Pask integrates hormonal signaling with histone modification via Wdr5 phosphorylation to drive myogenesis
Peer review process
This article was accepted for publication as part of eLife's original publishing model.
History
- Version of Record updated
- Version of Record published
- Accepted
- Received
Decision letter
-
Peter TontonozReviewing Editor; University of California, United States
In the interests of transparency, eLife includes the editorial decision letter and accompanying author responses. A lightly edited version of the letter sent to the authors after peer review is shown, indicating the most substantive concerns; minor comments are not usually included.
Thank you for submitting your article "PASK integrates hormonal signaling with histone modification via Wdr5 phosphorylation to drive myogenesis" for consideration by eLife. Your article has been favorably evaluated by K VijayRaghavan (Senior Editor) and three reviewers, one of whom, Peter Tontonoz, is a member of our Board of Reviewing Editors. The following individuals involved in review of your submission have agreed to reveal their identity: Bradley B Olwin (Reviewer #2); Elizaveta V Benevolenskaya (Reviewer #3).
The reviewers have discussed the reviews with one another and the Reviewing Editor has drafted this decision to help you prepare a revised submission.
Summary:
In the manuscript the authors provide a comprehensive analysis of the role for PASK in regulating satellite cell fate decisions. The authors find that PASK via phosphorylation of Wdr5 alters methylation of the Myog gene promoter altering accessibility for MyoD, initiating a feed-forward loop required for Myogenin induction irreversibly committing the myoblast to terminal differentiation. The data are thorough, comprehensive and the approach elegant and convincing. The data provide a significant step forward in understanding commitment to terminal differentiation and will have a major impact on research in skeletal muscle. If broadly applicable, the data will impact general stem cell research.
Essential revisions:
1) The authors claim that PASK is dispensable for iPSC reprograming but necessary for differentiation. Several prior studies have suggested that WDR5 is important for self-renewal, proliferation and reprogramming through promotion of H3K4me3, and that loss of WDR5 function leads to activation of genes that promote differentiation (Ang et al. Cell 2011, Yang et al. eLife 2014, Wang et al. Nature 2011). This issue should at least be addressed in the Discussion.
2) Are there any effects on proliferation rate due to loss of WDR5 phosphorlyation under proliferative culture conditions?
3) PASK knockdown had no effect on H3K4me3 signature at the MyoD promoter (or expression of MyoD) although it did influence H3K4me3 and MyoD occupancy at MyoG promoter to drive the expression of MyoG in response to differentiation cues. While MyoD is required for the driving myogenesis, is it specifically necessary for PASK dependent changes in trimethylation of H3K4 at the MyoG promoter? In other words, given the multiple positive reinforcing feedback loops in the proposed model, could it be that MyoD acts cooperatively with WDR5 to change chromatin accessibility? The hierarchical relationship between these factors can be clarified. For example, are there differences in the observed changes in chromatin signatures with pask inhibition or deletion upon MyoD knockdown?
https://doi.org/10.7554/eLife.17985.040Author response
Essential revisions:
1) The authors claim that PASK is dispensable for iPSC reprograming but necessary for differentiation. Several prior studies have suggested that WDR5 is important for self-renewal, proliferation and reprogramming through promotion of H3K4me3, and that loss of WDR5 function leads to activation of genes that promote differentiation (Ang et al. Cell 2011, Yang et al. eLife 2014, Wang et al. Nature 2011). This issue should at least be addressed in the Discussion.
This is an interesting and very important question that we also probed in a limited way during our studies. In C2C12 myoblasts, Wdr5 silencing resulted in stunted cell proliferation (Author response image 1). When myoblasts were induced to differentiate, control myoblasts efficiently differentiated, however, Wdr5 silenced myoblasts failed to form multi-nucleated myofibers (Author response image 2)and induce myogenin and MHC- two markers of differentiation we have used in this manuscript (Author response image 3). More important for this question, we did not see any myogenin or MHC expression at Day 0 (in proliferative condition) when Wdr5 was silent. Thus, at least in myoblasts in our hands, Wdr5 knockdown is not sufficient to cause exit from self-renewal and differentiation. Our data show that Wdr5 is required for both the proliferative phase and differentiation process in myoblasts. This is not surprising since Wdr5 is an essential intermediate in many transcriptional events during both proliferation and differentiation states. For example, in myoblasts, Wdr5 has been shown to associate with Pax7 and regulate myoblast proliferation. During differentiation, MyoD and other transcription factors are known to recruit MLL, KAT2A-B and Set complexes of which Wdr5 is a core member to the Myog promoter to promote differentiation. We have included the data here, but have not included it in the manuscript per se, because it seemed a bit tangential to the logic of the already complex story. At the discretion of the editor and reviewers, we could include it in the paper.

