Methylated cis-regulatory elements mediate KLF4-dependent gene transactivation and cell migration
Figures
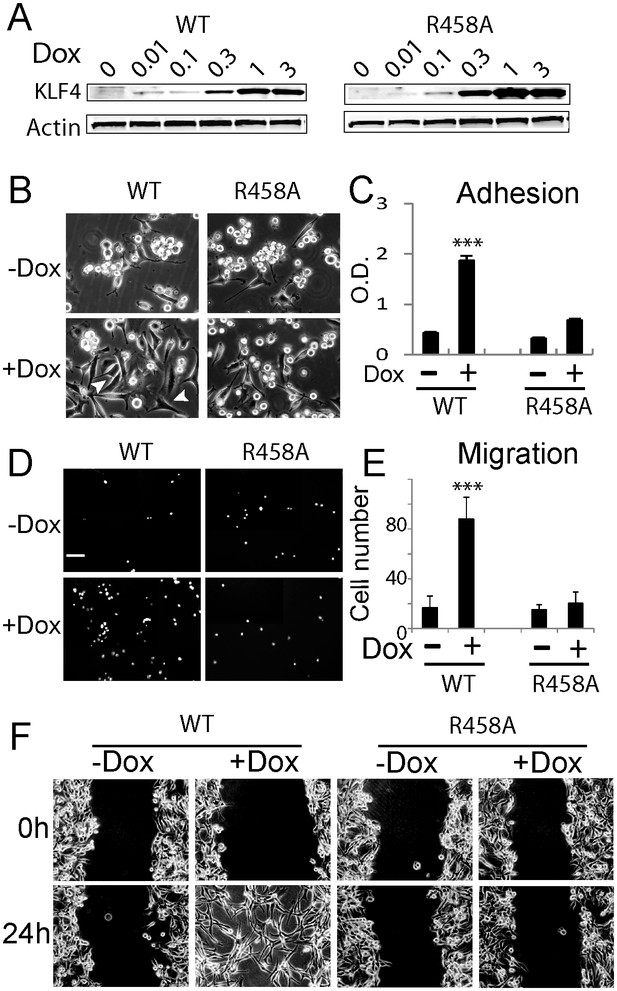
Methyl CpG-dependent KLF4 binding activity promoted GBM cell adhesion and migration.
(A) Induced expression of KLF4 WT and R458A in human U87 GBM cells upon doxycycline (Dox) treatment. Cells were transfected with lentivirus harboring tet-on KLF4 WT or R458A constructs and selected with antibiotics. Stable cell lines were treated with Dox for 48 hr before immunoblotting. Immunoblotting showing induced expression of KLF4 WT and R458A in human U87 GBM cells upon doxycycline (Dox) treatment. (B and C) KLF4 WT but not R458A promoted GBM cell adhesion. Cells were pre-treated with Dox for 48 hr and plated for 2 hr before washing. (D and E) KLF4 WT promoted GBM cell migration in broyden chamber transwells. Cells were pretreated with Dox for 48 hr, plated on transwells containing 0.1% FCS and migrating towards 10% FCS. After 3 hr, migrating cells were stained with DAPI and five field / transwell were counted. (F) KLF4 WT but not R458A promoted GBM cell migration in wound healing assays. Cells were treated with Dox for 5 days till confluence. A scratch was made and cells were maintained in 0.1% FCS medium overnight, cell proliferation was inhibited by mitomycin C. Microphotographs were taken 0 hr and 24 hr after scratching. Bar = 25 µm. ***p<0.001.
