https://doi.org/10.7554/eLife.15828.021
-
Supplementary file 2
GDC-0810 and fulvestrant mouse pharmacokinetic data.
(A) GDC-0810 mouse pharmacokinetics (B) Fulvestrant plasma concentrations.
- https://doi.org/10.7554/eLife.15828.022
-
Supplementary file 3
Primer sequences.
(A) Transcriptional Real-time PCR Oligonucleotide Sequence (B) ER-ChIP Real-time PCR Oligonucleotide Sequence.
- https://doi.org/10.7554/eLife.15828.023