Wdr5 silencing affects proliferation rate in C2C12 myoblasts.
https://doi.org/10.7554/eLife.17985.026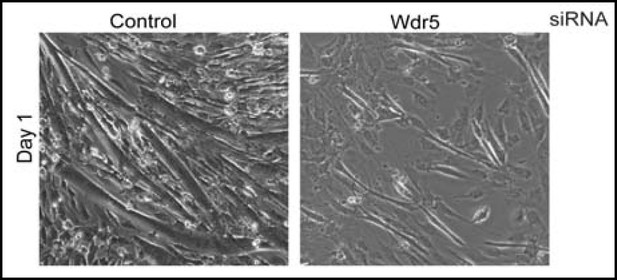
Wdr5 silencing suppresses myotube formation in C2C12 myoblasts.<Author response image 2>
https://doi.org/10.7554/eLife.17985.027
Wdr5 is required expression of myogenin (MyoG) and myosin (MHC) during differentiation in C2C12 myoblasts.
https://doi.org/10.7554/eLife.17985.0282) Are there any effects on proliferation rate due to loss of WDR5 phosphorlyation under proliferative culture conditions?
We do not see any effect on proliferation rate upon either PASK inhibition (Manuscript Figure 1—figure supplement 4F) or Wdr5 S49A expression in myoblasts (Author response image 4).

C2C12 cells were infected with retrovirus expressing GFP (Control) or WT, S49A or S49E‐Wdr5 cDNAs. 24 hrs after infection, puromycin selection was performed for four days.
1000 cells from each samples were plated into 96 well plates and after two days, total cell number was counted for each cell types.
3) PASK knockdown had no effect on H3K4me3 signature at the MyoD promoter (or expression of MyoD) although it did influence H3K4me3 and MyoD occupancy at MyoG promoter to drive the expression of MyoG in response to differentiation cues. While MyoD is required for the driving myogenesis, is it specifically necessary for PASK dependent changes in trimethylation of H3K4 at the MyoG promoter? In other word.s given the multiple positive reinforcing feedback loops in the proposed model, could it be that MyoD acts cooperatively with WDR5 to change chromatin accessibility? The hierarchical relationship between these factors can be clarified. For example, are there differences in the observed changes in chromatin signatures with pask inhibition or deletion upon MyoD knockdown?
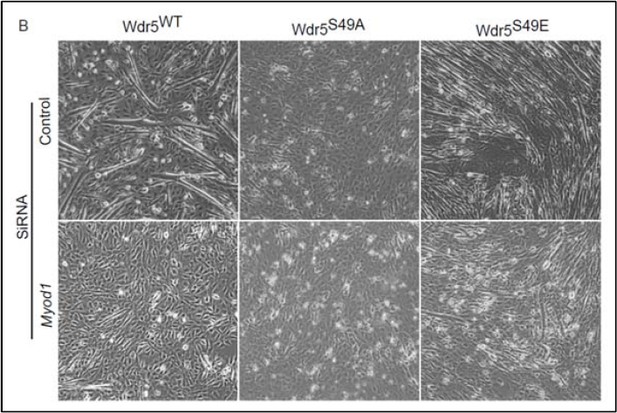
This is a very interesting mechanistic question. While the interplay between MyoD and pWdr5 is an important and complex area, we have tried to address this question partially here. MyoD is extensively involved in remodeling of the Myog promoter at the onset of differentiation by recruiting MLL complexes, HATs and SWI/SNF complexes. MyoD serves a pioneering function at the Myog promoter to activate its transcription in response to differentiation signaling (Cao et al., 2006). While MyoD occupies Myog promoter during the proliferative phase, signaling inputs are required to stimulate chromatin remodeling and activation of MyoG transcription (Cao et al., 2006). On the other hand, inhibition of MyoD aborts the differentiation process. Thus, we hypothesize that MyoD acts synergistically with signaling inputs such as those that activate the PASK-Wdr5 pathway to remodel Myog promoter. We tested this by examining levels of H3K4me3 on the Myog promoter in control or Myod1- silenced C2C12 before and after the onset of differentiation. This was done in cells expressing WT, S49A and S49E Wdr5 as a surrogate for activation or inactivation of PASK signaling (Author response image 5). We observed that knockdown of Myod1 partially prevented H3K4me3 induction on the Myog promoter in all samples, including Wdr5S49E. We also examined the effect of Myod1 knockdown on differentiation in Wdr5 WT, S49A or S49E cells. Again, we noticed that in non-targeting siRNA (Control) samples, Wdr5S49E expressing cells made robust myofibers and Wdr5S49A expressing cells were defective in myotube formation (Author response image 6). Myod1 knockdown, however, significantly suppressed myotube formation in Wdr5S49E expressing cells. Hence, we think that Wdr5 phosphorylation is not sufficient to overcome the loss of MyoD function and that MyoD acts synergistically with phosphorylated WDR5 to induce epigenetic changes at the Myog promoter.

H3K4me3 ChIP was performed from C2C12 myoblasts after control or MyoD knockdown expressing WT, S49A or S49E WDR5 mutants at Day 0 and Day 1 of differentiation.
* P<0.05 between Wdr5S49E vs. Wdr5WT at Day 0 in control samples.