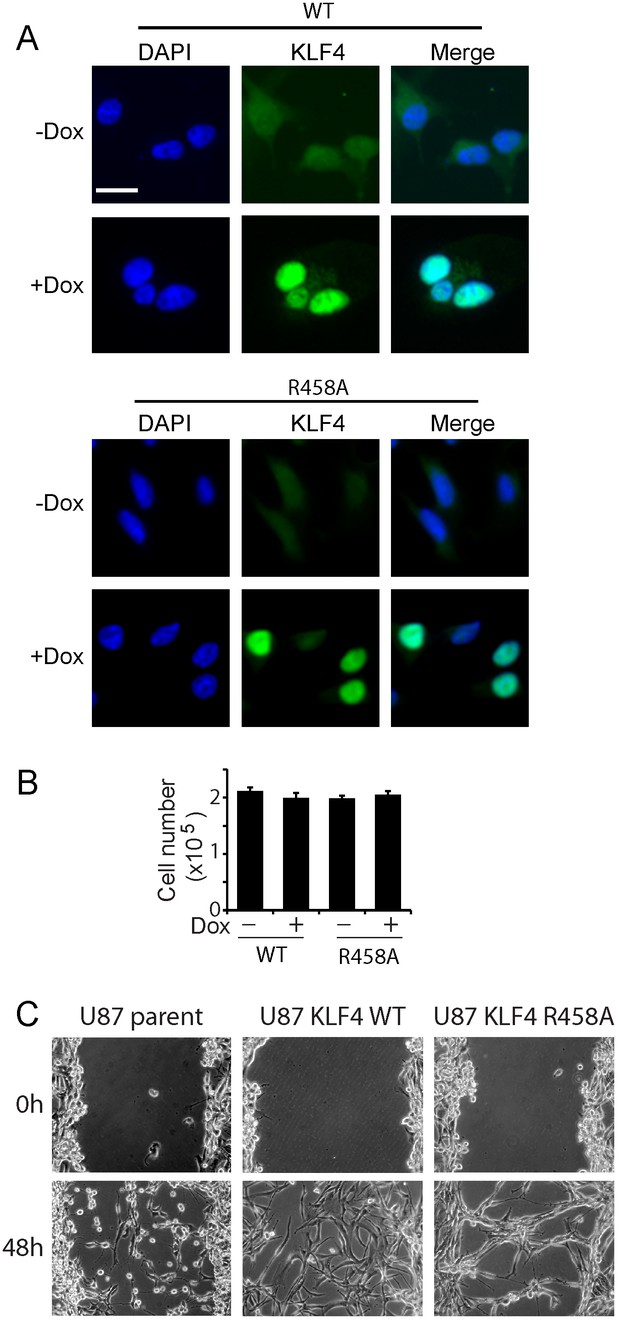
Expression of KLF4 WT and R458A mutant did not affect cell growth.
(A) Immunocytochemistry staining of KLF4 cellular location. Human U87 GBM cells express low level of endogenous KLF4 protein, which was evenly spread in cytoplasma and nucleus. Dox induction of KLF4 WT and R458A expression resulted in strong KLF4 staining in the nucleus. Bar = 10 µm. (B) KLF4 WT and R458A had no effect on cell growth 5 days post Dox (1 µg/ml) treatment as determined by trypan blue staining. (C) Scratch assays for 48 hr in KLF4 WT expressing cells (+Dox) in comparison with non-transfected U87 parent cells and KLF4 R458A expressing cells. Bar = 25 µm.
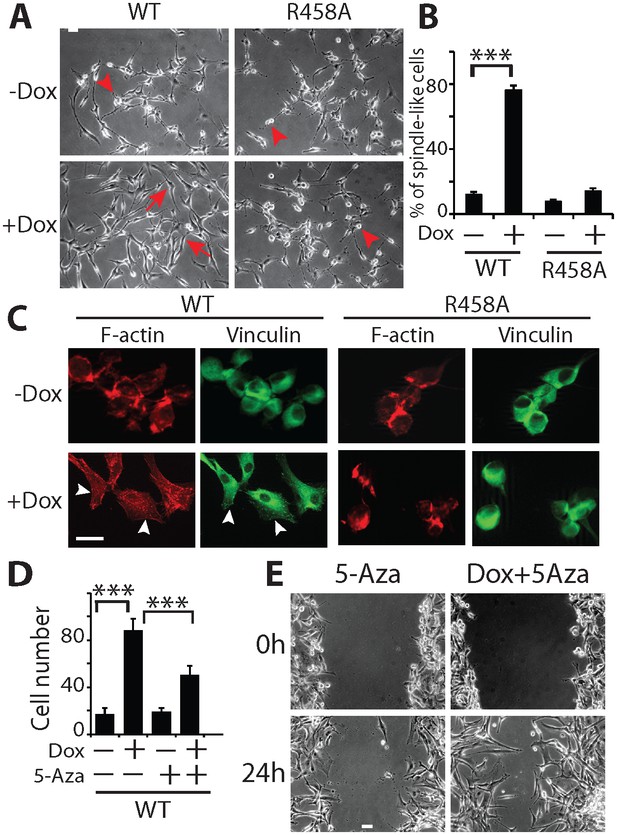
KLF4 WT-induced cell migration is methylation dependent.
(A) Expression of KLF4 WT but not R458A induced cell morphology changes in U87 cells (+Dox, 48 hr). The control and R458A expressing cells showed round and short cell body (arrowheads), whereas KLF4 WT induced elongated, spindle-like cell shape (arrows). Bar = 25 µm. (B) Quantification of cell morphology changes after Dox treatment for 48 hr. More than 80% of the KLF4 WT expressing cells showed elongated, spindle-like cell morphology, whereas most KLF4 R458A expressing cells remained as round and short cells. (C) F-actin and vinculin staining in control, KLF4 WT and R458A-expressing cells. KLF4 WT induced actin stress fiber formation and focal adhesion formation (arrowheads). Bar = 10 µm. (D) Pre-treatment of the cells with DNA methyltransferase inhibitor 5-Aza (1µmol/L, 10 days) blocked KLF4 WT-induced cell migration in transwell assays. (E) Scratch assays indicating that 5-Aza blocked wound healing induced by KLF4 WT. Bar = 25 µm. ***p<0.001.
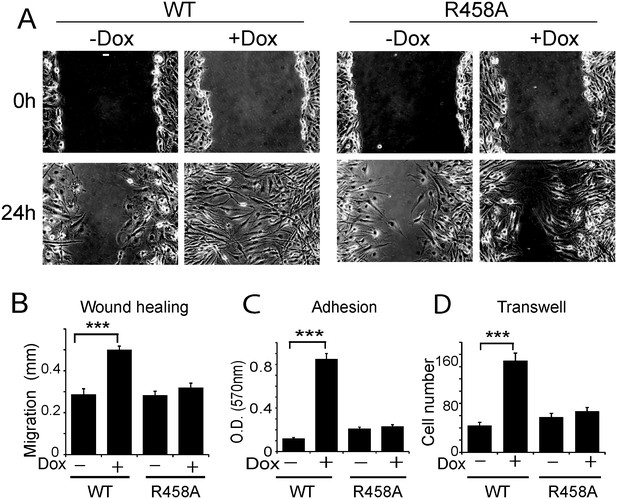
Methyl CpG-dependent KLF4 binding activity promoted adhesion and migration of human U373 GBM cells.
(A and B) mCpG-dependent KLF4 binding promoted U373 cell migration in wound healing assays. Cell migration distance was plotted in (B). Bar = 25 µm. (C) KLF4 WT but not R458A promoted U373 GBM cell migration in transwell assays. (D) KLF4-WT but not R458A promoted adhesion of U373 GBM cells, as measured by MTT assays (***p<0.001).
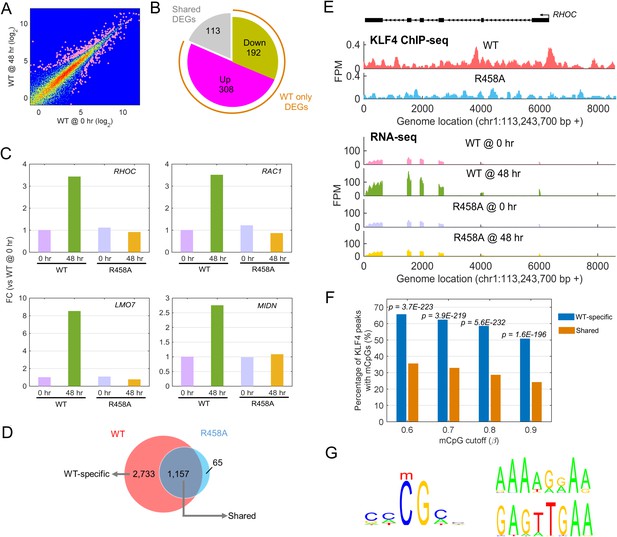
Identify transcriptional network regulated by KLF4-mCpG interactions.
(A) RNA-seq data before (0 hr) and after (48 hr) KLF4 WT induction. The pink dots were determined as differential expressed genes (DEGs) (p<0.001). (B) Overlap between DEGs in KLF4 WT and R458A cells, showing that a total of 613 genes were significantly regulated by KLF4 WT, 115 of which were also significantly regulated by KLF4 R458A. Among the rest 500 genes significantly regulated by KLF4 WT but not R458A (WT only DEGs), 308 of them were up-regulated by KLF4 WT only. (C) Four examples of KLF4 WT only DEGs. (D) Overlap between KLF4 WT and R458A KLF4 ChIP-seq peaks (48 hr +Dox), indicating that ~2733 peaks can be only bound by KLF4 WT; ~1157 peaks bound by both KLF4 WT and R458A, whereas R458A alone only bound a few new sites. (E) ChIP-Seq for KLF4 WT and R458A on and surrounding RHOC promoter as an example. RNA-seq at the same region was also shown, pre and post KLF4 WT and R458A induction, respectively. (F) Percentage of ChIP-seq peaks with mCpGs evaluated by whole genome bisulfite sequencing analysis. A significant enrichment was observed for methylated CpG in KLF4 WT-specific peaks (Blue bar) as compared to KLF4 R458A shared peaks (orange bar). (G) Motifs identified for KLF4-mCpG binding in KLF4 WT-specific peaks (Left) and for KLF4 R458A shared peaks (Right), respectively.
-
Figure 3—source data 1
Mapped reads for all the RNA-sequencing experiments.
- https://doi.org/10.7554/eLife.20068.007
-
Figure 3—source data 2
Mapped reads for the ChIP-sequencing experiments.
- https://doi.org/10.7554/eLife.20068.008
-
Figure 3—source data 3
Chromosol location of KLF4 WT-specific, shared, and mutant-specific peaks.
- https://doi.org/10.7554/eLife.20068.009
-
Figure 3—source data 4
Methylated 6-mer cis motifs identified in KLF4 WT-specific peaks.
- https://doi.org/10.7554/eLife.20068.010
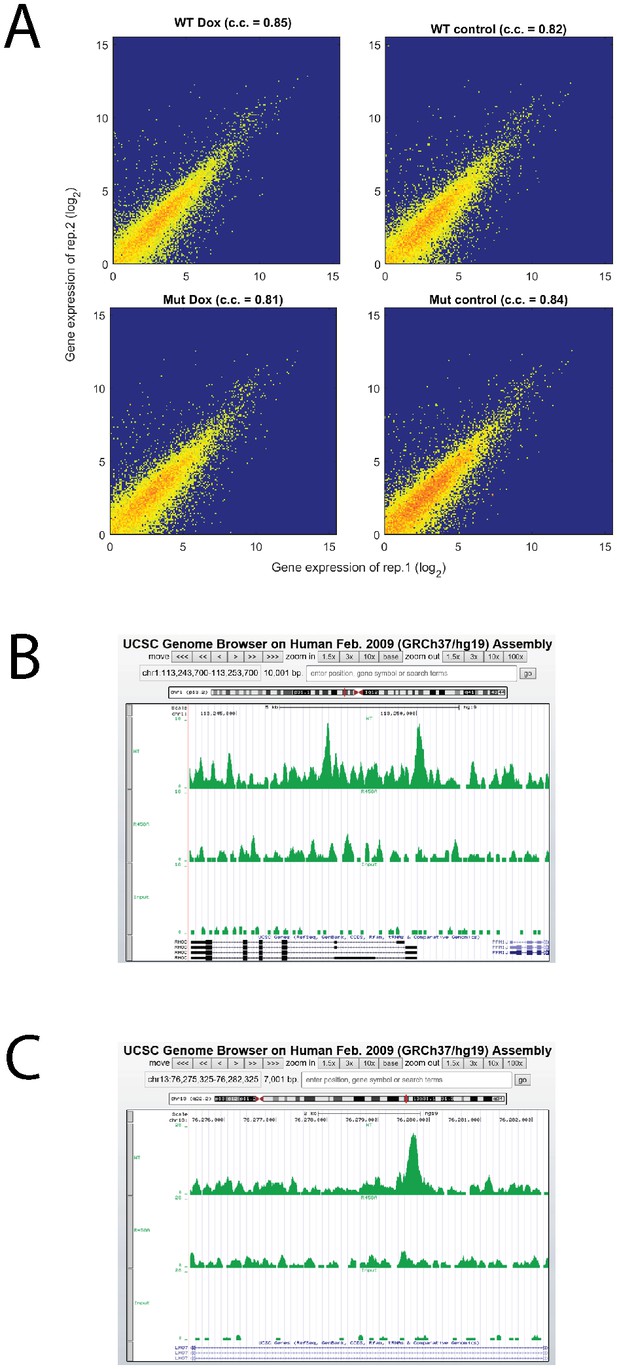
Analysis of RNA-seq and ChIP-seq data.
(A) High reproducibility of RNA-seq replicate. (B, C) Screenshots of RHOC and LMO7 ChIP-seq together with input line.
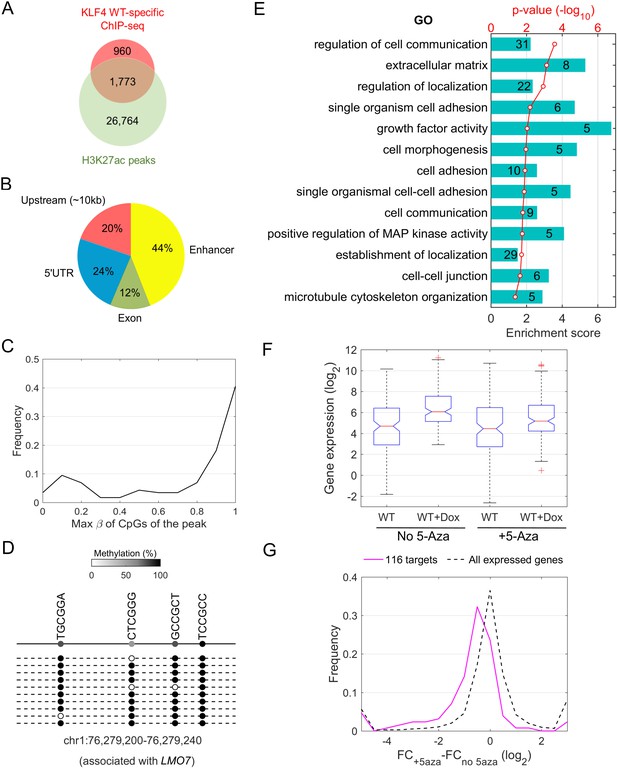
Downstream targets of KLF4-mCpG interactions.
(A) Overlap between KLF4 WT-specific ChIP peaks (2733) and the enhancer mark H3K27ac ChIP-seq peaks. A total of 1733 loci were identified. (B) A total of 116 KLF4-mCpG direct targets were identified in a serial of genome-wide studies. The overlap between WT-specific binding peaks (2733) and the 308 WT-only upregulated genes indicated that 20%, 24% and 12% of these genes were activated by KLF4 binding to mCpGs in gene upstream, 5’UTR and exon region, respectively. The overlap between enhancer regions in KLF4 WT-binding sites further identified 44% genes were activated by KLF4 binding to mCpGs in the enhancer regions. (C) Methylation level distribution of cis-elements in KLF4 binding peaks associated with 116 target genes. (D) Bisulfite sequencing confirmed DNA methylation in some of the KLF4 WT-specific binding peaks. (E) Gene ontology analysis of direct targets of KLF4-mCpG indicated that these targets have been implicated in cell adhesion, migration, cytoskeleton arrangement and cell binding activities. (F) Boxplot of gene expression for WT, WT+Dox, WT + 5 Aza, and WT + 5-Aza+Dox (G) Histogram of FC (after and before Dox) difference with and without 5-Aza for 116 target genes and all expressed genes, respectively.
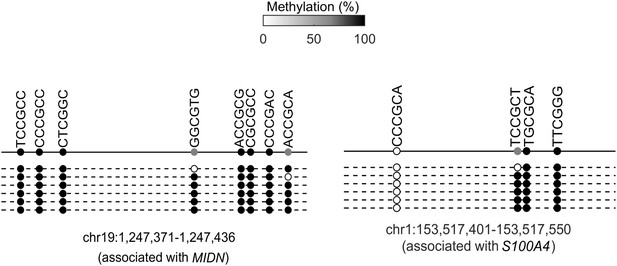
Bisulfite sequencing of the cis-regulatory elements of additional KLF4-mCpG direct targets.
https://doi.org/10.7554/eLife.20068.013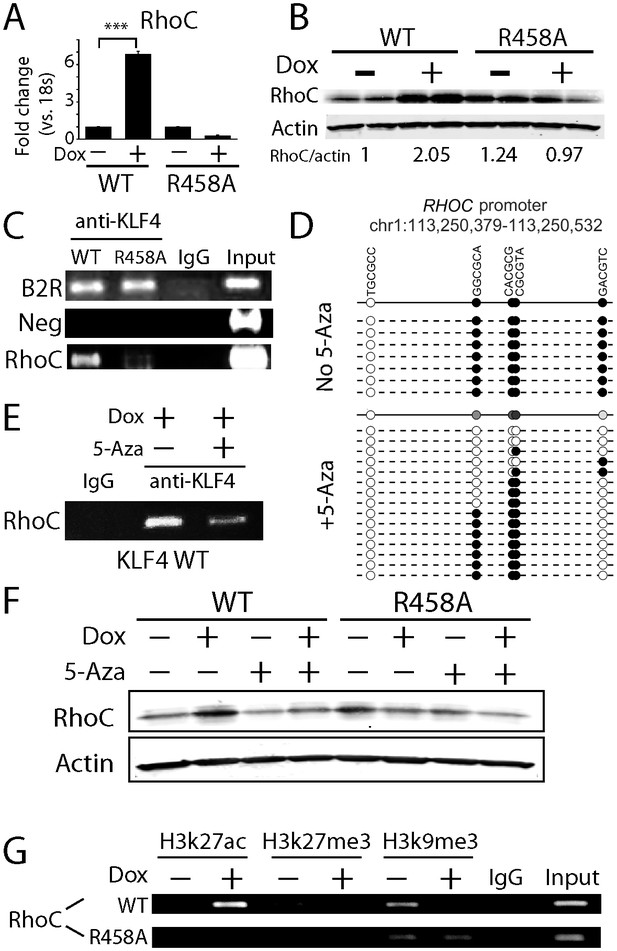
KLF4-mCpG binding activity activates RHOC expression by chromatin remodeling.
(A) Real-time PCR indicated that RHOC RNA was significantly upregulated by KLF4 WT but not R458A (+Dox, 48 hr, ***p<0.001). (B) RHOC protein expression was upregulated by KLF4 WT only; fold changes were listed under the blots. (C) A KLF4 antibody was used to precipitate crosslinked genomic DNA from cells expressing KLF4 WT and R458A. Rabbit IgG was used to control for non-specific binding. De-crosslinked DNA samples were served as the input for PCR. The RHOC promoter was only enriched in the ChIP’ed samples from KLF4 WT expressing cells. In contrast, a known KLF4 binding site on B2R promoter was used as a positive control, and detected in the ChIP’ed samples from both KLF4 WT and R458A expressing cells. A non-promoter sequence was selected as a negative control (Neg) and no band was detected. (D) Bisulfite sequencing of RHOC promoter region: each row represents one sequenced clone; each column represents one CpG site; filled circles stand for methylation. All 7 clones showed 100% methylation at four CpG sites in the RHOC promoter (~507 bp upstream from TSS). The DNMT inhibitor 5-Aza pretreatment partially reversed DNA methylation in the RHOC promoter region. (E) ChIP-PCR indicated that 5-Aza greatly abolished KLF4 WT binding to RHOC promoter region when hypomethylated. (F) Immunobloting analysis showed that KLF4 WT-induced RHOC up-regulation was blocked by pretreatment with 5-Aza. (G) KLF4-mCpG interactions in RHOC promoter region triggered histone mark changes. Tet-on KLF4 WT cells were treated with Dox for 48 hr, and genomic DNA from cells before and after Dox treatment were ChIP’ed with antibodies recognizing different histone marks and amplified for the RHOC promoter region. KLF4-mCpG interactions were associated with an increase in the active mark H3K27ac and a decreased in the repressive marks H3K27me3 and H3K9me3. Disrupted KLF4-mCpG interactions in KLF4 R458A expressing cells did not show changes in these histone marks in the RHOC promoter region.
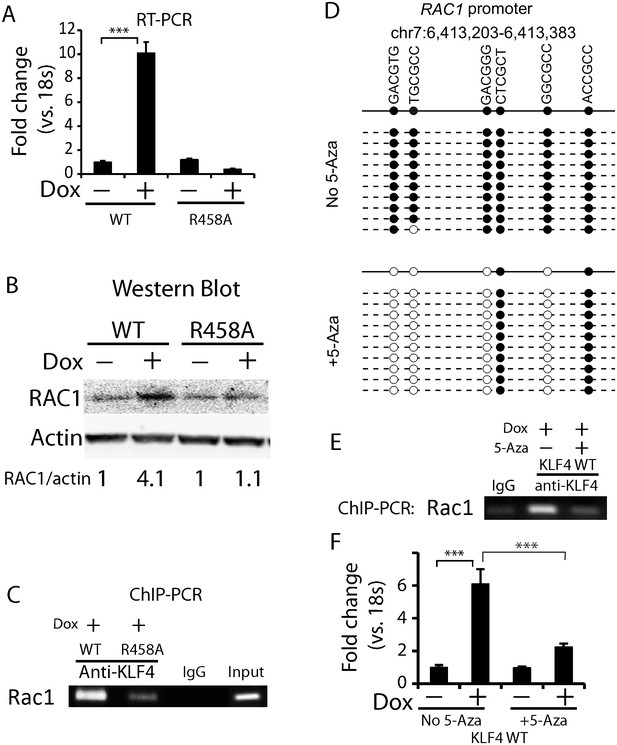
Methylated DNA in the cis-regulatory region of RAC1 determined KLF4 binding and RAC1 upregulation.
(A, B) RT-PCR and Western blot showed RAC1 was significantly up-regulated by KLF4 WT at both the mRNA and protein level. (C) ChIP-PCR showing KLF4 and RAC1 promoter binding. Compared to KLF4 R458A cells, there is an enrichment of KLF4-RAC1 promoter binding in KLF4 WT expressing cells. (D) Bisulfite sequencing of the RAC1-KLF4 binding site before and after 5-Aza treatment. RAC1 promoter was heavily methylated, whereas 5-Aza abolished methylation on these sites. (E) ChIP-PCR of KLF4-RAC1 promoter interactions in the presence of 5-Aza in KLF4 WT cells. 5-Aza treatment prevented KLF4 binding to the RAC1 promoter region when demethylated. (F) RT-PCR showing 5-Aza reversed KLF4 WT-mediated RAC1 up-regulation.
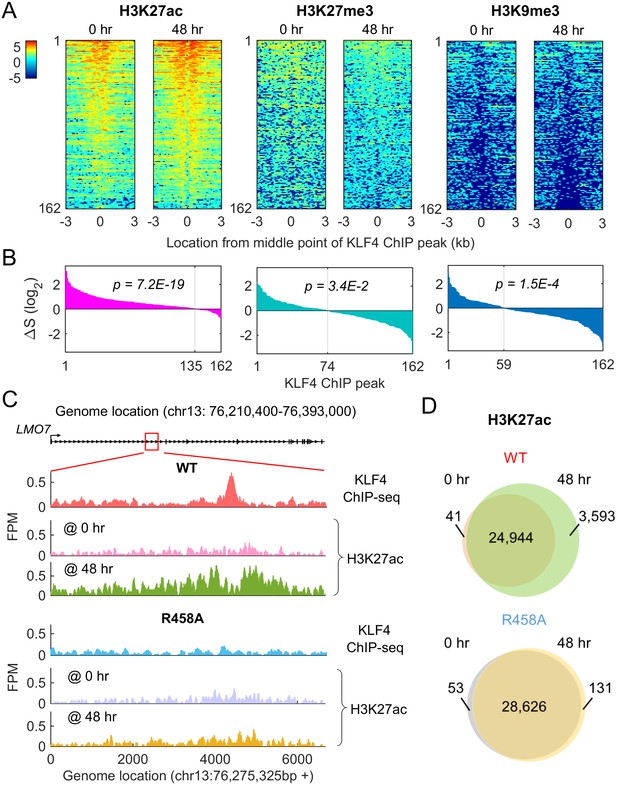
Methyl-mCpG dependent KLF4 binding activity triggers chromatin remodeling to activate gene expression.
(A) Heatmaps of histone mark signals, H3K27ac, H3K27me3, and H3K9me3, before and after KLF4-mCpG interactions (0 vs. 48 hr), respectively, ±3 kb surrounding 162 KLF4 ChIP peaks, which were associated with the 116 KLF4-mCpG direct targets. The peaks were sorted by their average signals at 48 hr for each histone mark. (B) The signal difference of histone marks between 48 hr and 0 hr, sorted from minimum to maximum. Over 83% of the 162 peaks had increased H3K27ac signals (p=7.2E-19), whereas 54.3% (p=3.4E-2) and 63.6% (p=1.5E-4) of the peaks had decrease in H3K27me3 and H3K9me3 signals, respectively. (C) Stronger H3K27ac signals were accumulated surrounding the KLF4 WT ChIP-seq peak on gene LMO7, at 48 hr after KLF4 WT induction; whereas no significant change in H3K27ac signals was observed in R458A expressing cells, as the R458A mutation abolished KLF4-mCpG binding ability. (D) Genome-wide analysis of dynamic changes of H3K27ac peaks before and after KLF4 WT or R458A induction, respectively. A total of 3593 new H3K27ac peaks appeared after KLF4 WT induction (upper panel), whereas only 131 new peaks were generated in KLF4 R458A expressing cells (lower panel), indicating that mCpG-dependent KLF4 binding activity caused chromatin remodeling to activate gene expression.
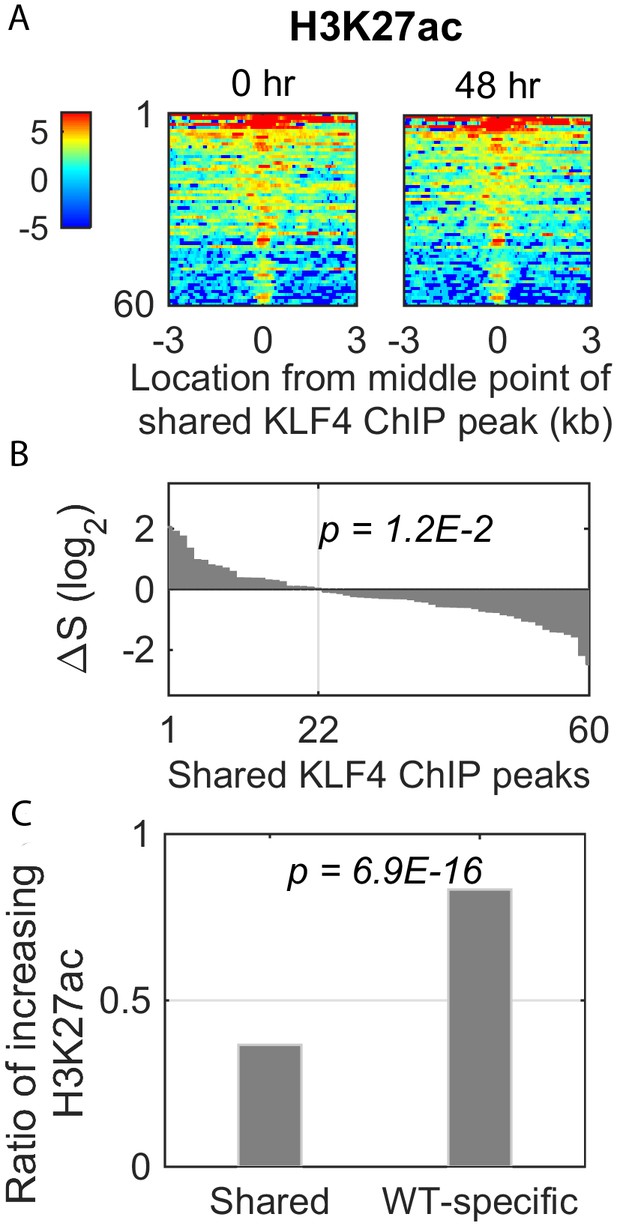
Dynamic changes of histone mark H3K27ac signal in the shared KLF4 peaks.
(A) Heatmaps of H3K27ac signals around 60 shared peaks, corresponding to up-regulated genes by KLF4 WT and R458A. (B and C) Only 36% (22/60) of the shared peaks was associated with an increase in H3K27ac, whereas there were more than 83% (135/162) of the WT-specific peaks associated with increased H3K27ac.
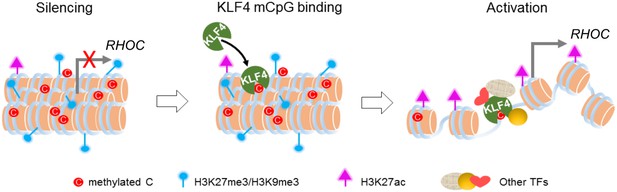
Working model of KLF4 binds to methylated cis-regulatory elements, followed by chromatin remodeling and transcription activation.
https://doi.org/10.7554/eLife.20068.018Additional files
-
Supplementary file 1
Direct downstream targets of KLF4-mCpG binding activity in GBM cells.
- https://doi.org/10.7554/eLife.20068.019
-
Supplementary file 2
Primer sequences used for bisulfite-PCR.
- https://doi.org/10.7554/eLife.20068.020





